Abstract
We have developed a simple shell-less chick embryo culture system to study glucose-induced malformations. This system involves the culturing of chick embryos from the second day to the fifth day of incubation, with associated yolk and thick and thin albumen outside the egg shell. The system allows the observation of embryonic development of chicks in a glass bowl. Developing embryos at 24 h, 48 h and 72 h incubation, corresponding to the Hamberger Hamilton (HH) stages from 7 to 21, were treated with two concentrations of glucose (50 mM and 100 mM) for 24 h. Glucose treatment resulted in a mortality rate of over 70% in younger embryos. Furthermore, a variety of malformations such as retarded growth, abnormal heart development, macrosomia, exencephaly, etc. were observed in older embryos, which were similar to those reported in mammalian embryos as a consequence of diabetic pregnancy. The glucose-induced malformations were found to be concentration- and stage-dependent, thus emphasizing the roles of the degree of hyperglycemia and the stage of embryonic development in diabetic growth anomalies. Here we demonstrate for the first time that the present system can be used (i) for experiments at early stages of chick embryo development and (ii) for assessing the effects of acute glucose toxicity similar to those reported for mammalian embryos in a hyperglycemic environment.
Keywords: chick embryo, shell-less culture, malformations, glucose, mammalian embryos
Introduction
The degree of hyperglycemia in diabetes varies depending on the degree of deterioration of the metabolic state. After a long period of metabolic derangement, secondary diabetic complications, such as retinopathy, neuropathy, nephropathy and cardiomyopathy may occur. A large array of animal models has been developed to study different aspects of diabetes, such as the db/db mouse, the ob/ob mouse, the sand rat and the Goto Kakisaki rat.
Previous studies to detect fetal malformations in subjects with diabetes have been carried out using mammalian models for two main reasons: (i) both embryonic and adult tissues are challenged in a similar way by the diabetic environment and (ii) the time span for the induction of embryonic damage is remarkably short compared with the time needed for the development of organ complications in the diabetic individual [1]. Such studies have been carried out in vivo or in vitro. However, the use of mammalian embryos requires invasive investigations in pregnant animals that may implicate tissue damage and provoke ethical considerations. Therefore, it would be interesting to investigate glucose-induced malformations by observing developing embryos without invasive interventions. We focused on the development of a system to cultivate chick embryos outside their egg shells. Existing techniques of shell-less chick embryo cultures have inherent limitations depending on the experimental design and the aim of the study [2-4]. These techniques made use of Petri dishes [5], plastic culture tripods with plastic wraps [6], recipient avian eggshells [4] and transparent plastic cups with clear polythene films [7] permitting various degrees of survival rate. However, none of these systems have reported on the successful culturing of early embryonic stages during active organogenesis.
Hence, we aimed to develop a simple model of shell-less chick embryo culture to enable the observation of glucose-induced malformations in early stages of embryo development. Excessive glucose reportedly has a negative impact on fetal growth, including the development of the central nervous system [8-10], macrosomia, rotational defects, growth retardation, and embryonic dysmorphogenesis [11-14]. Moreover, complications of later development and congenital malformations have been reported to be more prevalent in infants of diabetic mothers than under conditions of normal pregnancy [13-17]. In chick embryos, hyperglycemia has been reported to cause abnormalities and mortality in early stages [18]. Our model of a shell-less chick embryo culture is able to ascertain effects of glucose treatment on developing chick embryos at various stages, and to identify disturbances of growth and a variety of malformations depending on the concentration of glucose and the stage of development.
Materials and methods
Fertilized RIR (Rhode Island Red strain) eggs weighing 58-60g and unfertilized eggs obtained from a local hatchery were washed with water and allowed to air dry. Fertilized eggs were incubated at 37.5°C with a relative humidity of 70-80% for 24 h before culturing.
Preparation of shell-less embryo culture
The entire embryo culture was processed under sterile conditions. The assembly of our model for the shell-less chick embryo culture consisted of a transparent glass bowl (8.5 cm surface diameter, a base diameter of 4.5 cm and a depth of 5 cm) and its cover. Incubated eggs were allowed to cool for 25-30 min, then wiped with 70% alcohol under laminar flow to minimize contamination from the shell surface. The eggs were kept in a horizontal position to assure that the embryo was properly positioned.
Thin albumen from unfertilized eggs was poured into a sterile bowl. This albumen acted as a shock-absorber, provided a cushion for the culture and it limited desiccation. The fertilized eggs were then cracked from above with the help of a scalpel approximately 3-3.5 cm from the narrow end and the contents of the eggs were gently released over the albumen cushion in the bowl. It was found that the chance of embryo survival was greater if the eggs’ contents were transferred without damaging the embryo or the yolk, and if the embryo was kept in an upright position.
Each bowl was then covered with a lid, and cultures were incubated at 37.5°C and 80% humidity. The chick embryos aged 24 h (approximate HH stages 7-9), 48 h (HH stages 12-14) and 72 h of incubation (HH stages 19-21) were used for the experiments following Hamberger Hamilton staging [19].
Treatment with glucose
Shell-less chick embryos at the selected stages of development were treated with either 50 μl glucose of 30 mM, 50 mM [20] or 100 mM concentrations prepared in 0.9% saline. Control embryos were treated with 50 μl of 0.9% saline only. The osmolarity of the glucose solutions was assessed using a Micro Osmometer (Precision Systems, USA) and corrected by the addition of distilled water following the protocol described by Torchinsky et al. [21]. The solutions were added from a distance of approximately 1.5 cm above the blastodisc and embryos were then incubated for 24 h. Particular care was taken to transfer the cultures into the incubator within 3-4 min of treatment.
Evaluation of embryonic mortality and morbidity
Embryos were observed after 24 h exposure to glucose treatment. A ring of Whattman paper no.2 was disposed around the blastodisc. The vitelline membrane was cut around the ring, and the ring - along with the embryonic disc - was placed into a Petri dish containing 0.9% saline. The yolk adhering to the embryonic disc was washed with 0.9% saline and the embryo was examined under the stereobinocular microscope. Only live embryos with an evident heartbeat were examined to assess the stage of development and any developmental malformations. Less developed embryos and those with abnormalities were regarded as malformed. Embryos were then fixed with acid-ethanol fixative (acetic acid: absolute alcohol 1:3) and stored in 70% ethanol for detailed investigations.
Results
The survival rate of the embryos varied according to the duration of the culture period. After the first 3 days following culture, 140-142 of the 150 embryos exhibited normal development (94% survival rate). By days 5 to 12 following culture, development was continued in 85-90 of 140-142 surviving embryos (60% survival rate). Approximately 35% of the cultures had to be discarded by day 12 for a variety of reasons, such as leakage of the yolk, adherence of the embryo to the glass surface of the bowl and arrested growth. Those embryos whose membranes were spreading smoothly over the yolk albumen surface (5%) survived until 17 days of development and subsequently died. The movement of the limbs, eyes, beak etc. could be observed clearly (Figure 1).
Figure 1. Developing chick embryos in shell-less culture.
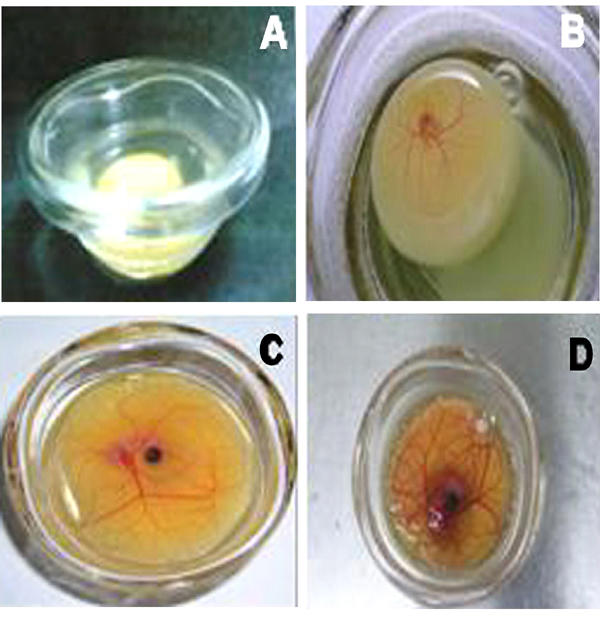
1A: 1 day, B: 5 days, C: 10 days and D: 15 days after culturing.
The osmolarity of the 50 mM and 100 mM glucose solutions prepared by the addition of D-glucose to the 0.9% saline was observed to be 331 ± 6 mOsmoles/l and 386 ± 8, respectively. We adjusted the osmolarity of glucose solutions by adding distilled water [21] to match the plasma osmolarity of the chick, i.e. 280 mOsmoles/l [22]. Consequently, isosmotic glucose solutions were applied to the embryos. Embryos treated with 30 mM glucose neither exhibited any mortality nor any visible abnormalities. However, glucose treatment of 50 mM and 100 mM to the embryos cultured for 24 h (approximate HH stages 7-9) resulted in 70% and 90% mortality, respectively. The surviving embryos showed abnormal development of the neural tube, somites, retarded growth and exencephaly (Figure 2 and Table 1). Similar treatment of the chick embryos cultured for 48 h (approximate HH stages 12-14) resulted in 50% and 70% mortality, respectively. A variety of malformations, such as retarded growth, embryonic dysmorphogenesis, exencephaly, an enlarged heart and blood vessels and abnormal somites were observed in the surviving embryos (Figure 3 and Table 1). Chick embryos cultured for 72 h (approximate HH stages 18-20) and treated in a similar manner resulted in 10% and 15% mortality, respectively. Surviving embryos mainly showed retarded growth, abnormal flexures, torsions, macrosomia and abnormal limbs and tail development (Figure 4 and Table 1). Thus, it can be seen from the results that similar glucose treatment affects the embryonic development differentially depending upon the age of the embryo, exhibiting higher mortality in younger embryos and higher morbidity in older embryos.
Figure 2. Effects of glucose on developing chick embryos (HH 7-9).
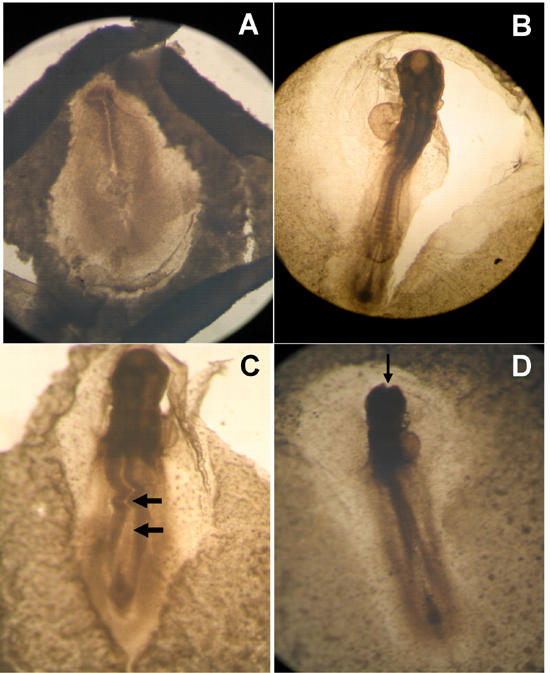
A: Untreated control embryo. B: Embryo treated with 0.9% saline exhibiting normal development. C: Embryo treated with 50 mM glucose. Abnormal development of neural tube and somites are prominent. D: Embryo treated with 100 mM glucose exhibiting exencephaly i.e. inhibition of neural tube closure.
Table 1. Stages of incubation and malformations of the embryos exposed to 50 mM and 100 mM glucose.
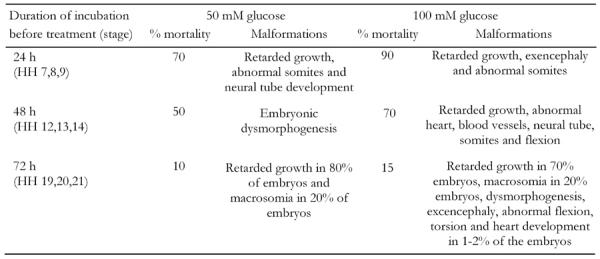
Embryos were incubated for different time intervals to obtain embryos at various developmental stages and then were treated with glucose (50 and 100 mM) for 24, 48 and 72 h. Embryos were then evaluated for their mortality and morbidity under a stereobinocular microscope. Each group included approximately 30-50 embryos.
Figure 3. Effects of glucose on chick embryos (HH 12-14).
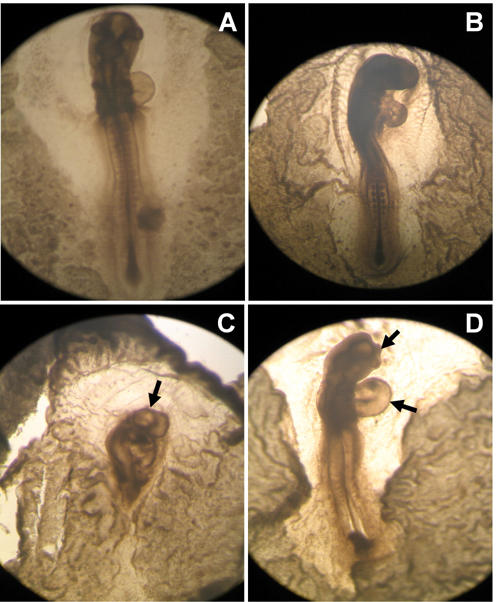
A: Untreated control embryo. B: Embryo treated with 0.9% saline indicating normal development. C: Embryo treated with 50 mM glucose exhibiting embryonic dys-morphogenesis. D: Embryo treated with 100 mM glucose exhibiting abnormal development of brain and heart.
Figure 4. Effects of glucose on chick embryos (HH 18-20).
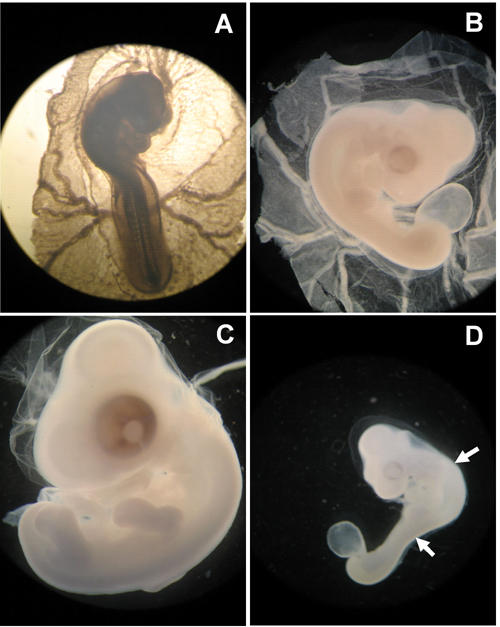
A: Untreated control embryo. B: Embryo treated with 0.9% saline developed normally. C: Embryo treated with 50 mM glucose showing macrosomia. D: Embryo treated with 100 mM glucose. Abnormal flexion and torsion are prominent.
Discussion
In the present investigation, we have developed a model of a shell-less chick embryo culture and evaluated its potential for detecting glucose-induced malformations. Since the existing techniques of shell-less chick embryo cultures do not permit observations of early stages of active organogenesis, we developed a simple method to serve this purpose. Our technique is beneficial compared to the previously developed techniques as it does not require any sophisticated equipment, media or sera [2-4]. It is inexpensive in terms of instrumentation and incubator space [5-7]. It permits studies in the early stages of development, allowing immediate observations of a large number of embryos at the same time. It has the advantage of providing easy access to the embryo and extra-embryonic membranes for the observation of morphogenesis and growth as well as for the application of different agents under study and inoculation of viruses in specific extra-embryonic membranes, such as the yolk sac, amnion and chorioallantoic membrane (Figure 1C). Previously developed shell-less culture systems made use of late-stage embryos, leading to a limited embryonic survival rate. This has proved to be a severe limitation for their use for experiments at early embryo stages [2]. The systems were expensive in terms of the laboratory ware and incubator space as well as the high level of skill and practice required. This has prevented their introduction at graduate and undergraduate levels. Against this background, our model could be used not only for research purposes but could also easily be introduced in school, college and university curricula for the teaching of embryology. Although our primary purpose was to study glucose-induced malformations we have used this model for a variety of other studies because of the high survival rate of the embryos even after the first week. For example, we have studied the onset and progression of angiogenesis and anti-angiogenesis by applying known and unknown angiogenic factors on chorioallantoic membrane using this model (data not shown). Thus our model may serve as an in-vitro model suitable for the fields of developmental biology, toxicology, teratology, virology and almost all aspects of biomedical research.
Glucose has been reported to be a teratogen for young vertebrate embryos [8, 23, 24]. However, there have been few studies on glucose-induced malformations observed at different stages of development using the chick embryo as a model. Our results demonstrate that glucose treatment has a lethal effect in the early stages of embryogenesis, which is concentration- and age-dependent. Thus, younger embryos are more frequently and more severely affected than older embryos, and a higher concentration of glucose led to a greater incidence of exencephaly at the ages tested. We compared the malformations obtained due to hyperglycemia alone against those obtained due to the combined effects of hyperglycemia and hyperosmolarity and did not find any significant difference. Therefore, the present study incorporates the data representing the combined effect of hyperglycemia and hyperosmolarity. These results are comparable to those reported for in vivo studies with induced diabetes in mice [25] and in vitro studies on mouse and rat embryos [9, 26].
We were also able to observe that the number of malformed embryos decreased when hyperglycemia was induced only at later stages of the embryonic development (HH 19 to HH 21). The ultimate fate of the embryos affected at these early stages is difficult to determine since cultures were maintained for only 24 h. However, the observed embryonic dysmorphogenesis and retarded growth in combination with CNS malformations suggest that these embryos may not have survived had they been similarly affected in vivo. Moreover, a large number of stillbirths occurred amongst the fetuses of diabetic mothers and this may also be related to early severe defects which are life-threatening [24]. Our observations indicate that hyperglycemia-induced malformations that appear after the closure of the neural tube (approximate HH stages 19-21) are not fatal. An example for these malformations is macrosomia, which is comparable to the incidence of large babies born by diabetic mothers [27].
In the present investigation, we have shown for the first time that hyperglycemic malformations in mammalian embryos can be demonstrated and examined in the developing chick embryo. Nevertheless, it is difficult to transfer the results obtained here using chick embryos in shell-less culture systems directly to the human fetus considering the metabolism, excretion and biochemical mechanisms of glucose utilization during embryonic life. However, it is interesting to note that similar results have been reported for the human fetus. The hyperglycemic state has been shown to be associated with increased risk for embryonic dysmorphogenesis [28]. Increased incidence of malformations in the offspring of diabetic mothers with elevated HbA1c levels during early human pregnancy [29] and similar observations in rodent diabetic pregnancies [30-32] support these findings. Furthermore, since the time required for the induction of malformations is very short (24 h) in chick embryos as compared to mammalian embryos, e.g. 10-12 days for mouse embryos [33] and 2-5 weeks in human embryos [34], the system is time-efficient. Since the use of chick embryos in stages of up to 10 days of incubation does not evoke severe ethical issues [35] its usage for the study of glucose-induced malformations during embryonic development would be an appropriate alternative to other animal experiments.
To assess and compare the effects of gestational diabetes on the embryos of different species, one must consider the different periods in developmental stages. In this regard, a 21-day incubation period of the chick is comparable to the 21-day gestation period of mice. The first 4-5 days of incubation in chicks correspond to 2-5 weeks of post-conceptional age in human embryos [36] and 12-15 days of rodent pregnancy [37]. The shell-less culture system for chick embryos reported here, therefore, may also be used to help improve understanding of the pathophysiology of gestational diabetes, although the technique has limitations as it is an ex-ovo system. Admittedly, gestational diabetes is a multifactorial process where the diabetic state is associated with several other parameters, such as pregnancy hormones, ketosis etc. Hence, the present shell-less culture system may only partly simulate the conditions of gestational diabetes.
In summary, it can be argued from the present results that the shell-less chick embryo culture system described offers great potential for various aspects of research into embryogenesis and glucose-induced malformations.
Acknowledgments
The authors wish to thank the Director of the NCCS for providing all the facilities to carry out this work. Thanks also to Drs. Sanjeev Galande, NCCS Pune, and Surendra Ghaskadbi, ARI Pune, for the photography of the shell-less chick embryo cultures and the embryonic malformations. The financial assistance provided by the UGC to teacher fellow S.D. is gratefully acknowledged.
References
- 1.Eriksson UJ. In: Sima AF, Shafrir E (Eds.). Chronic complications in diabetes. Animal models and chronic complications. Harwood; Amsterdam: 2000. Fetal malformations in diabetes. [Google Scholar]
- 2.Auerbach R, Kubai L, Knighton D, Folkman J. A simple procedure for the long- term cultivation of chick embryos. Dev Biol. 1974;41(2):391–394. doi: 10.1016/0012-1606(74)90316-9. [DOI] [PubMed] [Google Scholar]
- 3.Dunn BE, Fitzharris TP, Barnett BD. Effects of varying chamber construction and embryo pre-incubation age on survival and growth of chick embryos in shell-less culture. Anat Rec. 1981;199:33–43. doi: 10.1002/ar.1091990105. [DOI] [PubMed] [Google Scholar]
- 4.Perry MM. A complete culture system for the chick embryo. Nature. 1988;331:70–72. doi: 10.1038/331070a0. [DOI] [PubMed] [Google Scholar]
- 5.Auerbach R, Kubai L, Sidky Y. Angiogenesis induction by tumors, embryonic tissues and lymphocytes. Cancer Res. 1976;36:3435–3440. [PubMed] [Google Scholar]
- 6.Dunn BE, Boone MA. Growth and mineral content of cultured chick embryos. Poultry Sci. 1977;56:662–672. doi: 10.3382/ps.0560662. [DOI] [PubMed] [Google Scholar]
- 7.Shusei H, Hideo N. Establishment of a chick embryo shell-less culture system and its use to observe changes in behavior caused by nicotine and substances from cigarette smoke. Toxicology Letters. 2001;119:95–102. doi: 10.1016/s0378-4274(00)00300-3. [DOI] [PubMed] [Google Scholar]
- 8.Cockroft DL, Coppola PT. Teratogenic effects of excess glucose on head-fold rat embryos in culture. Teratology. 1977;16:141–146. doi: 10.1002/tera.1420160205. [DOI] [PubMed] [Google Scholar]
- 9.Phelan SA, Ito M, Loeken MR. Neural tube defects in embryos of diabetic mice: role of the Pax-3 gene and apoptosis. Diabetes. 1997;46:1189–1197. doi: 10.2337/diab.46.7.1189. [DOI] [PubMed] [Google Scholar]
- 10.Suzuki N, Svensson K, Eriksson UJ. High glucose concentrations inhibits migration of rat cranial neural crest cells in vitro. Diabetologia. 1996;39:401–411. doi: 10.1007/BF00400671. [DOI] [PubMed] [Google Scholar]
- 11.Hod M, Star S, Passonneau JV, Unterman TG, Freinkel N. Glucose induced dysmorphogenesis in cultured rat conceptus: prevention by supplementation with myoinositol. Israel J Med Sci. 1990;26:541–544. [PubMed] [Google Scholar]
- 12.Zusman I, Ornoy A, Yaffe P, Shafrir E. Effects of glucose and streptozotocin-diabetic and non-diabetic rats on the in vitro development of preimplantation mouse embryos. Israel J Med Sci. 1985;21:359–365. [PubMed] [Google Scholar]
- 13.Pederson J. Problems and management. Copenhagen; Munksgaard: 1977. The pregnant diabetic and her newborn. 2nd Ed. [Google Scholar]
- 14.Hod M, Star S, Passonneau JV, Unterman TG, Freinkel N. Effect of hyperglycemia on sorbitol and myoinositol content of cultured rat conceptus: failure of aldose reductase inhibitors to modify myoinositol depletion and dysmorphogenesis. Biochem Biophys Res Comm. 1986;140:974–980. doi: 10.1016/0006-291x(86)90731-x. [DOI] [PubMed] [Google Scholar]
- 15.Pedersen LM, Tygstrup I, Pedersen J. Congenital malformations in newborn infants of diabetic women. Lancet. 1964;1:1124–1126. doi: 10.1016/s0140-6736(64)91805-7. [DOI] [PubMed] [Google Scholar]
- 16.Eriksson UJ, Dahlstrom E, Larsson KS, Hellerstrom C. Increased incidence of congenital malformations in the offsprings of diabetic rats and their prevention by maternal insulin therapy. Diabetes. 1982;31:1–6. doi: 10.2337/diab.31.1.1. [DOI] [PubMed] [Google Scholar]
- 17.Solar NG, Walsh CH, Malnis JM. Congenital malformations in infants of diabetic mothers. Quart J Med. 1976;178:303–313. [PubMed] [Google Scholar]
- 18.Hughes AF, Freeman RB, Fadem T. The teratogenic effects of sugars on the chick embryo. J Embryol Exp Morphol. 1974;32:661–674. [PubMed] [Google Scholar]
- 19.Hamburger V, Hamilton HL. A series of normal stages in the development of chick embryos. J Morphol. 1951;88:49–92. [PubMed] [Google Scholar]
- 20.Hernandez-Sanchez C, Rubio E, Serna J, de la Rosa EJ, de Pablo F. Unprocessed proinsulin promotes cell survival during neurulation in the chick embryo. Diabetes. 2002;51(3):770–777. doi: 10.2337/diabetes.51.3.770. [DOI] [PubMed] [Google Scholar]
- 21.Torchinsky A, Brokhman I, Shepshelovich J, Orenstein H, Savion S, Zaslavsky Z, Koifman M, Dierenfeld H, Fein A, Toder V. Increased TNF-alpha expression in cultured mouse embryos exposed to teratogenic concentrations of glucose. Reproduction. 2003;125(4):527–534. doi: 10.1530/rep.0.1250527. [DOI] [PubMed] [Google Scholar]
- 22.Larger E, Marre M, Corvol P, Gasc JM. Hyperglycemia-induced defects in angiogenesis in the chicken chorioallantoic membrane model. Diabetes. 2004;53(3):752–761. doi: 10.2337/diabetes.53.3.752. [DOI] [PubMed] [Google Scholar]
- 23.Garnham EA, Beck F, Clarke CA, Stanisstreet M. Effects of glucose on rat embryos in culture. Diabetologia. 1983;25:291–295. doi: 10.1007/BF00279946. [DOI] [PubMed] [Google Scholar]
- 24.Sadler TW. Effects of maternal diabetes on early embryogenesis II: Hyperglycemia-induced exencephaly. Teratology. 1980;21:349–356. doi: 10.1002/tera.1420210311. [DOI] [PubMed] [Google Scholar]
- 25.Watanabe G, Ingalls TH. Congenital malformations in the offspring of alloxan diabetic mice. Diabetes. 1963;12:66–72. doi: 10.2337/diab.12.1.66. [DOI] [PubMed] [Google Scholar]
- 26.Sadler TW. Effects of maternal diabetes on early embryogenesis. I. The teratogenic potential of diabetic serum. Teratology. 1980;21:339–347. doi: 10.1002/tera.1420210310. [DOI] [PubMed] [Google Scholar]
- 27.Leipold H, Worda C, Gruber CJ, Kautzky-Willer A, Husslein PW, Bancher-Todesca D. Large-for-gestational-age newborns in women with insulin treated gestational diabetes under strict metabolic control. Wien Klin Wochenschr. 2005;117(15-16):521–525. doi: 10.1007/s00508-005-0404-1. [DOI] [PubMed] [Google Scholar]
- 28.Karlsson K, Kjellmer I. The outcome of diabetic pregnancies in relation to the mother’s blood sugar level. Am J Obstet Gynecol. 1972;112:213–230. doi: 10.1016/0002-9378(72)90118-4. [DOI] [PubMed] [Google Scholar]
- 29.Greene MF, Hare JW, Cloherty JP, Benacerraf BR, Soeldner JS. First-trimester hemoglobin A1 and risk for major malformation and spontaneous abortion in diabetic pregnancy. Teratology. 1989;39:225–231. doi: 10.1002/tera.1420390303. [DOI] [PubMed] [Google Scholar]
- 30.Baker L, Egler JM, Klein SM, Goldman AS. Meticulous control of diabetes during organogenesis prevents congenital lumbosacral defects in rats. Diabetes. 1981;30:955–959. doi: 10.2337/diab.30.11.955. [DOI] [PubMed] [Google Scholar]
- 31.Giavini E, Brocca ML, Prati M, Roversi GG, Vismara C. Effects of streptozotocin-induced diabetes on fetal development of rat. Teratology. 1986;34:81–88. doi: 10.1002/tera.1420340111. [DOI] [PubMed] [Google Scholar]
- 32.Styrud J, Thunberg L, Nybacka O, Eriksson UJ. Correlations between maternal metabolism and deranged development in the offsprings of normal and diabetic rats. Pediatr Res. 1995;37:343–353. doi: 10.1203/00006450-199503000-00015. [DOI] [PubMed] [Google Scholar]
- 33.Mills JL, Baker L, Goldman AS. Malformations in infants of diabetic mothers occur before the seventh gestational week. Implications for treatment. Diabetes. 1979;28(4):292–293. doi: 10.2337/diab.28.4.292. [DOI] [PubMed] [Google Scholar]
- 34.Eriksson UJ, Bone AJ, Turnbull DM, Baird JD. Timed irruption of insulin therapy in diabetic BB/E rat pregnancy: effect on maternal metabolism and fetal outcome. Acta Endocrinol. 1989;120(6):800–810. doi: 10.1530/acta.0.1200800. [DOI] [PubMed] [Google Scholar]
- 35.West DC, Thompson WD, Sells PG, Burbridge MF. In: Murry C (Ed). Angiogenesis protocols. Humana Press; Totowa, New Jersey: Angiogenesis assays using chick chorioallantoic membrane; p. 122. [Google Scholar]
- 36.Kucera P, Burnad MB. Routine teratogenicity test that uses chick embryos in vitro. Teratog Carcinog Mutagen. 1987;7(5):427–447. doi: 10.1002/tcm.1770070502. [DOI] [PubMed] [Google Scholar]
- 37.Eriksson RSM, Thunberg L, Eriksson UJ. Effects of interrupted insulin treatment on fetal outcome of pregnant diabetic rats. Diabetes. 1989;38:764–772. doi: 10.2337/diab.38.6.764. [DOI] [PubMed] [Google Scholar]


