Abstract
Comparison of the inflammatory response of Sigmodon hispidus cotton rats to pulmonary infection with wild-type 5 adenovirus (Ad5) or with a viral mutant, in which the early region 1B gene encoding a 55-kDa protein, Ad5dl110 (dl110), was deleted, indicated that the inflammation in animals infected with dl110 was markedly reduced compared with the inflammation in animals infected with wild-type Ad5, although both viruses replicated to the same extent. Comparable experiments done with C57BL/6 mice yielded identical results, even though only the early phase of gene expression essential for viral replication occurs in mice. Cytokine analysis of infected mouse lungs indicated that tumor necrosis factor-α and IL-6 were produced in relatively large quantities in wild-type Ad5-infected mice and at significantly lower levels in dl110-infected mice during the early stages of infection.
Keywords: cytokines
The unexpected but exciting discovery that pulmonary infection of cotton rats with type 5 adenovirus (Ad5) induced a pneumonia that was pathologically the same as that seen after a similar infection of humans (1) opened an unexplored field of the molecular pathogenesis of adenovirus pneumonia. Virologists had provided critical fundamental data on the structure of adenovirus virions, the means by which they entered cells, and the basic events in viral replication (2). However, nothing was known of the basic mechanism by which adenoviruses produce disease. Different types of adenoviruses induce different diseases including respiratory infections, ocular infections, gastrointestinal diseases, cystitis, and, less frequently, other types of disease (2), but the mechanisms responsible for induction of these diseases were unknown. The finding of the production of pneumonia in cotton rats has allowed research to begin in this field (1). Intranasal inoculation of Ad5 results in infection of epithelial cells lining the entire respiratory tract from the nasal cavities to the cells lining the pulmonary alveoli (3, 4). Major investigations have been done on the replication of Ad5 in alveolar epithelial cells. It is important to emphasize that the virus productively replicates in these cells, kills the cells, but does not lyse the infected cells in vivo or in vitro. Hence, it is incorrect to call the Ad5 infection a lytic infection as is done frequently in the literature.
It was a surprising discovery that only the early phase of Ad5 infection is required to produce the Ad5 pneumonia, i.e., the late-phase reactions for synthesis of the viral structural proteins and formation of virions were not essential to induce the pulmonary inflammation (5). This finding led to the demonstration that Ad5 could induce a similar pneumonia in mice (6). Previously, it had been shown that the early phase of viral replication occurred in infected cells in culture, but infectious progeny virus was not produced (7). Thus, it is now possible to determine whether cytokines are induced as a promoter of the early phases of Ad5 pulmonary infection (5).
One of the earliest investigations of molecular pathogenesis in cotton rats determined whether the so-called “nonessential” early region 3 (E3) was truly without important function, because it comprises 10% of the total genome but was not essential for viral replication. When cotton rats were intranasally infected with a mutant virus in which the entire E3 was deleted, the virus replicated like wild-type (Wt) Ad5, but it induced a pneumonia that was markedly greater than that induced in WtAd5-infected animals (8). It was then determined that the gene encoding a 19-kDa protein near the 5′ end of the E3 markedly suppressed cellular expression of the MHC class I antigen and therefore markedly reduced cytotoxic T cells from attacking infected cells (8). This phenomenon allowed the adenovirus to remain latent in infected cells, as was apparent from the initial isolation of an adenovirus (9). These findings led to an investigation as to whether any other early genes were critical to the induction of pulmonary inflammation.
Experiments were performed with a mutant in which the gene encoding the E1B 55-kDa protein had been deleted [designated Ad5dl110 (dl110)]. This protein had been shown to be essential to shut off host protein synthesis in productive infections (10, 11), which suggested that it may play a role in viral pathogenesis. It has also been shown that the E1B 55-kDa protein interacts with the host p53 protein (12); however, dl110 lacks the region of the 55-kDa protein that interacts with p53. It is the objective of the investigation described in this paper to present the results of experiments with cotton rats and C57BL/6N mice establishing an essential role for the E1B 55-kDa protein in the production of Ad5 pneumonia.
These studies are important, not only in understanding the molecular mechanism by which Ad5 produces pneumonia, but in opening opportunities to construct effective vectors for gene therapy. The original adenovirus vector employed for gene therapy used a mutant in which the entire E3, as well as E1A, had been deleted (13). This vector produced marked inflammation in recipients. The dl110 mutant might provide the basis for a suitable vector, because E4 can be deleted without producing a deleterious effect (H.S.G., L.L.M., and G.A.P., unpublished data). This deletion would provide space for the insertion of the gene to be expressed. Despite these advantages, adenovirus vectors have one damaging drawback in that they induce an immune response that does not permit the Ad vector to persist for a long period and that does not allow the Ad vector to be administered a second time.
Our goal is to describe the role of the E1B 55-kDa protein in the production of pulmonary inflammation and to show that the deletion of this gene produces a virus that replicates like the WtAd5 virus but with markedly reduced pulmonary inflammation, compared with WtAd5, in cotton rats and mice.
MATERIALS AND METHODS
Cells Lines and Viruses. Monolayer cultures of KB and 293 cell lines were grown in DMEM supplemented with 10% (vol/vol) FCS. WtAd5 was propagated in human KB cells grown in MEM supplemented with 10% (vol/vol) human serum. The dl110 mutant was propagated in monolayer cultures of 293 cells.
Indirect immunofluorescent assays on 293 cells were used to determine viral infectivity titers. Polyclonal rabbit antiserum prepared with intact, purified virus was employed for these assays.
Animals.
An inbred strain of C57BL/6N mice and an inbred strain of Sigmodon hispidus cotton rats, obtained from Virion Systems, were used for this investigation. Cotton rats aged 3–6 weeks were employed in these experiments, because previous experiments had shown that the age of the animals was not critical (5); however, animals of the same age were used in each experiment. Animals were killed for histologic examination, and their lungs were inflated with 10% (vol/vol) neutral buffered formalin, fixed in formalin for at least 24 h, and embedded in paraffin for sectioning. Histologic sections were stained with hematoxylin and eosin. Control lung sections were obtained from mock-infected animals. For cytokine assays, lungs from infected C57BL/6N mice were processed as described below.
Histologic Assays.
The degree of pulmonary inflammation in lungs from infected and control animals was determined without knowledge of the inoculum or day of death (i.e., scored “blind”). In each experiment, histologic slides were shuffled and then laid out beside the microscope with the slide identification placed such that it could not be observed during the scoring. The histologic lung sections were scored according to the number of lobes involved, the degree of bronchiolar epithelial cell damage, and the extent of alveolar, perivascular, and peribronchial inflammatory response, as previously described (5). In a number of experiments, two of the authors scored the slides, and the scores were compared and then averaged. For the majority of experiments, only one of the investigators (H.S.G.) scored the histologic slides.
Viral Assays.
Plaque assays in 293 cells were employed to quantify viral stocks for animal innocula. To determine viral titers of infected animals, immunofluorescent assays were used by employing polyclonal rabbit antiserum prepared with intact purified virus.
Cytokine Assays.
Lungs were removed rapidly from C57BL/6N mice and immediately frozen in liquid nitrogen. Frozen samples were tared and then homogenized at 4°C in five weight volumes of physiologic saline with a Polytron homogenizer at its top speed for 1 min. The homogenate was centrifuged at 12,000 × g for 20 min at 4°C, and the supernatant was removed by decanting. Tumor necrosis factor-α (TNF-α) bioactivity was determined immediately on the cell-free supernatants by using the WEHI 164 clone 13 assay as described (6). Recombinant TNF-α was used as a standard, and the sensitivity of the assay was approximately 50 pg/ml of lung homogenate. Murine IL-6 was determined by using commercially available ELISA reagents (Enogen, Boston, MA). The sensitivity of the assay was 12 pg/ml.
RESULTS
Inflammatory Responses in Cotton Rats Infected with WtAd5 or dl110.
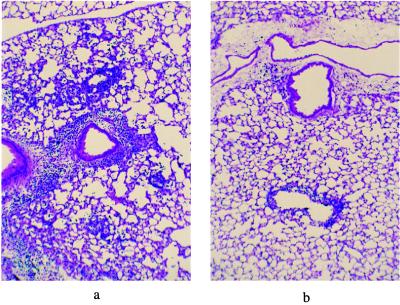
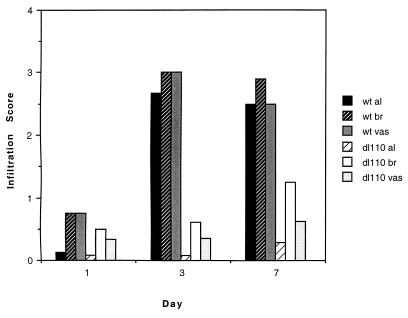
The pulmonary inflammatory responses in cotton rats infected with either WtAd5 or dl110 were markedly different: WtAd5 produced a severe pneumonia consisting of a lymphocytic infiltration in the alveoli and in the perivascular, peribronchial, and peribronchiolar regions. This pneumonia is very similar to the one that adenoviruses produce in humans (1, 3). In contrast, the dl110 mutant virus effected a minimal inflammatory response in all regions noted above. Fig. 1 illustrates the histologic inflammatory response to WtAd5 as compared with that induced by dl110. Fig. 2 presents the actual marked differences in the scores of all aspects of pulmonary inflammation in animals infected with WtAd5 or dl110.
Figure 1.
Histology (×50 magnification) of the inflammatory response of cotton rats infected with 109 plaque-forming units of WtAd5 virus (a) and with 109 plaque-forming units of dl110 mutant virus (b). The quantitative differences in the inflammatory responses to WtAd5 and dl110 are presented in Fig. 2.
Figure 2.
Scores of the inflammatory responses of cotton rats to WtAd5 virus and to the dl110 mutant. al, alveolar; br, bronchiolar; vas, perivascular.
Replication of WtAd5 and dl110 in Cotton Rat Lungs.
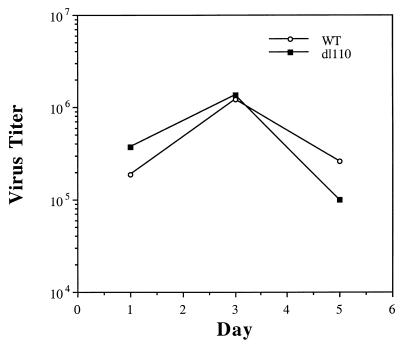
Despite the marked differences in the pulmonary responses to WtAd5 and dl110, the Wt virus and dl110 mutant multiplied to the same extent in the cotton rat lungs (Fig. 3). The Wt virus and dl110 mutant also multiplied to the same extent in 293 cell cultures (data not shown), although replication of dl110 was about 10-fold less in HeLa cells (10).
Figure 3.
Replication of the WtAd5 virus and dl110 mutant in cotton rat lungs after infection with 109 plaque-forming units of each virus.
Inflammatory Response of dl110 vs. WtAd5 in C57BL/6N Mice.
Because only the early events of adenovirus replication are required to produce the pulmonary inflammatory response (6), it was possible to examine the pneumonias that WtAd5 virus and dl110 produced in C57BL/6N mice, the mouse strain most sensitive to the adenovirus inflammatory response (6). In addition to examination of the inflammatory response, experiments in mice permitted examination of the cytokine response to each virus, which has proved to be an important aspect of the inflammatory response to adenoviruses (6). Plotting the alveolar, perivascular, and peribronchial pathological responses to WtAd5 virus and dl110 (Fig. 2) revealed a marked decrease in the inflammatory responses in the dl110-infected mice, similar to the responses produced after similar infection of cotton rats.
Cytokine Response.
After intranasal administration of both the Wt and dl110 adenoviruses, there was a monophasic appearance of TNF-α and IL-6 in the lungs of the infected mice (Table 1). The amount of IL-6 peaked on the first day after infection and declined thereafter, whereas TNF-α levels peaked on the second day. This early appearance of proinflammatory cytokines is consistent with earlier observations (5) and shows that, in early and transient innate immune responses, proinflammatory cytokines dominate. The concentration of TNF-α in the lungs of mice infected with the dl110 mutant was approximately one-half of the peak concentrations developed in mice infected with Wt virus (P < 0.05, as measured by one-way ANOVA), proportional to the reduced inflammatory response that occurred in these animals.
Table 1.
Cytokine levels in WtAD5- and dl110-infected C57BL/6 mice
| Cytokine | Virus | Cytokine levels at day after infection, units
|
||||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 5 | 7 | ||
| TNF-α | Wt-Ad5 | 213.3 | 1,227.0 | 500.0 | 50.0 | 50.0 |
| dl110 | 138.5 | 689.0 | 50.8 | 50.0 | 50.0 | |
| IL-6 | Wt-Ad5 | 1,885.3 | 1,268.3 | 887.0 | 123.5 | 128.0 |
| dl110 | 1,770.0 | 849.3 | 112.8 | 82.8 | 180.0 | |
DISCUSSION
The mutant virus constructed to eliminate the gene encoding the Ad5 E1B 55-kDa protein markedly reduced the pulmonary inflammatory response compared with that induced by the WtAd5 virus. It is noteworthy that, although the dl110 virus effected a markedly reduced inflammatory response to the viral infection, dl110 replicated to the same extent as the WtAd5 virus (Fig. 3). When C57BL/6N mice were infected with either WtAd5 virus or dl110, the alveolar, peribronchial, and perivascular inflammatory responses showed the same differences as those produced in cotton rats (Figs. 1 and 2), although, in contrast to the cotton rat, no infectious virus was produced in the mice. It is important to note, however, that dl110 induced a significantly lower proinflammatory cytokine response compared with the WtAd5 virus (Table 1).
Previous studies have suggested the potential mechanism(s) by which dl110 induces a markedly reduced inflammatory response as compared with the WtAd5 virus. It was previously shown that the Ad5 E1B 55-kDa protein travels into the nucleus and is critical to shutting off host protein synthesis after Ad5 infection (10, 11). Thus, if host protein synthesis is eradicated, the cells die and an inflammatory response ensues. If the E1B 55-kDa protein were not expressed, however, host protein synthesis would continue; the cells would not die; and, therefore, the inflammatory response would be markedly reduced. It should also be noted that the complete E1B 55-kDa protein (which is not present in dl110) interacts with the host cell p53 protein (12), but there is no evidence to indicate that this interaction would influence the inflammatory response to Ad5 infection. The reduced proinflammatory cytokine response is probably also an important factor in the reduced inflammatory response that dl110 induced compared with the response to WtAd5 infection.
The markedly reduced inflammatory response to dl110 infection suggests a potential use of this mutant as an adenovirus vector for gene therapy. As noted above, previous use of Ad5 as a vector in which the entire E3 was deleted resulted in an inflammatory response in the therapy recipients (13). It is important to note that an adenovirus vector in which the E3 is deleted should not be constructed. It is now possible to use the dl110 mutant as a basic vector in which the entire E4 is also deleted. Deletion of the entire E4 does not induce any inflammatory response, but it does allow a large space for the insertion of a desired gene for therapeutic purposes (H.S.G., L.L.M., and G.A.P., unpublished data). Such a vector should effectively express the desired gene product but not induce any undesirable inflammatory response. It must be noted, however, that such a vector could only be used once, and its expression would not be prolonged because of the immunological response to the adenovirus capsid protein, the hexon, that generates the immunologically effective neutralizing antibodies (14).
Acknowledgments
We thank Wai Wong, Miriam E. R. Darnell, and Susan Johnson for technical assistance and David D. Porter for assistance in the preparation of the histology figure. The initial stages of this research at the College of Physicians and Surgeons, Columbia University, was supported by National Institutes of Health Grant AI12052 (to H.S.G.).
ABBREVIATIONS
- Ad5
type 5 adenovirus
- Wt
wild type
- En
early region n
- dl110
Ad5 mutant dl110
- TNF-α
tumor necrosis factor-α
References
- 1.Pacini D L, Dubovi E J, Clyde W A., Jr J Infect Dis. 1984;150:92–97. doi: 10.1093/infdis/150.1.92. [DOI] [PubMed] [Google Scholar]
- 2.Ginsberg H S, editor. The Adenoviruses. New York: Plenum; 1984. [Google Scholar]
- 3.Ginsberg H S, Prince G A. In: Concepts in Viral Pathogenesis. Notkins A L, Oldstone M B A, editors. New York: Springer; 1989. pp. 275–281. [Google Scholar]
- 4.Prince G A, Porter D D, Jenson A B, Horswood R L, Chanock R M, Ginsberg H S. J Virol. 1993;67:101–111. doi: 10.1128/jvi.67.1.101-111.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ginsberg H S, Horswood R L, Chanock R M, Prince G A. Proc Natl Acad Sci USA. 1990;87:6191–6195. doi: 10.1073/pnas.87.16.6191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ginsberg H S, Moldawer L L, Sehgal P B, Redington M, Kilian P L, Chanock R M, Prince G A. Proc Natl Acad Sci USA. 1991;88:1651–1655. doi: 10.1073/pnas.88.5.1651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Eggerding F A, Pierce W C. Virology. 1986;148:97–113. doi: 10.1016/0042-6822(86)90406-x. [DOI] [PubMed] [Google Scholar]
- 8.Ginsberg H S, Lundholm-Beuchamp U, Horswood R L, Pernis B, Wold W S M, Chanock R M, Prince G A. Proc Natl Acad Sci USA. 1989;86:3823–3827. doi: 10.1073/pnas.86.10.3823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rowe W P, Huebner R J, Gilmore R J, Parrott R N, Ward T G. Proc Soc Exp Biol Med. 1953;84:570–573. doi: 10.3181/00379727-84-20714. [DOI] [PubMed] [Google Scholar]
- 10.Babiss L E, Ginsberg H S. J Virol. 1984;50:202–212. doi: 10.1128/jvi.50.1.202-212.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Babiss L E, Ginsberg H S, Darnell J E., Jr Mol Cell Biol. 1985;5:2552–2558. doi: 10.1128/mcb.5.10.2552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sarnow P, Ho T S, Williams J, Levine A J. Cell. 1982;28:367–396. doi: 10.1016/0092-8674(82)90356-7. [DOI] [PubMed] [Google Scholar]
- 13.Crystal R G, McElvaney N G, Rosenfeld M A, Chu C S, Mastrangeli A, Hay J G, Brody S L, Jaffe H A, Eissa N T, Danel C. Nat Genet. 1994;8:42–51. doi: 10.1038/ng0994-42. [DOI] [PubMed] [Google Scholar]
- 14.Wilcox W C, Ginsberg H S. Proc Soc Exp Biol Med. 1963;114:307. doi: 10.3181/00379727-114-28579. [DOI] [PubMed] [Google Scholar]