Abstract
Background
Psoriatic arthritis may progress to joint damage. Joint damage may be assessed clinically, by identifying deformed, fused, or flail joints, or radiologically, by recording erosions, joint space narrowing, ankylosis, lysis, or surgery. The relation between clinical and radiological damage is unclear.
Objective
To study the ordering of clinical and radiological damage detection, and the clinical features associated with the type of damage detected first.
Methods
The University of Toronto psoriatic arthritis database was used to relate clinical and radiological damage in the hand joints in 655 patients followed prospectively between 1978 and 2003. Generalised estimating equations were used to fit logistic regression models to identify factors that predict classification of damage by radiographic assessment first.
Results
The majority of the joints were not informative, as they either had evidence of damage by both methods at entry, or remained undamaged. Of the remainder, 81% of the joints showed radiological damage first and 19% had clinical damage first. Development of radiological damage first was related to previous detection of swollen joints, and was inversely related to duration of arthritis.
Conclusions
Radiological damage is often detected before clinical damage is observed. Clinical inflammation often precedes the detection of radiological damage.
Keywords: psoriatic arthritis, radiological damage, joint damage, prognosis
Psoriatic arthritis is as an inflammatory arthritis, usually seronegative (for rheumatoid factor), associated with psoriasis. It has been reported in 6–39% of patients with psoriasis.1,2,3,4,5 The initial description of psoriatic arthritis by Wright suggested that it was milder than rheumatoid arthritis.6 Subsequent studies suggested patients with psoriatic arthritis fared better than patients with rheumatoid arthritis.7 Psoriatic arthritis was thus considered a benign disease, with short lived synovitis that did not lead to residual damage in the majority of patients. This may partly be explained by our observation that patients with psoriatic arthritis have a lower frequency of fibromyalgia and a lower pain threshold in both actively inflamed joints and fibromyalgia tender points than patients with rheumatoid arthritis; thus both patients and physicians have underestimated the degree of inflammation.8 However, in the past 15 years it has become clear that psoriatic arthritis may be more severe than previously thought. Recent studies have shown that 67% of the patients seen in a psoriatic arthritis clinic have at least one erosion documented at presentation to the clinic.9,10 Indeed, of 129 patients identified as having psoriatic arthritis in an early arthritis clinic, 47% developed joint erosions by two years.11 Moreover, some 20% of the patients registered in a longitudinal psoriatic arthritis clinic had more than five deformed joints at presentation, and 11% had markedly reduced functional class.9
We have previously shown that, after 10 years of follow up, 55% of the patients with psoriatic arthritis developed five or more damaged joints.12 Others have also documented progression of deformity and radiological damage in psoriatic arthritis over time.13,14 The presence of five or more swollen joints at presentation to the clinic and a high level of drug use before presentation were found in one study to be predictors of progression of clinical damage, while a low erythrocyte sedimentation rate (ESR) was protective.15 Polyarticular presentation predicted progression of both clinical and radiological damage in another study.14 Indeed, actively inflamed joints at any visit were predictive of progression of clinical damage at subsequent visits, such that for each actively inflamed joint detected at one visit, there was a 4% increased risk of progression of damage in a subsequent visit.16 In addition, the HLA antigens HLA‐B27 (in the presence of HLA‐DR7), HLA‐B39, and HLA‐DQw3 (in the absence of HLA‐DR7) were identified as risk factors for progression of clinical damage, while HLA‐B22 was protective.17
In our previous studies we used clinical damage—determined by the presence of a limitation of range of movement of more than 20% of the range not related to the presence of joint effusion, joint deformities, subluxation, loosening, or ankylosis—as the outcome measure, as it was measured at every visit. However, the radiological damage has been considered a more traditional method of assessing damage in patients with arthritis. The relation between radiological damage (erosions, joint space narrowing, ankylosis, and joint lysis) and clinical damage (deformities, limitation of movement, ankylosis, and loosening of the joints) is unclear. Some patients appear to have evidence of clinical deformity without obvious changes on their radiographs, whereas others show erosive changes with joint space narrowing without obvious deformities. We therefore aimed to determine whether the detection of clinical damage preceded or followed radiological damage in our patients with psoriatic arthritis, and to identify disease related features that were associated with the development of clinical and radiological joint damage.
Methods
The database used was based on the cohort of patients registered in the Psoriatic Arthritis Clinic at the University of Toronto. This clinic registry began accruing patients in 1978 and is now the largest registry of patients with psoriatic arthritis with detailed prospective follow up. Upon entry to the University of Toronto psoriatic arthritis clinic, detailed demographic and historical data are recorded in a standardised form.9
For the present study we restricted eligibility to patients who were accrued between 1978 and 2003. There were 655 patients in this dataset. Patients are scheduled for semi‐annual clinical assessment (which include a clinical history, physician examination, and laboratory evaluation according to a standard protocol) and biannual radiographic assessment. Joints are classified on clinical examination as being normal, deformed, ankylosed, flail, or having been surgically replaced. Joints which show a reduction in movement by more than 20% of the range but have a balottable swelling suggesting that fluid may be contributing to the limitation of movement are not considered damaged, as once the inflammation resolves the range of movement may be restored. This definition was based on a workshop in which the rheumatologists of the University of Toronto Rheumatic Disease Unit participated in 1978, at the initiation of the psoriatic arthritis clinic. The assessment of clinically damage joints has been proved reliable both by physicians who trained at the University of Toronto psoriatic arthritis clinic18 and by physicians from across Canada who participated in an exercise to assess patients with psoriatic arthritis.19 Radiographic examination based on a modified Steinbrocker scoring system20 classifies joints into the following categories: normal (with possible soft tissue swelling); surface or pocket erosions; erosion and joint space narrowing; disorganisation (including total ankylosis, pencil in cup change, or total joint destruction); or as having required surgery. We define a joint as damaged clinically or radiologically if it is anything other than normal. In this analysis we considered only the joints of the hands (28 joints altogether, 14 in each hand: the metacarpophalangeal, posterior interphalangeal, and distal interphalangeal joints including thumbs and fingers). The assessments of radiological damage have proved reliable in our clinic.20
We examined all data relating to these joints in all patients over the period of observation in our analysis. The data were used to define, if possible, the method that first detected damage. This is primarily determined by clinic visits at which both clinical and radiological damage are assessed. As clinical damage is assessed at all clinic visits while x rays are taken biannually, only visits in which both clinical and radiological damage were assessed were considered in the analysis. When information on both radiological and clinical damage was available, then four scenarios were possible:
if radiological damage was first seen at a visit when clinical damage was not seen then it is known that radiological damage was detected first;
if radiological damage was not seen at a visit when x rays were taken but clinical damage was observed, then it is known that clinical damage was detected first;
if the first detection of both types of damage was at the same visit then detection was regarded as simultaneous;
if radiological damage was detected at a visit but clinical damage had been seen at a previous visit when no x rays were taken, then it was not possible to determine the order as radiological damage could have been present earlier.
If, in addition to these four scenarios, clinical damage was detected but no concurrent or subsequent radiological assessment was done, then the order of detection could not be determined.
Joints which were first classified as damaged by both methods are not informative, as it was not possible to determine which method had detected damage first. We therefore excluded such joints from the analysis, as well as joints for which the order of detection could not be determined. Joints which were not classified as damaged by either method continued to be tracked over eligible follow up visits. If at the end of the follow up a joint was not classified as damaged by any method then this joint was excluded from the analysis as it was not informative.
We were thus able to characterise each joint for each patient into one of the following categories: A, never detected as damaged by either method; B, first detected as damaged by clinical assessment; C, first detected as damaged by radiological assessment; D, first detected as damaged by both clinical and radiological assessments or impossible to determine.
The percentage of joints in each category was computed. Outcomes A and D represent concordance and B and C represent discordance. To help identify patient characteristics that predict characterisation of damage by radiographic assessment before clinical assessment, the discordant outcomes B and C are informative and the outcomes A and D lead to the joint being excluded from the analysis as uninformative. For each informative joint, a binary response was constructed which took the value 1 if a radiological assessment led to a classification of damage first (case C) and 0 if a clinical assessment led to the classification of damage first (case B).
As joints from the same patient may behave in a more similar fashion than joints from different patients, methods for dealing with cluster correlated binary data are required. We therefore used generalised estimating equations (GEE)21 to fit logistic regression models to identify factors that predict classification of damage by radiographic assessment first. An exchangeable working correlation structure between the cross sectional visit data was used. This working assumption appears sensible in the light of the irregular spacing of clinic visits, but the use of GEE methodology provides an analysis that is robust to departures from this assumption.
In addition to the primary analysis of cross sectional visit data, a descriptive longitudinal analysis of time to damage and the gap between damage detection by the different methods was done using Kaplan–Meier methodology.
Results
Table 1 provides a summary of the characteristics of the 655 patients included in this analysis.
Table 1 Patient characteristics at clinic entry.
| No of patients in the analysis | 655 | |
| Mean age at clinic entry (range) | 43.5 (15.5 to 87.5) | |
| Mean age at onset of psoriasis (range) | 28.9 (0 to 84) | |
| Mean age at onset of arthritis (range) | 35.9 (9 to 86) | |
| Mean duration of arthritis (SEM) | 7.5 (2.9) | |
| Mean duration of psoriasis (SEM) | 14.6 (3.5) | |
| ⩾40 years old at onset of psoriasis | 150 (22.9%) | |
| ⩾60 years old at onset of psoriatic arthritis | 34 (5.2%) | |
| Family history of psoriasis | 264 (40.5%) | |
| Family history of psoriatic arthritis | 65 (10%) | |
| Actively inflamed joints | ||
| No with no actively inflamed joints | 43 (6.6%) | |
| No with 1–5 actively inflamed joints | 181 (27.6%) | |
| No with ⩾5 actively inflamed joints | 431 (65.8%) | |
| Deformed Joints | ||
| No with no deformed joints | 402 (61.4%) | |
| No with 1–5 deformed joints | 138 (21.1%) | |
| No with ⩾5 deformed joints | 115 (17.5%) | |
| Mean No of active joints (SEM) | 10.1 (3.1) | |
| Mean No of deformed joints (SEM) | 3.2 (2.8) |
Table 2 gives the number of patients with various patterns of observed damage at clinic entry.
Table 2 Patterns of damage at clinic entry for 655 patients.
| No damage | 224 (34.2%) | |
| Clinical damage only | 40 (6.1%) | |
| Radiological damage only | 220 (33.6%) | |
| Both radiological and clinical damage observed in all damaged joints | 16 (2.4%) | |
| Clinical damage only or both observed in all damaged joints | 18 (2.8%) | |
| Radiological damage only or both observed in all damaged joints | 58 (8.9%) | |
| Clinical damage only or radiological damage only in all damaged joints | 17 (2.6%) | |
| All patterns of damage observed | 62 (9.5%) |
The numbers of joints in categories A, B, C and D, as defined in Methods, were 14 298, 565, 2370, and 1107, respectively. Of the joints that became damaged, simultaneous detection by clinical and radiological means could not be ruled out for 27% (1107 of 4042). Of the remainder, 81% (2370 of 2935) were seen first on x rays and 19% were first detected clinically.
Of the 2370 joints detected first radiologically, 1447 showed surface erosions only, 716 showed erosions and joint space narrowing, and 207 were in other categories. For the 565 joints detected first clinically, 492 were deformed, 45 ankylosed, and 28 in the other categories.
Table 3 presents the results of univariate GEE logistic regression analyses of the relations between the factors listed in the methods and the probability of having radiological damage detected first. The variable recent history of effusions is coded 1 if a joint was swollen on the two previous clinic visits, or on the previous visit if data from clinic visit 2 were being used. Observations from clinic visit 1 were excluded for any model involving this variable. The drug treatment level was determined by the highest level of treatment the patient was receiving at the time the joint under consideration was detected as damaged (the order is: no drug treatment→non‐steroidal anti‐inflammatory drugs→disease modifying antirheumatic drugs→intra‐articular steroids).
Table 3 Single factor analysis.
| Description | OR | p Value (Z) | |
|---|---|---|---|
| Age at first clinic visit (y) | 1.00 | 0.64 | |
| Duration of psoriasis at clinic entry (y) | 0.99 | 0.14 | |
| Duration of psoriatic arthritis at clinic entry (years) | 0.98 | 0.035 | |
| Family history of psoriasis (yes/no) | 0.82 | 0.25 | |
| Family history of psoriatic arthritis (yes/no) | 1.30 | 0.39 | |
| ESR at clinic entry (abnormal/normal) | 0.80 | 0.27 | |
| Rheumatoid factor at clinic entry (present/absent) | 1.12 | 0.66 | |
| Drug treatment | |||
| NSAIDs | 1 | 0.66* | |
| DMARDs | 0.90 | ||
| IAS | 0.67 | ||
| None | 0.84 | ||
| Antinuclear antibody (present/absent) | 0.85 | 0.41 | |
| Age at onset of psoriasis (y) | 1.00 | 0.43 | |
| Age at onset of arthritis (y) | 1.01 | 0.34 | |
| History of activity before damage (present/absent) | 0.92 | 0.50 | |
| History of effusion before damage (present/absent) | 1.12 | 0.43 | |
| Recent history of effusion (present/absent) | 1.73 | 0.002 | |
| Current effused joint (yes/no) | 1.24 | 0.16 | |
| Duration of psoriasis at time of damage (y) | 0.99 | 0.03 | |
| Duration of psoriatic arthritis at time of damage (y) | 0.98 | 0.01 | |
| ESR at time of damage (abnormal/normal) | 0.88 | 0.55 |
*Likelihood ratio test.
ESR, erythrocyte sedimentation rate; IAS, intra‐articular steroids; DMARD, disease modifying antirheumatic drug; NSAID, non‐steroidal anti‐inflammatory drug; OR, odds ratio; y, years.
Only four variables achieved significance levels below 0.05. These were arthritis duration at time of clinic entry (p = 0.04), arthritis duration at time of damage detection (p = 0.01), duration of psoriasis at time of damage detection (p = 0.03), and recent history of effusions (p = 0.002).
When the four variables identified in the univariate analyses were considered in a multivariate model, only arthritis duration at clinic entry (OR = 0.97/year, p = 0.01) and recent history of effusions (OR = 1.72, p = 0.002) retained significance. In addition, there was some evidence (p = 0.04) of an interaction between swelling at the time of damage detection and recent history of effusions defined as swelling in the previous two clinic visits. The estimated odds ratios for recent effusion history from a model including this interaction were 1.39 with no current swelling and 3.34 with current swelling.
No qualitative changes in these results were observed when analysis was restricted to patients with arthritis duration at clinic visit of less than two years, or when restricted to patients with no damage at first clinic visit. There was also no evidence that the type of damage detected in a patient's joints at the first visit influenced the pattern of subsequent damage development.
A descriptive analysis was also undertaken of the gap between radiological and clinical damage detection at the joint level. No adjustment for the correlation between joints in the same patient was made as no significance tests are presented. The data for this analysis are necessarily restricted to joints that had no damage detected by either method on clinic entry, as otherwise the relevant timings cannot be determined.
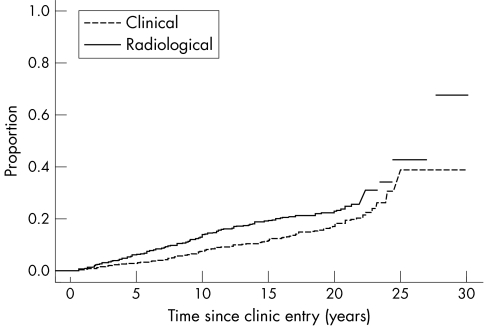
Figure 1 presents two curves for the time to first damage in individual joints, detected radiologically and detected clinically. The time scale is the time since clinic entry in years. It can be seen that although radiologically detected damage does occur earlier, the curves track fairly closely together. These estimated curves take account of the interval censoring introduced by observations only being made at clinic visits for clinical damage and biannual clinic visits for radiological damage.
Figure 1 Progression of joint damage over time (in years) in a psoriatic arthritis clinic.
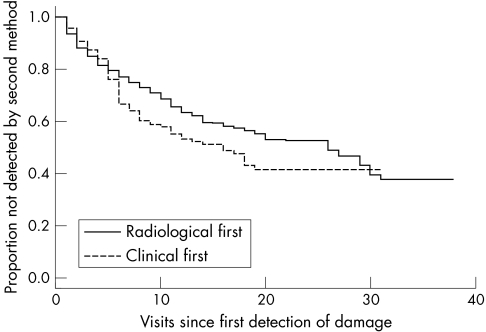
Figure 2 presents data on the number of subsequent visits to a joint being detected by clinical or radiological assessment, given that it was detected first by the other method. Long gaps can occur and slightly greater delays occur for clinical following radiological detection compared with radiological following clinical detection.
Figure 2 Survival of damage‐free joints over time (in visits) in a psoriatic arthritis clinic.
Discussion
The evaluation of patients with psoriatic arthritis includes assessment of joint inflammation—namely the number of tender and swollen joints—and the assessment of joint damage, comprising both clinical and radiological damage.22 The clinical assessment of actively inflamed and damaged joints has been shown to be reliable in our own clinic, as well as among Canadian rheumatologists.18,19 Clinical damage can be assessed at each clinical encounter with the patient, whereas radiological damage requires the patient to undergo a series of radiographs. The relation between clinical and radiological damage, and the role of joint inflammation in the development of damage, has not been addressed previously. We aimed to study clinical and radiological damage in patients with psoriatic arthritis followed in a longitudinal clinic, and to relate the means of detection of damage to disease related features. We found that in 81% of the informative joints damage was first detected radiologically, whereas in 19% of the joints damage was first detected clinically.
However, patients are often reluctant to undergo radiographic assessment because of fear of excessive radiation. Thus it is important to determine whether information can be gained from the assessment of clinical damage. Considering all categories of clinical and radiological damage, clinical damage was detected at the same time or before radiological damage in 41% of the cases ((565+1107)/4042). It is possible that we have underestimated clinical damage in some patients as we do not assign damage to a joint that is clinically swollen. This was done on purpose as we have seen situations in which patients appeared to have a flexion contracture associated with joint swelling which disappeared once the joint was injected with corticosteroids and the inflammation resolved. We did not want to assign a joint to a damage state erroneously as the underlying assumption is that damage does not recover. If we underestimated the number of joints with damage, there might be an even greater correlation between the development of clinical and radiological damage.
This study shows that, based on informative joints, radiographic detection occurred first in the majority of patients, and that the presence of effusions is associated with the development of radiological damage in these joints. It should be noted, however, that the detection of clinical damage followed closely. As the evaluation of clinical damage can be carried out at each patient encounter and does not require further effort on the part of the patient, it may be a worthwhile tool both in the management of patients with psoriatic arthritis in the clinic and as an outcome measure in clinical trials.
Acknowledgements
The University of Toronto Psoriatic Arthritis Program is supported by the Krembil Foundation.
References
- 1.Wright V, Moll J M H. Psoriatic arthritis. In: Seronegative polyarthritis. Amsterdam: North Holland Publishing Co, 1976169–223.
- 2.Gladman D D, Rahman P. Psoriatic arthritis. In: Ruddy S, Harris ED, Sledge CB, Budd RC, Sergent JS, editors. Textbook of rheumatology. 5th ed. Philadelphia, WB Saunders Co 20011071–1079.
- 3.Shbeeb M, Uramoto K M, Gibson L E, O'Fallon W M, Gabriel S E. The epidemiology of psoriatic arthritis in Olmsted County, Minnesota, USA, 1982–1991. J Rheumatol 2000271247–1250. [PubMed] [Google Scholar]
- 4.Zachariae H. Prevalence of joint disease in patients with psoriasis: implications for therapy. Am J Clin Dermatol 20034441–447. [DOI] [PubMed] [Google Scholar]
- 5.Green L, Meyers O L, Gordon W, Briggs B. Arthritis in psoriasis. Ann Rheum Dis 198140366–369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wright V. Psoriatic arthritis: a comparative study of rheumatoid arthritis and arthritis associated with psoriasis. Ann Rheum Dis 196120123–131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Coulton B L, Thomson K, Symmons D P, Popert A J. Outcome in patients hospitalized for psoriatic arthritis. Clin Rheumatol 19898261–265. [DOI] [PubMed] [Google Scholar]
- 8.Buskila D, Langevitz P, Gladman D D, Urowitz S, Smythe H A. Patients with rheumatoid arthritis are more tender than those with psoriatic arthritis. J Rheumatol 1992191115–1119. [PubMed] [Google Scholar]
- 9.Gladman D D, Shuckett R, Russell M L, Thorne J C, Schachter R K. Psoriatic arthritis (PSA) – an analysis of 220 patients. QJM 198762127–141. [PubMed] [Google Scholar]
- 10.Torre Alonso J C, Rodrigues Perez A, Arribas Castrillom J M, Ballina Garcia J, Riestra Noriega J L, Lopez Larrea C. Psoriatic arthritis (PA): a clinical immunologic and radiological study. Br J Rheumatol 199130245–250. [DOI] [PubMed] [Google Scholar]
- 11.Kane D, Stafford L, Bresnihan B, Fitzgerald O. A prospective, clinical and radiological study of early psoriatic arthritis: an early synovitis clinic experience. Rheumatology 2003421460–1468. [DOI] [PubMed] [Google Scholar]
- 12.Gladman D D. The natural history of psoriatic arthritis. Baillieres Clin Rheumatol 19948379–394. [DOI] [PubMed] [Google Scholar]
- 13.McHugh N J, Balachrishnan C, Jones S M. Progression of peripheral joint disease in psoriatic arthritis: a 5‐yr prospective study. Rheumatology (Oxford) 200342778–783. [DOI] [PubMed] [Google Scholar]
- 14.Queiro‐Silva R, Torre‐Alonso J C, Tinture‐Eguren T, Lopez‐Lagunas I. A polyarticular onset predicts erosive and deforming disease in psoriatic arthritis. Ann Rheum Dis 20036268–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gladman D D, Farewell V T, Nadeau C. Clinical indicators of progression in psoriatic arthritis (PSA): multivariate relative risk model. J Rheumatol 199522675–679. [PubMed] [Google Scholar]
- 16.Gladman D D, Farewell V T. Progression in psoriatic arthritis: role of time varying clinical indicators. J Rheumatol 1999262409–2413. [PubMed] [Google Scholar]
- 17.Gladman D D, Farewell V T, Kopciuk K, Cook R J. HLA markers and progression in psoriatic arthritis. J Rheumatol 199825730–733. [PubMed] [Google Scholar]
- 18.Gladman D D, Farewell V, Buskila D, Goodman R, Hamilton L, Langevitz P.et al Reliability of measurements of active and damaged joints in psoriatic arthritis. J Rheumatol 19901762–64. [PubMed] [Google Scholar]
- 19.Gladman D D, Cook R J, Schentag C, Feletar M, Inman R I, Hitchon C.et al The clinical assessment of patients with psoriatic arthritis: results of a validation study of the SpondyloArthritis Research Consortium of Canada (SPARCC). J Rheumatol 2004311126–1131. [PubMed] [Google Scholar]
- 20.Rahman P, Gladman D D, Cook R J, Zhou Y, Young G, Salonen D. Radiological assessment in psoriatic arthritis. Br J Rheumatol 199837760–765. [DOI] [PubMed] [Google Scholar]
- 21.Zeger S L, Liang K Y. Longitudinal data analysis for discrete and continuous outcomes. Biometrics 198642121–130. [PubMed] [Google Scholar]
- 22.Gladman D D, Helliwell P, Mease P J, Nash P, Ritchlin C, Taylor W. Assessment of patients with psoriatic arthritis. A review of currently available measures. Arthritis Rheum 20045024–35. [DOI] [PubMed] [Google Scholar]