Abstract
The placenta constitutes a physical and immunological barrier against invading infectious agents and has been suggested to be a pregnancy-specific component of the innate immune system. The aim of this study was to investigate the presence and regulation of Toll-like receptors-2 and -4 (TLR2 and TLR4) in the human placenta, because these receptors are believed to be important for immune responses against pathogens. Twenty-eight placentas from normal term pregnancies were analysed with immunohistochemistry, which showed a strong immunoreactivity for TLR2 and TLR4 in the villous and the intermediate trophoblasts. The regulation of TLR2 and TLR4 by microbial stimulus was assessed by incubating explants of term chorionic villi with zymosan or lipopolysaccharide (LPS) and analysed with real-time reverse transcriptase–polymerase chain reaction. Stimulation with zymosan and LPS readily induced interleukin (IL)-6 and IL-8 cytokine production in the placenta cultures, whereas TLR2 and TLR4 mRNA and protein expression remained at the same high level as in unstimulated explants. These data suggests a novel mechanism for the fetoplacental unit to interact with micro-organisms.
Introduction
Although the placenta constitutes a physical barrier for invading infectious agents, infections still occur by this route and therefore additional mechanisms to protect the fetus are necessary. Recent findings suggest that the placenta, and in particular the trophoblast, plays an important role in the innate immune system. Defensin transcripts have been detected in placenta,1 which indicate that it has the capacity to battle an infection. Further, it was recently demonstrated that the trophoblasts are involved in the immune response against placental infection with Listeria monocytogenes.2
Toll-like receptors (TLR) have been shown to be important to the innate immune response.3 Toll was first discovered in Drosophila (dToll) where it has a function in development of dorsoventral polarity. Drosophila lacks an adaptive immune response but is resistant to fungal infection through the action of Toll. Upon infection, the Toll ligand Spätzle is processed into a biologically active form, and by binding to Toll induces a signalling cascade, resulting in the production of the antimicrobial peptide Drosomycin.4,5 All TLRs belong to the interleukin-1 receptor (IL-1R)/TLR super family, which has a conserved region in the cytosolic domain called a Toll/IL-1R (TIR) domain.6 Today more than 10 members of the TLR family can be found in the human and mouse databases3 and these (TLR 1–10) have been characterized.6–10 Janeway and colleagues discovered the first human homologue to dToll, which they termed TLR4.11 Through the study of mutant mice that are hyporesponsive or non-responsive to lipopolysaccharide (LPS), TLR4 has been shown to function primarily in the recognition of Gram-negative bacteria.12,13 TLR4 mutations have also been associated to endotoxin hyporesponsiveness in humans.14 TLR2, on the other hand, is believed to be involved in the recognition of a wide variety of infectious pathogens and their products. These include yeast cell walls,15,16 mycobacteria,17 whole Gram-positive bacteria,18 lipoproteins,19,20 glycolipids21 and peptidoglycan.22
So far, studies of TLRs have mainly been performed in animal models and cell cultures. The distribution and function of TLR2 and TLR4 in human placenta during pregnancy and their importance for the fetus is unknown. In this study in human placenta we investigate the cellular distribution and expression of TLR2 and TLR4, two receptors that might have crucial functions in the battle against fetoplacental infection.
Materials and methods
Placentas
Placentas from 28 normal term pregnancies (13 vaginal deliveries and 15 elective caesarean sections) were collected from the delivery unit of Karolinska Hospital and Söder's Hospital in Stockholm, Sweden. The mothers were 24–44 years old (median 31·5 years) and with one to four children. The weight of the placentas ranged from 468 g to 1030 g (median of 669·5 g), and the babies (19 girls, 9 boys) weighed 2750–4920 g (median 3650 g). The placentas were collected immediately after delivery and kept refrigerated at 4° until studied. For tissue culture experiments, fresh placental tissue explants were cut from five of the placentas from elective caesarean sections. The study was approved by the Ethics Committees at Karolinska Hospital and Söder's Hospital in Stockholm, Sweden. The mothers gave their informed consent to the study.
Processing and immunohistochemical staining of placental slices
Three placental slices were taken in a circular fashion around the umbilical cord insertion. The slices spanned from the fetal membranes to the decidua. The slices were washed thoroughly in sterile NaCl. From all placentas two slices were frozen on dry ice and stored at −70°, and from 15 of the placentas one slice was additionally formalin fixed and paraffin embedded. After having screened available antibodies against TLR2 and TLR4 it was found that the rabbit anti-human TLR2 (anti-hTLR2) antisera raised against a 16-amino acid residue (YDLSTLYSLTERVKRC) in the variable extra cellular domain of the mature hTLR2, custom made at Innovagen (dilution 1/300, Lund, Sweden) worked on cryosections, while goat anti-human TLR4 (anti-hTLR4) (dilution 1/20, concentration 10 µg/ml, Santa Cruz Biotechnology Inc., Santa Cruz, CA) did so on the formalin fixed, paraffin embedded sections. The cryosections (7 µm thick) were put on glass slides and fixed in acetone. From the formalin fixed, paraffin embedded placentas 4 µm sections were deparaffinized in xylol for 30 min, followed by rehydration in ethanol. Antigen retrieval on these sections was performed by microwave irradiation for 3×4 min in sodium citrate (10 mm, pH 6·0) and the sections were allowed to cool for 20 min. The ABC-ELITE (Vector Laboratories Inc., Burlingame, CA) immunohistochemical staining was then performed as previously described.23 The primary antibodies were absorbed against 25% human serum for 2 hr before being added to the specimen. As secondary antibodies, biotin-labelled goat anti-rabbit (dilution 1/200, Vector Laboratories Inc.) and horse anti-goat (dilution 1/400, Vector Laboratories Inc.) were used for anti-TLR2 and anti-TLR4 staining, respectively. The stainings were developed by incubating the sections with three-amino-9-ethylcarbazole (AEC) as substrate. Staining without the primary antibody and staining with the primary antibodies preincubated with blocking peptides specific for the anti-TLR2 (Innovagen) and anti-TLR4 (Santa Cruz Biotechnology Inc.) antibodies were used as negative controls. No homology to other TLRs or other proteins could be seen when performing a National Center for Biotechnology Information (NCBI) non-redundant GenBank search [Chemical Database Service (CDS) translations, the Protein Data Bank (PDB), SwissProt, Protein Information Resource (PIR), Protein Research Foundation (PRF)] with the epitope specific for the anti-TLR2 antibody.
Evaluation of the immunohistochemical stainings
The sections were 2–3 cm long and represented the whole placental thickness from amnion to the decidua. Haematoxylin and eosin stainings of the cryosections revealed good morphology. A minimum of one section per placenta slice was stained for each antibody.
Explant processing and stimulation
Pieces of chorionic villi were taken from fresh placental tissue from five of the elective caesarean sections. The tissue was thoroughly washed in sterile phosphate-buffered saline (PBS) to remove blood and cut into centimetre-sized pieces with scissors. Slices spanning through the whole placental tissue were also snap frozen as described above for immunohistochemical stainings. The explants were incubated two by two in a 12-well plate (Costar, Cambridge, UK) with 2 ml RPMI-1640 (Life Technologies, Täby, Sweden) supplemented with gentamicine (25 µg/ml), penicillin (100 IU/ml), streptomycin (100 µg/ml), l-glutamine (2 mm) (Merck, Darmstadt, Germany), and 10% heat inactivated fetal calf serum (FCS; Hyclone Laboratories Inc., Logan, UT). Different concentrations of LPS (Escherichia coli K-12, W3110),24 or zymosan (Sigma-Aldrich, Stockholm, Sweden) were added to the medium. The stimulation was carried out for 2, 4 and 6 hr at 37° in 6% CO2 atmosphere. As controls, explants were incubated as described above in medium without any stimulation. After culture the explants were washed in sterile PBS, frozen on dry ice and stored at −70° until used for immunohistochemistry and RNA preparation.
IL-6 and IL-8 determination
IL-6 and IL-8 levels were determined in explant culture supernatants using the sandwich enzyme-linked immunoassay technique (ELISA) according to the manufacturer's instructions (Quantikine® human IL-6/IL-8; R & D Systems, Abingdon, Oxon, UK). All supernatants were centrifuged to remove particulate matter and kept frozen at −20° until analysis. All samples were added in duplicate. The optical density was determined using a microplate reader (SLT-Labinstruments, Salzburg, Austria) set at 450 nm. The detection limit was 3 pg/ml and 31 pg/ml for IL-6 and IL-8, respectively.
RNA preparation and real time reverse transcriptase–polymerase chain reaction (RT–PCR)
Total RNA was isolated with RNeasy Protect Mini Kit (Qiagen, Hilden, Germany) according to the manufacturer's instructions, or with an Ultraspec II RNA isolation system (Biotecx, Houston TX), as previously described.25 cDNA was prepared and mRNA levels was quantified using the Roche LightCycler Instrument (Roche, Diagnostics Scand AB, Bromma, Sweden). For each PCR reaction 2 µl each of the standard cDNA (1 : 1, 1 : 10, 1 : 100, 1 : 1000) and the unknown samples diluted to 1 : 10 (to put the cross-point value approximately in the middle of the corresponding standard curve), were mixed with 2 µl of nucleotide triphosphates (NTPs), Hot-start Taq polymerase, reaction buffer and SYBR Green I dye (LightCycler DNA Master SYBR Green I kit) and placed into individual LightCycler glass capillaries. The reactions were supplemented with 3 mm Mg2+ and 0·5 µl of each gene specific primer (sense and antisense). Primer-pairs for the human β-actin house-keeping gene were purchased from Clontech (Palo Alto, CA) and gene-specific primers for TLR2 (GenBank no: XM_003304) and TLR4 (Gene bank no: NM_003266) (sequences: (TLR2) 5′-GCC TCT CCA AGG AAG AAT CC-3′, 5′-TCC TGT TGT TGG ACA GGT CA-3′ (TLR4) 5′-AAG CCG AAA GGT GAT TGT TG-3′, 5′-CTG AGC AGG GTC TTC TCC AC-3′) were custom made at DNA Technology AIS (Aarhus, Denmark). Forty-five cycles of PCR reaction (5-s denaturation at 95°; 10-s annealing at 60°; and extension at 72°) was run until the cross-point was attained for the highest cDNA dilution sample. Melting curve analyses were performed after the completion of cycling to control for the specificity of the PCR products obtained. To ensure that equal amounts of total RNA were loaded in each cDNA synthesis reaction, the TLR-primer-generated fluorescence data were normalized to the fluorescence values generated by human β-actin primers from the same total RNA sample. Aliquots of the reaction product were run on 2% agarose gel containing ethidium bromide to establish the PCR product size and thereby control for the specificity of amplification.
Results
Expression of TLR2 and TLR4 in the human placenta
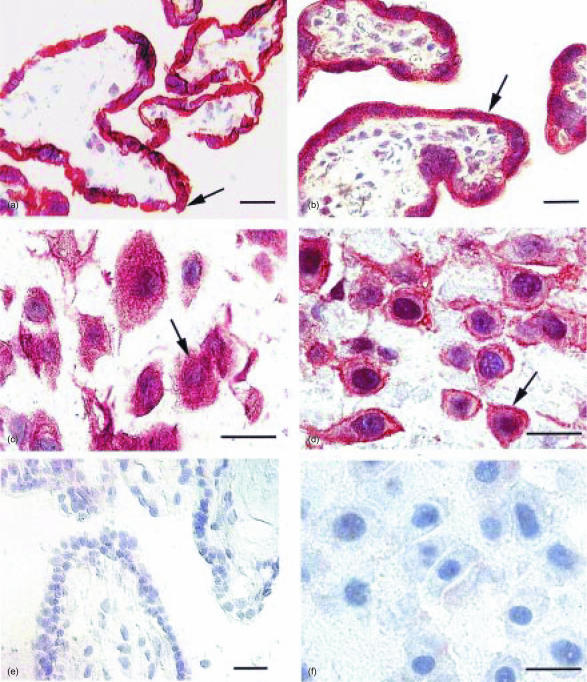
TLR2 (Fig. 1a) and TLR4 (Fig. 1b) were found to be highly and continuously expressed on trophoblasts covering the peripheral chorionic villi, while no immunoreactive cells were present in the villous core. Intermediate trophoblasts in free cell islands, the cell columns and the decidua were found to express both TLR2 (Fig. 1c) and TLR4 (Fig. 1d). The intermediate trophoblast was identified by their characteristic morphology, staining pattern with haematoxylin and eosin and immunoreactivity for human placental lactogen26 (data not shown). The trophoblasts covering the peripheral chorionic villi had a characteristic homogenous cytoplasmic staining and there was no striking difference in the amount and distribution of TLR2+ and TLR4+ cells between the placentas investigated. The staining was extinct when the anti-TLR2 and anti-TLR4 antibodies had been preincubated with blocking peptides specific for anti-TLR2 (Fig. 1e) and anti-TLR4 (Fig. 1f), respectively.
Figure 1.
TLR2 and TLR4 are highly expressed in the human placenta. Acetone-fixed cryostat sections (7 µm thick) of frozen slices (a,c,e) and formalin fixed, paraffin imbedded sections (4 µm) (b,d,f) were stained with the ABC Elite method, with a rabbit anti-hTLR2 (a,c) or a goat anti-hTLR4 antibody (b,d), respectively. A counterstaining was made with Mayer's haematoxylin. Positive cells are seen as brown/red (examples are indicated by arrows). (a) TLR2+ and (b) TLR4+ trophoblasts covering the peripheral chorionic villi. (c) TLR2+ intermediate trophoblasts and (d) TLR4+ intermediate trophoblasts. Staining was extinct when the anti-TLR2 and anti-TLR4 antibodies had been preincubated with blocking peptides specific for anti-TLR2 (e) and anti-TLR4 (f), respectively, neither in chorionic villi (e), nor in cells in the decidua (f). Scale bar 25 µm.
TLR2 and TLR4 expression after zymosan and LPS stimulation
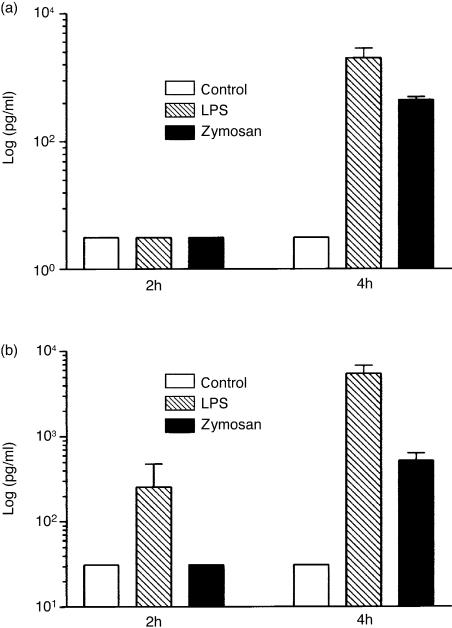
TLR2 and TLR4 expression have been shown to be regulated by stimuli of microbial origin.12,13,15–22 Therefore we stimulated fresh placental explants with different concentrations of zymosan and LPS or incubated them with medium alone for 2, 4 and 6 hr. This treatment induced release of IL-6 after 4 hr of stimulation with LPS and zymosan (Fig. 2a) and of IL-8 already after 2 hr of stimulation with LPS and after 4 hr with zymosan (Fig. 2b) as measured in culture supernatants with ELISA.
Figure 2.
IL-6 and IL-8 are secreted by placental explants after stimulation with LPS and zymosan. The presence of IL-6 (a) and IL-8 (b) were measured in culture supernatants, with ELISA, after incubation with 10 ng/ml of LPS and 3 µg/ml of zymosan. Data are given as means±SEM of experiments from four different placentas. Where SE bars are missing the errors are too small to show.
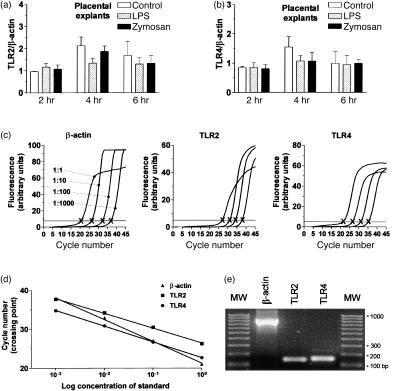
Neither stimulation with zymosan, nor with LPS, however, led to a difference in TLR2 or TLR4 mRNA expression compared to unstimulated controls. In Fig. 3(a,b) the mRNA expression of TLR2 and TLR4 after stimulation with 10 ng of LPS and 3 µg of zymosan is shown. Higher concentrations of LPS (100 ng/ml, 1 µg/ml) and zymosan (30 µg/ml, and 300 µg/ml) did not alter the levels of TLR2 and TLR4 mRNA expression (data not shown). Real time RT–PCR reaction products were run on 2% agarose gel to establish the PCR product size, and thereby control for specificity of amplification. All amplicons had the expected size (Fig. 3e). The immunohistochemical staining on cryostat sections of parallel placental explants from five placentas incubated with LPS, zymosan or medium alone showed no difference on the protein level for TLR2 between stimulated and unstimulated placental explants compared to uncultured placental slices from the same placenta (data not shown).
Figure 3.
mRNA levels for TLR2 and TLR4 in human term placenta remain high after stimulation with zymosan or LPS. Real-time RT–PCR was performed on mRNA isolated from placental explants (a, b). Relative abundance is shown of mRNA for TLR2 (a) and TLR4 (b) from cultured explants stimulated with LPS (10 ng/ml) and zymosan (3 µg/ml) in comparison with unstimulated controls. The levels of TLR2 and TLR4 were normalized to β-actin expression in the same total RNA preparation to even out the slight variation introduced by the cDNA synthesis step. The results are shown as means±SEM of (a) TLR2 mRNA expression and (b) TLR4 mRNA expression from experiments with five different placentas. (c) Representative fluorescence profiles showing the increase in SYBR Green I fluorescence as a function of PCR cycle number and cDNA dilution factor with β-actin, TLR2 and TLR4 primers. Horizontal line drawn through the log-linear part of the fluorescence curves defines the crossing point (X), the number of PCR cycles necessary to produce an apparent increase in fluorescence. (d) Crossing points for the indicated gene-specific PCR reactions with control cDNA are plotted against the dilution factor of the cDNA to produce a standard linear regression. (e) Estimation of PCR product size by 2% agarose gel electrophoresis. Molecular weight standards (MW) contain 10 bands ranging from 100 to 1000 bp with 100 bp increments. All amplicons had the expected size.
Discussion
TLR protein expression has been described in tissues like intestinal epithelium27 and in human bladder urothelium24 indicating that mucosal linings have the capacity to respond to micro-organisms. mRNA for TLR1–TLR10 has been reported to be present in the human placenta6,7,28 and the ontogeny of TLR2 and TLR4 mRNA in murine placenta as well as in fetal murine lung and liver has been studied.29 At present there are no reports on the cellular distribution of TLR2 and TLR4 in the human placenta. Our study showed a strong immunoreactivity for TLR2 and TLR4 in term placentas in the trophoblast covering the peripheral chorionic villi, which constitutes the frontal barrier between the mother and the fetus. Notably, in a study in mice infected with the Gram-positive bacterium L. monocytogenes, the observation that the trophoblasts produce factors that primarily recruit maternal neutrophils to the site of infection is indicative of the trophoblast as a pregnancy-specific component of the innate immune system.2 Thus, the trophoblasts seem to have an important function in organizing the maternal immune response against bacterial infections. Our observations on TLR2 and TLR4 expression by the trophoblasts support these assumptions.
A co-operation between TLR2 and TLR4 has been proposed, where costimulation of the receptors is needed for a reliable signalling. A synergy has also been observed between LPS, the ligand for TLR4 and a ligand for TLR2, muramyl dipeptide, in the release of tumour necrosis factor from murine RAW 264·7 macrophage cells (American Type Culture Collection) cells.30 Several earlier studies have also shown that injection of muramyl peptides into mice modified their responses to endotoxin.31–35 The coexpression of TLR2 and TLR4 on both villous trophoblasts and intermediate trophoblasts could therefore be a safety mechanism, preventing inappropriate, potentially fatal reactions from these cells.
Using real-time RT–PCR we found that mRNA levels for TLR2 and TLR4 in chorionic villi explants were high but unchanged after exposure in vitro to either LPS or zymosan. To ensure that placental explants had retained their capacity to react to stimulation with zymosan and LPS we measured the release of the pro-inflammatory cytokine IL-6 and the chemokine IL-8 in the culture supernatants. Both IL-6 and IL-8 can be produced by human trophoblasts after stimulation with LPS.36 LPS and zymosan did induce IL-6 and IL-8 release by placental explants (Fig. 2), indicating that they responded to the stimuli. It is possible that the expression of TLR2 and TLR4 are normally up-regulated in utero as a defence mechanism that can readily be mobilized to protect the fetus from infections during the pregnancy and labour period. This could also explain why we do not see further up-regulation of these receptors following stimulation with LPS or zymosan.
In the Drosophila embryo a small number of genes are involved in the dorsoventral polarization37 and the signalling pathway centres on Toll.38,39 The high homology between Drosophila Toll and human TLRs might indicate a possible developmental role for these receptors in the mammalian placenta as in the Drosophila embryo. To further elucidate the importance of TLR2 and TLR4 in placenta, in vivo experiments using animal models are required.
In summary, a strong immunoreactivity for TLR2 and TLR4 was found in the villous and the intermediate trophoblasts in human term placenta. The protein and mRNA expression for TLR2 and TLR4 were unaltered after zymosan and LPS stimulation of placental explants, although these stimuli readily induced IL-6 and IL-8 production. We believe that the TLRs could be important immune regulators of placental infection, but they may also be needed for maturation of fetal and neonatal immune responses. Recently, the importance of TLRs in the activation of antigen-specific T helper type 1 (Th1) but not Th2 immune responses, as well as that a lack of TLR signalling leads to increased immunoglobulin E responses, have been reported.40 This makes further studies on expression and regulation of TLRs in atopic and nonatopic individuals particularly interesting.
Acknowledgments
We thank Hojjatollah Eshagi for excellent technical assistance with immunohistochemistry, Jia-Xin Wang for preparing slides and Malin Emilson for assistance with mRNA isolation and real-time RT–PCR. We would also like to thank Berit Legestam, Lotta Blomberg and the staff at the Delivery Unit of Karolinska Hospital and Söder's Hospital for help to collect the placenta material.
This work was supported by grants from Karolinska Institutet, the Swedish Asthma and Allergy Association's Research Foundation, the Swedish Medical Research Council (grant no. 16X-7924), the Swedish Council for Work Life Research, the Swedish Foundation for Health Care Sciences and Allergy Research, the Hesselman's Foundation, Magnus Bergwall's Foundation, Konsul Th C Bergh's Foundation, Tore Nilson's Foundation and Apotekare Hedberg's Foundation.
References
- 1.Svinarich DM, Gomez R, Romero R. Detection of human defensins in the placenta. Am J Reprod Immunol. 1997;38:252–5. doi: 10.1111/j.1600-0897.1997.tb00511.x. [DOI] [PubMed] [Google Scholar]
- 2.Guleria I, Pollard JW. The trophoblast is a component of the innate immune system during pregnancy. Nat Med. 2000;6:589–93. doi: 10.1038/75074. [DOI] [PubMed] [Google Scholar]
- 3.Akira S, Takeda K, Kaisho T. Toll-like receptors: critical proteins linking innate and acquired immunity. Nat Immunol. 2001;2:675–80. doi: 10.1038/90609. [DOI] [PubMed] [Google Scholar]
- 4.Lemaitre B, Nicolas E, Michaut L, Reichhart JM, Hoffmann JA. The dorsoventral regulatory gene cassette spatzle/Toll/cactus controls the potent antifungal response in Drosophila adults. Cell. 1996;86:973–83. doi: 10.1016/s0092-8674(00)80172-5. [DOI] [PubMed] [Google Scholar]
- 5.Levashina EA, Langley E, Green C, Gubb D, Ashburner M, Hoffmann JA, Reichhart JM. Constitutive activation of toll-mediated antifungal defense in serpin-deficient Drosophila. Science. 1999;285:1917–9. doi: 10.1126/science.285.5435.1917. [DOI] [PubMed] [Google Scholar]
- 6.Rock FL, Hardiman G, Timans JC, Kastelein RA, Bazan JF. A family of human receptors structurally related to Drosophila Toll. Proc Natl Acad Sci USA. 1998;95:588–93. doi: 10.1073/pnas.95.2.588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chuang TH, Ulevitch RJ. Cloning and characterization of a sub-family of human toll-like receptors: hTLR7, hTLR8 and hTLR9. Eur Cytokine Netw. 2000;11:372–8. [PubMed] [Google Scholar]
- 8.Du X, Poltorak A, Wei Y, Beutler B. Three novel mammalian toll-like receptors. Gene structure, expression, and evolution. Eur Cytokine Netw. 2000;11:362–71. [PubMed] [Google Scholar]
- 9.Hemmi H, Takeuchi O, Kawai T, et al. A Toll-like receptor recognizes bacterial DNA. Nature. 2000;408:740–5. doi: 10.1038/35047123. [DOI] [PubMed] [Google Scholar]
- 10.Takeuchi O, Hoshino K, Kawai T, Sanjo H, Takada H, Ogawa T, Takeda K, Akira S. Differential roles of TLR2 and TLR4 in recognition of gram-negative and gram-positive bacterial cell wall components. Immunity. 1999;11:443–51. doi: 10.1016/s1074-7613(00)80119-3. [DOI] [PubMed] [Google Scholar]
- 11.Medzhitov R, Preston-Hurlburt P, Janeway CA., Jr A human homologue of the Drosophila Toll protein signals activation of adaptive immunity. Nature. 1997;388:394–7. doi: 10.1038/41131. [DOI] [PubMed] [Google Scholar]
- 12.Hoshino K, Takeuchi O, Kawai T, Sanjo H, Ogawa T, Takeda Y, Takeda K, Akira S. Cutting edge: Toll-like receptor 4 (TLR4)-deficient mice are hyporesponsive to lipopolysaccharide: evidence for TLR4 as the Lps gene product. J Immunol. 1999;162:3749–52. [PubMed] [Google Scholar]
- 13.Poltorak A, He X, Smirnova I, et al. Defective LPS signaling in C3H/HeJ and C57BL/10ScCr mice: mutations in Tlr4 gene. Science. 1998;282:2085–8. doi: 10.1126/science.282.5396.2085. [DOI] [PubMed] [Google Scholar]
- 14.Arbour NC, Lorenz E, Schutte BC, et al. TLR4 mutations are associated with endotoxin hyporesponsiveness in humans. Nat Genet. 2000;25:187–91. doi: 10.1038/76048. [DOI] [PubMed] [Google Scholar]
- 15.Lin Y, Lee H, Berg AH, Lisanti MP, Shapiro L, Scherer PE. The lipopolysaccharide-activated toll-like receptor (TLR)-4 induces synthesis of the closely related receptor TLR-2 in adipocytes. J Biol Chem. 2000;275:24255–63. doi: 10.1074/jbc.M002137200. [DOI] [PubMed] [Google Scholar]
- 16.Underhill DM, Ozinsky A, Hajjar AM, Stevens A, Wilson CB, Bassetti M, Aderem A. The Toll-like receptor 2 is recruited to macrophage phagosomes and discriminates between pathogens. Nature. 1999;401:811–5. doi: 10.1038/44605. [DOI] [PubMed] [Google Scholar]
- 17.Means TK, Wang S, Lien E, Yoshimura A, Golenbock DT, Fenton MJ. Human toll-like receptors mediate cellular activation by Mycobacterium tuberculosis. J Immunol. 1999;163:3920–7. [PubMed] [Google Scholar]
- 18.Flo TH, Halaas O, Lien E, Ryan L, Teti G, Golenbock DT, Sundan A, Espevik T. Human toll-like receptor 2 mediates monocyte activation by Listeria monocytogenes, but not by group B streptococci or lipopolysaccharide. J Immunol. 2000;164:2064–9. doi: 10.4049/jimmunol.164.4.2064. [DOI] [PubMed] [Google Scholar]
- 19.Aliprantis AO, Yang RB, Mark MR, et al. Cell activation and apoptosis by bacterial lipoproteins through toll-like receptor-2. Science. 1999;285:736–9. doi: 10.1126/science.285.5428.736. [DOI] [PubMed] [Google Scholar]
- 20.Brightbill HD, Libraty DH, Krutzik SR, et al. Host defense mechanisms triggered by microbial lipoproteins through toll-like receptors. Science. 1999;285:732–6. doi: 10.1126/science.285.5428.732. [DOI] [PubMed] [Google Scholar]
- 21.Opitz B, Schroder NW, Spreitzer I, et al. Toll-like receptor-2 mediates Treponema glycolipid and lipoteichoic acid-induced NF-kappaB translocation. J Biol Chem. 2001;276:22041–7. doi: 10.1074/jbc.M010481200. [DOI] [PubMed] [Google Scholar]
- 22.Schwandner R, Dziarski R, Wesche H, Rothe M, Kirschning CJ. Peptidoglycan- and lipoteichoic acid-induced cell activation is mediated by toll-like receptor 2. J Biol Chem. 1999;274:17406–9. doi: 10.1074/jbc.274.25.17406. [DOI] [PubMed] [Google Scholar]
- 23.Ekström ES, Bengtsson Å, Svensson A, et al. Presence of CD30 (+) and CD30L (+) cells in human placenta and soluble CD30 levels in cord blood are independent of maternal atopy. Placenta. 2001;22:372–9. doi: 10.1053/plac.2000.0619. [DOI] [PubMed] [Google Scholar]
- 24.Bäckhed F, Söderhäll M, Ekman P, Normark S, Richter-Dahlfors A. Induction of innate immune responses by Escherichia coli and purified lipopolysaccharide correlate with organ- and cell-specific expression of Toll-like receptors within the human urinary tract. Cell Microbiol. 2001;3:153–8. doi: 10.1046/j.1462-5822.2001.00101.x. [DOI] [PubMed] [Google Scholar]
- 25.Kovacs AD, Cebers G, Cebere A, Liljequist S. Selective and AMPA receptor-dependent astrocyte death following prolonged blockade of glutamate reuptake in rat cerebellar cultures. Exp Neurol. 2002;174:58–71. doi: 10.1006/exnr.2001.7839. [DOI] [PubMed] [Google Scholar]
- 26.Benirscke K, Kaufmann P. 3. New York: Springer-Verlag; 1995. Pathology of the Human Placenta. [Google Scholar]
- 27.Cario E, Brown D, McKee M, Lynch-Devaney K, Gerken G, Podolsky DK. Commensal-associated molecular patterns induce selective toll-like receptor-trafficking from apical membrane to cytoplasmic compartments in polarized intestinal epithelium. Am J Pathol. 2002;160:165–73. doi: 10.1016/S0002-9440(10)64360-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zarember KA, Godowski PJ. Tissue expression of human Toll-like receptors and differential regulation of Toll-like receptor mRNAs in leukocytes in response to microbes, their products, and cytokines. J Immunol. 2002;168:554–61. doi: 10.4049/jimmunol.168.2.554. [DOI] [PubMed] [Google Scholar]
- 29.Harju K, Glumoff V, Hallman M. Ontogeny of Toll-like receptors Tlr2 and Tlr4 in mice. Pediatr Res. 2001;49:81–3. doi: 10.1203/00006450-200101000-00018. [DOI] [PubMed] [Google Scholar]
- 30.Beutler E, Gelbart T, West C. Synergy between TLR2 and TLR4: a safety mechanism. Blood Cells Mol Dis. 2001;27:728–30. doi: 10.1006/bcmd.2001.0441. [DOI] [PubMed] [Google Scholar]
- 31.Adeleye TA, Moreno C, Ivanyi J, Aston R. The modulation of tumour necrosis factor-alpha, interleukin-1 alpha and glucose levels with GMDP and other analogues of muramyl dipeptide. APMIS. 1994;102:145–52. doi: 10.1111/j.1699-0463.1994.tb04859.x. [DOI] [PubMed] [Google Scholar]
- 32.Beigneux A, Huang J, Le Contel C, Heremans H, Parant MA. Differential role of interferon-gamma in the potentiating effect of muramyl peptides for enhanced responses to lipopolysaccharide in mice: effect of cyclosporin A. J Interferon Cytokine Res. 1995;15:359–65. doi: 10.1089/jir.1995.15.359. [DOI] [PubMed] [Google Scholar]
- 33.Noso Y, Becker J, Riveau G, Audibert F, Chedid L. Production and enhanced anti-tumor activity of tumor necrosis factor in mice treated with cyclophosphamide. Jpn J Cancer Res. 1990;81:942–8. doi: 10.1111/j.1349-7006.1990.tb02671.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Parant MA, Pouillart P, Le Contel C, Parant FJ, Chedid LA, Bahr GM. Selective modulation of lipopolysaccharide-induced death and cytokine production by various muramyl peptides. Infect Immun. 1995;63:110–5. doi: 10.1128/iai.63.1.110-115.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Shimizu T, Iwamoto Y, Yanagihara Y, Ikeda K, Achiwa K. Combined effects of synthetic lipid A analogs or bacterial lipopolysaccharide with glucosaminylmuramyl dipeptide on antitumor activity against Meth A fibrosarcoma in mice. Int J Immunopharmacol. 1992;14:1415–20. doi: 10.1016/0192-0561(92)90013-b. [DOI] [PubMed] [Google Scholar]
- 36.Nakatsuka M, Asagiri K, Noguchi S, Habara T, Kudo T. Nafamostat mesilate, a serine protease inhibitor, suppresses lipopolysaccharide-induced nitric oxide synthesis and apoptosis in cultured human trophoblasts. Life Sci. 2000;67:1243–50. doi: 10.1016/s0024-3205(00)00716-5. [DOI] [PubMed] [Google Scholar]
- 37.St Johnston D, Nusslein-Volhard C. The origin of pattern and polarity in the Drosophila embryo. Cell. 1992;68:201–19. doi: 10.1016/0092-8674(92)90466-p. [DOI] [PubMed] [Google Scholar]
- 38.Belvin MP, Anderson KV. A conserved signaling pathway: the Drosophila toll-dorsal pathway. Ann Rev Cell Dev Biol. 1996;12:393–416. doi: 10.1146/annurev.cellbio.12.1.393. [DOI] [PubMed] [Google Scholar]
- 39.Morisato D, Anderson KV. Signaling pathways that establish the dorsal-ventral pattern of the Drosophila embryo. Annu Rev Genet. 1995;29:371–99. doi: 10.1146/annurev.ge.29.120195.002103. [DOI] [PubMed] [Google Scholar]
- 40.Schnare M, Barton GM, Holt AC, Takeda K, Akira S, Medzhitov R. Toll-like receptors control activation of adaptive immune responses. Nat Immunol. 2001;2:947–50. doi: 10.1038/ni712. [DOI] [PubMed] [Google Scholar]