Abstract
Objective
To examine whether smoking is a risk factor for rheumatoid nodules in early rheumatoid arthritis, and if so to determine the quantitative effect of smoking.
Methods
From a cohort (n = 1589) in a structured programme for follow up of newly diagnosed cases of rheumatoid arthritis (symptoms of swollen joints ⩽12 months), 112 individuals with rheumatoid nodules at inclusion were identified. Nodular patients were each compared with two age and sex matched controls without nodules from the same cohort. A detailed self administered tobacco use questionnaire was answered by 210 patients (63%).
Results
Seventy patients were current smokers, 71 former smokers, and 69 had never smoked. Current smoking and former smoking were more common in patients with rheumatoid nodules compared with controls (86% v 59%) in both sexes. Positive rheumatoid factor (RF) was found more often among cases with nodules than controls (78% v 64%). Using detailed information from the questionnaires with conditional logistic regression analyses, ever having smoked was associated with an increased risk of the presence of rheumatoid nodules (odds ratio (OR) = 7.3 (95% confidence interval, 2.3 to 23.6); p = 0.001). The risk of having nodules was not obviously dose dependent when smoking duration as well as smoking amount were examined. A stratified analysis showed that only RF positive smokers had an increased risk of rheumatoid nodules. Smoking was associated with rheumatoid nodules among both men (p = 0.006) and women (p = 0.001). Tobacco use other than smoking (n = 31) was not associated with an increased risk of nodules (OR = 0.8 (0.2 to 3.4); p = 0.813).
Conclusions
There is a strong association between smoking and rheumatoid nodules in early seropositive rheumatoid arthritis.
Keywords: rheumatoid arthritis, rheumatoid nodules, smoking
Rheumatoid arthritis is the most common inflammatory arthritis, affecting 0.5–1.0% of the population.1,2 Suggested aetiological and disease modifying factors include genetic markers and environmental exposure.3,4 The low concordance for rheumatoid arthritis in monozygotic twins highlights the importance of environmental factors.5 Epidemiological studies have suggested that smoking is an independent risk factor for rheumatoid arthritis, particularly rheumatoid factor (RF) positive disease.6,7 A recent survey found that smoking had a particular impact on the risk of rheumatoid arthritis in individuals carrying the rheumatoid arthritis associated HLA‐DRB1 genotype.8 Smoking also seems to influence the disease outcome.9,10 It is known that smoking influences the immune system, but its overall effect is not fully understood. Studies of rheumatoid patients have shown that smokers are significantly more likely to be RF positive.11
The development of subcutaneous nodules is the most common inflammatory extra‐articular manifestation in rheumatoid arthritis. With long term follow up, 30–40% of all patients with rheumatoid arthritis have been noted to develop nodules at any time in well defined samples.9,12,13,14 Rheumatoid nodules are often disturbing for the patients because they are painful, act as mechanical obstacles, and are sites of infection. Rheumatoid nodules have also been shown to be a marker for more severe disease, but often appear later in the disease. Studies have demonstrated a two‐ to fourfold increase in mortality in patients with extra‐articular manifestations, including rheumatoid nodules.14,15,16 The early presence of rheumatoid nodules has been suggested to be a predictor of severe extra‐articular manifestations.15 Previous studies have shown that smokers, especially those who have smoked for a long time, are more likely to develop rheumatoid nodules.9,12,17,18 Nodules are found predominantly in seropositive patients, but it has also been shown that current or previous smoking is associated with the presence of nodules even after controlling for RF positivity.12 Unlike the production of RF, the development of subcutaneous nodules does not occur in healthy individuals who smoke.11
Our aims in this study were to investigate the frequency of rheumatoid nodules in early rheumatoid arthritis and their relation to smoking. We also studied the quantitative effect of smoking and smoking length on the development of nodules, and the effect of other types of tobacco use.
Methods
The patients were recruited from the BARFOT register, which is a structured programme for follow up of newly diagnosed cases of rheumatoid arthritis.19,20 The BARFOT register (Better AntiRheumatic FarmacOTherapy) was established in 1992. Of the six participating outpatient rheumatology clinics, five are located in the south and west of Sweden (at the general hospitals in Helsingborg, Kalmar, Kristianstad, Mölndal, and the Spenshult Hospital for Rheumatic Diseases) and one clinic in the southern part of the Stockholm area (at Huddinge University Hospital). General practitioners in these areas are strongly recommended to refer patients with definite or suspected rheumatoid arthritis for specialised rheumatology attention early in the disease.
All patients are assessed by a rheumatologist. At the first visit the patient is asked about the duration of symptoms. If the symptoms of arthritis (swollen joints) have lasted for ⩽12 months the patient can be included in the BARFOT register. The patient's answer is documented in the register by the rheumatologist. A detailed history is obtained using a structured questionnaire, including smoking history (smoker, ex‐smoker, never smoked) and details of formal education. Joints (28 joint index) are examined for the presence of soft tissue swelling and tenderness. The presence of rheumatoid nodules is recorded, and patients complete the Swedish validated version of the Health Assessment Questionnaire (HAQ),21 which is scored from 0–3. Patients are asked to provide a blood sample and RF is measured at the local laboratory. The disease activity score (DAS28)22 is calculated and an x ray examination of the hands and feet is done.
In order to be included, the patients in the register also must meet at least four of seven items from the 1987 American College of Rheumatology (ACR) classification criteria for rheumatoid arthritis.23 The register covers all adults (ages ⩾16 years) who are registered with a rheumatologist in one of the six regions. This covers a population of approximately 1.5 million, predominantly of white European origin.
The study was approved by the research ethics committee of Lund University, and was carried out according to the Helsinki Declaration.
Cases and controls
The study design was a nested case–control investigation. From a cohort (n = 1589) in the BARFOT register, all patients with rheumatoid nodules at inclusion (n = 112; 7%) were consecutively enrolled between 1992 and 2001. Two controls without rheumatoid nodules at inclusion were selected from the same cohort. The controls (n = 224) were matched to the cases by sex, age at inclusion, disease duration, and when possible by geographical region.
Smoking history
As part of the above mentioned inclusion procedure, information about present and previous smoking was recorded. To obtain more detailed information on smoking exposure and other factors related to the extra‐articular manifestations of rheumatoid arthritis, an invitation was mailed to the 336 patients (174 men and 162 women), of whom 240 responded (71%) after two reminders. In all, 238 agreed to take part in the study and two refused (both with rheumatoid nodules). Sixty eight did not answer the letter (17 with rheumatoid nodules and 51 controls), three had moved abroad (all with rheumatoid nodules), and 25 had died (14 with rheumatoid nodules and 11 controls). The baseline data for non‐participants were not different from those of participants with one exception—those who had died were more often men and older at disease onset (table 1).
Table 1 Patient characteristics in the whole cohort (non‐participants and participants) and in the extensive smoking questionnaire group.
| All (n = 336) | Dead at the time of the questionnaire (n = 25; 7%) | Non‐participants* (n = 73; 22%) | Participants (n = 238; 71%) | Smoking questionnaire responders (n = 210; 63%) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Cases (RN) | 112 | 14 (12%) | 22 (20%) | 76 (68%) | 64 (57%) | |||||
| Controls | 224 | 11 (5%) | 51 (23%) | 162 (72%) | 146 (65%) | |||||
| Men | 174 (52%) | 18 (72%) | 37 (51%) | 119 (50%) | 108 (51%) | |||||
| Women | 162 (48%) | 7 (28%) | 36 (49%) | 119 (50%) | 102 (49%) | |||||
| Age (years) | 56 (14) | 67 (13) | 55 (17) | 56 (13) | 56 (12) | |||||
| RA duration (months) | 6.3 (3.4) | 5.2 (2.8) | 6.1 (3.6) | 6.5 (3.4) | 6.6 (3.3) | |||||
| DAS28 | 5.18 (1.49) | 4.99 (1.04) | 5.14 (1.15) | 5.15 (1.35) | 5.18 (1.34) | |||||
| HAQ | 0.97 (0.64) | 1.19 (0.74) | 1.01 (0.59) | 0.94 (0.64) | 0.94 (0.62) | |||||
| RF+ | 220 (65%) | 14 (56%) | 45 (62%) | 161 (68%) | 143 (68%) | |||||
| Smoking data | ||||||||||
| Ever smoked | 222 (66%) | 18 (72%) | 43 (59%) | 161 (68%) | 141 (67%) | |||||
| Current smokers | 118 (35%) | 10 (40%) | 25 (34%) | 83 (35%) | 70 (33%) | |||||
| Former smokers | 104 (31%) | 8 (32%) | 18 (25%) | 78 (33%) | 71 (34%) | |||||
| Never smoked | 69 (21%) | 0 | 0 | 69 (29%) | 69 (33%) | |||||
| Missing data | 45 (13%) | 7 (28%) | 30 (41%) | 8 (3%) | 0 |
Values are mean (SD) or n (%).
*Includes patients who moved abroad, n = 3, and who refused to participate, n = 2
DAS28, 28 joint disease activity score; HAQ, Health Assessment Questionnaire; RA, rheumatoid arthritis; RF+, rheumatoid factor positive; RN, rheumatoid nodules.
Questionnaire for detailed smoking history
At the baseline assessment, patients were asked whether they smoked currently or in the past or if they had never smoked, but there was no information on smoking years, smoking amount, or type of tobacco used. Thus a questionnaire24 was mailed to the 238 patients who agreed to take part in the study, in which they were asked for details on tobacco use.
The patients were asked to return their answers in a prepaid envelope. Completed questionnaires were obtained from 210 patients (63%), 64 cases with rheumatoid nodules and 146 controls, after two reminders.
Data on tobacco history included type of tobacco use (cigarettes, cigars, pipe, snuff) and for current or former smokers, years of smoking before the onset of rheumatoid arthritis. Patients who had stopped smoking within 12 months before inclusion were classified as smokers and those who had stopped smoking one year before inclusion as former smokers. Subjects who had ever smoked cigarettes on a regular basis (more than three cigarettes a week) were asked about the number of cigarettes or packs per day or week. Cigarette consumption was quantified as total packs (one pack = 20 cigarettes) and pack years. One pack year is equivalent to 20 cigarettes smoked a day for one year. One patient smoked cigars as well as cigarettes and was analysed as a cigarette smoker. Four patients smoked a pipe and cigarettes and were analysed as cigarette smokers. Ten patients were exclusively pipe smokers and were only analysed for smoking years, not tobacco amount. Thirty one patients used snuff (25 current users and six former users).
Smoking status was classified using three methods: (1) never smoked/ex‐smoker/current smoker; (2) never smoked and categories of number of years smoked, divided into tertiles (<26 years, 26–35 years, and >35 years); (3) never smoked and tertiles of number of cigarette packs (<5001 packs/13.7 pack years, 5001–9500 packs/13.7–26.0 pack years, and >9500 packs/26.0 pack years).
Statistical analysis
Data were analysed using the SPSS software (SPSS 11.1). Odds ratios (OR) with 95% confidence intervals (CI) were calculated using conditional logistic regression models, taking into account the matched design of the study in all analyses except for the stratified analyses by RF status. In these latter analyses, ordinary logistic regression was used. When calculating the odds ratios, each tertile of ever smokers was compared with never smokers as a reference. Formal education level was defined as low (⩽12 years of education) or high (>12 years of education), and used as a covariate in the multivariate analysis.
Results
In all 336 patients were studied, with 210 responders to the questionnaire (table 1).
Among the responders 70 were current smokers, 71 former smokers, and 69 had never smoked (table 1). Among both cases and controls, women had more often never smoked. Age did not differ between responders and non‐responders (except for the 25 patients who died), or between cases and controls, and was the same as in the whole BARFOT material. Further comparison of responders and non‐responders showed only minor differences, with one exception—none of the non‐responders was identified as a never smoker at inclusion in the BARFOT register. Some patients with tobacco use other than cigarettes were identified: one cigar smoker, 14 pipe smokers, and 31 who used snuff tobacco.
When the patients were subdivided as cases and controls and according to sex (table 2), the frequency of current and former smokers was higher in patients with rheumatoid nodules than in the controls (86% v 59%) in both sexes. Positive RF was found more often in rheumatoid nodules cases than in the controls (78% v 64%). Details on the smoking history of cases and controls are given in table3.
Table 2 Demographic, disease activity score, health assessment questionnaire score, smoking, and rheumatoid factor data on all patients and controls who completed the smoking questionnaire.
| Cases (RN) | Controls | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| All (n = 64) | Male (n = 34) | Female (n = 30) | All (n = 146) | Male (n = 74) | Female (n = 72) | |||||||
| Ever smoked (n = 141) | 55 (86%) | 31 (91%) | 24 (80%) | 86 (59%) | 51 (69%) | 35 (49%) | ||||||
| Smoker (n = 70) | 25 | 13 | 12 | 45 | 21 | 24 | ||||||
| Former smoker (n = 71) | 30 | 18 | 12 | 41 | 30 | 11 | ||||||
| Never smoked (n = 69) | 9 (14%) | 3 (9%) | 6 (20%) | 60 (41%) | 23 (31%) | 37 (51%) | ||||||
| Age (years) | 56 (12) | 58 (10) | 55 (12) | 56 (12) | 56 (12) | 56 (13) | ||||||
| RA duration (months) | 6.8 (3.3) | 6.5 (3.4) | 7.2 (3.3) | 6.4 (3.0) | 6.0 (2.9) | 6.7 (3.2) | ||||||
| DAS28 | 5.44 (1.26) | 5.16 (1.24) | 5.74 (1.23) | 5.06 (1.37) | 4.87 (1.55) | 5.26 (1.11) | ||||||
| HAQ | 1.06 (0.63) | 0.98 (0.56) | 1.18 (0.68) | 0.87 (0.60) | 0.68 (0.52) | 1.09 (0.61) | ||||||
| RF+ | 50 (78%) | 26 (76%) | 24 (80%) | 93 (64%) | 50 (67%) | 43 (60%) | ||||||
Values are n (%) or mean (SD).
DAS28, 28 joint disease activity score; HAQ, Health Assessment Questionnaire; RA, rheumatoid arthritis; RF+, rheumatoid factor positive; RN, rheumatoid nodules.
Results from the baseline cohort
When analysing the odds ratio for ever being exposed to smoking in the cohort (n = 336) based on data from the inclusion in the BARFOT register, smoking was associated with rheumatoid nodules (OR = 8.8 (95% CI, 3.3 to 23.4); p<0.001). The risk of rheumatoid nodules was increased for current smokers (OR = 8.4 (3.0 to 23.2); p<0.001 v never smokers) as well as former smokers (OR = 9.6 (3.2 to 29.1); p<0.001 v never smokers) when analysed separately. Smoking was associated with rheumatoid nodules among men (OR = 12.9 (2.1 to 80.6); p = 0.006) as well as among women (OR = 7.3 (2.3 to 23.7); p = 0.001). Patients with rheumatoid nodules were more often RF positive (OR = 4.0 (1.9 to 8.4), p<0.001) and had a higher DAS score (5.52 v 5.06; p = 0.014).
Results from the questionnaires
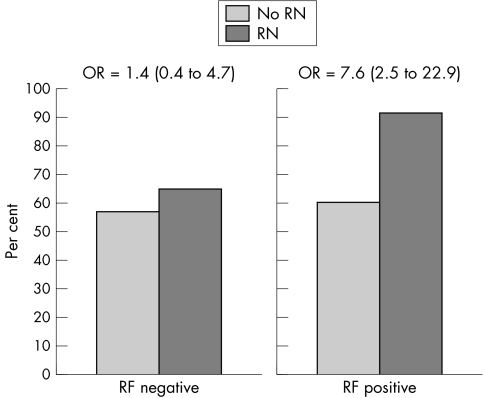
When analysing the detailed information obtained from the questionnaires (n = 210) and using conditional logistic regression analyses, we found that ever having smoked was associated with an increased risk of the presence of rheumatoid nodules (OR = 7.3 (95% CI, 2.3 to 23.6); p = 0.001). To evaluate whether the effect of smoking on the development of rheumatoid nodules was confounded by RF seropositivity or DAS we controlled for these variables in a multivariate conditional logistic regression model and found smoking to be an independent risk factor of rheumatoid nodules development (OR = 6.7 (2.17 to 20.7)). The association between smoking and nodules remained significant when adjusting for formal education (data not shown). To investigate a possible interaction between RF and smoking status the subjects were divided into those with positive RF and those with negative RF. A stratified analysis was carried out, showing an increased risk of rheumatoid nodules in the group of patients with RF positivity, but not among RF negative patients (tables 4 and 5). The odds ratio for developing rheumatoid nodules for ever smokers were 7.6 (2.5 to 22.9) and 1.4 (0.4 to 4.7) in RF seropositive and seronegative individuals, respectively (fig 1).
Table 4 Relative risk of having rheumatoid nodules in ever smokers compared with never smokers by duration of smoking, stratified for being rheumatoid factor positive or negative.
| Smoking years | RF positive patients (74 male, 66 female) | RF negative patients (32 male, 35 female) | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cases (n) | Controls (n) | OR | 95% CI | Cases (n) | Controls (n) | OR | 95% CI | |||||||||
| 0 | 4 | 37 | 1.0 (ref) | 5 | 23 | 1.0 (ref) | ||||||||||
| 1 to 25 | 12 | 18 | 6.8 | 1.9 to 24.6 | 4 | 12 | 1.2 | 0.2 to 5.9 | ||||||||
| 26 to 35 | 19 | 17 | 10.9 | 3.2 to 37.3 | 2 | 8 | 1.4 | 0.2 to 9.2 | ||||||||
| >35 | 15 | 18 | 8.5 | 2.4 to 30.2 | 3 | 10 | 1.4 | 0.2 to 8.5 | ||||||||
| Total | 50 | 90 | 14 | 53 | ||||||||||||
CI, confidence interval; OR, odds ratio; ref, reference; RF, rheumatoid factor.
Table 5 Relative risk of having rheumatoid nodules in ever smokers compared with never smokers by number of smoked cigarette packs, stratified as rheumatoid factor positive or negative.
| Packs | RF positive patients (68 men, 66 women) | RF negative patients (30 men, 35 women) | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cases | Controls | OR | 95% CI | Cases | Controls | OR | 95% CI | ||||||||||
| 0 | 4 | 37 | 1.0 (ref) | 5 | 23 | 1.0 (ref) | |||||||||||
| 1 to 5000 | 15 | 11 | 12.9 | 3.5 to 47.3 | 5 | 13 | 1.7 | 0.4 to 7.2 | |||||||||
| 5001 to 9500 | 14 | 20 | 6.8 | 1.9 to 23.9 | 3 | 8 | 1.4 | 0.2 to 8.1 | |||||||||
| 9501 to 37 000 | 15 | 18 | 8.3 | 2.4 to 29.6 | 1 | 7 | 0.6 | 0.1 to 6.7 | |||||||||
| Total | 48 | 86 | 14 | 51 | |||||||||||||
CI, confidence interval; OR, odds ratio; ref, reference; RF, rheumatoid factor.
Figure 1 Frequencies of former or current smokers among patients with rheumatoid arthritis (n = 210), with (n = 64) and without (controls, n = 146) rheumatoid nodules, by the presence of abnormal serum levels of rheumatoid factor. RF, rheumatoid factor; RN, rheumatoid nodules.
The risk of having nodules was not dose dependent when we examined smoking duration (table 4) or smoking amount (table 5). The majority of ever smokers (93%) had a smoking history of more than 10 years. The mean duration and amount of smoking in both the current smokers and the former smokers were high (table 3). Only two individuals had smoked less than five years and only one individual had consumed less than 600 packs (1.6 pack years).
Table 3 Smoking history of cases and controls.
| Current smokers (n = 70) | Former smokers (n = 71) | Ever smoked (n = 141) | ||||
|---|---|---|---|---|---|---|
| Smoking years before rheumatoid arthritis | 34.4 (10.8) | 25.5 (11.4) | 29.8 (11.9) | |||
| Smoking pack ( = 20 cigarettes) | 9105 (6606) | 7385 (6455) | 8285 (6566) | |||
| Pack year ( = 20 cigarettes daily for one year) median | 24.9 (18.0) | 20.2 (17.7) | 22.7 (17.9) | |||
| Years stopped smoking before rheumatoid arthritis | 15.7 (8.9) |
Values are mean (SD).
Time after smoking cessation (median 14 years, range 2 to 40), when analysed by quartiles of non‐smoking years before rheumatoid arthritis, did not affect the risk of rheumatoid nodules in former smokers (data not shown).
Tobacco use except for smoking (n = 31; 25 current and six former snuff takers, with median years of using snuff 15 and 5, respectively) was not associated with an increased risk of developing nodules (OR = 0.8 (95% CI, 0.2 to 3.4); p = 0.813).
Discussion
This was a case–control study of a well characterised group of patients with newly diagnosed rheumatoid arthritis. The median disease duration was six months and no patient had been treated with methotrexate or other disease modifying anti‐rheumatic drug (DMARD). We found that having smoked at any time was a significant risk factor for rheumatoid nodules early in the disease. Previous studies of rheumatoid nodules have mainly investigated long standing rheumatoid arthritis in patients treated with DMARDs including methotrexate, which is known to increase the risk for developing rheumatoid nodules.9,12,25,26 In a study of patients with early inflammatory polyarthritis18 the frequency of rheumatoid nodules was similar at baseline among never smokers, ex‐smokers, and current smokers, but smokers had developed more nodules after three years. This could be explained by a more heterogeneous group at baseline, with only 67% satisfying the ACR criteria for rheumatoid arthritis, compared with our patients who all fulfilled the criteria.
The BARFOT register includes about 60% of expected cases of early rheumatoid arthritis (incidence rate 25/100 000 adults), which could introduce a selection bias. It is possible that those with milder disease will be less likely to seek medical care, which would overestimate the number of smokers, if smoking is associated with more severe disease. On the other hand, a selective loss of cases among smokers because of their excess mortality would lead to an underestimation. Overall, we think it is unlikely that such selection bias would have had a major impact on the association of smoking with early rheumatoid nodules.
Various studies have shown that smoking is a predictor of the development of rheumatoid arthritis.3,6,8,27,28 Studies have also suggested that smoking influences overall rheumatoid arthritis severity.8,12,17,26 In only one study did smoking seem to be protective against rheumatoid arthritis.29 This may be explained by the fact that current smokers were compared with a combined group of never smokers and ex‐smokers.
The prevalence of smokers in our study cohort was much higher than the population based rates for Sweden, which confirms that patients with rheumatoid arthritis often are or have been smokers. About 20% of the general population in Sweden are smokers,30 in comparison with 67% ever smokers (33% current, 34% former) among the patients studied.
The mechanism by which smoking influences rheumatoid arthritis is unclear. Smoking has effects on the immune system both in the lung and systemically.4 Smoking has been associated with positive RF reactions in the absence of diagnosed rheumatoid arthritis.11 We confirmed the observations of other studies9,11,12,18,31 which found that patients who smoked were more likely to be RF positive than non‐smokers. The correlation between smoking and RF makes it difficult to determine the independent predictive value of each of these factors in relation to rheumatoid nodules. Controlling for RF in a multivariate analyses did not weaken the association, in accordance with a previous study of long standing rheumatoid arthritis.12 However, after the subjects were divided into two groups—those with seropositive and those with seronegative rheumatoid arthritis—a stratified analyses showed an association of smoking and rheumatoid nodules only among patients with seropositive RF. This is compatible with the possibility that RF, or some closely associated factor, may be a path variable mediating the effect of smoking and the risk of developing rheumatoid nodules.
Rheumatoid arthritis is more prevalent in women than in men (65% in the whole BARFOT material), but in this study we found equal numbers of male and female patients with rheumatoid nodules, supporting earlier findings that rheumatoid nodules are more common in men than in women with rheumatoid arthritis.32 The association between rheumatoid nodules and smoking was at least as strong in men as in women.
The evidence for the effect of the duration or intensity of smoking, or both, is conflicting.4,7,28,31,33,34,35 Some9,26 have suggested that smoking may be more important in the initiation of rheumatoid arthritis than in the perpetuation of the disease process. The lack of a dose related effect of smoking (as reflected by cessation, smoking duration, and pack years) on the occurrence of rheumatoid nodules in our study may add some weight to this idea. However, as the vast majority in our study had a high smoking exposure, a dose–response effect in the lower range cannot be excluded.
The association between rheumatoid nodules and smoking, even decades after smoking cessation, is in accord with other studies.28,31 The long lag between previous smoking and the development of rheumatoid arthritis with rheumatoid nodules cannot be understood unless there are long lasting and perhaps irreversible effects of smoking on the pathophysiology of rheumatoid nodule development, or unless smoking is linked to some other factor to which there is continued exposure.
The use of snuff tobacco did not increase the risk of rheumatoid nodules, which may suggest that some factor inhaled in smoke is importance, rather than a general effect of nicotine. However, the number of individuals using snuff was limited, and more studies are needed before firm conclusions can be drawn.
The risk of developing rheumatoid nodules can be affected by DMARDs. Accelerated nodulosis has been reported in particular with methotrexate,36 but also with azathioprine37 and ciclosporine A.38 Thus DMARD treatment could be a confounder in studies of rheumatoid nodules, but in our study none of the patients had ever used DMARDs.
One limitation of this study is that the detailed smoking history from the separate questionnaire was collected retrospectively. However, information on current, former, or never smoker status was also obtained at the time of disease onset, and in comparing this information with the smoking history questionnaire data only three patients were misclassified (data not shown). Thus the reliability of data from the smoking questionnaire seems to be good and the range and type of associations were the same using smoking exposure data from both sources.
Conclusions
There is an association between smoking and rheumatoid nodules in early rheumatoid arthritis in patients with positive RF. In contrast to others we did not find a quantitative relation between smoking extent and rheumatoid nodules. Smoking is a risk factor for the development of rheumatoid nodules in susceptible subjects, possibly mediated through increased RF production.
Acknowledgements
This study was supported by the Centre for Clinical Research Dalarna, Falun, Sweden, the Swedish Rheumatism Association and the Spenshult Hospital for Rheumatic Diseases, Halmstad, Sweden.
The BARFOT study group
M Ahlmén, J Bratt, K Forslind, I Hafström, C Keller, I Leden, B Lindell, I Petersson, B Svensson, A Teleman, J Theander.
Abbreviations
BARFOT - Better AntiRheumatic FarmacOTherapy study group
DAS28 - 28 joint disease activity score
HAQ - Health Assessment Questionnaire
RF - rheumatoid factor
Footnotes
1Members of the BARFOT study group are listed at the end of the article.
References
- 1.Gabriel S E. The epidemiology of rheumatoid arthritis. Rheum Dis Clin North Am 200127269–281. [DOI] [PubMed] [Google Scholar]
- 2.Simonsson M, Bergman S, Jacobsson L T, Petersson I F, Svensson B. The prevalence of rheumatoid arthritis in Sweden. Scand J Rheumatol 199928340–343. [DOI] [PubMed] [Google Scholar]
- 3.Silman A J, Newman J, MacGregor A J. Cigarette smoking increases the risk of rheumatoid arthritis. Results from a nationwide study of disease‐discordant twins. Arthritis Rheum 199639732–735. [DOI] [PubMed] [Google Scholar]
- 4.Voigt L F, Koepsell T D, Nelson J L, Dugowson C E, Daling J R. Smoking, obesity, alcohol consumption, and the risk of rheumatoid arthritis. Epidemiology 19945525–532. [PubMed] [Google Scholar]
- 5.Silman A J, MacGregor A J, Thomson W, Holligan S, Carthy D, Farhan A.et al Twin concordance rates for rheumatoid arthritis: results from a nationwide study. Br J Rheumatol 199332903–907. [DOI] [PubMed] [Google Scholar]
- 6.Symmons D P, Bankhead C R, Harrison B J, Brennan P, Barrett E M, Scott D G.et al Blood transfusion, smoking, and obesity as risk factors for the development of rheumatoid arthritis: results from a primary care‐based incident case‐control study in Norfolk, England. Arthritis Rheum 1997401955–1961. [DOI] [PubMed] [Google Scholar]
- 7.Hutchinson D, Shepstone L, Moots R, Lear J T, Lynch M P. Heavy cigarette smoking is strongly associated with rheumatoid arthritis (RA), particularly in patients without a family history of RA. Ann Rheum Dis 200160223–227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Padyukov L, Silva C, Stolt P, Alfredsson L, Klareskog L. A gene–environment interaction between smoking and shared epitope genes in HLA‐DR provides a high risk of seropositive rheumatoid arthritis. Arthritis Rheum 2004503085–3092. [DOI] [PubMed] [Google Scholar]
- 9.Saag K G, Cerhan J R, Kolluri S, Ohashi K, Hunninghake G W, Schwartz D A. Cigarette smoking and rheumatoid arthritis severity. Ann Rheum Dis 199756463–469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Turesson C, O'Fallon W M, Crowson C S, Gabriel S E, Matteson E L. Extra‐articular disease manifestations in rheumatoid arthritis: incidence trends and risk factors over 46 years. Ann Rheum Dis 200362722–727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tuomi T, Heliovaara M, Palosuo T, Aho K. Smoking, lung function, and rheumatoid factors. Ann Rheum Dis 199049753–756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wolfe F. The effect of smoking on clinical, laboratory, and radiographic status in rheumatoid arthritis. J Rheumatol 200027630–637. [PubMed] [Google Scholar]
- 13.Hirsch R, Lin J P, Scott W W, Ma L D, Pillemer S R, Kastner D L.et al Rheumatoid arthritis in the Pima Indians: the intersection of epidemiologic, demographic, and genealogic data. Arthritis Rheum 1998411464–1469. [DOI] [PubMed] [Google Scholar]
- 14.Turesson C, O'Fallon W M, Crowson C S, Gabriel S E, Matteson E L. Occurrence of extra‐articular disease manifestations is associated with excess mortality in a community based cohort of patients with rheumatoid arthritis. J Rheumatol 20022962–67. [PubMed] [Google Scholar]
- 15.Turesson C, Jacobsson L, Bergstrom U, Truedsson L, Sturfelt G. Predictors of extra‐articular manifestations in rheumatoid arthritis. Scand J Rheumatol 200029358–364. [DOI] [PubMed] [Google Scholar]
- 16.Gabriel S E, Crowson C S, Kremers H M, Doran M F, Turesson C, O'Fallon W M.et al Survival in rheumatoid arthritis: a population‐based analysis of trends over 40 years. Arthritis Rheum 20034854–58. [DOI] [PubMed] [Google Scholar]
- 17.Houssien D A, Scott D L, Jonsson T. Smoking, rheumatoid factors, and rheumatoid arthritis. Ann Rheum Dis 199857175–176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Harrison B J, Silman A J, Wiles N J, Scott D G, Symmons D P. The association of cigarette smoking with disease outcome in patients with early inflammatory polyarthritis. Arthritis Rheum 200144323–330. [DOI] [PubMed] [Google Scholar]
- 19.Svensson B, Schaufelberger C, Teleman A, Theander J. Remission and response to early treatment of RA assessed by the Disease Activity Score. BARFOT study group. Better Anti‐rheumatic Farmacotherapy. Rheumatology (Oxford) 2000391031–1036. [DOI] [PubMed] [Google Scholar]
- 20.Keller C, Hafstrom I, Svensson B. Bone mineral density in women and men with early rheumatoid arthritis. Scand J Rheumatol 200130213–220. [DOI] [PubMed] [Google Scholar]
- 21.Ekdahl C, Eberhardt K, Andersson S I, Svensson B. Assessing disability in patients with rheumatoid arthritis. Use of a Swedish version of the Stanford Health Assessment Questionnaire. Scand J Rheumatol 198817263–271. [DOI] [PubMed] [Google Scholar]
- 22.van Gestel A M, Prevoo M L, van't Hof M A, van Rijswijk M H, van de Putte L B, van Riel P L. Development and validation of the European League Against Rheumatism response criteria for rheumatoid arthritis. Comparison with the preliminary American College of Rheumatology and the World Health Organization/International League Against Rheumatism Criteria. Arthritis Rheum 19963934–40. [DOI] [PubMed] [Google Scholar]
- 23.Arnett F C, Edworthy S M, Bloch D A, McShane D J, Fries J F, Cooper N S.et al The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum 198831315–324. [DOI] [PubMed] [Google Scholar]
- 24.Manthorpe R, Benoni C, Jacobsson L, Kirtava Z, Larsson A, Liedholm R.et al Lower frequency of focal lip sialadenitis (focus score) in smoking patients. Can tobacco diminish the salivary gland involvement as judged by histological examination and anti‐SSA/Ro and anti‐SSB/La antibodies in Sjogren's syndrome? Ann Rheum Dis 20005954–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mattey D L, Dawes P T, Fisher J, Brownfield A, Thomson W, Hajeer A H.et al Nodular disease in rheumatoid arthritis: association with cigarette smoking and HLA‐DRB1/TNF gene interaction. J Rheumatol 2002292313–2318. [PubMed] [Google Scholar]
- 26.Mattey D L, Hutchinson D, Dawes P T, Nixon N B, Clarke S, Fisher J.et al Smoking and disease severity in rheumatoid arthritis: association with polymorphism at the glutathione S‐transferase M1 locus. Arthritis Rheum 200246640–646. [DOI] [PubMed] [Google Scholar]
- 27.Hernandez Avila M, Liang M H, Willett W C, Stampfer M J, Colditz G A, Rosner B.et al Reproductive factors, smoking, and the risk for rheumatoid arthritis. Epidemiology 19901285–291. [DOI] [PubMed] [Google Scholar]
- 28.Stolt P, Bengtsson C, Nordmark B, Lindblad S, Lundberg I, Klareskog L.et al Quantification of the influence of cigarette smoking on rheumatoid arthritis: results from a population based case‐control study, using incident cases. Ann Rheum Dis 200362835–841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hazes J M, Dijkmans B A, Vandenbroucke J P, de Vries R R, Cats A. Lifestyle and the risk of rheumatoid arthritis: cigarette smoking and alcohol consumption. Ann Rheum Dis 199049980–982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Statistics Sweden (www.tobaksfakta.org)
- 31.Heliovaara M, Aho K, Aromaa A, Knekt P, Reunanen A. Smoking and risk of rheumatoid arthritis. J Rheumatol 1993201830–1835. [PubMed] [Google Scholar]
- 32.Gorman J D, David‐Vaudey E, Pai M, Lum R F, Criswell L A. Lack of association of the HLA‐DRB1 shared epitope with rheumatoid nodules: an individual patient data meta‐analysis of 3,272 Caucasian patients with rheumatoid arthritis. Arthritis Rheum 200450753–762. [DOI] [PubMed] [Google Scholar]
- 33.Karlson E W, Lee I M, Cook N R, Manson J E, Buring J E, Hennekens C H. A retrospective cohort study of cigarette smoking and risk of rheumatoid arthritis in female health professionals. Arthritis Rheum 199942910–917. [DOI] [PubMed] [Google Scholar]
- 34.Reckner Olsson A, Skogh T, Wingren G. Comorbidity and lifestyle, reproductive factors, and environmental exposures associated with rheumatoid arthritis. Ann Rheum Dis 200160934–939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Criswell L A, Merlino L A, Cerhan J R, Mikuls T R, Mudano A S, Burma M.et al Cigarette smoking and the risk of rheumatoid arthritis among postmenopausal women: results from the Iowa Women's Health Study. Am J Med 2002112465–471. [DOI] [PubMed] [Google Scholar]
- 36.Combe B, Didry C, Gutierrez M, Anaya J M, Sany J. Accelerated nodulosis and systemic manifestations during methotrexate therapy for rheumatoid arthritis. Eur J Med 19932153–156. [PubMed] [Google Scholar]
- 37.Langevitz P, Maguire L, Urowitz M. Accelerated nodulosis during azathioprine therapy. Arthritis Rheum 199134123–124. [DOI] [PubMed] [Google Scholar]
- 38.Spadaro A, Fiore D, Iagnocco A, Riccieri V, Sili Scavalli A, Coari G.et al Rheumatoid nodules and cyclosporin A treatment. Int J Clin Pharmacol Res 19941475–78. [PubMed] [Google Scholar]