SYNOPSIS
Objective
Men have higher mortality rates than women for most causes of death. This study was conducted to determine the contribution of specific causes of death to the sex difference in years of potential life lost (YPLL).
Methods
The authors examined data from the National Health Interview Survey with linked mortality data through 1997. Using survival analysis estimates, a stochastic simulation model to simulate death events for cohorts of white, African American, and Latino adults was created.
Results
YPLL from all causes were greater among men than women. Homicide, motor vehicle accidents, and suicide accounted for 33% of YPLL sex difference among whites, 36% among African Americans, and 52% among Latinos. For all three racial/ethnic groups, cardiovascular disease (principally ischemic heart disease) was the second largest contributor to the sex difference in YPLL (29% among whites, 23% among African Americans, and 25% among Latinos). Lung cancer was also important among whites and African Americans, accounting for 15% and 17% of the sex difference in YPLL from all causes, respectively.
Conclusions
Ischemic heart disease, lung cancer, and traumatic deaths account for as much as three-quarters of the excess YPLL among men, suggesting that a few modifiable behaviors such as the use of tobacco, alcohol, and drugs and violence may account for much of the shorter life expectancy among men.
While mortality rates have dropped for both men and women over the last century, women have consistently had longer life expectancy. In 1900, women lived on average two more years than men,1 and most recent estimates from 2002 indicate this gap is now just over five years.2 The sex differential in life expectancy appears to be attributable to a broad range of causes. Specifically, men have higher relative death rates from ischemic heart disease, cancer, stroke, diabetes, liver disease, kidney disease, pneumonia, and accidents.3 This suggests that a variety of biologic and behavioral mechanisms might be involved in the different health trajectories of men and women.
Studies examining specific causes of death can be useful in identifying the relative impact of various diseases on sex differences in life expectancy. This information is important because it can suggest which mechanisms are more likely to account for sex differences in life expectancy and also help direct future studies toward those diseases that have the biggest impact. However, previous studies examining sex differences in cause-specific mortality are limited because they have largely examined mortality rates and mortality rate ratios,4 which do not completely account for the burden of premature death.5 Vital statistics reports have examined years of potential life lost (YPLL) for men and women,6 but these reports estimated YPLL occurring over a one-year period rather than for a cohort of individuals followed over their lifetime. This “cross-sectional” estimate of YPLL is potentially misleading.7
In the present study, we examined death data from a nationally representative sample of adults in the U.S. and estimated the sex differences in YPLL over a lifetime. We also examined whether sex differences in YPLL vary by race or ethnicity.
METHODS
Overview
The goal of the study was to estimate differences in YPLL before age 75 from specific causes of death between men and women. YPLL are the years a person would have lived (up to an age cutoff) had he or she not died, with the number of years attributed to a specific cause.5 To estimate YPLL, we first estimated the hazard of death from specific causes using national data. We then used estimated survival times to simulate mortality events in a stochastic simulation model for a cohort of 25-year-olds followed until death. Similar methods are described elsewhere.8
Data
We analyzed data from the National Health Interview Survey (NHIS), an annual cross-sectional study of a nationally representative sample of households, the primary purpose of which was to assess the prevalence of illness, disability, and chronic disease among the U.S. population.9 National Death Index data through December 1997 has been linked to NHIS participants surveyed from 1986 to 1994.10 These nine cross-sectional samples include 1,009,997 children and adults. We examined data on non-Latino whites (whites), non-Latino African Americans (African Americans), and Latinos of all races and excluded 36,795 individuals (3.6%) from other racial/ethnic groups. Because we were primarily interested in adults, we also excluded an additional 361,487 individuals (35.8%) younger than 25 years old. We excluded an additional 20,650 individuals (2.0%) because they had inadequate identifying information, which precluded reliable linkage to death certificates, and 5,810 individuals (0.6%) because of missing key demographic data.
Data analysis
We used NHIS data to estimate the hazard of death from specific causes. We examined the single underlying cause of death derived from all the causes listed on the death certificate.11 Causes of death were then coded using the International Classification of Diseases-9 (ICD-9).12 We examined deaths from cardiovascular disease (ischemic heart disease, cerebrovascular stroke, hypertension, congestive heart failure, other atherosclerotic disease, and other cardiovascular disease), cancer (colon cancer, other gastrointestinal cancers, gynecologic cancers, prostate cancer, lung cancer, breast cancer, hematologic malignancies, and other cancer), infection (pneumonia, acquired immunodeficiency syndrome [AIDS], and other infection), lung disease, diabetes mellitus, liver disease, and trauma (motor vehicle accident, suicide, homicide, and other accidents). All other causes of death were categorized as “other causes.”
To estimate cause-specific death hazards, we used parametric survival analysis rather than a Cox proportional hazards model, which is more commonly used, because the latter method treats the hazard function as a nuisance parameter.13 To find the best fitting parametric distribution for each cause of death, we compared various distributions with the non-parametric hazard curve using the Kaplan-Meier method.13 For almost all of the causes of death, we used a Weibull distribution.13 For traumatic deaths and HIV, which did not fit standard parametric distributions, we used a piecewise exponential model.14 Covariates in each model were sex, self-reported race/ethnicity (white vs. African American vs. Latino), and education (less than high school vs. high school graduate or more), as well as all two- and three-way interactions. Competing risks of death from other causes were treated as non-informative censoring.
Because death data were available only through 1997, the data do not accurately account for more recent trends in AIDS mortality. Therefore, we used national AIDS surveillance data to determine the trends in AIDS mortality that occurred from 1994 to 2002.15 According to these estimates, AIDS mortality over this time period dropped by 23% to 85%, depending on the age, gender, and race/ethnicity of the population. Using these estimates, we proportionally adjusted the hazard estimates of AIDS mortality for the decline in AIDS mortality that occurred after 1997, stratified by sex, race/ethnicity, and 10-year age categories. For the survival analyses, we used analytic weights that account for the sampling methods used in NHIS,9,10 sample loss because some individuals could not be linked to the National Death index8 and the combining of nine different NHIS samples.8
Simulation model
We used a stochastic simulation model to simulate death events for a cohort of 25-year-olds. For each person in the cohort, we used the estimated survival functions (based on the person's sex, race/ethnicity, and education level) to simulate the age at which the person would die for each of the 25 specific causes of death, assuming that death is a non-competing event. This process yields 25 possible death events, but of course, each cause of death is a competing event (e.g., a person cannot die of cancer after they have already died of a stroke). Thus, the age and specific cause of death was determined based on the earliest of the simulated mortality events. This process was then repeated for each person in the cohort. The cohort consisted of six sex/race/ethnicity strata (white men and women, African American men and women, and Latino men and women) comprised of 80,000 people in each stratum, which was the minimum size to yield consistent and reliable estimates, given our model design and parameters.
Finally, we calculated YPLL as the number of years lived with no premature death (which we set at 75 years) minus the actual age of death. For those dying at age 75 or later, YPLL equals zero. Estimates were made separately for each sex/race/ethnicity strata. We reported the difference in YPLL between men and women for each race/ethnicity group, standardized to the education distribution of the U.S. adult population based on 2000 NHIS data. We also calculated YPLL using ages 65 and 85 as the age cutoff. Results were very similar in these sensitivity analyses; thus, we present only the results for YPLL before age 75.
In addition to calculating YPLL, we also estimated the potential gain in life expectancy (PGLE) that would occur if a particular cause of death were eliminated. PGLE is superior to YPLL because it better accounts for competing causes of death.16 As with previous analyses,8 the PGLE results (not shown) were similar to the YPLL results. Thus, for simplicity's sake we have presented only the YPLL results.
To estimate standard errors, we used a simulation method approach17 in which we randomly drew 1,000 sets of parameter estimates, β, from each of the survival analyses. Each set of parameter estimates was drawn using the variance-covariance matrix of the estimated models. Thus, each draw captures the uncertainty around β. For each set of parameters, we ran the simulation and estimated the gender difference in YPLL using the methods described above. This yielded 1,000 estimates of YPLL, from which we calculated confidence intervals using the percentile method. To compare YPLL among men and women, we also estimated two-sided p-values by examining the centile for the male-female difference in YPLL equal to zero. We used STATA version 8.018 and SAS version 8.0219 software for all analyses.
RESULTS
Sex differences in YPLL for major categories of death
For all three racial/ethnic groups, men lost more years of potential life before age 75 than women from all causes of death combined. The greatest difference was among African Americans (2,886 years per 1,000 individuals; 95% confidence interval [CI] 2,606, 3,231), followed by Latinos (2,406 years; 95% CI 1,929, 2,879) and whites (1,845 years; 95% CI 1,731, 1,954) (see Table 1). With just a few exceptions, YPLL was significantly greater among men than women from almost all of the major causes of death that we examined. These included cardiovascular disease, cancer, infection, lung disease, diabetes, liver disease, and traumatic deaths. For all three racial/ethnic groups, trauma contributed the most to the sex difference in YPLL, accounting for 33% (618/1,845 years per 1,000 individuals) of the YPLL due to all causes among whites, 36% (1,032/2,886) among African Americans, and 52% (1,245/2,406) among Latinos. Cardiovascular disease accounted for approximately one-quarter of the sex difference in YPLL for all three racial/ethnic groups. Cancer accounted for 15% of the sex difference in YPLL among whites and 20% among African Americans, but only 2% among Latinos.
Table 1.
Years of potential life lost (per 1,000 persons) by major causes of death and race/ethnicity

NOTES: Columns may not sum to the totals shown because of rounding. Years of potential life lost before age 75 (per 1,000 persons) = Σ(75 − age at death among those dying before age 75) × 1,000/n, where n=total number of persons at risk of dying. Percentages may not add to 100% due to rounding error.
p<0.002
p<0.01
p<0.05
Sex differences in YPLL from specific causes of death
We examined specific types of traumatic deaths, including motor vehicle accidents, suicide, and homicide and found different patterns across the three racial/ethnic groups (Table 2). Among African Americans, men lost 539 more years of potential life per 1,000 individuals than women due to homicide, which accounted for 19% of the total YPLL disparity. In comparison, the male-female difference in YPLL due to homicide was 481 years per 1000 (20%) among Latinos, but only 41 years (2%) among whites. In contrast, suicide accounted for 15% of the sex disparity in YPLL among whites, 10% among Latinos, and 5% among African Americans.
Table 2.
Years of potential life lost (per 1,000 persons) by specific types of traumatic deaths and race/ethnicity

NOTES: Columns may not sum to the totals shown because of rounding. Years of potential life lost before age 75 (per 1,000 persons) = Σ(75 − age at death among those dying before age 75) × 1,000/n, where n=total number of persons at risk of dying. Some columns may not adds up due to rounding error.
p<0.002
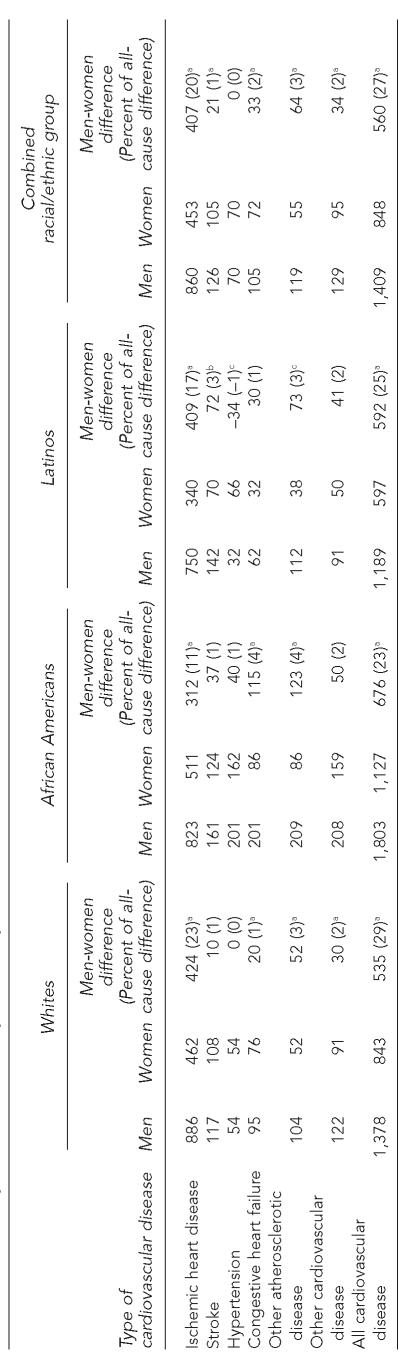
Cardiovascular disease is the second largest contributor to the sex difference in YPLL, most of which is due to ischemic heart disease (Table 3). Among whites, ischemic heart disease accounted for 23% (424/1,845) of the male-female difference in YPLL from all causes, compared to 11% among African Americans and 17% among Latinos. The remainder of the cardiovascular causes of death, which included cerebrovascular strokes, hypertension, and congestive heart failure, contributed much less to the sex difference in YPLL. Similar patterns in the contribution of cardiovascular disease were observed for all three racial/ethnic groups.
Table 3.
Years of potential life lost (per 1,000 persons) from cardiovascular causes of death
NOTES: Columns may not sum to the totals shown because of rounding. Years of potential life lost before age 75 (per 1,000 persons) = Σ(75 − age at death among those dying before age 75) × 1,000/n, where n=total number of persons at risk of dying. Some columns may not adds up due to rounding error.
p<0.002
p<0.01
p<0.05
As expected, YPLL from breast and gynecologic cancers was almost entirely among women and YPLL from prostate cancer was restricted to men (Table 4). For the remainder of the specific types of cancers that we examined, which included colon and other gastrointestinal tract cancers, lung cancer, and hematologic cancers, men lost more years of potential life than women. Among whites, lung cancer had the largest impact, accounting for 15% of the excess YPLL (from all causes) among men for whites, 17% for African Americans, but only 4% among Latinos. Colon cancer accounted for only 1% of the male-female difference in YPLL for all three racial/ethnic groups.
Table 4.
Years of potential life lost (per 1,000 persons) by specific types of cancer and race/ethnicity

NOTES: Columns may not sum to the totals shown because of rounding. Years of potential life lost before age 75 (per 1,000 persons) = Σ(75 − age at death among those dying before age 75) × 1,000/n, where n=total number of persons at risk of dying. Some columns may not adds up due to rounding error.
p<0.002
Impact of HIV
Adjusted for more recent estimates in HIV mortality, YPLL from HIV among white men was 45 years per 1,000 individuals and six years among white women. This difference of 39 years per 1,000 individuals (95% CI 26, 41) represents 2% of the sex difference in YPLL from all causes. In comparison, African American men lost 378 and African American women lost 168 years of potential life from HIV, (difference of 210 years; 95% CI 185, 218), and Latino men and women lost 190 and 71 years, respectively (difference of 119 years; 95% CI 102, 129). Thus, HIV accounted for 7% of the sex difference in YPLL among African Americans and 5% among Latinos. We estimate that prior to 1997 (i.e., no adjustment for recent AIDS mortality trends), HIV accounted for 9% of the sex difference in YPLL among whites, 21% among African Americans, and 19% among Latinos.
DISCUSSION
Numerous hypotheses have been suggested as to why women live longer than men.4,20 One explanation is that men have an innate and/or acquired greater physiologic susceptibility to disease. Men also have worse health behaviors. They are more likely to use tobacco, alcohol and illicit drugs, drive without a seatbelt or while intoxicated, use firearms, exhibit violent behavior, eat a poor diet, live a sedentary lifestyle, and have high-risk sexual behaviors.21,22 Men may also be more commonly exposed to environmental hazards and unsafe working conditions.23 Furthermore, numerous psychosocial factors could explain sex differences in health, including differences in coping strategies, response to stress, and willingness to seek medical care.23
Most, if not all, of these explanations seem plausible, given that men appear to have higher mortality rates from a broad range of causes.3 However, we found that a few specific causes of death account for most of the sex difference in YPLL. Traumatic deaths, which include homicide, suicide, and accidental deaths, account for one-third of the sex difference in YPLL among whites and African Americans and 50% of the difference among Latinos. These differences in traumatic deaths are primarily due to homicide among African Americans and Latinos, while suicide and motor vehicle accidents are important among whites.
These findings suggest that violence and use of firearms, as well as drug and alcohol use, are important contributors to the sex difference in mortality and that public health efforts targeting these factors could dramatically close the sex gap in life expectancy. Men are much more likely than women to use drugs and alcohol,21,24 and approximately 75% of homicides are related to the use of drugs or alcohol or the selling of illicit drugs.25 Furthermore, about half of all deaths from motor vehicle accidents among men are related to the use of alcohol, compared to 33% among women.26 Although women are almost twice as likely to be diagnosed with major depression and three times more likely to attempt suicide than men, men are more likely to successfully commit suicide.27 This difference occurs partly because men are more likely to use effective methods such as firearms or hanging, and tend to use drugs and alcohol, which increase impulsive behavior.28
Among deaths related to specific diseases, ischemic heart disease and lung cancer contribute most to the YPLL difference between men and women. This suggests that higher rates of smoking among men may explain why they have shorter life expectancy. Recent estimates indicate that 26% of men have used tobacco compared with 21% of women.29 Still, it is unlikely that tobacco use alone completely explains the sex differences for these two causes. One might expect that sex differences in YPLL from other smoking-related deaths, such as other atherosclerotic diseases and chronic lung disease, would be larger than they are. In addition, the contribution of lung cancer varies substantially across the three racial/ethnic groups and does not appear consistent with racial/ethnic variations in smoking. Among Latinos, lung cancer accounts for 4% of total sex difference in YPLL, compared to 15% among whites and 17% among African Americans. Rates of smoking, however, are similar among men of different racial/ethnic groups (26% among whites and African Americans and 24% among Latinos), while Latino women are much less likely to smoke (13%) compared with white women (22%) and African American women (21%).29 Thus, Latinos have the greatest sex difference in smoking rates, yet have the smallest sex difference in YPLL from lung cancer.
Our study also found that HIV contributed substantially to the mortality difference between men and women prior to 1997. Since then, HIV mortality has dropped dramatically as the result of more effective medications and the use of multiple drug regimens.16,30 We estimate that this change has led to a substantial drop in the contribution of HIV to sex difference in mortality for all three racial/ethnic groups.
The causes of death that contribute most to the sex difference in YPLL vary by race/ethnicity. Cancer accounted for only 2% of the sex difference in YPLL among Latinos, compared to 15% among whites and 20% among African Americans. These findings are not likely to be explained by differences in smoking rates, as mentioned previously, nor by differences in cancer screening. Latinos are less likely to receive mammography, Pap smears, and colon cancer screening.31,32 Whether Latinos have less exposure to carcinogens or have innate or acquired protection from cancer is unknown.
The sex difference in YPLL attributable to suicide is much larger among whites than African Americans. Some possible explanations for this are that whites, compared to African Americans, are more likely to become depressed, have a poor response to treatment, and/or receive worse treatment. Evidence does not support these explanations, however. Whites appear to have equal or lower rates of depression.33–35 Whites also tend to receive better mental health care35,36 partly because of better access to care35 and greater acceptance of medications and counseling.37 Whites do not appear less responsive to treatment,31 but evidence is limited. Whites may also have higher suicide rates because they tend to have more severe depression,38 though it is not clear whether this finding is related to detection bias. Other factors, such as religion and cultural norms (e.g., the acceptability of suicide), may also protect African Americans from committing suicide.
The results of this study should be interpreted in light of some limitations. First, we examined the underlying cause of death, determined as the single cause from among all of the causes listed on the death certificate that ultimately caused the person's death. Though the underlying cause was determined using a commonly used algorithm,11 death is often attributable to more than one cause. In addition, the death certificate data on cause of death may be inaccurate.
Other limitations include the fact that we examined Latinos as a single ethnic group. Results may differ according to immigration status and country of origin.39–42 We were also unable to examine the impact of tobacco, drug and alcohol use, or other potential mediating factors. The survival and simulation models used in this study assume that the risks of dying from various causes are independent from each other. Of course, many causes of death that we examined share similar risk factors and violate this assumption. Thus, caution is warranted in comparing the relative contribution of causes that share similar risk factors, e.g., ischemic heart disease and stroke. While we adjusted for recent trends in HIV mortality, we did not adjust for potential temporal changes that might have occurred since 1997 for other causes of death. Finally, we estimated YPLL without taking into account disability or quality of life. Although there is some debate on this topic,18 some evidence suggests that in spite of having longer life expectancy, women also have more comorbid conditions, greater disability, and worse quality of life.4 Thus, adjusting YPLL for quality of life may decrease the apparent difference in YPLL between men and women and may also alter the study results regarding the relative importance of various diseases to the sex difference in YPLL.
Achieving equal life expectancy between men and women has seemed rather daunting, given that numerous and diverse factors were thought to be involved.16 In the present study, we have found that just a few causes—homicide, suicide, motor vehicle accidents, ischemic heart disease, and lung cancer—account for 71% of the total YPLL gap between men and women among whites, 63% among African Americans, and 73% among Latinos. Thus, a few factors such as violence and the use of tobacco, alcohol, and drugs may be responsible for much of the sex difference in life expectancy. Public health researchers should focus future studies on understanding how much these behaviors, as well as cardiovascular risk factors, contribute to life expectancy differences between men and women. Future studies will also need to take race/ethnicity into account, which is clearly associated with differences in causes of death between men and women. Our results also suggest that the sex gap in life expectancy might be substantially reduced if clinicians and public health officials targeted risky health behaviors among adolescents and young adults, who have the highest rates of traumatic deaths.
REFERENCES
- 1.Arias E. United States life tables, 2000. Natl Vital Stat Rep. 2002;51(3):1–38. [PubMed] [Google Scholar]
- 2.Kochanek KD, Smith BL. Deaths: preliminary data for 2002. Natl Vital Stat Rep. 2004;52(13):1–47. [PubMed] [Google Scholar]
- 3.Arias E, Anderson RN, Kung HC, Murphy SL, Kochanek KD. Deaths: final data for 2001. Natl Vital Stat Rep. 2003;52(3):1–115. [PubMed] [Google Scholar]
- 4.Verbrugge LM, Wingard DL. Sex differentials in health and mortality. Health Matrix. 1987;5(2):3–19. [PubMed] [Google Scholar]
- 5.Gardner JW, Sanborn JS. Years of potential life lost (YPLL)--what does it measure? Epidemiology. 1990;1:322–9. doi: 10.1097/00001648-199007000-00012. [DOI] [PubMed] [Google Scholar]
- 6.Freid VM, Prager K, MacKay AP, Xia H. Health United States. Hyattsville (MD): National Center for Health Statistics; 2003. Chartbook on trends in the health of Americans. [Google Scholar]
- 7.Lee WC. Quantifying the future impact of disease on society: life table-based measures of potential life lost. Am J Public Health. 1997;87:1456–60. doi: 10.2105/ajph.87.9.1456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wong MD, Shapiro MF, Boscardin WJ, Ettner SL. Contribution of major diseases to disparities in mortality. N Engl J Med. 2002;347:1585–92. doi: 10.1056/NEJMsa012979. [DOI] [PubMed] [Google Scholar]
- 9.National Center for Health Statistics. Springfield (VA): National Technical Information Service; 1994. National Health Interview Survey, Part 1 (database) [Google Scholar]
- 10.National Center for Health Statistics. Hyattsville (MD): U.S. Department of Health and Human Services; 2000. National Health Interview Surveys, 1986-1997: multiple cause of death, 1986-1997. [Google Scholar]
- 11.Minino AM, Smith BL. Deaths: preliminary data for 2000. Natl Vital Stat Rep. 2001;49(12):1–40. [PubMed] [Google Scholar]
- 12.5th ed. Los Angeles: Practive Management Information Corporation; 1999. International Classification of Diseases, 9th Revision. [Google Scholar]
- 13.Klein J, Moeschberger M. New York: Springer-Verlag; 1997. Survival analysis: techniques for censored and truncated data. [Google Scholar]
- 14.Friedman M. Piecewise exponential models for survival data with covariates. Ann Statistics. 1982;10:101–13. [Google Scholar]
- 15.Centers for Disease Control and Prevention (US) HIV/AIDS surveillance report. 2002;14:1–48. [Google Scholar]
- 16.Lai D, Hardy RJ. Potential gains in life expectancy or years of potential life lost: impact of competing risks of death. Int J Epidemiol. 1999;28:894–8. doi: 10.1093/ije/28.5.894. [DOI] [PubMed] [Google Scholar]
- 17.King G, Tomz M, Wittenberg J. Making the most of statistical analyses: improving interpretation and presentation. Am J Political Science. 2000;44:341–55. [Google Scholar]
- 18.Stata Corp LP. College Station (TX): Stata Corp; 2003. STATA: version 8.0. [Google Scholar]
- 19.SAS Institute, Inc. Cary (NC): SAS Institute, Inc.; 2003. SAS: version 8.02. [Google Scholar]
- 20.Williams DR. The health of men: structured inequalities and opportunities. Am J Public Health. 2003;93:724–31. doi: 10.2105/ajph.93.5.724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Holtzman D, Powell-Griner E, Bolen JC, Rhodes L. State- and sex-specific prevalence of selected characteristics--Behavioral Risk Factor Surveillance System, 1996 and 1997. MMWR Surveill Summ. 2000;49(SS-06):1–39. [PubMed] [Google Scholar]
- 22.Grunbaum JA, Kann L, Kinchen S, Ross J, Hawkins J, Lowry R, et al. Youth risk behavior surveillance--United States, 2003. MMWR Surveill Summ. 2004;53(SS-02):1–96. [PubMed] [Google Scholar]
- 23.Verbrugge LM. The twain meet: empirical explanations of sex differences in health and mortality. J Health Soc Behav. 1989;30:282–304. [PubMed] [Google Scholar]
- 24.Warner LA, Kessler RC, Hughes M, Anthony JC, Nelson CB. Prevalence and correlates of drug use and dependence in the United States. Results from the National Comorbidity Survey. Arch Gen Psychiatry. 1995;52:219–29. doi: 10.1001/archpsyc.1995.03950150051010. [DOI] [PubMed] [Google Scholar]
- 25.McLaughlin CR, Daniel J, Joost TF. The relationship between substance use, drug selling, and lethal violence in 25 juvenile murderers. J Forensic Sci. 2000;45:349–53. [PubMed] [Google Scholar]
- 26.Voas RB, Tippetts S, Fisher DA. Landover, MD:: Pacific Institute for Research and Evaluation; [cited 2006 Jul 13]. Ethnicity and alcohol-related fatalities: 1990 to 1994. Available from: URL: http://www.nhtsa.dot.gov/people/injury/alcohol/ethnicity/ethnicity.html#T5. [Google Scholar]
- 27.Sloan DM, Kornstein SG. Gender differences in depression and response to antidepressant treatment. Psychiatr Clin North Am. 2003;26:581–94. doi: 10.1016/s0193-953x(03)00044-3. [DOI] [PubMed] [Google Scholar]
- 28.Moller-Leimkuhler AM. The gender gap in suicide and premature death or: why are men so vulnerable? Eur Arch Psychiatry Clin Neurosci. 2003;253:1–8. doi: 10.1007/s00406-003-0397-6. [DOI] [PubMed] [Google Scholar]
- 29.Cigarette smoking among adults–United States, 2000. MMWR Morb Mortal Wkly Rep. 2002;51(29):642–5. [PubMed] [Google Scholar]
- 30.Palella FJ, Jr, Delaney KM, Moorman AC, Loveless MO, Fuhrer J, Satten GA, et al. Declining morbidity and mortality among patients with advanced human immunodeficiency virus infection. HIV Outpatient Study Investigators. N Engl J Med. 1998;338:853–60. doi: 10.1056/NEJM199803263381301. [DOI] [PubMed] [Google Scholar]
- 31.Coughlin SS, Uhler RJ, Richards T, Wilson KM. Breast and cervical cancer screening practices among Hispanic and non-Hispanic women residing near the United States-Mexico border, 1999-2000. Fam Community Health. 2003;26:130–9. doi: 10.1097/00003727-200304000-00006. [DOI] [PubMed] [Google Scholar]
- 32.Bolen JC, Rhodes L, Powell-Griner EE, Bland SD, Holtzman D. State-specific prevalence of selected health behaviors, by race and ethnicity--Behavioral Risk Factor Surveillance System, 1997. MMWR Surveill Summ. 2000;49(SS-02):1–60. [PubMed] [Google Scholar]
- 33.Oquendo MA, Ellis SP, Greenwald S, Malone KM, Weissman MM, Mann JJ. Ethnic and sex differences in suicide rates relative to major depression in the United States. Am J Psychiatry. 2001;158:1652–8. doi: 10.1176/appi.ajp.158.10.1652. [DOI] [PubMed] [Google Scholar]
- 34.Dunlop DD, Song J, Lyons JS, Manheim LM, Chang RW. Racial/ethnic differences in rates of depression among preretirement adults. Am J Public Health. 2003;93:1945–52. doi: 10.2105/ajph.93.11.1945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rollman BL, Hanusa BH, Belnap BH, Gardner W, Cooper LA, Schulberg HC. Race, quality of depression care, and recovery from major depression in a primary care setting. Gen Hosp Psychiatry. 2002;24:381–90. doi: 10.1016/s0163-8343(02)00219-0. [DOI] [PubMed] [Google Scholar]
- 36.Miranda J, Duan N, Sherbourne C, Schoenbaum M, Lagomasino I, Jackson-Triche M, Wells KE. Improving care for minorities: can quality improvement interventions improve care and outcomes for depressed minorities? Results of a randomized, controlled trial. Health Serv Res. 2003;38:613–30. doi: 10.1111/1475-6773.00136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Cooper LA, Gonzales JJ, Gallo JJ, Rost KM, Meredith LS, Rubenstein LV, et al. The acceptability of treatment for depression among African-American, Hispanic, and white primary care patients. Med Care. 2003;41:479–89. doi: 10.1097/01.MLR.0000053228.58042.E4. [DOI] [PubMed] [Google Scholar]
- 38.Zhang AY, Snowden LR. Ethnic characteristics of mental disorders in five U.S. communities. Cultur Divers Ethnic Minor Psychol. 1999;5:134–46. doi: 10.1037/1099-9809.5.2.134. [DOI] [PubMed] [Google Scholar]
- 39.Shai D, Rosenwaike I. Violent deaths among Mexican-, Puerto Rican- and Cuban-born migrants in the United States. Soc Sci Med. 1988;26:269–76. doi: 10.1016/0277-9536(88)90249-3. [DOI] [PubMed] [Google Scholar]
- 40.Serow WJ, Cowart ME, Camezon J. Epidemiologic transition theory and aging: Hispanic populations of North America and the Caribbean. J Health Hum Serv Adm. 1998;20:333–47. [PubMed] [Google Scholar]
- 41.Shai D. Cancer mortality in Cuba and among the Cuban-born in the United States: 1979-81. Public Health Rep. 1991;106:68–73. [PMC free article] [PubMed] [Google Scholar]
- 42.Singh GK, Siahpush M. All-cause and cause-specific mortality of immigrants and native born in the United States. Am J Public Health. 2001;91:392–99. doi: 10.2105/ajph.91.3.392. [DOI] [PMC free article] [PubMed] [Google Scholar]