Introduction
Of the many significant advances in the field of immunology that have occurred in the last 30 years arguably the most important, at least from a T-cell biologists point of view, has been the definition of major histocompatibility complex (MHC) restriction1 and the crystal structure of human leucocyte antigen (HLA) molecules.2–4 These demonstrations have enabled detailed study of the way in which T cells recognize antigen and also how this process differs from that in B cells. With a few notable exceptions, production of specific immunoglobulin by B cells is T-cell dependent. Furthermore, recruitment of non-lymphoid effector cells into areas of inflammation is a process largely marshalled by the T lymphocyte. It follows therefore that inhibition of T-cell function may have beneficial outcomes in situations in which detrimental immune responses occur and give rise to pathology.
Suppression of T-cell function has been achieved with the use of pharmacological agents, such as glucocorticosteroids and cyclosporin, and more recently with biological agents, such as monoclonal antibodies directed against CD3 and CD4. Whilst achieving the desired aim of immunosuppression, these approaches suffer from serious weaknesses, the most obvious of which is a lack of specificity for the antigen(s) which drive the inflammatory process. The same may be said to be true of other therapeutic approaches currently being developed which aim to neutralize the effects of individual mediators in the inflammatory cascade. Here, a notable success has been the use of monoclonal antibodies to tumour necrosis factor (TNF) to treat patients with rheumatoid arthritis.5 However, the long-term consequences of removing individual mediators remain to be seen. Of concern is the recent report of subjects receiving anti-TNF therapy developing a lupus-like syndrome thought to be a direct result of the therapy itself.6
The challenge of introducing specificity into strategies aimed at modulating T-cell responses is one that has been met with considerable success in animal models by the use of peptide epitopes from the antigen in question. Peptide-induced tolerance has been demonstrated in models of experimental autoimmune encephalomyelitis (EAE),7,8 collagen-induced arthritis9 and, more recently, in models of allergic disease. Briner and colleagues sensitized mice to the cat allergen Fel d I and subsequently demonstrated the ability of allergen-derived peptides to inhibit T-cell cytokine and antibody production.10 Hoyne and colleagues demonstrated the ability of peptides from the house dust mite allergen Der p II to down-regulate T-cell and antibody responses to challenge with intact protein and also to prevent sensitization by prior administration.11,12 Bauer administered a dominant T-cell epitope of Bet v I to CBA/J mice either prior to or following sensitization with the whole allergen. No change in antibody isotype was observed but T-cell proliferative responses were inhibited by both treatments.13 More recently, mice sensitized to bee venom allergen phospholipase A2 (PLA2), were treated by intranasal administration of a mixture of three long peptides spanning the entire molecule. A marked reduction in specific immunoglobulin E (IgE) was observed coupled with a decreased interleukin-4 (IL-4) to interferon-γ (IFN-γ) ratio. Pre-administration of peptide resulted in a failure to develop IgE sensitization to subsequent immunization with the whole molecule.14
In addition to animal models, the ability of high-dose peptides to modulate human T-cell responses has also been demonstrated. In vitro studies with human T cells were performed by Lamb and colleagues, who demonstrated the induction of antigen-specific non-responsiveness following the incubation of T-cell clones with supraoptimal doses of specific ligand in the absence of professional antigen-presenting cells.15 Collectively, these studies provided a logical basis for the translation of a peptide-based therapeutic approach in man.
Whole Allergen Immunotherapy
Allergic diseases have, in some quarters, been regarded as the ‘poor relation’ of immunology; a group of diseases which are not particularly severe and can be managed adequately with conventional pharmacotherapy. The reality is that allergic sensitization is increasing in prevalence at an alarming rate. Furthermore, sensitization to previously innocuous food stuffs, such as peanuts and tree nuts, have increased dramatically and represent a significant threat to life. There is therefore a significant unmet need in relation to disease-modifying therapies (as opposed to palliation) in atopic disorders.
In fact, disease-modifying therapy is available in the form of conventional whole allergen immunotherapy often referred to as SIT (specific immunotherapy). First described by Freeman and Noon working at St Mary's Hospital in London, SIT involves administration, initially by injection but more recently by sublingual or swallow, of increasing doses of whole allergen extract. This approach is widely used but due to a number of drawbacks, is unlikely ever to be adopted on a large scale.
The primary problems associated with this form of therapy are the risk of systemic IgE-mediated reactions resulting from cross-linking by intact allergen, the relatively crude and unstandardized extracts employed and also the length of time that effective desensitization takes. For example, subcutaneous or intramuscular injections are given at least monthly for 3 or more years.
Allergic Diseases as a Model for the Development of Immunological Therapies
It is, however, the ability to induce disease in allergic subjects in the clinical laboratory that separates this group of diseases from others driven by T cells. By careful selection of patients, it is possible to elicit exacerbations of disease in relative safety by challenge of the subject with the molecule to which they are allergic. Importantly, there is no evidence that such experimental challenges lead to any permanent worsening of the diseases and, moreover, challenge can be performed in a single day, allowing measurements of tissues (bronchial, nasal skin biopsies) and body fluids [e.g. blood, bronchoalveolar lavage fluid (BALF)] prior to despatching the patient with no ill effects. Such an approach is not possible in other areas. In autoimmunity for example, antigens remain incompletely defined (despite obvious HLA disease associations) and induction of disease exacerbation is unethical and impractical. Similar concerns are applicable to allograft transplantation. It follows then, that allergic disorders offer the only safe and ethical system in which to develop and evaluate novel forms of antigen-specific immunological therapy which, as described earlier, is the logical pinnacle of disease-modifying approaches.
The model of allergen challenge has been successfully employed to study allergic and non-allergic asthma and to define the presence of increased numbers of CD4+ T cells and eosinophils in areas of allergic inflammation, both at baseline and following allergen challenge. Furthermore, the challenge model has enabled evaluation of therapeutic strategies directed against the T cell and has provided useful samples for the investigation of immunological mechanisms in pathogenesis.16,17
The Role of the T cell in Asthma: Direct T-cell Activation with Peptides
We recently took the allergen challenge model a step further by injecting subjects with allergic asthma with peptides derived from the allergen to which they were sensitive, in this case Fel d I, the major allergen of cats. The object of our experiment was to activate human antigen-specific T cells in vivo and attempt to induce symptoms of asthma, which would be both T-cell-dependent and IgE-independent since peptides were designed to be too short to cross-link adjacent IgE molecules on the surface of mast cells and basophils.
In the study, peptides were administered intradermally to allergic asthmatic subjects, with moderate disease severity, resulting in the induction of isolated late asthmatic reactions in a proportion of individuals(18). This is unlike the response to whole allergen challenge. Following whole allergen challenge in the skin, or lung or elsewhere in an allergic subject, a bimodal response can be observed. The two phases are comprised of a type I hypersensitivity reaction (IgE-mast cell-mediated) peaking at 15 min (example: wheal and flare reaction) and a form of type IV hypersensitivity with an onset of 2–4 hr, peaking at 6–9 hr and requiring 24 hr or more to resolve. In the asthmatic, both early- and late-phase responses can lead to airway narrowing.
In the peptide challenge model, each of the three injected peptides was capable of inducing peripheral blood mononuclear cell proliferation in a percentage of the subjects. The ability to induce isolated late asthmatic reactions did not correlate with peptide-induced proliferative responses since the latter may be dose-dependent and the dose administered in the study was the lowest dose demonstrated to induce late asthmatic reactions. T-cell responses to two of the three peptides were shown to be MHC-restricted and subjects experiencing late asthmatic reactions were shown to express HLA-DR molecules associated with peptide restriction. Thus, using human and murine fibroblasts transfected with individual HLA-DR molecules, we were able to determine that one of the three peptides could be presented to allergen-specific T-cell lines by DRB1*0405 and 0408, whereas another of the peptides was restricted by DRB1*0101 and both 1301 and 1302.
The Induction of Antigen-Specific T-Cell Hyporesponsiveness with Peptides
The ability to design peptides which, theoretically, lack the three-dimensional structure required to interact with and cross-link specific IgE, leads to the notion that peptide immunotherapy may supersede conventional whole allergen immunotherapy. Peptide-based immunotherapy was recently evaluated in individuals with allergic disease induced by cats. Approximately 95% of subjects allergically sensitized to cats have IgE directed against the major cat allergen Fel d I. Epitope mapping studies by Mark and colleagues19 and Counsell and colleagues,20 defined the majority of T-cell proliferative responses to be induced by the N terminus of chain 1 (Fel d I is a heterodimer) of the molecule. Based upon these findings, two relatively large peptides, termed IPC-1 and IPC-2, were evaluated by a number of groups. Norman and colleagues21 administered four injections of peptide or placebo to 95 cat-sensitive patients in three dose groups (7·5 µg, 75 µg and 750 µg per injection). Clinical efficacy was observed only at the highest dose of peptide (4 × 750 µg). Both nasal and lung symptom scores improved in response to peptide therapy, although a significant placebo effect was observed. Treatment was associated with a significant incidence of adverse events, which occurred a few minutes to several hours after peptide injection. Further studies from the same group22 demonstrated reduced IL-4 production in IPC-1/IPC-2-specific T-cell lines following therapy. A similar reduction in IL-4 production was reported by Pène and colleagues.23 Peripheral blood mononuclear cells were stimulated with cat dander extract before and after treatment with IPC-1/IPC-2. A significant reduction (compared to background levels) of IL-4 production was observed in the high-dose group (individual dose 750 µg; total dose 1500–4500 µg) but not in the lower dose groups. A reduction in allergen PD20 [the provocative dose of inhaled allergen giving a 20% fall in forced expiratory volume in 1s (FEV1)] was also observed in response to both high (1500–4500 µg) and medium (individual dose 75 µg; total dose 150–450 µg) dose regimens when compared to baseline, but not when compared to placebo. The variability in dose between each group arose as a result of a staggered dose protocol in which a number of individuals received weekly injections for 2, 4, or 6 weeks. Simons and colleagues performed a double-blind, parallel group study administering weekly injections, for 4 weeks, of 250 µg of the same IPC-1 and IPC-2 preparation to 42 (21 active and 21 placebo) individuals with cat-induced rhinitis and/or asthma. The investigators observed late-onset symptoms of rhinitis, asthma and pruritis following treatment.24 No change in cytokine secretion patterns of peripheral blood mononuclear cells was observed before and after therapy. Furthermore, no change in early- and late-phase skin responses to whole allergen were recorded at several time-points (2, 6 and 24 weeks) after treatment. In a multicentre study of 133 patients, Maguire and colleagues reported an improvement in pulmonary function in subjects receiving eight injections of 750 µg IPC-1/IPC-2, who had diminished baseline 1-s forced expiratory volume (FEV1) but only at a single time-point (3 weeks) post-therapy25. Injections were given in two blocks of four, with two injections per week. A large number of adverse events were reported in this study, including some that required the use of adrenaline. In keeping with other studies evaluating IPC-1/IPC-2, late-onset adverse reactions diminished with successive doses of peptide, suggesting the induction of hyporesponsiveness. Thus, a relatively modest improvement in symptom scores combined with frequent adverse reactions resulted in cat and ragweed vaccines being withdrawn from clinical trials.
At least some of the adverse events were likely to be due to cross-linking of IgE by the peptides which, by virtue of their relatively large size, may have been able to bridge adjacent IgE molecules on the surface of mast cells. A second, more common, manifestation was symptoms of breathlessness and wheeze with an onset several hours after peptide administration. Our own work has confirmed that these symptoms were probably T-cell dependent, IgE-independent, isolated late asthmatic reactions.
Norman and co-workers reported a diminution in late adverse events with successive peptide administration compatible with the induction of hyporesponsiveness.21 In our own model, subjects who developed isolated late asthmatic reactions were rechallenged several weeks after their initial reaction. The second injection failed to elicit bronchospasm in the lung unless it was administered several months after the first. We have subsequently been able to demonstrate a ‘window’ of hyporesponsiveness which occurs between approximately 2 and 8 weeks after a single peptide injection (unpublished observations). From 8 weeks on, the ability to react to peptide challenge gradually returns but takes 4–6 months to reach baseline. It may be that a series of injections of increasing peptide dose may extend the refractory period and result in protection from rechallenge for a period of a year or more.
Peptides Induce Allergen-Specific Hyporesponsiveness to Challenge With Whole Antigen
Rechallenge demonstrated reduced responses to peptides in the lung. More relevant for therapy was the question of whether the induction of peptide-specific hyporesponsiveness would also result in reduced responses to challenge with intact allergen. Cat-allergic asthmatics were challenged cutaneously (intradermal administration) with whole cat dander allergen extract, before and after a single injection of 5 µg of each of 12 peptides in a mixture. The 12 peptides spanned the majority of the Fel d I molecule in order to enhance the chances of providing T-cell epitopes for the majority of the population. The results demonstrated that the magnitude of the cutaneous late-phase response to allergen could be markedly reduced even after a single administration of peptide. Furthermore, several subjects, who had received peptide but had not experienced a late asthmatic reaction, also displayed equivalent reductions in the size of the cutaneous late response to whole allergen, suggesting that late asthmatic reactions are not required to achieve T-cell hyporesponsiveness to whole allergen. No change was observed in the cutaneous early response to whole allergen after a single peptide challenge. However, in a subsequent study employing multiple peptide administrations, the magnitude of the IgE-mast cell-dependent early response was also reduced (unpublished data).
Protection from rechallenge with whole allergen was also achieved by Muller and colleagues who treated five subjects allergic to bee venom, with a mixture of three peptides from the major venom allergen phospholipase A2.26 Each of the study subjects had experienced a previous moderately severe systemic reaction following a wild bee sting. Peptides were administered at weekly intervals at increasing doses. The ‘induction’ phase consisted of seven injections (0·1, 1·0, 3·0, 6·0, 12·0, 25·0 and 50·0 µg). ‘Maintenance’ dose was three injections of 100 µg. A total dose of 397·1 µg was administered. Post peptide therapy, all five were able to tolerate challenge with 10 µg of purified PLA2. Furthermore, when subjects were challenged with a wild bee sting in the clinical laboratory, three experienced no reaction whilst the remaining two experienced mild localized reactions at the sting site. Analysing the in vitro T-cell responses to both peptides and whole allergen, the authors went on to demonstrate that the induction of clinical hyporesponsiveness was accompanied by reductions in T-cell proliferative responses and inflammatory cytokines. Subsequent studies revealed an increase in allergen-specific IL-10 in these subjects (Akdis and Blaser, personal communication), which was accompanied by an increase in allergen-specific IgG4 concentrations.
Antigen-specific immune regulation versus immune deviation
Many studies evaluating mechanisms in successful whole allergen immunotherapy have described a relative shift in T helper cytokine phenotype from T helper type 2 (Th2) towards Th1.27 In general terms, an increase in the ratio of IL-4:IFN-γ was observed, together with a concomitant change in the ratio of allergen-specific IgE:IgG in favour of the latter.
Based upon the thesis that shifting allergic responses towards Th1 is the predominant mechanism underlying the success of conventional immunotherapy, other approaches have been developed which, it is hoped, will achieve the same end but by different means. For example, mycobacteria (Mycobacterium vaccae; SR Pharma, London, UK) and bacterial DNA [immunostimulatory sequences (ISS); Dynavax Corp., Berkeley, CA, USA] are currently being evaluated in clinical trials.
However, few if any of the studies analysing immune deviation in immunotherapy have focused on ‘regulatory’ cytokines such as IL-10 and transforming growth factor-β (TGF-β), or indeed, regulatory cell fate-determining pathways such as the Notch system.28,29 Akdis and colleagues provided extensive evidence for a major role for IL-10 in establishing PLA2-specific hyporesponsiveness both in allergic individuals undergoing successful whole allergen or peptide immunotherapy and also in hyperimmune bee keepers.30,31 Our own studies with peptides derived from Fel d I suggest that whilst pro-inflammatory cytokines are decreased after peptide therapy, IL-10 production is markedly increased (unpublished observations).
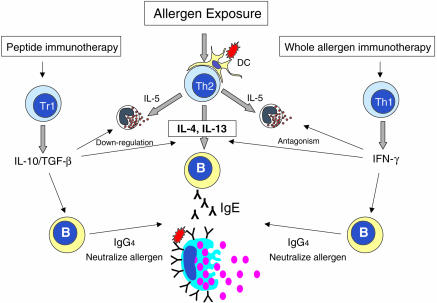
It appears then, that either two different mechanisms exist in conventional aeroallergen immunotherapy (Th2 to Th1 shift) versus peptide therapy or whole venom allergen immunotherapy (induction of IL-10) or, that mechanisms are similar (Fig. 1). It is conceivable that the different route of exposure to venom creates a qualitatively different response and that highly soluble peptides, which do not require antigen processing and the accompanying activation of antigen-presenting cells, lead to a different pathway of immune deviation (i.e. IL-10). However, it is quite apparent from the literature that regulatory cytokines have not been analysed in studies of whole aeroallergen immunotherapy. Until such investigations are performed, division or unification of mechanisms will not be possible.
Figure 1.
In the atopic subject, allergen challenge results in the generation and activation of Th2 cells that release IL-4, IL-5 and IL-13. IL-5 acts on the bone marrow to release eosinophils that traffic to the site of challenge. IL-4 and IL-13 act as isotype switch factors for B cells resulting in IgE production. IgE primes tissue mast cells for cross-linking and activation by allergen, leading to smooth muscle contraction, erythema and pruritis. Whole allergen immunotherapy induces a change in the ratio of allergen-specific IL-4:IFN-γ in favour of the latter. IFN-γ antagonizes the actions of IL-4 and Th2 cells in addition to promoting isotype switching to IgG subclasses. IgG competes with IgE for allergen, resulting in reduced mast cell activation. Peptide immunotherapy appears to induce IL-10 (TGF-β)-secreting regulatory T cells (Tr). Regulatory cytokines suppress Th2 cells and the actions of Th2 cytokines. IL-10 promotes IgG4 isotype switching leading to competition with IgE for allergen.
Peptide Immunotherapy in Autoimmune Diseases
Whilst induction of Th1 responses may appeal to those attempting to reverse allergic responses, such a strategy would be an anathema to those charged with ameliorating the majority of autoimmune diseases. The concept, then, of an approach, such as the use of peptides, which appears to induce antigen-specific regulatory responses (IL-10, Notch), provides a framework upon which unification of therapeutic approaches may be hung. To date, the only autoimmune disease to have been addressed with peptides is multiple sclerosis. Like many autoimmune diseases, multiple sclerosis has common HLA associations, such as with HLA-DR2. In DR2-positive sufferers, the major myelin basic protein epitope spans residues 93–99. Karin and colleagues32 performed alanine scanning analysis of residues 87–99 in order to determine T-cell receptor and MHC contact residues and to identify altered peptide ligands which may have therapeutic benefit by virtue of their ability to induce reduced IFN-γ and TNF-α. Altered peptide ligands were evaluated in vivo in a rat model of experimental allergic encephalomyelitis (EAE). Substitution of alanine for lysine at position 91 resulted in a peptide with properties favourable for use in therapy. Subsequently a peptide substituted at several residues was developed and evaluated in clinical trials.33,34
Both studies were terminated prior to completion due to the high frequency of adverse events, predominantly hypersensitivity reactions and disease exacerbations. Analysis of responses in vitro revealed considerable cross-reactivity among T cells which were capable of recognizing both the native and the altered peptide. However, modulation of the T-cell responses toward a Th2 phenotype, believed to be protective in multiple sclerosis, was also observed.
Our own studies have indicated that disease exacerbation, in the case of asthmatics the induction of a late asthmatic reaction, can occur following intradermal administration of doses of peptide as low as 1 µg. In other studies with cat peptides (described above), allergic symptoms were also elicited with microgram quantities of peptide and in both sets of studies, were dose dependent in terms of both frequency and magnitude. It is interesting therefore to note that in the two multiple sclerosis studies described in which an altered peptide ligand was employed, patients were administered doses of between 5 and 50 mg of peptide on a weekly and then monthly basis. It is not surprising therefore that exacerbations were observed.
It is unclear from the published studies how such dosing regimes were selected. However, it is apparent that considerably higher doses of peptide are required to induce ‘tolerance’ in mice and rats than in humans. Karin administered doses of 2 mg of altered peptide ligand to ameliorate EAE in rats.32 Hoyne and colleagues employed multiple administrations totalling several hundred micrograms to render mice hyporesponsive to house dust mite proteins.11,12
The studies of Muller and those from our own laboratory suggest that antigen-specific hyporesponsiveness in humans can be achieved with dramatically lower doses. Indeed, taking differences in body weight into account, there may be a difference of approximately five orders of magnitude in the dose of peptide required to induce significant reductions in both in vivo and in vitro parameters of reactivity between murine and human systems.
Conclusions
Antigen-specific approaches to therapy offer the best chance of modifying disease processes without impinging on the rest of the immune response. Not only do currently available palliative approaches fail to address the underlying pathogenesis of disease, they also fail to address the issue of disease incidence. The incidence of allergic diseases including asthma is increasing rapidly, particularly in developed countries. Failure to develop disease-modifying approaches to therapy will give rise to greater morbidity, mortality and economic burden. Peptide immunotherapy, particularly in allergic diseases, may offer the prospect of regulating immune responses in a specific way, with a substantially reduced risk of IgE-mediated adverse events. Years of evaluation of whole allergen immunotherapy have demonstrated that antigen-specific modulation is achievable, although the risk associated with the use of whole allergen molecules is considerable. The use of short peptide sequences with negligible IgE cross-linking capacity may give rise to safer and more efficacious immunotherapy for both allergic and autoimmune diseases.
Acknowledgments
I am indebted to my colleagues Professor A. B. Kay, Dr Mimi Haselden, Dr Bill Oldfield, Dr Cezmi Akdis and Dr Kurt Blaser for their contributions to this work which was funded by the National Asthma Campaign.
References
- 1.Zinkernagel RM, Doherty PC. Restriction of in vitro T cell-mediated cytotoxicity in lymphocytic choriomeningitis within a syngeneic or semiallogeneic system. Nature. 1974;248:701–2. doi: 10.1038/248701a0. [DOI] [PubMed] [Google Scholar]
- 2.Bjorkman PJ, Saper MA, Samraoui B, Bennett WS, Strominger JL, Wiley DC. Structure of the human class I histocompatibility antigen, HLA-A2. Nature. 1987;329:506–12. doi: 10.1038/329506a0. [DOI] [PubMed] [Google Scholar]
- 3.Bjorkman PJ, Saper MA, Samraoui B, Bennett WS, Strominger JL, Wiley DC. The foreign antigen binding site and T cell recognition regions of class I histocompatibility antigens. Nature. 1987;329:512–18. doi: 10.1038/329512a0. [DOI] [PubMed] [Google Scholar]
- 4.Brown JH, Jardetzky TS, Gorga JC, Stern LJ, Urban RG, Strominger JL, Wiley DC. Three-dimensional structure of the human class II histocompatibility antigen HLA-DR1. Nature. 1993;364:33–9. doi: 10.1038/364033a0. [DOI] [PubMed] [Google Scholar]
- 5.Feldmann M, Maini RN. Anti-TNF-α therapy of rheumatoid arthritis. What Have We Learned? Annu Rev Immunol. 2001;19:163–96. doi: 10.1146/annurev.immunol.19.1.163. [DOI] [PubMed] [Google Scholar]
- 6.Charles PJ, Smeenk RJ, De Jong J, Feldmann M, Maini RN. Assessment of antibodies to double-stranded DNA induced in rheumatoid arthritis patients following treatment with infliximab, a monoclonal antibody to tumor necrosis factor alpha: findings in open-label and randomized placebo-controlled trials. Arthritis Rheum. 2000;43:2383–90. doi: 10.1002/1529-0131(200011)43:11<2383::AID-ANR2>3.0.CO;2-D. [DOI] [PubMed] [Google Scholar]
- 7.Clayton JP, Gammon GM, Ando DG, Kono DH, Hood L, Sercarz EE. Peptide-specific prevention of experimental allergic encephalomyelitis. Neonatal tolerance induced to the dominant T cell determinant of myelin basic protein. J Exp Med. 1989;169:1681–91. doi: 10.1084/jem.169.5.1681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gaur A, Wiers B, Liu A, Rothbard J, Fathman CG. Amelioration of autoimmune encephalomyelitis by myelin basic protein synthetic peptide-induced anergy. Science. 1992;258:1491–4. doi: 10.1126/science.1279812. [DOI] [PubMed] [Google Scholar]
- 9.Staines NA, Harper N, Ward FJ, Malmstrom V, Holmdahl R, Bansal S. Mucosal tolerance and suppression of collagen-induced arthritis CIA induced by nasal inhalation of synthetic peptide 184–198 of bovine type II collagen CII expressing a dominant T cell epitope. Clin Exp Immunol. 1996;103:368–75. doi: 10.1111/j.1365-2249.1996.tb08289.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Briner TJ, Kuo MC, Keating KM, Rogers BL, Greenstein JL. Peripheral T-cell tolerance induced in naive and primed mice by subcutaneous injection of peptides from the major cat allergen Fel d I. Proc Natl Acad Sci USA. 1993;90:7608–12. doi: 10.1073/pnas.90.16.7608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hoyne GF, O'Hehir RE, Wraith DC, Thomas WR, Lamb JR. Inhibition of T cell and antibody responses to house dust mite allergen by inhalation of the dominant T cell epitope in naive and sensitized mice. J Exp Med. 1993;178:1783–8. doi: 10.1084/jem.178.5.1783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hoyne GF, Askonas BA, Hetzel C, Thomas WR, Lamb JR. Regulation of house dust mite responses by intranasally administered peptide: transient activation of CD4+ T cells precedes the development of tolerance in vivo. Int Immunol. 1996;8:335–42. doi: 10.1093/intimm/8.3.335. [DOI] [PubMed] [Google Scholar]
- 13.Bauer L, Bohle B, Jahn-Schmid B, Wiedermann U, DaSeries A, Renz H, Kraft D, Ebner C. Modulation of the allergic immune response in BALB/c mice by subcutaneous injection of high doses of the dominant T cell epitope from the major birch pollen allergen Bet v 1. Clin Exp Immunol. 1997;107:536–41. doi: 10.1046/j.1365-2249.1997.d01-953.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Astori M, von Garnier C, Kettner A, Dufour N, Corradin G, Spertini F. Inducing tolerance by intranasal administration of long peptides in naive and primed CBA/J mice. J Immunol. 2000;165:3497–505. doi: 10.4049/jimmunol.165.6.3497. [DOI] [PubMed] [Google Scholar]
- 15.Lamb JR, Skidmore BJ, Green N, Chiller JM, Feldmann M. Induction of tolerance in influenza virus-immune T lymphocyte clones with synthetic peptides of influenza hemagglutinin. J Exp Med. 1983;157:1434–47. doi: 10.1084/jem.157.5.1434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Khan LN, Kon OM, Macfarlane AJ, Meng Q, Ying S, Barnes NC, Barry KA. Attenuation of the allergen-induced late asthmatic reaction by cyclosporin A is associated with inhibition of bronchial eosinophils, interleukin-5, granulocyte macrophage colony-stimulating factor, and eotaxin: am. J Respir Crit Care Med. 2000;162:1377–82. doi: 10.1164/ajrccm.162.4.9911117. [DOI] [PubMed] [Google Scholar]
- 17.Macfarlane AJ, Kon OM, Smith SJ, et al. Basophils, eosinophils, and mast cells in atopic and nonatopic asthma and in late-phase allergic reactions in the lung and skin. J Allergy Clin Immunol. 2000;105:99–107. doi: 10.1016/s0091-6749(00)90184-2. [DOI] [PubMed] [Google Scholar]
- 18.Haselden BM, Kay AB, Larche M. Immunoglobulin E-independent major histocompatibility complex-restricted T cell peptide epitope-induced late asthmatic reactions. J Exp Med. 1999;189:1885–94. doi: 10.1084/jem.189.12.1885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mark PG, Segal DB, Dallaire ML, Garman RD. Human T and B cell immune responses to Fel d 1 in cat-allergic and non-cat-allergic subjects. Clin Exp Allergy. 1996;26:1316–28. [PubMed] [Google Scholar]
- 20.Counsell CM, Bond JF, Ohman JL, Greenstein JL, Garman RD. Definition of the human T-cell epitopes of Fel d 1, the major allergen of the domestic cat. J Allergy Clin Immunol. 1996;98:884–94. doi: 10.1016/s0091-6749(96)80004-2. [DOI] [PubMed] [Google Scholar]
- 21.Norman PS, Ohman JL, Long AA, et al. Treatment of cat allergy with T-cell reactive peptides. Am J Respir Crit Care Med. 1996;154:1623–8. doi: 10.1164/ajrccm.154.6.8970345. [DOI] [PubMed] [Google Scholar]
- 22.Marcotte GV, Braun CM, Norman PS, Nicodemus CF, Kagey-Sobotka A, Lichtenstein LM, Essayan DM. Effects of peptide therapy on ex vivo T-cell responses. J Allergy Clin Immunol. 1998;101:506–13. doi: 10.1016/S0091-6749(98)70358-6. [DOI] [PubMed] [Google Scholar]
- 23.Pene J, Desroches A, Paradis L, Lebel B, Farce M, Nicodemus CF, Yssel H, Bousquet J. Immunotherapy with Fel d 1 peptides decreases IL-4 release by peripheral blood T cells of patients allergic to cats. J Allergy Clin Immunol. 1998;102:571–8. doi: 10.1016/s0091-6749(98)70294-5. [DOI] [PubMed] [Google Scholar]
- 24.Simons FE, Imada M, Li Y, Watson WTKT HayGlass. Fel d 1 peptides: effect on skin tests and cytokine synthesis in cat-allergic human subjects. Int Immunol. 1996;8:1937–454. doi: 10.1093/intimm/8.12.1937. [DOI] [PubMed] [Google Scholar]
- 25.Maguire P, Nicodemus C, Robinson D, Aaronson D, Umetsu DT. The safety and efficacy of ALLERVAX CAT in cat allergic patients. Clin Immunol. 1999;93:222–31. doi: 10.1006/clim.1999.4795. [DOI] [PubMed] [Google Scholar]
- 26.Muller U, Akdis CA, Fricker M, Akdis M, Blesken T, Bettens F, Blaser K. Successful immunotherapy with T-cell epitope peptides of bee venom phospholipase A2 induces specific T-cell anergy in patients allergic to bee venom. J Allergy Clin Immunol. 1998;101:747–54. doi: 10.1016/S0091-6749(98)70402-6. [DOI] [PubMed] [Google Scholar]
- 27.Rolland J, O'Hehir R. Immunotherapy of allergy: anergy, deletion, and immune deviation. Curr Opin Immunol. 1998;10:640–5. doi: 10.1016/s0952-7915(98)80082-4. [DOI] [PubMed] [Google Scholar]
- 28.Hoyne GF, Dallman MJ, Lamb JR. T-cell regulation of peripheral tolerance and immunity: the potential role for Notch signalling. Immunology. 2000;100:281–8. doi: 10.1046/j.1365-2567.2000.00073.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hoyne GF, Le RI, Corsin-Jimenez M, et al. Serrate1-induced notch signalling regulates the decision between immunity and tolerance made by peripheral CD4 (+) T cells. Int Immunol. 2000;12:177–85. doi: 10.1093/intimm/12.2.177. [DOI] [PubMed] [Google Scholar]
- 30.Akdis CA, Joss A, Akdis M, Blaser K. Mechanism of IL-10-induced T-cell inactivation in allergic inflammation and normal response to allergens. Int Arch Allergy Immunol. 2001;124:180–2. doi: 10.1159/000053704. [DOI] [PubMed] [Google Scholar]
- 31.Akdis CA, Akdis M, Blesken T, Wymann D, Alkan SS, Muller U, Blaser K. Epitope-specific T cell tolerance to phospholipase A2 in bee venom immunotherapy and recovery by IL-2 and IL-15 in vitro. J Clin Invest. 1996;98:1676–83. doi: 10.1172/JCI118963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Karin N, Binah O, Grabie N, et al. Short peptide-based tolerogens without self-antigenic or pathogenic activity reverse autoimmune disease. J Immunol. 1998;160:5188–94. [PubMed] [Google Scholar]
- 33.Bielekova B, Goodwin B, Richert N, et al. Encephalitogenic potential of the myelin basic protein peptide (amino acids 83–99) in multiple sclerosis: results of a phase II clinical trial with an altered peptide ligand. Nat Med. 2000;6:1167–75. doi: 10.1038/80516. [DOI] [PubMed] [Google Scholar]
- 34.Kappos L, Comi G, Panitch H, Oger J, Antel J, Conlon P, Steinman L. Induction of a non-encephalitogenic type 2 T helper-cell autoimmune response in multiple sclerosis after administration of an altered peptide ligand in a placebo-controlled, randomized phase II trial. The Altered Peptide Ligand Relapsing MS Study Group. Nat Med. 2000;6:1176–82. doi: 10.1038/80525. [DOI] [PubMed] [Google Scholar]