Abstract
Objectives
Patients with rheumatoid arthritis have an increased risk for cardiovascular disease (CVD). The prevalence of metabolic syndrome (MetS)—a major contributor to CVD—in a cohort of patients with rheumatoid arthritis and its relationship with rheumatoid arthritis related factors is investigated here.
Methods
200 outpatients with rheumatoid arthritis (147 women and 53 men), with a mean (standard deviation (SD)) age of 63 (11) years, and 400 age and sex‐matched controls were studied. MetS was assessed according to the adult treatment panel III criteria and rheumatoid arthritis disease activity by the disease activity score of 28 joints (DAS28). A standard clinical evaluation was carried out, and a health and lifestyle questionnaire was completed.
Results
The overall prevalence of MetS was 44% in patients with rheumatoid arthritis and 41% in controls (p = 0.5). Patients with rheumatoid arthritis were more likely to have low high‐density lipoprotein cholesterol compared with controls (p = 0.02), whereas controls were more likely to have increased waist circumference or raised blood pressure (p = 0.001 and 0.003, respectively). In multivariate logistic regression analysis adjusting for demographics and rheumatoid arthritis treatment modalities, the risk of having moderate‐to‐high disease activity (DAS28>3.2) was significantly higher in patients with MetS compared with those with no MetS components (OR 9.24, 95% CI 1.49 to 57.2, p = 0.016).
Conclusion
A high, albeit comparable to the control population, prevalence of MetS was found in middle‐to‐older aged patients with rheumatoid arthritis. The correlation of rheumatoid arthritis disease activity with MetS suggests that the increased prevalence of coronary heart disease in patients with rheumatoid arthritis may, at least in part, be attributed to the inflammatory burden of the disease.
The metabolic syndrome (MetS) describes a constellation of major risk factors for cardiovascular diseases (CVDs) such as atherogenic dyslipidaemia, obesity, hypertension and diabetes.1 These associated risk factors have been previously called syndrome X2 or the insulin resistance syndrome.3 In Europeans, the presence of the MetS was associated with an approximate twofold increased risk for incident cardiovascular morbidity and mortality,4 whereas a 2.1‐fold increase for initial stroke was found in participants of the Framingham offspring study in North America.5
Insulin resistance is believed to be the key pathogenetic factor of the metabolic syndrome.3,6 However, MetS is influenced by a complex interplay between multiple genetic variations and environmental factors.7 To enable international comparison of studies, the World Health Organization (WHO) in 1999 published a working definition of the metabolic syndrome.8 Alternative definitions have been subsequently proposed by the European Group for the Study of Insulin Resistance (EGIR)9 and the Adult Treatment Panel III of the National Cholesterol Education Program (NCEP ATPIII).1 All definitions include a measure of blood pressure, triglycerides, HDL cholesterol and fasting glucose. They differ with respect to the selection of cut‐off points and the measure of obesity. In contrast with the WHO and EGIR definitions, in which the presence of hyperinsulinaemia as an indicator of insulin resistance is the starting point, the NCEP ATPIII definition is based only on the number of abnormalities. The NCEP ATP III criteria—which were developed for clinical use—are superior in terms of simplicity, requiring fasting glucose assessment instead of oral glucose test or measurement of insulin resistance by the euglycaemic clamp. The over‐riding concern when assessing MetS is to predict people at high risk for CVD; to this end, Dekker et al4 have recently compared the available definitions for MetS in a European cohort and found that definitions including information on fasting insulin level—like the EGIR or WHO—were no better predictors of CVD risk.
Patients with rheumatoid arthritis are at increased risk for CVD, which accounts for about half of all deaths in these patients.10 This is attributed to accelerated atherosclerosis as a result of both systemic inflammatory burden and the high prevalence of traditional risk factors for CVD.11,12,13 Insulin resistance, one of the key components of the MetS,6 is also common among patients with rheumatoid arthritis.14,15 Moreover, chronic inflammation has been implicated in its pathogenesis.16,17
Although several studies have established the increased risk for CVD and the increased prevalence of insulin resistance in patients with rheumatoid arthritis,15 the overall prevalence of the MetS, a more global indicator of the risk of CVD, has not been studied. This is important in view of the increased morbidity and mortality from CVD. To this end, we sought to assess the prevalence of MetS in a cohort of patients with rheumatoid arthritis and explore the possible relationships of MetS with disease‐related factors.
Our results suggest that although the prevalence of MetS in patients with rheumatoid arthritis is not different from that in a relative risk population, the prevalence of MetS correlates with disease activity. These results corroborate previous findings that implicate the inflammatory burden of the disease in morbidity from CVD.
Patients and methods
Study population
We studied 200 consecutive outpatients meeting the American College of Rheumatology criteria for the classification of rheumatoid arthritis18 that were followed by the Department of Rheumatology, University Hospital of Heraklion, Heraklion, Greece. For each patient with rheumatoid arthritis, two people without rheumatoid arthritis or other chronic inflammatory diseases were matched for age and sex. All controls were residents of rural and semi‐urban areas in one county (Heraklion) of the island of Crete, recruited following public invitation by primary care physicians of seven primary healthcare centres throughout the county, during the same period as the recruitment of patients with rheumatoid arthritis. We used controls from the general population—rather than from hospital—to minimise the possibility of bias, considering that health‐seeking behaviour is not a major issue in the present study. All participants signed informed consent.
MetS
MetS was defined according to ATPIII criteria1 that required the presence of three or more of the following conditions: large waist circumference (>88 cm in women and >102 cm in men), increased triglyceride level (⩾150 mg/dl), low level of high‐density lipoprotein cholesterol (HDL‐C) (<50 mg/dl in women and <40 mg/dl men), increased fasting glucose (⩾110 mg/dl), and increased systolic or diastolic blood pressure (⩾130/85 mm Hg) or self‐reported use of drugs for hypertension.
Assessment of the metabolic syndrome
A detailed health and lifestyle questionnaire was completed for all participants, including history of hypertension, coronary heart disease (CHD; including myocardial infraction and angina pectoris documented by medical reports), family history of premature (<60 years) CHD, drugs taken and use of tobacco. The waist circumference was measured at the umbilical level according to the WHO guidelines, with the person breathing silently.19
Blood samples were taken the morning after 10 h of overnight fasting. Serum concentrations of total count, HDL‐C, and triglycerides were measured using an automated chemistry analyser (Olympus AU‐600, Diamond Diagnostics, Hollinston, Massachusetts, USA) with reagents from the same manufacturer. Low‐density lipoprotein cholesterol (LDL‐C) was calculated according to the Friedewald formula except for samples with serum triglycerides >400 mg/dl (4.5 mmol/l), for which LDL‐C could not be determined with the method used. Serum glucose concentration was determined by standard methods in routine use. For patients with rheumatoid arthritis, high‐sensitivity C reactive protein (hs‐CRP), erythrocyte sedimentation rate (ESR) and rheumatoid factor levels were additionally measured (hs‐CRP by immune nephelometry (BN II analyser, Dade Behring, USA) and ESR with an automated analyser (Ves‐matic 20, Diesse Diagnostica, Senese, Italy)).
All laboratory analyses were carried out in the Department of Clinical Chemistry of the University Hospital of Heraklion, which participates in several quality assurance programmes and has full clinical pathology accreditation .
Rheumatoid arthritisdisease characteristics
Rheumatoid arthritis disease activity was assessed by the DAS28 index. A score of DAS28 ⩽3.2 is considered as the cut‐off for low disease activity, whereas DAS28 >5.1 is considered as the limit for high disease activity.20 Rheumatoid arthritis disease‐related data, such as disease duration, extra‐articular manifestations (as a marker of disease severity) and physical activity, were recorded.
Statistical analysis
Continuous variables are reported as mean (standard deviation (SD). Variables were compared using either the t test or χ2 test as appropriate. The relationship between DAS28 and the number of MetS components was examined using linear regression, treating DAS28 as the dependent variable and age, sex and MetS components (0, 1–2 or ⩾3) as independent variables. Logistic regression (backward selection) was also carried out to calculate odds ratios (OR) for moderate–high DAS28 (ie, DAS28 >3.2) according to MetS components, controlling for the possible effects of age, sex, body mass index (BMI ⩽25, 25.1–29.9, ⩾30 kg/m2), tobacco use and rheumatoid arthritis treatment modalities (use of biological treatment, corticosteroids and methotrexate). p Values <0.05 (two tailed) were considered significant. SPSS V.10.0 was used for all statistical calculations.
Results
Demographics
Among the 200 patients with rheumatoid arthritis, 147 (73.5%) were women and 53 (26.5%) were men, with a mean (SD) age of 63 (11) years. Similarly, 294 (73.5%) of the controls were women and 106 (26.5%) were men, with a mean (SD) age of 63 (11) years. The mean age of men was higher than that of women both among patients with rheumatoid arthritis (67 (8) v 62 (11), p = 0.003) and among controls (67 (8) v 61 (12), p = 0.001).
Prevalence of MetS
The overall prevalence of MetS was 44% in patients with rheumatoid arthritis and 41% in controls (p = 0.5) (table 1). The prevalence rates in men were 39.6% and 37.7% (p = 0.8), respectively, whereas those in women were 45.6% and 42.2% (p = 0.5), respectively. When the prevalence of MetS was analysed in various age groups, no significant differences were depicted between patients with rheumatoid arthritis and controls.
Table 1 Prevalence of the metabolic syndrome in patients with rheumatoid arthritis and controls.
| Age group (years) | Study population | Women | Men | Total | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| n | % | p Value* | n | % | p Value* | n | % | p Value* | ||
| <60 | RA | 20/58 | 34.5 | 0.7 | 4/10 | 40 | 0.2 | 24/68 | 35.3 | 0.4 |
| Controls | 37/116 | 31.9 | 3/18 | 16.7 | 40/134 | 29.9 | ||||
| 60–70 | RA | 27/53 | 50.9 | 0.2 | 9/25 | 36 | 1 | 36/78 | 46.2 | 0.3 |
| Controls | 42/106 | 39.6 | 19/52 | 36.5 | 61/158 | 38.6 | ||||
| >70 | RA | 20/36 | 55.6 | 0.5 | 8/18 | 44.4 | 0.7 | 28/54 | 51.9 | 0.4 |
| Controls | 45/72 | 62.5 | 18/36 | 50 | 63/108 | 58.3 | ||||
| Total | RA | 67/147 | 45.6 | 0.5 | 21/53 | 39.6 | 0.8 | 88/200 | 44 | 0.5 |
| Controls | 124/294 | 42.2 | 40/106 | 37.7 | 164/400 | 41 | ||||
RA, rheumatoid arthritis.
*Difference between patients with RA and controls.
Metabolic syndrome‐related features
The two leading components of MetS in the study population were large waist circumference (71% in patients and 83% in controls, p = 0.001) and increased blood pressure (66% in patients and 78% in controls, p = 0.003; table 2). Patients with rheumatoid arthritis were more likely to have low HDL‐C compared with controls (32% v 23%, p = 0.02). The frequency of raised glucose levels or hypertriglyceridaemia was not different between groups, whereas large waist circumference and raised blood pressure were more prevalent in controls (p = 0.001 and 0.003, respectively; table 2).
Table 2 Distribution of metabolic syndrome components, cardiovascular disease risk factors and coronary heart disease in the study population.
| Variable | Women | Men | All patients | ||||||
|---|---|---|---|---|---|---|---|---|---|
| RA (n = 147; %) | Controls (n = 294; %) | p Value* | RA (n = 53; %) | Controls (n = 106; %) | p Value | RA (n = 200; %) | Controls (n = 400; %) | p Value | |
| Fasting glucose ⩾110 mg/dl | 45 (30.6) | 94 (32) | 0.8 | 14 (26.4) | 38 (35.8) | 0.2 | 59 (29.5) | 132 (33) | 0.4 |
| Triglycerides ⩾150 mg/dl | 44 (29.9) | 80 (27.2) | 0.5 | 19 (35.8) | 32 (30.2) | 0.5 | 63 (31.5) | 112 (28) | 0.4 |
| HDL‐C <50 mg/dl (♀)/40 mg/dl (♂) | 47 (32) | 74 (25.2) | 0.1 | 17 (32.1) | 17 (16) | 0.02 | 64 (32) | 91 (22.8) | 0.02 |
| Waist circumference>88 cm (♀)/>102 cm (♂) | 110 (74.8) | 267 (90.8) | 0.001 | 32 (60.4) | 65 (61.3) | 0.9 | 142 (71) | 332 (83) | 0.001 |
| Arterial blood pressure ⩾130/85 mm Hg | 97 (66) | 219 (74.5) | 0.06 | 35 (66) | 91 (85.8) | 0.004 | 132 (66) | 310 (77.5) | 0.003 |
| Total cholesterol ⩾200 mg/dl | 95 (64.6) | 227 (77.2) | 0.005 | 31 (58.5) | 75 (70.8) | 0.1 | 126 (63) | 302 (75.5) | 0.001 |
| LDL‐C ⩾130 mg/dl | 82 (55.8) | 194 (66.2) | 0.03 | 25 (47.2) | 61 (57.5) | 0.2 | 107 (53.5) | 255 (63.9) | 0.01 |
| Family history of CHD | 32 (21.8) | 84 (28.6) | 0.1 | 9 (17.0) | 36 (34) | 0.04 | 41 (20.5) | 120 (30) | 0.02 |
| Current smoking | 16 (10.9) | 25 (8.5) | 0.4 | 14 (26.4) | 30 (28.3) | 0.9 | 30 (15) | 55 (13.8) | 0.6 |
| CHD | 10 (6.8) | 10 (3.4) | 0.1 | 13 (24.5) | 19 (17.9) | 0.3 | 23 (11.5) | 29 (7.3) | 0.09 |
CHD, coronary heart disease; CVD, cardiovascular diseases; HDL‐C, high‐density lipoprotein cholesterol; LDL‐C, low‐density lipoprotein cholesterol; MetS, metabolic syndrome; RA, rheumatoid arthritis.
Significant (p<0.05) differences are highlighted in bold.
*Difference between patients with rheumatoid arthritis and controls.
CHD and traditional CVD risk factors
A trend for higher prevalence of CHD was seen in patients with rheumatoid arthritis compared with controls (11.5% v 7.3%, p = 0.09; table 2). It has been proposed that the increased risk of CHD may precede the clinical onset of rheumatoid arthritis,21 and that there is a preclinical phase of rheumatoid arthritis during which inflammatory activity and serological disturbances occur.22 For this reason, and to avoid any biases in the study, we included all CHD incidents irrespective of whether they occurred before or after the clinical onset of rheumatoid arthritis. Controls were more likely to have hypercholesterolaemia (defined as total cholesterol ⩾200 mg/dl, 76% v 63%, p = 0.001), high LDL cholesterol levels (LDL‐C ⩾130 mg/dl, 64% v 54%, p = 0.01) and family history of CHD (30% v 21%, p = 0.02). On the contrary, smoking status was comparable between the two groups.
Patients with rheumatoid arthritis; MetS and disease characteristics
Among patients with rheumatoid arthritis, women with MetS had a higher prevalence of CHD than those without MetS (11.9% v 2.5%, p = 0.02; table 3). As expected, patients with MetS had higher BMI than those without MetS (30.7 (5.2) v 27.1 (4.7) kg/m2, p = 0.001). No significant differences were observed in traditional CVD risk factors such as family history of CHD, high LDL‐C levels and hypercholesterolaemia (table 3). Smokers were more prevalent among patients without MetS than among those with MetS (19.6% v 9.1%, p = 0.03).
Table 3 Prevalence of cardiovascular disease risk factors and coronary heart disease among patients with rheumatoid arthritis.
| Variable | Women with RA | Men with RA | All RA | ||||||
|---|---|---|---|---|---|---|---|---|---|
| MetS−* (n = 80) | MetS+† (n = 67) | p Value‡ | MetS− (n = 32) | MetS+ (n = 21) | p Value‡ | MetS− (n = 112) | MetS+ (n = 88) | p Value‡ | |
| Age (years), mean (SD) | 59.2 (12.7) | 64.3 (9.1) | 0.006 | 66 (8.3) | 67.5 (7.6) | 0.5 | 61.1 (12) | 65.1 (8.8) | 0.01 |
| BMI (kg/m2) | 27.4 (5.1) | 31.4 (5.5) | 0.001 | 26.6 (3.4) | 28.7 (3.5) | 0.04 | 27.1 (4.7) | 30.7 (5.2) | 0.001 |
| Hypercholesterolaemia, n (%) | 54 (67.5) | 41 (61.2) | 0.4 | 17 (53.1) | 14 (66.7) | 0.3 | 71 (63.4) | 55 (62.5) | 0.9 |
| High LDL, n (%) | 49 (61.3) | 33 (49.3) | 0.1 | 13 (40.6) | 12 (57.1) | 0.2 | 62 (55.4) | 45 (51.1) | 0.5 |
| Family history of CHD, n (%) | 14 (17.5) | 18 (26.9) | 0.2 | 6 (18.8) | 3 (14.3) | 0.7 | 20 (17.9) | 21 (23.9) | 0.4 |
| Current smoking, n (%) | 13 (16.3) | 3 (4.5) | 0.02 | 9 (28.1) | 5 (23.8) | 0.6 | 22 (19.6) | 8 (9.1) | 0.03 |
| CHD, n (%) | 2 (2.5) | 8 (11.9) | 0.02 | 7 (21.9) | 6 (28.6) | 0.6 | 9 (8.0) | 14 (15.9) | 0.08 |
CHD, coronary heart disease; CVD, cardiovascular diseases; HDL, high‐density lipoprotein; LDL, low‐density lipoprotein; MetS, metabolic syndrome; RA, rheumatoid arthritis.
Significant (p<0.05) differences are highlighted in bold.
*MetS−, no metabolic syndrome.
†MetS+, metabolic syndrome.
‡Difference between patients with rheumatoid arthritis, with and without MetS.
The association between individual MetS components and the DAS28 was next assessed by linear regression analysis, adjusting for sex and age. We found a significant correlation with systolic blood pressure (β = 0.254, p = 0.01) and a trend for an inverse correlation with HDL‐C (β = −0.148, p = 0.056), whereas no relationship was observed with diastolic blood pressure, triglycerides, waist circumference or glucose levels.
Next we explored the correlation of rheumatoid arthritis disease activity with MetS. Low disease activity (DAS28⩽3.2) correlated with the absence of MetS; 16.5% of the patients without MetS had DAS28⩽3.2 compared with only 2.8% of those with MetS (p = 0.003). Both ESR and CRP correlated with the number of MetS components present (β = 0.193, p = 0.006 and β = 0.158, p = 0.029, respectively). Furthermore, patients with MetS showed a trend for higher ESR than those without the syndrome (35.4 (19.8) v 30.8 (18.1), p = 0.08; table 4).
Table 4 Disease‐related factors among patients with rheumatoid arthritis according to the presence of MetS.
| Variable | Women with RA | Men with RA | All RA | ||||||
|---|---|---|---|---|---|---|---|---|---|
| MetS−* (n = 80) | MetS+† (n = 67) | p Value‡ | MetS− (n = 32) | MetS+ (n = 21) | p Value | MetS− (n = 112) | MetS+ (n = 88) | p Value | |
| RF positivity, n (%) | 28 (35) | 21 (31.3) | 0.7 | 13 (40.6) | 9 (42.9) | 0.8 | 41 (36.6) | 30 (34.1) | 0.8 |
| RA duration, years, mean (SD) | 9.7 (8.6) | 9.4 (10.1) | 0.8 | 8.3 (9.3) | 11.3 (14.8) | 0.4 | 9.3 (8.8) | 9.8 (11.4) | 0.7 |
| Extra‐articular manifestations, n (%) | 12 (15) | 16 (23.9) | 0.2 | 6 (18.8) | 4 (19) | 0.9 | 18 (16.1) | 20 (22.7) | 0.3 |
| Biological treatment, n (%) | 34 (42.5) | 20 (29.9) | 0.1 | 14 (43.8) | 5 (23.8) | 0.1 | 48 (42.9) | 25 (28.4) | 0.04 |
| NSAID, n (%) | 3 (3.8) | 6 (9) | 0.2 | 1 (3.1) | 0 | 0.4 | 4 (3.6) | 6 (6.8) | 0.3 |
| Steroids, n (%) | 24 (30.0) | 11 (16.4) | 0.05 | 9 (28.1) | 4 (19) | 0.5 | 33 (29.5) | 15 (17) | 0.04 |
| Methotrexate, n (%) | 54 (67.5) | 39 (58.2) | 0.2 | 20 (62.5) | 15 (71.4) | 0.5 | 74 (66.1) | 54 (61.4) | 0.5 |
| hs‐CRP, mg/dl, mean (SD) | 0.6 (0.5) | 0.7 (0.7) | 0.3 | 0.9 (0.8) | 0.9 (0.9) | 0.8 | 0.7 (0.6) | 0.8 (0.8) | 0.4 |
| ESR, mm/h, mean (SD) | 31.9 (18.6) | 38.1 (20.8) | 0.06 | 27.9 (16.4) | 27.0 (13.5) | 0.8 | 30.8 (18.1) | 35.4 (19.8) | 0.08 |
ESR, erythrocyte sedimentation rate; HDL, high‐density lipoprotein; hs‐CRP, high‐sensitivity C reactive protein; LDL, low‐density lipoprotein; MetS, metabolic syndrome components; NSAID, non‐steroidal anti‐inflammatory drug; RA, rheumatoid arthritis; RF, rheumatoid factor.
Significant (p<0.05) differences are highlighted in bold.
*MetS−: no metabolic syndrome.
†MetS+: metabolic syndrome.
‡Difference between patients with rheumatoid arthritis with and without MetS.
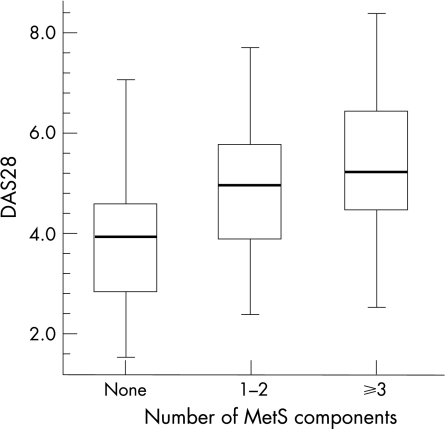
A significant relationship was found between the DAS28 index and the number of MetS components present (fig 1). The median DAS28 in patients with no MetS components present (n = 21) was 3.99 (1.5), compared with 4.99 (1.35) in patients with 1–2 MetS components present (n = 91) and 5.49 (1.28) in patients with MetS (n = 88; ie, ⩾3 components; β coefficient for the correlation between DAS28 and the number of MetS components was 0.237; p = 0.001).
Figure 1 Correlation of DAS28 with the number of metabolic syndrome (MetS) components present. Patients were grouped depending on the number of MetS components into three groups: those with no MetS components present (n = 21), those with 1–2 MetS components present (n = 91) and those with ⩾3 components (n = 88). A significant linear correlation of DAS28 with the number of MetS components present was found after controlling for age and sex effects (β correlation coefficient 0.237, p = 0.001) or after controlling for demographics (age, sex), rheumatoid arthritis treatments (MTX, steroids, biologicals) and behavioural characteristics (smoke and BMI; β coefficient = 0.231, p = 0.003).
Other disease‐related factors, including disease duration and disease severity (such as extra‐articular manifestations), were comparable between patients with and without MetS (table 4). In reference to drugs for rheumatoid arthritis, both biologicals and steroids were more frequently given in patients without MetS compared with those with MetS (42.9% v 28.4%, p = 0.04 and 29.5% v 17%, p = 0.04, respectively). In multivariate analysis, adjusting for demographics (age, sex), treatment (methotrexate, biologicals, steroids) and behavioural factors (smoking and BMI), the risk for having moderate‐to‐high DAS28 (>3.2) was found to be significantly higher in patients with MetS compared with those without MetS (OR 9.24, 95% CI 1.49 to 57.2, p = 0.016; table 5). In this multivariate‐adjusted model, rheumatoid arthritis‐related treatments (MTX, biologics and steroids) were not independent predictors of (or were not significantly associated with) moderate‐to‐high DAS28.
Table 5 OR (95% CI) for moderate‐to‐high disease activity score of 28 joints (DAS28>3.2) in patients with rheumatoid arthritis according to the presence of metabolic syndrome components.
| Metabolic syndrome | Increased disease activity (DAS28 >3.20) | |||
|---|---|---|---|---|
| Age‐adjusted and sex‐adjusted* | Multivariate‐adjusted† | |||
| Components | OR (95% CI) | p Value | OR (95% CI) | p Value |
| None | 1.00 (reference) | — | 1.00 (reference) | — |
| 1–2 | 2.44 (0.70 to 8.44) | 0.160 | 1.82 (0.49 to 6.72) | 0.370 |
| ⩾3 | 12.1 (2.1 to 71.1) | 0.006 | 9.24 (1.49 to 57.2) | 0.016 |
| p for trend 0.004 | p for trend 0.011 | |||
DAS28, disease activity score of 28 joints; MetS, metabolic syndrome.
*Logistic regression for increased DAS28 (>3.20) including age, sex and number of MetS risk factors (0, 1–2 or ⩾3) as independent variables.
†Logistic regression for increased DAS28 (>3.20) including age, sex, number of MetS risk factors (0, 1–2, or ⩾3), use of biologicals/steroids/methotrexate, smoking and body mass index (<25, 25.1–29.9, ⩾30 kg/m2) as independent variables.
Discussion
In this study of middle‐to‐old aged people, MetS was found to be common both in patients with rheumatoid arthritis (44%) as well as in age‐matched and sex‐matched controls (41%), and to correlate with disease activity. The prevalence of MetS was comparable between patients and controls both in the whole study population and in the different age groups. This is higher than what has recently been reported for the Greek adult population in the region of Attica (ATTICA study, prevalence 19.8%). Such a discrepancy may be attributed to differences in the selection criteria of the study population: participants in the ATTICA study were free of CHD, stroke, diabetes mellitus or any atherosclerotic disease, with a significantly lower mean age than our study population.23
The prevalence of MetS in our population was also assessed according to the criteria recently proposed by the American Heart Association/National Heart, Lung, and Blood Institute.24 As expected, prevalence was higher compared with the NCEP ATPIII criteria, yet no significant difference was found between patients and controls (data not shown).
When rheumatoid arthritis disease‐related factors were examined for a possible correlation with the presence of MetS, no significant differences were found regarding disease duration. Accordingly, in previous studies there was no relationship between disease duration and risk of CHD in patients with rheumatoid arthritis.21,25 It has been proposed that the increased risk of CHD may precede the clinical onset of rheumatoid arthritis,21 and that there is a preclinical phase of rheumatoid arthritis during which inflammatory activity and serological disturbances occur.22 Other disease‐related factors, such as extra‐articular manifestations and rheumatoid factor seropositivity, did not correlate with the presence of MetS.
In this study, a significant correlation between DAS28 and individual components of MetS—namely, systolic blood pressure (p = 0.01) and a trend towards an inverse correlation with HDL‐C (p = 0.056)—was found. Significantly more patients without MetS a had low DAS28 index (⩽3.2) compared with patients with MetS (16.5% v 2.8%, p = 0.003). A significant linear correlation (β = 0.237, p = 0.001) was found between the DAS28 and the number of MetS components present (fig 1). Moreover, in a multivariate model assessment, the risk of having moderate‐to‐high DAS28 was found to be significantly higher for patients with MetS compared with those without MetS components (p = 0.016) even after adjusting for rheumatoid arthritis treatments, demographics and behavioural factors (table 5). On the contrary, the use of biologicals, steroids or MTX was not associated with a higher risk of DAS28. Taken together, these data suggest that rheumatoid arthritis disease activity correlates with MetS, implicating a significant role for the inflammatory burden in the evolution of metabolic disturbances in patients with rheumatoid arthritis.
Metabolic syndrome is considered a proinflammatory state, whereby the expanded adipose tissue is thought to represent a source of proinflammatory cytokines (ie, interleukin 6 and tumour necrosis factor (TNF)α).26 There is increasing evidence that insulin resistance, the basic metabolic disturbance of MetS, is not only associated with the abundance of proinflammatory cytokines but also is the direct result of this burden.27,28 TNFα interferes with insulin signalling, both inducing down regulation of the insulin‐responsive glucose transporter expression in adipocytes and muscle,29 and reducing the tyrosine kinase activity of the insulin receptor.30 Our data, which correlate DAS28, a combined inflammatory index of rheumatoid arthritis, with MetS, further support the role of chronic inflammation in insulin resistance/MetS development.
The results of this study must be interpreted within the limitations of the methods, the major limitation being the small number of patients studied, especially men. Also, we did not have information on inflammatory markers such as hs‐CRP in both groups, and hence we were unable to directly examine the possible relationship of these factors with the presence of MetS. We cannot exclude the possibility of patient selection bias, as our centre is a tertiary referral centre. At the time of assessment, the vast majority of the patients (97.5%) were on treatment for rheumatoid arthritis and about 40% of them were taking anti‐TNF agents. Published reports support a favourable effect of anti‐TNF treatment on insulin resistance in patients with rheumatoid arthritis or ankylosing spondylitis, and an increase in HDL cholesterol.31,32 We can say that as almost 40% of our patients were on anti‐TNF treatment, we may have underestimated the prevalence of MetS. We are currently prospectively investigating the effect of anti‐TNF treatment on MetS.
In summary, the prevalence of MetS appears to be high—albeit comparable—in patients with rheumatoid arthritis and in controls. Rheumatoid arthritis disease activity correlates with the presence of MetS, implicating a role for inflammation in MetS. Larger studies are required to clarify the importance of MetS in this population.
Acknowledgements
We thank Professor Manolis Kogevinas for his invaluable assistance in the analysis of the data and for the critical review of the manuscript. We also thank Dr Styliani Maragaki, Charalambos Linardakis, Irini Flouri, Argyro Dimaka, RN, Zacharenia Papadopoulou, RN and Stauroula Lanara, RN, for patient care.
Abbreviations
BMI - body mass index
CHD - coronary heart disease
CVD - cardiovascular disease
DAS28 - disease activity score of 28 joints
EGIR - European Group for the Study of Insulin Resistance
ESR - erythrocyte sedimentation rate
HDL‐C - high‐density lipoprotein cholesterol
hs‐CRP - high‐sensitivity C reactive protein
LDL‐C - low‐density lipoprotein cholesterol
MetS - metabolic syndrome
NCEP ATPIII - Adult Treatment Panel III of the National Cholesterol Education Program
TNF - tumour necrosis factor
WHO - World Health Organization
Footnotes
Funding: This work was supported in part by a grant from the AutoCure project and the Hellenic Society of Rheumatology.
Competing interests: None declared.
References
- 1.Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol In Adults (Adult Treatment Panel III) Executive summary of the third report of The National Cholesterol Education Program (NCEP). JAMA 20012852486–2497. [DOI] [PubMed] [Google Scholar]
- 2.Haffner S M, Valdez R A, Hazuda H P, Mitchell B D, Morales P A, Stern M P. Prospective analysis of the insulin‐resistance syndrome (syndrome X). Diabetes 199241715–722. [DOI] [PubMed] [Google Scholar]
- 3.Reaven G M. Banting lecture 1988. Role of insulin resistance in human disease. Diabetes 1988371595–1607. [DOI] [PubMed] [Google Scholar]
- 4.Dekker J M, Girman C, Rhodes T, Nijpels G, Stehouwer C D, Bouter L M.et al Metabolic syndrome and 10‐year cardiovascular disease risk in the Hoorn Study. Circulation 2005112666–673. [DOI] [PubMed] [Google Scholar]
- 5.Najarian R M, Sullivan L M, Kannel W B, Wilson P W, D'Agostino R B, Wolf P A. Metabolic syndrome compared with type 2 diabetes mellitus as a risk factor for stroke: the Framingham Offspring Study. Arch Intern Med 2006166106–111. [DOI] [PubMed] [Google Scholar]
- 6.Hanley A J, Karter A J, Festa A, D'Agostino R, Jr, Wagenknecht L E, Savage P.et al Factor analysis of metabolic syndrome using directly measured insulin sensitivity: The Insulin Resistance Atherosclerosis Study. Diabetes 2002512642–2647. [DOI] [PubMed] [Google Scholar]
- 7.Daskalopoulou S S, Mikhailidis D P, Elisaf M. Prevention and treatment of the metabolic syndrome. Angiology 200455589–612. [DOI] [PubMed] [Google Scholar]
- 8.World Health Organization Definition, diagnosis and classification of diabetes mellitus: report of a WHO consultation. Part 1: Diagnosis and classification of diabetes mellitus. Geneva, Switzerland: World Health Organization (WHO/NCD/NCS/99, 2) 1999
- 9.Balkau B, Charles M A. Comment on the provisional report from the WHO consultation. European Group for the study of Insulin Resistance (EGIR). Diabet Med 199916442–443. [DOI] [PubMed] [Google Scholar]
- 10.Stevens R J, Douglas K M, Saratzis A N, Kitas G D. Inflammation and atherosclerosis in rheumatoid arthritis. Expert Rev Mol Med 200571–24. [DOI] [PubMed] [Google Scholar]
- 11.del Rincon I D, Williams K, Stern M P, Freeman G L, Escalante A. High incidence of cardiovascular events in a rheumatoid arthritis cohort not explained by traditional cardiac risk factors. Arthritis Rheum 2001442737–2745. [DOI] [PubMed] [Google Scholar]
- 12.Gabriel S E, Crowson C S, O'Fallon W M. Comorbidity in arthritis. J Rheumatol 1999262475–2479. [PubMed] [Google Scholar]
- 13.McEntegart A, Capell H A, Creran D, Rumley A, Woodward M, Lowe G D. Cardiovascular risk factors, including thrombotic variables, in a population with rheumatoid arthritis. Rheumatology (Oxford) 200140640–644. [DOI] [PubMed] [Google Scholar]
- 14.Dessein P H, Stanwix A E, Joffe B I. Cardiovascular risk in rheumatoid arthritis versus osteoarthritis: acute phase response related decreased insulin sensitivity and high‐density lipoprotein cholesterol as well as clustering of metabolic syndrome features in rheumatoid arthritis. Arthritis Res 20024R5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dessein P H, Joffe B I, Stanwix A, Botha A S, Moomal Z. The acute phase response does not fully predict the presence of insulin resistance and dyslipidemia in inflammatory arthritis. J Rheumatol 200229462–466. [PubMed] [Google Scholar]
- 16.Xu H, Barnes G T, Yang Q, Tan G, Yang D, Chou C J.et al Chronic inflammation in fat plays a crucial role in the development of obesity‐related insulin resistance. J Clin Invest 20031121821–1830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Festa A, D'Agostino R, Jr, Howard G, Mykkanen L, Tracy R P, Haffner S M. Chronic subclinical inflammation as part of the insulin resistance syndrome: the Insulin Resistance Atherosclerosis Study (IRAS). Circulation 200010242–47. [DOI] [PubMed] [Google Scholar]
- 18.Arnett F C, Edworthy S M, Bloch D A, McShane D J, Fries J F, Cooper N S.et al The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum 198831315–324. [DOI] [PubMed] [Google Scholar]
- 19.WHO Expert Committee Physical status: the use and interpretation of anthropometry. WHO Tech Rep Ser 1995854424–438. [PubMed] [Google Scholar]
- 20.van Gestel A M, Haagsma C J, van Riel P L. Validation of rheumatoid arthritis improvement criteria that include simplified joint counts. Arthritis Rheum 1998411845–1850. [DOI] [PubMed] [Google Scholar]
- 21.Maradit‐Kremers H, Crowson C S, Nicola P J, Ballman K V, Roger V L, Jacobsen S J.et al Increased unrecognized coronary heart disease and sudden deaths in rheumatoid arthritis: a population‐based cohort study. Arthritis Rheum 200552402–411. [DOI] [PubMed] [Google Scholar]
- 22.Nielen M M, van Schaardenburg D, Reesink H W, van de Stadt R J, van der Horst‐Bruinsma I E, de Koning M H.et al Specific autoantibodies precede the symptoms of rheumatoid arthritis: a study of serial measurements in blood donors. Arthritis Rheum 200450380–386. [DOI] [PubMed] [Google Scholar]
- 23.Panagiotakos D B, Pitsavos C, Chrysohoou C, Skoumas J, Tousoulis D, Toutouza M.et al Impact of lifestyle habits on the prevalence of the metabolic syndrome among Greek adults from the ATTICA study. Am Heart J 2004147106–112. [DOI] [PubMed] [Google Scholar]
- 24.Grundy S M, Cleeman J I, Daniels S R, Donato K A, Eckel R H, Franklin B A.et al Diagnosis and management of the metabolic syndrome: an American Heart Association/National Heart, Lung, and Blood Institute scientific statement. Curr Opin Cardiol 2006211–6. [DOI] [PubMed] [Google Scholar]
- 25.Maradit‐Kremers H, Nicola P J, Crowson C S, Ballman K V, Gabriel S E. Cardiovascular death in rheumatoid arthritis: a population‐based study. Arthritis Rheum 200552722–732. [DOI] [PubMed] [Google Scholar]
- 26.Trayhurn P, Wood I S. Adipokines: inflammation and the pleiotropic role of white adipose tissue. Br J Nutr 200492347–355. [DOI] [PubMed] [Google Scholar]
- 27.Neuschwander‐Tetri B A, Caldwell S H. Nonalcoholic steatohepatitis: summary of an AASLD single topic conference. Hepatology 2003371202–1219. [DOI] [PubMed] [Google Scholar]
- 28.Eckel R H, Grundy S M, Zimmet P Z. The metabolic syndrome. Lancet 20053651415–1428. [DOI] [PubMed] [Google Scholar]
- 29.Stephens J M, Pekala P H. Transcriptional repression of the GLUT4 and C/EBP genes in 3T3‐L1 adipocytes by tumor necrosis factor‐alpha. J Biol Chem 199126621839–21845. [PubMed] [Google Scholar]
- 30.Hotamisligil G S, Budavari A, Murray D, Spiegelman B M. Reduced tyrosine kinase activity of the insulin receptor in obesity‐diabetes. Central role of tumor necrosis factor‐alpha. J Clin Invest 1994941543–1549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kiortsis D N, Mavridis A K, Vasakos S, Nikas S N, Drosos A A. Effects of infliximab treatment on insulin resistance in patients with rheumatoid arthritis and ankylosing spondylitis. Ann Rheum Dis 200564765–766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Vis M, Nurmohamed M T, Wolbink G, Voskuyl A E, de Koning M, van de S R.et al Short term effects of infliximab on the lipid profile in patients with rheumatoid arthritis. J Rheumatol 200532252–255. [PubMed] [Google Scholar]