Abstract
Overexpression of amphiregulin has been shown to induce psoriasiform changes in the skin of transgenic mice shortly after birth. Therefore, amphiregulin has been suggested as a target for anti-psoriatic therapy. To test this theory, a humanized monoclonal antibody capable of neutralizing human amphiregulin was examined for anti-proliferative effects in the human skin-severe combined immunodeficient (SCID) mouse transplant model. The anti-amphiregulin antibody reduced epidermal thickness of transplanted psoriatic skin and also inhibited the hyperplastic response that developed in nonpsoriatic skin after transplantation. The same antibody also suppressed keratinocyte proliferation in monolayer culture in a dose-dependent manner. Under the same conditions in which keratinocyte proliferation was inhibited, the antibody had little effect on proliferation of human dermal fibroblasts and no effect on type I procollagen production by these cells. Taken together, these data indicate an important role for amphiregulin in psoriatic hyperplasia and suggest that inhibition of amphiregulin activity could be an efficacious therapeutic strategy for psoriasis. These data also suggest that the hyperplastic response occurring in nonpsoriatic human skin on transplantation to the SCID mouse is mediated, in large part, by amphiregulin.
Psoriasis is an inflammatory skin disease, affecting 2 to 3% of the population.1–4 It is characterized by excessive keratinocyte proliferation, leading to a significant thickening of the epidermis, expansion of epidermal rete pegs into papillary dermal space, abnormalities in the differentiation process, and continuous shedding of the thickened epidermis. The etiology of the disease is complex and not well understood. T cells are almost certainly involved in the initiation and maintenance of psoriatic lesions.5–8 Activated T cells in the region of the dermal-epidermal junction are thought to drive the hyperplastic proliferative response through elaboration of TH1 cytokines including tumor necrosis factor-α, interferon-γ, interleukin-6, and interleukin-8.6,7,9 The prominent role of immune cells in psoriatic pathology has biased current therapeutic development efforts toward manipulating the immune system, resulting in the undesirable consequences of immunosuppression.
Abnormalities in keratinocyte function also appear to be important to the overall pathophysiology of the disease. Keratinocytes from psoriatic lesional skin have been shown to be less responsive to the growth-inhibitory effects of interferon-γ than normal keratinocytes10 and differences in cytokine generation between normal and psoriatic keratinocytes have been documented.10–12 A prominent role for epidermal growth factor (EGF) receptor function in psoriatic hyperplasia is strongly suggested. Past studies13 demonstrated that treatment of psoriatic lesional skin in organ culture with an antibody directed against the EGF receptor ameliorated abnormal histological features. Conversely, when skin from nonpsoriatic individuals or nonlesional skin from individuals with psoriasis was maintained in organ culture and treated with EGF, histological features similar to those of psoriatic lesional skin develop. Additionally, several ligands for the EGF receptor, including transforming growth factor-α, heparin-binding EGF and amphiregulin, are elevated in psoriatic lesional skin relative to control skin.14–18
Amphiregulin may be the key EGF receptor ligand in psoriasis. Studies by Cook and colleagues19,20 have demonstrated that in two amphiregulin-overexpressing transgenic mouse models, psoriasiform changes are observed in the skin shortly after birth. In one of the models, targeting amphiregulin overexpression to the basal epithelial cells, the animals also develop a synovitis, mimicking the changes seen in early-stage psoriatic arthritis.20 As a result of these past studies, amphiregulin has been suggested as a target for anti-psoriatic therapy. Based on this, we have in the present work examined a humanized anti-amphiregulin antibody for ability to suppress psoriatic hyperplasia in human skin transplanted to severe-combined immunodeficient (SCID) mice. Humanized antibodies have demonstrated therapeutic efficacy for a variety of conditions that include both infectious and immune-mediated diseases, as well as cancer. Antibody mediated cytokine-neutralization is an attractive strategy when elevated levels of the cytokine make a contribution to the pathology of the disease. This strategy has been unequivocally validated for anti-tumor necrosis factor-α agents in rheumatoid arthritis and Crohn’s disease.21 Our results suggest that an anti-amphiregulin strategy may have therapeutic efficacy in human psoriasis.
Materials and Methods
Generation of the Murine Anti-Human Amphiregulin Monoclonal Antibody
Human recombinant amphiregulin was obtained from R&D Systems (Minneapolis, MN). The recombinant protein was the 98-amino acid long form22 expressed in Escherichia coli. BALB/c mice were immunized and boosted twice with the recombinant protein. Seven days after the second boost, serum titers against human recombinant amphiregulin were determined using a standard enzyme-linked immunosorbent assay (ELISA). Splenocytes were isolated from mice with the highest sera titer against human amphiregulin and fused with SP2/0 cells. The supernatants of the resulting hybridomas were screened by ELISA to identify those specific for human amphiregulin. Specificity of antibodies for human amphiregulin was confirmed by examining all supernatants positive for binding to amphiregulin for reactivity with EGF and heparin-binding EGF by ELISA. No supernatants exhibited reactivity with these related members of the EGF family. Reactivity of antibodies with native amphiregulin was confirmed by testing hybridoma supernatants on ELISA plates coated with goat anti-human amphiregulin antibody that had captured amphiregulin produced by phorbol ester-stimulated MCF-7 cells. All antibodies demonstrating reactivity with and specificity for native human amphiregulin were purified and assessed for the ability to inhibit amphiregulin-mediated proliferation of murine 3T3 fibroblasts and human keratinocytes. One antibody of the panel, designated PAR34 (a murine IgG2b, k), possessed potent amphiregulin-neutralizing activity and was chosen for humanization.
Humanization of the PAR34 Murine Anti-Human Amphiregulin Monoclonal Antibody
PAR34 was humanized using techniques previously described.23 Briefly, the heavy and light chain variable regions of PAR34 were cloned and sequenced. The human heavy and light V region framework used as acceptors for the PAR34 complementarity determining regions (CDRs) were chosen based on sequence homology to the original murine antibody. The computer programs ABMOD and ENCAD24 were used to construct a molecular model of the variable regions. Amino acids in the humanized V regions predicted to have contact with complementarity determining regions (CDRs) were substituted with the corresponding residues of PAR34. The amino acids in the humanized V region that were found to be rare in the same V region subgroup were changed to the consensus amino acids to eliminate potential immunogenicity. The humanized light and heavy chain variable region genes were constructed and expressed as described to result in a humanized version with the isotype of IgG1, κ.23 High-yielding transfectants were expanded and used to produce purified humanized PAR34 (HuPAR34).
Functional comparison of PAR34 and HuPAR34 revealed that both murine PAR34 and HuPar34 bound specifically to human amphiregulin, but exhibited no reactivity to human EGF and heparin-binding EGF by ELISA. Biacore analysis of the murine and humanized versions revealed calculated kd values for PAR34 and HuPAR34 of 0.406 ± 0.082 nmol/L and 0.534 ± 0.050 nmol/L, respectively, indicating that the affinity of HuPAR34 to amphiregulin is ∼1.3-fold lower than that of PAR34, well within the twofold to threefold difference commonly seen on humanization. Comparison of the antibody’s capacity to inhibit amphiregulin-mediated proliferation of murine 3T3 cells indicated a similar difference with PAR34 exhibiting an IC50 of 25 ng/ml, while HuPAR34 displayed an IC50 of 55 ng/ml. Thus the humanized version of PAR34 (HuPAR34) retained the amphiregulin-neutralizing activity of the original murine antibody and all further studies involved only HuPAR34.
Other Reagents
MSL-109 (human IgG1, κ), a human antibody to glycoprotein H of cytomegalovirus25 was used as an isotype-matched control antibody for the anti-amphiregulin. Monoclonal antibodies to human CD3 (T-cell marker) and human von Willebrand factor (endothelial cell marker) were obtained from DAKO (Carpinteria, CA) and used for immunostaining of transplanted tissue.
Human Tissue
Six-mm punch biopsies of psoriatic lesional skin (four biopsies per volunteer) were obtained from four individuals with active psoriasis on the trunk or hip. None of the tissue donors were on therapy at the time of biopsy and none had been on systemic therapy for a period of at least 6 months. Six-mm punch biopsies of skin were also obtained from four nonpsoriatic volunteers (four biopsies per volunteer). The use of human tissue in this study was approved by the University of Michigan Institutional Review Board, and biopsies were obtained after receiving written informed consent from the skin donors.
Transplantation Procedure
SCID mice (CB-17 strain; Taconic Farms Inc., Germantown, NY) were used as tissue recipients. One 6-mm punch biopsy was transplanted onto the dorsal surface of a recipient mouse as described previously.26,27 Briefly, after mice were anesthetized, skin from the dorsal region was shaved. Mouse skin was surgically removed to size and replaced with the human tissue. The human tissue was secured to the back of the mice with absorbable sutures (4-0 Dexon S; Davis-Geck, Manati, Puerto Rico). The transplants were then bandaged with Xeroform petrolatum dressing (Kendall Co., Mansfield, MA) for 3 to 4 days. The animals were maintained in a pathogen-free environment throughout the preparation and treatment phases. Treatment was initiated 1 to 2 weeks after transplantation, depending on how rapidly the tissue healed. The human skin-SCID mouse transplant model has been used previously to study the pathophysiology of psoriasis28–31 and to assess potential anti-psoriatic agents.26,27,31
In Vivo Treatment Protocol
Normal human skin and psoriatic lesional plaque skin transplanted onto SCID mice were treated with humanized anti-amphiregulin or the irrelevant control antibody (MSL-109). Briefly, after allowing the tissue to heal (1 to 2 weeks), mice were treated by an intraperitoneal injection of 200 μg of antibody (∼10 mg/kg) per animal every 3 days for 28 days. At the end of the treatment period, animals were sacrificed. The transplanted skin with a small amount of surrounding mouse skin was removed and fixed in 10% buffered formalin. The skin was examined histologically after staining with hematoxylin and eosin. Tissue sections were visualized by light microscopy at ×200 magnification. Epidermal area of each tissue section was captured in equal segments (normally three to four segments across a typical tissue section) and epidermal thickness in each area was assessed at four to five points. From these values, mean epidermal thickness was determined in a blinded manner. Before transplantation, a small piece of donor tissue was fixed in 10% buffered formalin and used for zero-time assessment of epidermal thickness.
In addition to quantitative evaluation, skin grafts were also evaluated histologically for characteristic features of psoriasis including epidermal hyperplasia and rete peg formation. Tissue sections were also stained with an antibody to human CD3 to identify T cells in the tissue and with an antibody to human von Willebrand factor to identify capillaries. Staining was performed by the immunoperoxidase method using avidin-biotin complex staining as described previously.32
Human Epidermal Keratinocytes and Dermal Fibroblasts in Monolayer Culture
Epidermal keratinocytes and dermal fibroblasts were isolated from normal human skin as described previously.33 Keratinocytes were grown in keratinocyte growth medium, obtained from Cambrex (Walkersville, MD). Keratinocyte growth medium is a serum-free, low-Ca2+ (0.15 mmol/L) variant of MCDB-153 medium supplemented with EGF, insulin, and pituitary extract as growth supplements. Fibroblasts were grown in Dulbecco’s minimal essential medium supplemented with nonessential amino acids and 10% fetal bovine serum (Life Technologies, Inc., Grand Island, NY). Cells were grown at 37°C in an atmosphere of 5% CO2 and 95% air, and used at passages 3 to 4.
HuPAR34 and the control antibody (MSL-109) were examined for ability to inhibit keratinocyte proliferation in monolayer culture. Briefly, cells were plated in keratinocyte growth medium at 4 × 104 cells per well in wells of a 24-well dish. After allowing the cells to attach, the culture medium was removed and the cells were washed two times and then incubated in keratinocyte basal medium (Cambrex). Keratinocyte basal medium consists of the same basal medium as keratinocyte growth medium but is not supplemented with growth factors. Antibodies were added at the desired concentrations (5 to 50 μg/ml) and the cells incubated for 2 days. At the end of the incubation period, the cells were harvested with trypsin/ethylenediamine tetraacetic acid and counted. The assay conditions used were similar to those described previously34,35 and designed to allow autocrine stimulation of growth to occur. Amphiregulin is thought to be the major autocrine factor driving keratinocyte proliferation under these conditions.16,17,35,36 Fibroblast proliferation was examined in the same manner except that Dulbecco’s minimal essential medium-fetal bovine serum was used as plating medium and the Ca2+ concentration was increased to 1.4 mmol/L in the (keratinocyte basal medium) assay medium. Before harvest, fibroblast culture fluid was removed from each well and saved for assessment of type I procollagen (see below).
Type I Procollagen Assay
Fibroblast culture fluids were also assayed for type I procollagen by ELISA (Pan Vera Corp., Madison, WI) as described previously.37
Statistical Analysis
Differences between groups in experiments with two groups were analyzed using the Student’s t-test. Multiple group experiments were analyzed for statistical significance by analysis of variance followed by paired-group comparisons.
Results
Effects of Anti-Amphiregulin Antibody on Epidermal Thickness of Psoriatic and Nonpsoriatic Human Skin in the SCID Mouse Transplant Model
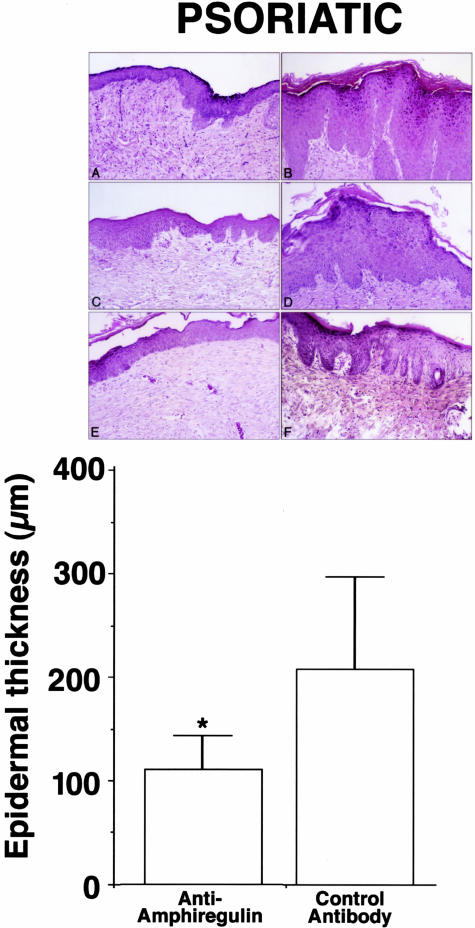
In the first series of studies, four psoriatic skin transplants were performed. Four mice were transplanted with skin from each volunteer. Two mice from each transplant were subsequently treated with the anti-amphiregulin antibody and two from each transplant were treated with the control antibody. Consistent with previous findings,26–28,30,31 the psoriatic skin maintained a hyperplastic phenotype through the period after transplantation and during treatment. Treatment of the transplant-bearing animals with the humanized anti-amphiregulin antibody, but not with the isotype-matched control antibody, suppressed the hyperproliferative conditions in the transplanted skin. The top panels of Figure 1 demonstrate histological features seen in transplanted psoriatic skin after treatment with either the anti-amphiregulin antibody (Figure 1; A, C, and E) or the isotype-matched control (Figure 1; B, D, and F). The bottom panel provides a quantitative comparison of epidermal thickness between the two groups.
Figure 1.
Effects of a monoclonal antibody to human amphiregulin on epidermal thickness of human psoriatic skin transplanted to SCID mice. Top: Histological appearance. A, C, and E: After transplantation and treatment with anti-amphiregulin; B, D, and F: after transplantation and treatment with a control antibody. Bottom: Quantitative measurements. Values shown are means and SDs based on four to six measurements per histological section in four histological sections per mouse from duplicate mice transplanted with skin samples from four psoriatic donors. Differences between groups were analyzed for statistical significance using the Student’s t-test. *, Difference from control antibody at P < 0.01 level.
In parallel studies, skin from four nonpsoriatic volunteers was transplanted to SCID mice and treated with the anti-amphiregulin and control antibodies. As expected, nonpsoriatic skin was much thinner than psoriatic plaque skin immediately on biopsy (62 ± 23 μm versus 357 ± 81 μm). Also consistent with previous results,26,27 normal skin underwent a hyperproliferative response after transplantation (316 ± 71 μm versus 62 ± 23 μm). As shown in Figure 2, treatment with anti-amphiregulin but not with the control antibody suppressed hyperplasia in the nonpsoriatic skin as effectively as it did psoriatic skin.
Figure 2.
Effects of a monoclonal antibody to human amphiregulin on epidermal thickness of human nonpsoriatic skin transplanted to SCID mice. Top: Histological appearance. A: After transplantation and treatment with anti-amphiregulin; B: after transplantation and treatment with a control antibody. Bottom: Quantitative measurements. Values shown are means and SDs based on four to six measurements per histological section in four histological sections per mouse from duplicate mice transplanted with skin samples from four nonpsoriatic donors. Differences between groups were analyzed for statistical significance using the Student’s t-test. *, Difference from control antibody at P < 0.01 level.
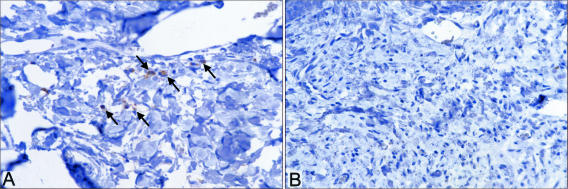
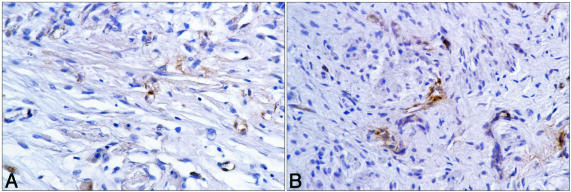
Psoriatic skin and nonpsoriatic skin were removed from mice after transplantation and treatment and examined for the presence of human T cells using antibody to CD3 as a marker. CD3+ cells were seen in sections from every psoriatic transplant examined; the number of cells per section ranged from 0 to 10 immunoreactive cells per ×400 high-power field). In contrast, none were observed in transplanted normal skin (Figure 3). There was no apparent difference between psoriatic tissue from animals treated with HuPAR34 and tissue from control antibody-treated animals. However, there were too few immunoreactive cells and too much variability to reliably determine whether there were differences between anti-amphiregulin-treated animals and animals treated with control antibody. Figure 4 demonstrates staining with antibody to human von Willebrand factor. Robust staining, indicative of a healthy microvasculature, could be seen in the dermis of both psoriatic and nonpsoriatic skin transplants after treatment with either antibody.
Figure 3.
Left: CD3+ cells (arrows) in a histological section of human psoriatic skin after transplantation to a SCID mouse (44 days after transplantation). Right: A lack of CD3+ cells in a histological section of nonpsoriatic skin after transplantation to a SCID mouse (44 days after transplantation).
Figure 4.
A: von Willebrand+ cells in histological section of human psoriatic skin after transplantation to a SCID mouse (44 days after transplantation). B: von Willebrand+ cells in a histological section of nonpsoriatic skin after transplantation to a SCID mouse (44 days after transplantation).
Effects of Anti-Amphiregulin Antibody on Epidermal Keratinocyte Proliferation in Monolayer Culture
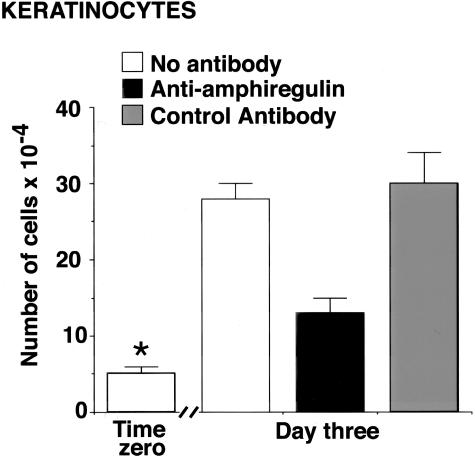
Based on the results obtained in the SCID mouse transplant model, in which treatment with an antibody to anti-amphiregulin but not with a control antibody reduced epidermal thickness, effects of the same two reagents on proliferation of human epidermal keratinocytes in monolayer culture were assessed. As shown in Figure 5, there was a significant reduction in proliferation of keratinocytes treated with anti-amphiregulin antibody, whereas no reduction was observed with the control antibody.
Figure 5.
Keratinocyte proliferation in the presence of anti-amphiregulin antibody and a control antibody. Values shown are means and SDs based on three separate experiments with 2 or 3 data points per experiment. Differences between groups were analyzed for statistical significance by analysis of variance followed by paired-group comparisons. *, Difference from untreated group at P < 0.01 level.
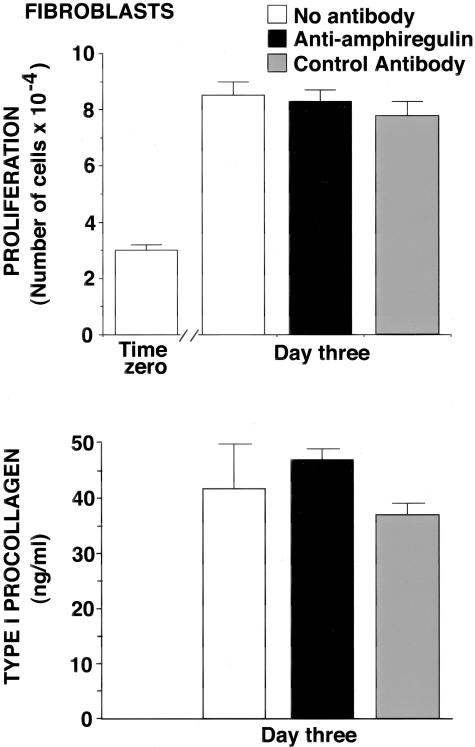
Effects of Anti-Amphiregulin Antibody on Dermal Fibroblast Proliferation and Procollagen Production in Monolayer Culture
In a final set of experiments, the anti-amphiregulin and control antibodies were examined for effects on human dermal fibroblast function. Neither antibody had an effect on proliferation (Figure 6, top). Likewise, neither reagent altered the level of type I procollagen secreted into the culture fluid (Figure 6, bottom).
Figure 6.
Top: Fibroblast proliferation in the presence of anti-amphiregulin antibody and a control antibody. Values shown are means and SDs based on three separate experiments with 2 or 3 data points per experiment. Differences between groups were analyzed for statistical significance by analysis of variance followed by paired-group comparisons. Bottom: Fibroblast production of type I procollagen in the presence of anti-amphiregulin antibody and a control antibody. Values shown are means and SDs based on three separate experiments with 2 to 3 data points per experiment. Differences between groups were analyzed for statistical significance by analysis of variance followed by paired-group comparisons. In neither assay were the anti-amphiregulin-treated cells different from untreated cells at P < 0.05.
Discussion
The data presented here demonstrate that intraperitoneal injection of a humanized anti-amphiregulin monoclonal antibody into SCID mice effectively reverses the epidermal hyperplasia present in human psoriatic lesional skin transplanted onto the mice. A control monoclonal antibody does not have this effect. It has been shown previously in two amphiregulin-overexpressing transgenic mouse models that psoriasiform changes develop in the skin shortly after birth.19,20 In one of the models (amphiregulin overexpressed in the basal epithelial layer), the animals also develop a synovitis, mimicking the changes seen in early-stage psoriatic arthritis.20 Additionally, it has been shown that amphiregulin is minimally expressed in the normal epidermis, but elevated in psoriatic lesional skin relative to control.18,38 Based on these past findings, amphiregulin has been suggested as an attractive target for anti-psoriatic therapy. The present results strongly support this suggestion.
How elevated amphiregulin contributes to epidermal hyperplasia in psoriasis is not fully understood. Based on the observations that suppression of amphiregulin not only reduced psoriatic hyperplasia, but also inhibited the hyperplasia that developed in nonpsoriatic skin after transplantation to SCID mice (Figure 2) and reduced autocrine keratinocyte proliferation in monolayer culture (Figure 5), a direct effect on keratinocyte function is strongly suggested. This is consistent with previous studies demonstrating the critical role for amphiregulin in autocrine keratinocyte proliferation.16,17,34–36 Activation of the EGF receptor is known to induce signaling through mitogen-activated protein kinase pathways, with subsequent effects on cell cycle control.39 Presumably, cytokines (eg, tumor necrosis factor-α, interferon-γ, and perhaps others) generated by activated T cells or other immunological/inflammatory cells in the developing psoriatic lesion trigger amphiregulin production in the epidermis, leading to autocrine or paracrine growth stimulation. It should be remembered, of course, that psoriasis is a complex pathophysiological process and that cytokines and growth factors generated by hyperplastic epidermal keratinocytes also influence immune cell function. For example, keratinocyte stimulation through the EGF receptor results in elaboration of interleukin-1α40 and interleukin-841 while down-regulating CCL2 (MCP-1), CCL5 (RANTES), and CXCL10 (IP-10) elaboration.41 The end result is an amplification loop involving epidermis and immune cell interactions. Presumably, growth factor/cytokine dysregulation also contributes to the inflammatory joint disease seen in many psoriatic patients. Thus, although amphiregulin has never been shown to directly influence immune cell function, inhibition of amphiregulin might be expected to down-regulate epithelial elaboration of cytokines and growth factors that do influence components of the immune/inflammatory system.
One might suggest, based on suppression of the hyperplastic response in transplanted nonpsoriatic skin and inhibition of keratinocyte proliferation in monolayer culture, that targeting amphiregulin in psoriasis would not provide the necessary specificity for an effective therapeutic approach. However, this may not necessarily be the case. Although keratinocyte proliferation in monolayer culture in the absence of exogenous growth factors appears to be dependent on autocrine production of amphiregulin (this report)16,17,34–36 and although the present findings strongly suggest that the hyperplastic response in normal skin transplanted to SCID mice also depends on elaboration of amphiregulin in the transplanted tissue, neither of these conditions represents quiescent normal skin. Past studies have shown that when normal human skin is maintained in organ culture under serum-free, growth factor-free conditions, the epidermis remains viable and healthy33 but does not undergo hyperplastic changes as it does after transplantation to SCID mice.26,27 Treatment with either a blocking antibody to the EGF receptor13 or an irreversible EGF receptor tyrosine kinase (EGF-RTK) antagonist42 has no apparent effect on histological features of the organ-cultured tissue. At the same time, blocking EGF receptor function suppresses hyperplasia in organ cultures of psoriatic skin13 as well as hyperplasia induced by retinoid treatment.42 Taken together these findings suggest that hyperplastic proliferation in human skin epidermis is dependent on an intact EGF receptor system whereas physiological proliferation occurs under conditions in which EGF receptor function is blocked. This suggestion is further supported by studies showing that although antibody to EGF receptor does not suppress physiological proliferation, antibody to insulin-like growth factor-1 is inhibitory under the same conditions.43 Blocking amphiregulin function in patients with psoriasis could be envisioned, therefore, to interfere with hyperplastic changes in the epidermis without affecting normal skin function.
A better indication of specificity may be the dermal fibroblast response. Despite its ability to suppress epidermal hyperplastic growth in the epidermis of transplanted normal skin and its ability to suppress autocrine keratinocyte growth in monolayer culture, the anti-amphiregulin antibody did not have a measurable effect on fibroblast function when used at doses that suppressed epidermal proliferation. In dermal fibroblast cultures, neither proliferation nor type I procollagen synthesis was altered. In this respect, targeting amphiregulin produces a different response than was observed previously when the potent corticosteroid, clobetasol propionate, was used in the same model. Treatment with this agent suppressed the psoriatic hyperplasia, but concomitantly produced thinning of the normal skin as well.26 Corticosteroids are known to be effective anti-psoriatic agents44,45 but a consequence of their continued use is dermal atrophy.46
In summary, previous studies16–20,38 have led to the suggestion that targeting amphiregulin may provide a useful approach to treatment of psoriasis. Our findings in the human skin-SCID mouse transplant model support this suggestion. Although it will be necessary to verify efficacy of an anti-amphiregulin approach in clinical studies, having positive findings in the transplant model provides hope for eventual success, and supports pursuing the clinical development of HuPAR34 as a potential treatment for psoriasis.
Footnotes
Address reprint requests to James Varani, Ph.D., Department of Pathology, University of Michigan, 1301 Catherine Rd., Box 0602, Ann Arbor, MI 48109. E-mail: varani@umich.edu.
Supported in part by the United States Public Health Service (grants DK59169 and AR49621 to J.V.).
References
- Krueger GC, Bergstresser PR, Lowe NJ, Voorhees JJ, Weinstein GD. Psoriasis. J Am Acad Dermatol. 1984;11:937–947. doi: 10.1016/s0190-9622(84)80018-3. [DOI] [PubMed] [Google Scholar]
- Fry L. Psoriasis. Br J Dermatol. 1988;119:445–461. doi: 10.1111/j.1365-2133.1988.tb03248.x. [DOI] [PubMed] [Google Scholar]
- Sander HM, Morris LF, Phillips CM, Harrison PE, Menter A. The annual cost of psoriasis. J Am Acad Dermatol. 1993;128:422–425. doi: 10.1016/0190-9622(93)70062-x. [DOI] [PubMed] [Google Scholar]
- Weiss SC, Kimball AB, Liewehr DJ, Blauvelt A, Turner ML, Emanuel EJ. Quantifying the harmful effect of psoriasis on health-related quality of life. J Am Acad Dermatol. 2002;47:512–518. doi: 10.1067/mjd.2002.122755. [DOI] [PubMed] [Google Scholar]
- Valdimarsson H, Baker BS, Jonsdottir I, Powles A, Fry L. Psoriasis: a T-cell mediated autoimmune disease mediated by streptococcal superantigen? Immunol Today. 1995;16:145–149. doi: 10.1016/0167-5699(95)80132-4. [DOI] [PubMed] [Google Scholar]
- Austin LM, Ozawa M, Kikuchi T, Walters IB, Kueuger JG. The majority of epidermal T cells in psoriasis vulgaris lesions can produce type 1 cytokines—interferon-γ, interleukin-2 and tumor necrosis factor-α—defining TC1 (cytotoxic T lymphocyte) and TH1 effector populations: a type 1 differentiation bias also measured in circulating blood T cells in psoriasis patients. J Invest Dermatol. 1999;113:101–108. doi: 10.1046/j.1523-1747.1999.00749.x. [DOI] [PubMed] [Google Scholar]
- Baker BS, Brent L, Valdimarsson H, Powles AV, al-Imara L, Walker M, Fry L. Is epidermal proliferation in psoriatic skin grafts on nude mice driven by T-cell derived cytokines? Br J Dermatol. 1992;126:105–110. doi: 10.1111/j.1365-2133.1992.tb07805.x. [DOI] [PubMed] [Google Scholar]
- Wong RL, Winslow CM, Cooper KD. The mechanisms of action of cyclosporin A in the treatment of psoriasis. Immunol Today. 1993;14:69–74. doi: 10.1016/0167-5699(93)90061-o. [DOI] [PubMed] [Google Scholar]
- Dustin ML, Singer KH, Tuck DT, Springer TA. Adhesion of T lymphoblasts to epidermal keratinocytes is regulated by interferon-gamma and is mediated by intercellular adhesion molecule-1. J Exp Med. 1988;167:1323–1340. doi: 10.1084/jem.167.4.1323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nickoloff BJ, Mitra RS, Elder JT, Fisher GJ, Voorhees JJ. Decreased growth inhibition by recombinant gamma interferon is associated with increased production of transforming growth factor alpha in keratinocytes cultured from psoriatic lesions. Br J Dermatol. 1989;121:161–174. doi: 10.1111/j.1365-2133.1989.tb01795.x. [DOI] [PubMed] [Google Scholar]
- Nickoloff BJ. The cytokine network in psoriasis. Arch Dermatol. 1991;127:871–884. [PubMed] [Google Scholar]
- Goebeler M, Toksoy A, Spandau U, Engelhardt E, Broker EB, Gillitzer R. The C-X-C chemokine Mig is highly expressed in the papillae of psoriatic lesions. J Pathol. 1998;184:89–95. doi: 10.1002/(SICI)1096-9896(199801)184:1<89::AID-PATH975>3.0.CO;2-Z. [DOI] [PubMed] [Google Scholar]
- Varani J, Kang S, Stoll S, Elder JT. Human psoriatic skin in organ culture: comparison with normal skin exposed to exogenous growth factors and effects of an antibody to the EGF receptor. Pathobiology. 1998;66:253–259. doi: 10.1159/000028031. [DOI] [PubMed] [Google Scholar]
- Gottlieb AB, Chang CK, Posnett DN, Fanelli B, Tam JP. Detection of transforming growth factor-alpha in normal, malignant and hyperproliferative human keratinocytes. J Exp Med. 1988;167:670–675. doi: 10.1084/jem.167.2.670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elder JT, Fisher GJ, Lindquist PB, Bennett GJ, Pittelkow MR, Coffey RJ, Ellingsworth L, Derynck R, Voorhees JJ. Overexpression of transforming growth factor-α in psoriatic epidermis. Science. 1989;243:811–814. doi: 10.1126/science.2916128. [DOI] [PubMed] [Google Scholar]
- Piepkorn M, Pittelkow MR, Cook PW. Autocrine regulation of keratinocytes: the emerging role of heparin-binding, epidermal growth factor-related growth factors. J Invest Dermatol. 1998;111:715–721. doi: 10.1046/j.1523-1747.1998.00390.x. [DOI] [PubMed] [Google Scholar]
- Piepkorn M, Predd H, Underwood R, Cook P. Proliferation-differentiation relationships in the expression of heparin-binding epidermal growth factor-related factors and erbB receptors by normal and psoriatic human keratinocytes. Arch Dermatol Res. 2003;295:93–101. doi: 10.1007/s00403-003-0391-x. [DOI] [PubMed] [Google Scholar]
- Cook PW, Pittelkow MR, Keeble WW, Graves-Deal R, Coffey RJ, Jr, Shipley GD. Amphiregulin messenger RNA is elevated in psoriatic epidermis and gastrointestinal carcinomas. Cancer Res. 1992;52:3224–3227. [PubMed] [Google Scholar]
- Cook PW, Piepkorn M, Clegg CH, Plowman GD, DeMay JM, Brown JR, Pittelkow MR. Transgenic expression of the human amphiregulin gene induces a psoriasis-like phenotype. J Clin Invest. 1997;100:2286–2294. doi: 10.1172/JCI119766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cook PW, Brown JR, Cornell KA, Pittelkow MR. Suprabasal expression of human amphiregulin in the epidermis of transgenic mice induces a severe, early-onset, psoriasis-like skin pathology: expression of amphiregulin in the basal epidermis is also associated with synovitis. Exp Dermatol. 2004;13:347–356. doi: 10.1111/j.0906-6705.2004.00183.x. [DOI] [PubMed] [Google Scholar]
- Palladino MA, Bahjat FR, Theodorakis EA, Moldawer LL. Anti-TNF-alpha therapies: the next generation. Nat Rev Drug Discov. 2003;2:736–746. doi: 10.1038/nrd1175. [DOI] [PubMed] [Google Scholar]
- Plowman GD, Green JM, McDonald VL, Neubauer MG, Disteche CM, Todaro GJ, Shoyab M. The amphiregulin gene encodes a novel epidermal growth factor-related protein with tumor-inhibitory activity. Mol Cell Biol. 1990;10:1969–1981. doi: 10.1128/mcb.10.5.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Queen C, Schneider WP, Selick HE, Payne PW, Landolfi NF, Duncan JF, Avdalovic NM, Levitt M, Junghans RP, Waldmann TA. A humanized antibody that binds to the interleukin 2 receptor. Proc Natl Acad Sci USA. 1989;186:10029–10033. doi: 10.1073/pnas.86.24.10029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levitt M. Molecular dynamics of native protein. I. Computer simulation of trajectories. J Mol Biol. 1983;168:595–617. doi: 10.1016/s0022-2836(83)80304-0. [DOI] [PubMed] [Google Scholar]
- Drobyski WR, Gottlieb M, Carrigan D, Ostberg L, Grebenau M, Schran H, Magid P, Ehrlich P, Nadler PI, Ash RC. Phase I study of safety and pharmacokinetics of a human anticytomegalovirus monoclonal antibody in allogeneic bone marrow transplant recipients. Transplantation. 1991;51:1190–1196. doi: 10.1097/00007890-199106000-00009. [DOI] [PubMed] [Google Scholar]
- Zeigler M, Chi Y, Tumas DB, Bodary S, Tang H, Varani J. Anti-CD11a ameliorates disease in the human psoriatic skin-SCID mouse transplant model: comparison of antibody to CD11a with cyclosporin A and clobetasol propionate. Lab Invest. 2001;81:1253–1261. doi: 10.1038/labinvest.3780339. [DOI] [PubMed] [Google Scholar]
- Ellis CN, Varani J, Fisher GJ, Pershadsingh HA, Benson SC, Kurtz TW. Troglitazone improves psoriasis and normalizes models of proliferative skin disease. Arch Dermatol. 2000;136:609–615. doi: 10.1001/archderm.136.5.609. [DOI] [PubMed] [Google Scholar]
- Nickoloff BJ, Kunkel SL, Burdick M, Strieter RM. Severe combined immunodeficiency mouse and human psoriatic skin chimeras. Validation of a new animal model. Am J Pathol. 1995;l46:580–588. [PMC free article] [PubMed] [Google Scholar]
- Wrone-Smith T, Nickoloff BJ. Dermal injection of immunocytes induces psoriasis. J Clin Invest. 1996;98:1878–1887. doi: 10.1172/JCI118989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilhar A, David M, Ullmann Y, Berkutski T, Kalish RS. T-lymphocyte dependence of psoriatic pathology in human psoriatic skin grafted to SCID mice. J Invest Dermatol. 1997;109:283–288. doi: 10.1111/1523-1747.ep12335758. [DOI] [PubMed] [Google Scholar]
- Dam TM, Kang S, Nickoloff BJ, Voorhees JJ. 1α25-dihydroxycholecalciferol and cyclosporin suppress induction and promote resolution of psoriasis in human skin grafts transplanted on to SCID mice. J Invest Dermatol. 1999;113:1082–1089. doi: 10.1046/j.1523-1747.1999.00811.x. [DOI] [PubMed] [Google Scholar]
- Varani J, Hattori Y, Chi Y, Schmidt T, Perone P, Zeigler ME, Fader DJ, Johnson TM. Collagenolytic and gelatinolytic matrix metalloproteinases and their inhibitors in basal cell carcinoma of skin: comparison with normal skin. Br J Cancer. 2000;82:657–665. doi: 10.1054/bjoc.1999.0978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Varani J, Perone P, Griffiths CEM, Inman D, Fligiel SEG, Voorhees JJ. All-trans retinoic acid stimulates events in organ-cultured skin that underlie repair. J Clin Invest. 1994;94:1747–1753. doi: 10.1172/JCI117522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pittelkow MR, Cook PW, Shipley GD, Derynck R, Coffey RJ., Jr Autonomous growth of human keratinocytes requires epidermal growth factor receptor occupancy. Cell Growth Differ. 1993;4:513–521. [PubMed] [Google Scholar]
- Cook PW, Mattox PA, Keeble WW, Pittelkow MR, Plowman GD, Shoyab M, Adelman JP, Shipley GD. A heparin sulfate-regulated human keratinocyte autocrine factor is similar or identical to amphiregulin. Mol Cell Biol. 1991;11:2547–2557. doi: 10.1128/mcb.11.5.2547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cook PW, Pittelkow MP, Shipley GD. Growth factor-independent proliferation of normal human neonatal keratinocytes: production of autocrine and paracrine mitogenic factors. J Cell Physiol. 1991;146:277–289. doi: 10.1002/jcp.1041460213. [DOI] [PubMed] [Google Scholar]
- Varani J, Warner RL, Mehrnaz G-K, Phan SH, Kang S, Chung JH, Wang ZQ, Datta SC, Fisher GJ, Voorhees JJ. Vitamin A antagonizes decreased cell growth and elevated collagen-degrading matrix metalloproteinases and stimulates collagen accumulation in naturally aged human skin. J Invest Dermatol. 2000;114:480–486. doi: 10.1046/j.1523-1747.2000.00902.x. [DOI] [PubMed] [Google Scholar]
- Piepkorn M. Overexpression of amphiregulin, a major autocrine growth factor for cultured human keratinocytes, in hyperproliferative skin diseases. Am J Dermatopathol. 1996;18:165–171. doi: 10.1097/00000372-199604000-00010. [DOI] [PubMed] [Google Scholar]
- Zeigler ME, Chi Y, Schmidt T, Varani J. Role of ERK and JNK pathways in regulating cell motility and matrix metalloproteinase 9 production in growth factor-stimulated human epidermal keratinocytes. J Cell Physiol. 1999;180:271–284. doi: 10.1002/(SICI)1097-4652(199908)180:2<271::AID-JCP15>3.0.CO;2-D. [DOI] [PubMed] [Google Scholar]
- Hobbs RM, Watt FM. Regulation of interleukin-1alpha expression by integrins and epidermal growth factor receptor in keratinocytes from a mouse model of inflammatory skin disease. J Biol Chem. 2003;278:19798–19807. doi: 10.1074/jbc.M300513200. [DOI] [PubMed] [Google Scholar]
- Mascia F, Mariani V, Girolomoni G, Pastore S. Blockade of the EGF receptor induces a deranged cytokine expression in keratinocytes leading to enhanced skin inflammation. Am J Pathol. 2003;163:303–312. doi: 10.1016/S0002-9440(10)63654-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Varani J, Zeigler M, Dame MK, Kang S, Fisher GJ, Voorhees JJ, Stoll SW, Elder JT. HB-EGF activation of keratinocyte ErbB receptors mediates epidermal hyperplasia, a prominent side effect of retinoid therapy. J Invest Dermatol. 2001;117:1335–1341. doi: 10.1046/j.0022-202x.2001.01564.x. [DOI] [PubMed] [Google Scholar]
- Tavakkol A, Varani J, Elder JT, Zouboulis CC. Maintenance of human skin in organ culture: role for insulin-like growth factor-1 receptor and epidermal growth factor receptor. Arch Dermatol Res. 1999;291:643–651. doi: 10.1007/s004030050469. [DOI] [PubMed] [Google Scholar]
- Greaves MW, Weinstein GD. Treatment of psoriasis. N Engl J Med. 1995;332:581–588. doi: 10.1056/NEJM199503023320907. [DOI] [PubMed] [Google Scholar]
- McKay IA, Winyard P, Leigh IM, Bustin SA. Nuclear transcription factors as potential targets for new modes of intervention in skin disease. Br J Dermatol. 1994;131:591–592. doi: 10.1111/j.1365-2133.1994.tb04968.x. [DOI] [PubMed] [Google Scholar]
- McMichael AJ, Griffiths CEM, Talwar HA, Finkel LJ, Rafal ES, Hamilton TA, Voorhees JJ. Concurrent application of tretinoin (retinoic acid) partially protects against corticosteroid-induced epidermal atrophy. Br J Dermatol. 1996;135:60–64. [PubMed] [Google Scholar]