Abstract
Parasitic nematode infections of humans and livestock continue to impose a significant public health and economic burden worldwide. Murine models of intestinal nematode infection have proved to be relevant and tractable systems to define the cellular and molecular basis of how the host immune system regulates resistance and susceptibility to infection. While susceptibility to chronic infection is propagated by T helper cell type 1 cytokine responses (characterised by production of IL-12, IL-18 and interferon-γ), immunity to intestinal-dwelling adult nematode worms is critically dependent on a type 2 cytokine response (controlled by CD4+T helper type 2 cells that secrete the cytokines IL-4, IL-5, IL-9 and IL-13). However, the immune effector mechanisms elicited by type 2 cytokines in the gut microenvironment that precipitate worm expulsion have remained elusive. This review focuses on new studies that implicate host intestinal epithelial cells as one of the dominant immune effector cells against this group of pathogens. Specifically, three recently identified type 2 cytokine-dependent pathways that could offer insights into the mechanisms of expulsion of parasitic nematodes will be discussed: (i) the intelectins, a new family of galactose-binding lectins implicated in innate immunity, (ii) the resistin-like molecules, a family of small cysteine-rich proteins expressed by goblet cells, and (iii) cytokine regulation of intestinal epithelial cell turnover. Identifying how the mammalian immune response fights gastrointestinal nematode infections is providing new insights into host protective immunity. Harnessing these discoveries, coupled with identifying what the targets of these responses are within parasitic nematodes, offers promise in the design of a new generation of anti-parasitic drugs and vaccines.
Keywords: Parasitic nematodes, Immune effector mechanisms, Intelectin, Resistin-like molecules, Intestinal epithelial cells
1. Introduction
Soil transmitted helminthiases continue to be the most prevalent chronic infectious diseases of humans, with an estimated 2 billion individuals infected worldwide (Colley et al., 2001). The major public health significance and economic impact of this group of pathogens is hard to quantify, although the World Health Organisation (WHO) has estimated that more than 1000 million people world-wide are infected with one or more of the major pathogen species of humans: Trichuris trichiura, Ascaris lumbriciodes and the hookworms Necator americanus and Ancylostoma duodenalis and that 39 million disability-adjusted life years (DALYs) are attributable to these four nematode species alone (Chan et al., 1994; Chan, 1997; Albonico et al., 1999; WHO, 2001). The most severe clinical symptoms of infection, including protein-losing enteropathy, chronic dysentery, anemia and rectal prolapse occur in the minority of heavily infected individuals (Symons, 1969; Bundy and Cooper, 1989; Grencis and Cooper, 1996; Albonico et al., 1999). However, significant detrimental clinical outcomes also occur in moderately infected individuals, including impaired nutritional status, growth retardation and lower educational achievement (Callender et al., 1992; Nokes et al., 1992; Nokes and Bundy, 1994). Beyond the direct consequences of infection, there is growing evidence from infected humans and murine model systems that chronic helminth infection can have a detrimental effect on the ability of individuals to respond to vaccination and can influence responses to other infections including malaria (Cooper et al., 2001; Chenine et al., 2005; Graham et al., 2005).
Intestinal nematode parasites are also a serious economic concern in the livestock industry. Haemonchus, Trichostrongylus and Ostertagia are the main disease-causing parasites, contributing significantly to reduced productivity and costing approximately US$1.5 billion annually in repeated anthelminthic treatments to control infections (Newton and Munn, 1999; Newton and Meeusen, 2003). Widespread reports of anthelminthic resistance and the growing costs of developing new anthelminthic drugs (Albonico et al., 2004; Lochnit et al., 2005) require the development of more long-lived immunological intervention strategies. The development of immunologically defined laboratory models of intestinal nematode infection such as Trichinella spiralis, Nippostrongylus brasiliensis, Heligmosomoides polygyrus and Trichuris muris has facilitated new insights into the immunological basis of susceptibility and resistance to infection under controlled laboratory conditions. By employing these nematode infection models, advances have been made in understanding the cellular and molecular interactions involved in the generation and regulation of immune responses during infection. Despite unique aspects of phylogeny, biochemistry, morphology and lifecycle strategies, a consensus has emerged that immunity to intestinal-dwelling nematode worms is critically dependent on a type 2 cytokine response (controlled by CD4+T helper type 2 cells that secrete the cytokines IL-4, IL-5, IL-9 and IL-13), while susceptibility to chronic infection is propagated by type 1 cytokine responses (characterised by production of IL-12, IL-18 and interferon (IFN)-γ) [for recent reviews, see (Finkelman et al., 1997; Maizels and Yazdanbakhsh, 2003; Cliffe and Grencis, 2004; Hayes et al., 2004; Maizels et al., 2004)]. IL-4 and IL-13, acting through shared and distinct pathways that are dependent on the type 2 cytokine-associated transcription factor STAT6, appear to be the most critical cytokines for immunity; while IL-4 stimulates fluid secretion in the gut, IL-13 promotes goblet cell responses and both cytokines induce smooth muscle contraction (McKenzie et al., 1998; Urban et al., 1998, 2001; Akiho et al., 2002; Zhao et al., 2003; Finkelman et al., 2004; Madden et al., 2004). Recent studies in livestock and human populations support a role for type 2 cytokines in host resistance to infection (Jackson et al., 2004; Pernthaner et al., 2005) and provide important indications that murine hosts are appropriate systems to model immune regulation of parasitic nematode infection.
Although the requirement for type 2 cytokines in worm expulsion is well established, the immune effector cells and molecular mechanisms elicited by this type of immune response have remained elusive. In the case of T. spiralis, clear evidence exists that mast cells can play an important role in worm expulsion (Knight et al., 2000, 2002; Brown et al., 2003; McDermott et al., 2003). However, expulsion of a number of intestinal nematode parasites—while dependent on type 2 cytokines—can occur in the absence of classical effector mechanisms associated with type 2 responses such as B cell activation, antibody production, eosinophilia and mastocytosis (Else and Finkelman, 1998; Betts and Else, 1999; Betts et al., 2000). In recent years this has led a number of groups to adopt global gene profiling and proteomic analyses of intestinal responses associated with worm expulsion in an attempt to identify novel type 2 cytokine-dependent immune effector mechanisms involved in immunity. In our own gene profiling studies of the intestinal response following T. muris infection, we found 171 genes that were upregulated greater than 1.5-fold over uninfected controls. Of the 10 genes exhibiting the most marked infection-induced upregulation, half of these were gut epithelial cell-specific genes (see Table 1). Specifically, intelectin, a family of calcium-dependent galactose-binding lectins (Komiya et al., 1998) and calcium-activated chloride channel 3, an epithelial ion channel implicated in mucus secretion (Gruber et al., 1998; Gyomorey et al., 2001; Leverkoehne and Gruber, 2002), were the two most highly upregulated genes. Pancreatic lipase-related protein 2 and pancreatic co-lipase, both members of the lipase family (Lowe, 2000) were also highly expressed. Resistin-like molecule-β,a small cysteine-rich goblet cell specific protein (Steppan et al., 2001), was in the top five epithelial-specific genes upregulated following T. muris infection. Interestingly, none of these dominant epithelial cell-specific genes had previously been associated with type 2 cytokine responses that are expressed around the period of worm expulsion.
Table 1.
Global gene profiling of intestinal epithelial gene expression following Trichuris muris infection of genetically resistant mice
| Gene | Fold induction |
|---|---|
| Intelectin | 83.63a |
| Mast cell protease 2 | 35.57 |
| Chloride channel calcium activated 3 | 10.56a |
| Pancreatic lipase-related protein 2 | 9.12a |
| Serine (or cysteine) proteinase inhibitor, clade F, member1 | 9.05 |
| Resistin like beta | 8.20a |
| Colipase, pancreatic | 6.07a |
| Carboxypeptidase A3, mast cell | 4.90 |
| Resistin like alpha | 4.82 |
| Fanconi anemia, complementation group C | 4.29 |
Genetically resistant BALB/c mice were infected with 150 T. muris eggs as previously described (Artis et al., 2002, 2004a,b). At day 18 p.i., a time point coincident with maximal type 2 cytokine responses and worm expulsion, RNA was isolated from naiï ve or infected intestinal tissue and microarray analysis carried out using a Massachusetts General Hospital mouse oligo chip containing 14,611 70-mer oligos. Spots were measured on an Agilent scanner and quantified by GenePix software. Numbers refer to fold induction of gene expression over nai ïve controls (four animals per group).
Denotes known intestinal epithelial gene.
In an independent microarray study, Knight and colleagues isolated epithelial cells from the intestine of naïve and T. spiralis-infected mice around the period of worm expulsion and identified 216 genes that were upregulated in the epithelial cell compartment; these included goblet cell genes such as resistin-like molecule-β (RELMβ) and other epithelial genes implicated in barrier function, ion exchange, tissue repair and metabolism (Knight et al., 2004). In elegant proteomic analyses of purified epithelial cells, Pemberton et al. demonstrated that the most marked change in protein expression within epithelial cells around the period of expulsion of T. spiralis was that of a newly described gene product, intelectin-2 (Pemberton et al., 2004b). More recently, Datta et al. performed similar analyses of epithelial gene expression around the period of expulsion of T. muris using a custom array containing 9000 genes. Many of the genes that were upregulated in the large intestinal epithelium were similar to those outlined in Table 1, including intelectin, calcium-activated chloride channel 3 and pancreatic lipase-related protein 2, as well as genes such as angiogenin-like protein and angiogenin-related protein (Datta et al., 2005). Therefore, in three independent studies using either T. muris or T. spiralis infection, a number of common epithelial-intrinsic genes were upregulated around the period of worm expulsion, specifically, intelectins, resistin-like molecules and machinery associated with ion exchange and barrier function.
The magnitude of the epithelial response highlighted by these microarray studies and the consistent pattern of gene expression suggest that epithelial cells themselves could be regarded as an immune effector cell against intestinal nematode parasites. Moreover, these results implicate the gene products expressed in epithelial cells in the coordinated host response to infection and potentially in contributing to worm expulsion and/or tissue repair. This review will focus on two of the most abundantly expressed and best-characterised gene products that were upregulated in the microarray results described above: (i) the intelectins, and (ii) the resistin-like molecules. In addition, a novel mechanism of worm expulsion involving immune-mediated changes in epithelial cell turnover will be discussed.
2. The intelectins
Lectins are found in mammals as either surface-bound receptors or in plasma where they exist as opsonins or agglutinins. This large family of molecules is conserved in evolution and performs a number of biological functions, including regulation of cell proliferation, recognition of tumor antigens and innate recognition of carbohydrates present in pathogen cell walls (Apostolopoulos and McKenzie, 2001; East and Isacke, 2002; Wang et al., 2004; Elola et al., 2005; Iwanaga and Lee, 2005). The mannose receptor is a well-characterised mammalian lectin that recognises mannosyl-containing residues of carbohydrates found in bacterial cell walls. Mannose receptors are found on phagocytic cells and are thought to play a critical role in innate immunity by enhancing uptake of bacteria (Ezekowitz et al., 1990; Stahl and Ezekowitz, 1998). Rather than binding mannose-rich subunits, the intelectin family is composed of calcium-dependent galactose-binding lectins. Intelectin-1 was first identified by sequence homology to the Xenopus egg lectin XL35. The frog egg lectin is released upon fertilisation of the egg, whereupon it binds to galactose-containing residues in the mucin covering the egg, precipitating hardening of the egg and preventing polyspermy (Nishihara et al., 1986). Subsequently, murine intelectin-1 was found to be expressed in paneth cells in the small intestine and was hypothesised to play a role in innate defense against bacterial infections (Komiya et al., 1998). Using both reverse transcription (RT)-PCR and proteomic approaches, a novel intelectin variant was identified in mice called intelectin-2, that exhibited 91% sequence identity to intelectin-1 and was found only in the small intestine (Pemberton et al., 2004a). There are also two known intelectins in the human genome: intelectin-1 (also called lactoferrin receptor) exists as a 120 kDa disulfide-linked homotrimer expressed in the thymus, heart and intestine, while human intelectin-2 expression is restricted to the small intestine (Lee et al., 2001; Suzuki et al., 2001; Tsuji et al., 2001). Studies demonstrating that recombinant human intelectin could bind to galactofuranosyl residue-containing molecules isolated from the bacterium Nocardia, support a role for intelectins in innate recognition of bacteria (Tsuji et al., 2001).
As discussed above, gene profiling analysis by a number of groups independently identified intelectin expression as a hallmark of parasitic nematode infection in the gastrointestinal (GI) tract. Subsequently, intelectin-1 expression was shown to be constitutive in the small intestine by two-dimensional gel electrophoretic studies, while upregulation of intelectin-2 was the most marked change in protein expression in the intestinal epithelium following infection with T. spiralis (Pemberton et al., 2004b). Critically, maximal intelectin-2 expression occurred around day 14 p.i., coincident with expression of type 2 cytokines and worm expulsion. These results were subsequently confirmed by RT-PCR and immunohistochemical staining identified paneth cells and goblet cells as the cellular source of intelectins following infection (Pemberton et al., 2004a). Gene profiling of intestinal responses following infection with T. muris also identified intelectin as the gene that exhibited most robust upregulation in genetically resistant mouse strains around the period of worm expulsion (see Table 1; Datta et al., 2005). We were able to confirm the microarray expression data by RT-PCR, finding that following T. muris infection, both intelectin-1 and intelectin-2 mRNA transcription were upregulated in the intestines of genetically resistant mice around the period of worm expulsion (Fig. 1A). In contrast, T. muris infection of the genetically susceptible AKR mouse strain was not associated with expression of either intelectin (unpublished observations). Immunohistochemical analysis demonstrated that there was little intelectin protein expression in the intestine of naïve mice (Fig. 1B) and identified goblet cells as the cellular source of intelectins following T. muris infection (Fig. 1C). Intelectins were localised to the exocrine vesicles within goblet cells (Fig. 1D), suggesting they are targeted for secretion into the intestinal lumen. The dominant expression of intelectins following T. spiralis and T. muris infections, coupled with the differential expression of intelectins in resistant and susceptible mouse strains following T. muris infection, implicate this family of glycoproteins in anti-nematode responses. Supporting this contention, Pemberton et al. recently found that certain inbred mouse strains naturally lack expression of the intelectin-2 gene. While most mouse strains express both intelectin-1 and intelectin-2 and rapidly expel T. spiralis, two strains that lack intelectin-2 (C57BL6 andC57BL10) exhibit a significant delay in worm expulsion (Pemberton et al., 2004a). While the delayed expulsion of T. spiralis in these mice is likely to be a multifactorial event and is unlikely to be due exclusively to the absence of intelectin-2, these results support a potential function for intelectins in contributing to rapid expulsion and host resistance to infection.
Fig. 1.
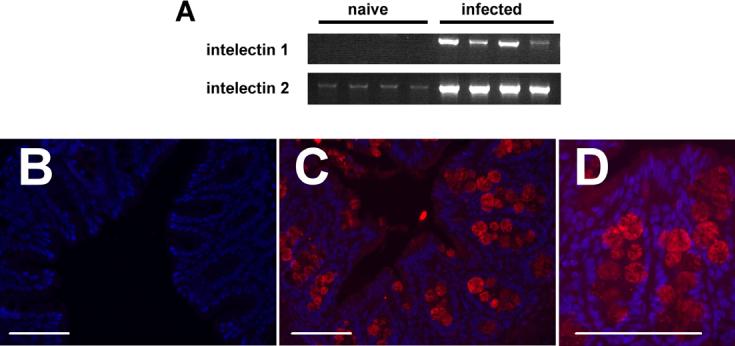
Intelectins are expressed by intestinal goblet cells following Trichuris muris infection. Genetically resistant BALB/c mice were infected with 150 T. muris eggs and cecal RNA or histologic sections prepared from naïve or infected animals on day 18 p.i. as previously described (Artis et al., 2002, 2004a,b). (A) reverse transcription-PCR analysis demonstrated robust upregulation of intelectin-1 and intelectin-2 in infected animals (details of primer sequences and methodology can be found in (Pemberton et al., 2004a)). (B)-(D) Immunofluorescent staining of intelectins (staining red) in naive (B) or infected mice (C) and (D). Paraformaldehyde-fixed tissues were wax-embedded and 5 μm sections stained using a polyclonal rabbit anti-intelectin1/2 antibody (a gift from Hugh Miller, University of Edinburgh, UK) following standard immunofluoresecence protocols. Intelectins are expressed within exocrine vesicles of intestinal goblet cells (D). Epithelial cell nuclei are stained in blue with DAPI. Blocking peptide confirmed specificity of staining. Bar, 100 μm.
The abundant expression of intelectins in the intestinal epithelium and their localisation to secretory paneth and goblet cells in the gut suggest they are likely to be present in the intestinal lumen at high concentrations around the period of worm expulsion. Given that intelectins bind galactofuranosyl-containing residues in bacterial cell walls, intelectins may bind similar carbohydrate residues in parasitic nematodes and impair worm fitness, potentially through influencing attachment and/or feeding behaviour or essential biological processes associated with cell proliferation and death that would render the worms more susceptible to immune-mediated expulsion. However, to date there is no evidence that intelectins directly bind to parasitic nematodes and the possibility that elevated intelectin expression following nematode infections in the GI tract may be secondary to enhanced exposure of the host to bacterial symbionts present in the gut lumen cannot be ruled out at present. Notwithstanding that, intelectins may contribute to anti-nematode responses indirectly, through binding to and altering the properties of other mucin glycoproteins that are secreted by intestinal goblet cells. Analogous to the Xenopus egg lectin, XL35, which binds to other glycoproteins resulting in hardening of the egg (Nishihara et al., 1986), there may be a similar cross-linking function for mammalian intelectins in the gut resulting in the formation of a ‘glycoprotein cement’ around the nematodes that precipitates their expulsion from the host. A recent study demonstrated that in addition to exposure to nematode parasites, intelectins are also expressed following Th2 cytokine production associated with airway inflammation (Kuperman et al., 2005). Therefore, in addition to potential direct effects on nematodes, the intelectins may also perform broader functions during type 2 inflammation including immunoregulation or tissue remodeling. Taken together, a role for the intelectins in anti-nematode responses is an attractive one that will require further investigation to define the functional contribution of this family of molecules in type 2 responses and host protective immunity.
3. Resistin-like molecules (RELMs)
Resistin (also called ADSF or found in inflammatory zone 3 (FIZZ3)) is a recently discovered 12.5 kDa adipocyte-derived hormone that plays a critical role in antagonising glucose homeostasis and insulin sensitivity (Holcomb et al., 2000; Kim et al., 2001; Steppan et al., 2001; Steppan and Lazar, 2004). Three other members of the resistin-like molecule family (RELMα (FIZZ1), RELMβ (FIZZ2), and RELMγ) were subsequently identified on the basis of an unusual and conserved C-terminal cysteine-rich domain (Holcomb et al., 2000; Steppan et al., 2001; Gerstmayer et al., 2003). The resistin gene family also exhibits unusual structural features including the formation of cysteine-rich multimeric structures that are conserved in all mammalian genomes analysed so far (Patel et al., 2004). Similar to the pattern of resistin expression, RELMα is expressed abundantly in adipocytes as well as the heart and lungs of naïve mice. It is also expressed at high levels in pulmonary epithelial cells, alveolar pneumocytes and the bronchoalveolar lavage fluid in a mouse model of pulmonary inflammation (Holcomb et al., 2000). Given its widespread distribution, it is not surprising that a number of functions have been proposed for RELMα including antagonising the action of insulin, inhibition of nerve growth factor-mediated gene expression of dorsal root ganglia, inhibition of adipocyte differentiation and stimulation of myofibroblast differentiation and expression of type I collagen (Holcomb et al., 2000; Blagoev et al., 2002; Rajala et al., 2003; Liu et al., 2004a). In contrast to the relatively broad expression of RELMα, RELMβ (FIZZ2) is expressed exclusively in goblet cells in the lung and GI tract (Steppan et al., 2001; Zimmermann et al., 2004) while RELMγ appears to exhibit species-specific expression patterns and can be found in nasal respiratory epithelial cells and hematopoietic cells (Gerstmayer et al., 2003; Schinke et al., 2004).
A number of research groups have identified RELMs as a dominant component of type 2 cytokine-dependent responses following nematode infection (see above). Loke et al. were the first to identify RELMα as one of the most abundantly expressed transcripts in alternatively activated macrophages that are elicited in the peritoneum following implantation of the tissue-dwelling filarial nematode, Brugia malayi (Loke et al., 2002). Alternatively activated macrophages express activation markers, although unlike their ‘classically’ activated counterparts that are promoted by proinflammatory cytokines like IFN-γ and perform microbicidal functions, alternatively activated macrophages fail to upregulate expression of inducible nitric oxide synthase. Rather, alternatively activated macrophages express high levels of arginase and have been implicated in immune suppression, responses to tissue-dwelling helminth parasites and tissue remodeling associated with type 2 cytokine responses (Mantovani et al., 2002; Raes et al., 2002a,b; Gordon, 2003; Nair et al., 2003; Noel et al., 2004; Park-Min et al., 2005; Rauh et al., 2005). RELMα expression in macrophages was found to be dependent on IL-4 and represented 2% of total mRNA in B. malayi-elicited alternatively activated macrophages (Loke et al., 2002; Maizels et al., 2004; Nair et al., 2005). This level of expression was second only to YM-1, a novel protein that shares homology to mammalian chitinases and has been implicated in cell-cell interactions and immune cell chemotaxis (Jin et al., 1998; Owhashi et al., 1998, 2000; Chang et al., 2001; Sun et al., 2001). Elevated expression of RELMα was also reported in the thoracic cavity of Litomosoides sigmodontis-infected mice (Nair et al., 2005) and was also recently identified as a dominant response in the gut following infection with the GI nematode parasites T. spiralis, N. brasiliensis and T. muris (Knight et al., 2004; Nair et al., 2005; Wang et al., 2005; and see Table 1). Therefore, expression of RELMα appears to be a critical component of the programmed profile of gene expression associated with multiple nematode infections in diverse anatomical sites.
Similarly, expression of RELMβ was found to be a common response in the GI tract following infection with T. spiralis, N. brasiliensis and T. muris (Artis et al., 2004b). Critically, maximal expression of RELMβ was associated with expression of type 2 cytokines and worm expulsion following all three infections despite differences in lifecycle strategies and biological niches favoured by these different species (Artis et al., 2004b). In the case of T. muris infection, RELMβ expression was only observed following infection of resistant mouse strains that develop type 2 cytokine responses but not in animals susceptible to chronic infections (Artis et al., 2004a,b;Wang et al., 2005), demonstrating that RELMβ expression is not a generic response to nematode infection; rather, its expression is restricted to environments in which protective type 2 responses dominate. Consistent with a restricted expression profile, there was no demonstrable upregulation of RELMβ mRNA or protein following protozoan infection in the GI tract (Artis et al., 2004b). Unlike RELMα that appears to be expressed in macrophages or epithelial cells in the GI tract, RELMβ expression appears to be restricted to goblet cells (Artis et al., 2004b) (see Fig. 2).
Fig. 2.

Resistin-like molecule (RELM)-β is expressed exclusively in intestinal goblet cells following T. muris infection. Genetically resistant BALB/c mice were infected with 150 T. muris eggs and histologic sections prepared from naïve or infected animals on day 18 p.i. as previously described (Artis et al., 2002, 2004a,b). Paraformaldehyde-fixed tissues were wax-embedded and 5 mm sections stained using a polyclonal rabbit anti-RELMβ antibody (Peprotech, USA) following standard immunofluoresecence protocols. (A) Immunofluorescent staining revealed that RELMβ (staining green) is localised to intestinal goblet cells. (B) RELMβ is expressed exclusively within exocrine vesicles of intestinal goblet cells. Epithelial cell nuclei are stained in blue with DAPI. Blocking peptide confirmed specificity of staining. Bar, 50 μm.
In vitro studies demonstrated that IL-4 and IL-13 could directly induce expression of RELMβ mRNA in a goblet celllike intestinal epithelial cell line (He et al., 2003; Artis et al., 2004b). Further, administration of rIL-13 to normallysusceptible AKR mice following infection with T. muris resulted in enhanced RELMβ expression and worm expulsion while blockade of IL-4 receptor signalling in normally resistant strains inhibited RELMβ responses (Artis et al., 2004b). Consistent with a role for type 2 cytokines in regulating expression, the RELMβ promoter contains numerous binding sites for the type 2 cytokine-associated transcription factor STAT6. Early reports suggested RELMβ expression was restricted to the GI tract, however it now clear that cells in the lung express this family member, either following infection with N. brasiliensis or the induction of type 2 cytokine-dependent airway inflammation (Zimmermann et al., 2004; Nair et al., 2005). There is limited analysis of RELMγ expression, although nematode infection-induced upregulation of RELMγ has been reported in the GI tract following T. muris infection (Wang et al., 2005).
The picture that emerges is one in which expression of RELMs occurs in multiple tissues following exposure to diverse nematode parasites. So what might be the functions of this family of molecules? There are a number of possibilities, including direct anti-parasitic effects, tissue repair and immune regulation. Evidence for a possible role for RELMβ as an effector molecule that directly targets nematode parasites comes from studies with T. muris and Strongyloides stercoralis. RELMβ binds specifically to structures associated with the lateral alae in both parasites. In the case of T. muris, the lateral alae are modified into a structure called the bacillary band (Fig. 3A and B). This band is composed of numerous epidermal glandular and non-glandular cells, some of which contain dendritic processes that run between pore cells and adjacent nerve cells and are implicated in neuro- and/or chemo-sensory functions (Gibbons, 2002; see Fig. 3C). In S. stercoralis, RELMβ binds to the lateral alae that overlie nerve-rich lateral cords and in vitro assays showed that RELMβ could inhibit nematode chemotaxis towards host tissue extract (Artis et al., 2004b). Therefore, coupled with other effector mechanisms, a possible function for RELMβ may be to disorient nematode parasites in their GI niche and contribute to their expulsion. A disorientation function for RELMs that might contribute to worm expulsion is supported by the demonstration that RELM family members can bind to neurons and influence their function in vertebrates (Holcomb et al., 2000). Further, it is noteworthy that many current anti-nematode drugs target worm neuro- and chemo-sensory functions, perhaps via a common pathway targeted by host-derived RELMs.
Fig. 3.
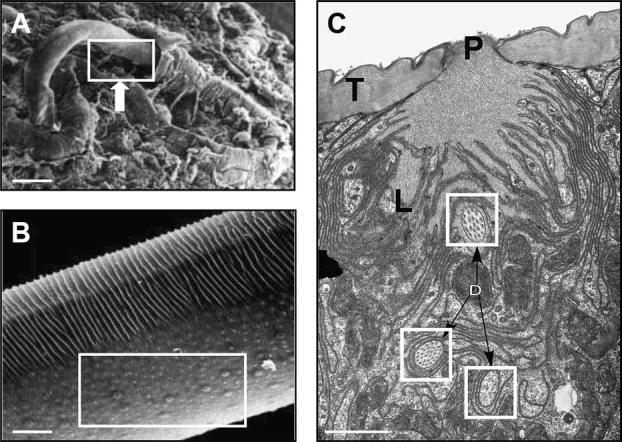
Ultrastructural analysis of the bacillary pores of T. muris. (A) SEM of T. muris entering host intestinal epithelial cells in infected mice. Bar, 100 μm. Genetically resistant BALB/c mice were infected with 150 T. muris eggs and tissue sections prepared from naïve or infected animals on day 14 p.i. as previously described (Artis et al., 2004a; Tilney et al., 2005). (B) Higher power analysis revealed the localisation of the bacillary pores to the ventral surface of the parasites (box). Bar, 10 μm. (C) TEM demonstrated that a subset of pores contains numerous dendritic processes (boxes) from adjoining adjacent nerve cells. T, tegument; P, pore opening, L, actin rich plical structures surrounding the pore opening. Bar, 1 μm. (Adapted, with permission, from Tilney et al., 2005.)
No receptors for any of the RELMs have been identified in mammalian genomes to date. However, it is possible that in addition to an anti-parasitic function, RELMs may also perform an autocrine or paracrine function on other mammalian cells. For instance, local expression of RELMβ in the lung and GI tract may influence cells of the innate and adaptive immune system to augment type 2 cytokine responses or sensitise the tissue to these cytokines and so promote host protective immunity. In addition, the putative role for RELMα in deposition of alpha smooth muscle actin and collagen in a lung model of fibrosis (Liu et al., 2004a), suggest RELMα may play a critical role in wound healing and tissue remodeling following nematode infection. Indeed, this repair response may also have a protective anti-parasitic effect, functioning to encapsulate nematode parasites or to alter the tissue, making it less permissive to chronic nematode infections. Nair et al. recently suggested an immuno-regulatory role for RELMs. In addition to expression in macrophages within the GI tract, RELMα is also expressed in B cells, dendritic cells and macrophages in the lymph nodes following exposure to B. malayi (Nair et al., 2005). Furthermore, in vitro activation with IL-4 induced RELMα expression in B cells, bone marrow-derived DC and bone marrow-derived macrophages (Nair et al., 2005). Therefore, in addition to potential anti-parasite activity and influencing tissue remodeling in the GI micro-environment, these studies highlight a possible role for RELMα in regulating innate and adaptive immune responses in secondary lymphoid tissues. The ability of RELMs to activate mitogen-activated protein kinases and suppressor-of-cytokine-signalling pathways (Kushiyama et al., 2005; Steppan et al., 2005) is consistent with their ability to influence host immune response genes. Supporting a broader regulatory role for this family of proteins, recent studies have highlighted that expression of RELMs is not restricted to nematode infection. For example, RELMα is highly expressed during chronic murine trypanosomiasis and in Schistosoma mansoni egg-induced granulomas (Raes et al., 2002a,b; Sandler et al., 2003; Noel et al., 2004). In addition to other infectious diseases, expression of RELMα and RELMβ has been reported in other inflammatory diseases including airway inflammation and cystic fibrosis (Stutz et al., 2003; Liu et al., 2004b; Norkina et al., 2004; Zimmermann et al., 2004). A dominant type 2 cytokine response characterises these conditions and although the function of each RELM family member is likely to be dictated by the infectious/inflammatory stimuli and tissue-specific location, these studies collectively implicate RELM proteins as effector and/or regulatory molecules in type 2 inflammation. The development of new reagents to block or overexpress the RELM proteins should provide important new insights into their biological functions.
4. Cytokine regulation of epithelial cell turnover: the ‘epithelial escalator’ model
In addition to profound changes in gene expression following exposure to nematode infection (Li et al., 1998; Knight et al., 2004; Datta et al., 2005; and Table 1), host intestinal epithelial cells also exhibit dramatic alterations in their proliferative behaviour, at least some of which are regulated by the immune system. The mammalian intestinal epithelium undergoes continuous and rapid homeostatic renewal and within each crypt unit forms a highly polarised proliferative hierarchy (Li et al., 1994; Potten, 1998; Marshman et al., 2002). Anchored pluripotent stem cells are found at the base of the crypts and following asymmetric division give rise to transit daughter cells that undergo continuous linear migration lumenally from the base of the crypts to the extrusion zone at the luminal surface. In the process of migration, epithelial cells undergo maturation and differentiation into a number of lineages including absorptive enterocytes, endocrine cells, M cells and goblet cells (Marshman et al., 2002). Proliferative indices suggest epithelial cells lose their ability to proliferate as they migrate with the bulk of mature differentiated cells undergoing apoptosis at the luminal surface. The balance of cell proliferation, migration and death is critical in maintaining epithelial integrity and tissue morphology in the gut.
A number of studies have demonstrated that epithelial cell proliferation can be severely disrupted following infection with parasitic nematodes such as N. brasiliensis, S. stercoralis, and Trichostrongylus colubriformis, implicating this dysregulated epithelial response in the crypt hyperplasia and villous atrophy commonly reported following nematode infection (Symons, 1978; Hoste, 1989; Hoste and Mariana, 1989; Artis et al., 1999). In the case of T. muris, cell position-linked proliferation analysis demonstrated that chronic infection of susceptible mouse strains was associated with a significant increase in the frequency of proliferating stem cells and transit cells in each crypt (Artis et al., 1999). The accumulation of proliferating epithelial cells during chronic infection suggested that either the epithelium was undergoing elevated proliferation or that proliferating cells were impaired in their migration up the crypt column, resulting in a build-up of proliferating epithelial cells in the crypt column. Critically, IFN-γ was found to play a critical role in the accumulation of proliferating epithelial cells during chronic infection, suggesting the anti-nematode immune response plays an important role in regulating epithelial cell turnover in the inflamed intestine (Artis et al., 1999).
In addition to comparing the epithelial cell proliferation between resistant and susceptible mouse strains following T. muris infection, a recent study by Cliffe et al. compared the rate of epithelial cell migration and turnover in mouse strains that develop type 2 cytokine responses and expel infection versus those that express type 1 cytokine responses and develop chronic infection. In striking contrast to genetically susceptible AKR mice, resistant mouse strains exhibited a much more rapid rate of epithelial cell migration up the crypt column around the period of worm expulsion (Cliffe et al., 2005). Critically, the protective type 2 cytokine, IL-13, was implicated in driving rapid migration and turnover of the epithelium, essentially creating an ‘epithelial escalator’ proposed to dislodge T. muris from its epithelial niche. IFN-γ-induced expression of the chemokine CXCL10 was found to reduce epithelial cell turnover in susceptible animals and blockade of this chemokine in infected AKR mice reversed the accumulation of proliferating epithelial cells and resulted in enhanced epithelial cell migration and turnover coupled with expulsion of T. muris (Cliffe et al., 2005). In addition, the same treatment strategy resulted in enhanced epithelial cell turnover and worm expulsion in infected sever combined immunodeficient mice (that lack T and B cells), providing the first demonstration that immunity to this nematode infection can occur in the absence of an adaptive immune response. Moreover, this finding suggests that regulation of epithelial cell turnover alone is sufficient to control worm expulsion (Cliffe et al., 2005). These studies suggest that earlier reports of elevated frequencies of proliferating cells during chronic nematode infection were not indicative of increased proliferation but rather the result of reduced migration of epithelial cells up the crypt column. Indeed, slowing the rate of epithelial cell turnover may be a strategy employed by parasitic nematodes to promote their own survival. The existence of nematode-derived factors that can influence host intestinal epithelial cell proliferation support this hypothesis (Rikihisa et al., 1984; Huby et al., 1999). Based on these new findings discussed above, it will be interesting to determine whether the ‘epithelial escalator’ model contributes to expulsion of other GI nematode parasites and whether chemicals known to influence epithelial turnover will prove effective in eradicating infection in chronically infected hosts. Based on the discussion above, it will also be interesting to determine whether blockade of CXCL10, in addition to altering epithelial cell turnover, also results in altered epithelial cell differentiation and gene expression including the intelectins and/or RELM family members. It is tempting to speculate that a combination of type 2 cytokine-driven dysregulation of epithelial cell turnover, coupled with profound changes in gene expression, provide an arsenal of host epithelial cell-intrinsic anti-nematode responses that precipitate worm expulsion and resistance to infection.
5. Concluding remarks
The molecules and pathways discussed in this review, and their potential functions in contributing to expulsion of intestinal nematode parasites, are areas of intense research and the development of new tools to deplete or enhance these pathways offer promise for future discoveries. Challenges that lie ahead include identifying how expression of these host molecules is regulated and the identification of nematode gene products that are targeted by the host response. Defining how different lifecycle stages of parasitic nematodes are influenced by these molecules and whether these pathways play distinct roles in primary versus secondary infection, will also be important. An additional task will be to determine whether the novel cytokine-induced epithelial genes identified in micro-arrays perform unique roles following infection with individual nematode species or if certain epithelial gene products exhibit a common protective role against multiple GI nematodes; identification of a single anti-nematode factor that acts against all nematode species would have important implications, particularly given the polyparasitism often reported in individuals exposed to nematode parasites. As we look forward, it will be important to extend these studies to humans and veterinary species that suffer the debilitating consequences of parasitic nematode infections. Notwithstanding these challenges, the recent data accumulated on cytokine regulation of gene expression in intestinal epithelial cells and the influence of cytokines on enterocyte function indicate these pathways are a critical component in host control of intestinal nematode infection. Harnessing these discoveries offers the potential to instruct the design of a new generation of intervention strategies.
Acknowledgements
Thanks to current lab members (Yurong Du, Katherine Guild, Meera Nair, Jackie Perriguoe, Katie Rook, Amy Troy and Colby Zaph) for insightful discussions and review of the manuscript. Additional thanks to Lewis Tilney for expertise in electron microscopy, to Peter White for performing and analyzing microarrays and to Hugh Miller and Alan Pemberton (University of Edinburgh) for providing the anti-intelectin antibody. Research in the lab is supported by the National Institutes of Health (AI61570); Molecular Biology Core, Morphology Core and Pilot Feasibility Program of the National Institute of Diabetes and Digestive and Kidney Diseases Center (DK50306); The Irvington Institute for Immunologic Research; and the Crohn’s and Colitis Foundation of America’s William and Shelby Modell Family Foundation Research Award.
References
- Akiho H, Blennerhassett P, Deng Y, Collins SM. Role of IL-4, IL-13, and STAT6 in inflammation-induced hypercontractility of murine smooth muscle cells. Am. J. Physiol. Gastrointest. Liver Physiol. 2002;282:G226–G232. doi: 10.1152/ajpgi.2002.282.2.G226. [DOI] [PubMed] [Google Scholar]
- Albonico M, Crompton DW, Savioli L. Control strategies for human intestinal nematode infections. Adv. Parasitol. 1999;42:277–341. doi: 10.1016/s0065-308x(08)60151-7. [DOI] [PubMed] [Google Scholar]
- Albonico M, Engels D, Savioli L. Monitoring drug efficacy and early detection of drug resistance in human soil-transmitted nematodes: a pressing public health agenda for helminth control. Int. J. Parasitol. 2004;34:1205–1210. doi: 10.1016/j.ijpara.2004.08.001. [DOI] [PubMed] [Google Scholar]
- Apostolopoulos V, McKenzie IF. Role of the mannose receptor in the immune response. Curr. Mol. Med. 2001;1:469–474. doi: 10.2174/1566524013363645. [DOI] [PubMed] [Google Scholar]
- Artis D, Potten CS, Else KJ, Finkelman FD, Grencis RK. Trichuris muris: host intestinal epithelial cell hyperproliferation during chronic infection is regulated by interferon-gamma. Exp. Parasitol. 1999;92:144–153. doi: 10.1006/expr.1999.4407. [DOI] [PubMed] [Google Scholar]
- Artis D, Shapira S, Mason N, Speirs KM, Goldschmidt M, Caamano J, Liou HC, Hunter CA, Scott P. Differential requirement for NF-kappa B family members in control of helminth infection and intestinal inflammation. J. Immunol. 2002;169:4481–4487. doi: 10.4049/jimmunol.169.8.4481. [DOI] [PubMed] [Google Scholar]
- Artis D, Villarino A, Silverman M, He W, Thornton EM, Mu S, Summer S, Covey TM, Huang E, Yoshida H, Koretzky G, Goldschmidt M, Wu GD, de Sauvage F, Miller HR, Saris CJ, Scott P, Hunter CA. The IL-27 receptor (WSX-1) is an inhibitor of innate and adaptive elements of type 2 immunity. J. Immunol. 2004a;173:5626–5634. doi: 10.4049/jimmunol.173.9.5626. [DOI] [PubMed] [Google Scholar]
- Artis D, Wang ML, Keilbaugh SA, He W, Brenes M, Swain GP, Knight PA, Donaldson DD, Lazar MA, Miller HR, Schad GA, Scott P, Wu GD. RELM{beta}/FIZZ2 is a goblet cell-specific immune-effector molecule in the gastrointestinal tract. Proc. Natl Acad. Sci. USA. 2004b;101:13596–13600. doi: 10.1073/pnas.0404034101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Betts CJ, Else KJ. Mast cells, eosinophils and antibody-mediated cellular cytotoxicity are not critical in resistance to Trichuris muris. Parasite Immunol. 1999;21:45–52. doi: 10.1046/j.1365-3024.1999.00200.x. [DOI] [PubMed] [Google Scholar]
- Betts J, deSchoolmeester ML, Else KJ. Trichuris muris: CD4+T cell-mediated protection in reconstituted SCID mice. Parasitology. 2000;121(Pt 6):631–637. [PubMed] [Google Scholar]
- Blagoev B, Kratchmarova I, Nielsen MM, Fernandez MM, Voldby J, Andersen JS, Kristiansen K, Pandey A, Mann M. Inhibition of adipocyte differentiation by resistin-like molecule alpha. Biochemical characterization of its oligomeric nature. J. Biol. Chem. 2002;277:42011–42016. doi: 10.1074/jbc.M206975200. [DOI] [PubMed] [Google Scholar]
- Brown JK, Donaldson DS, Wright SH, Miller HR. Mucosal mast cells and nematode infection: strain-specific differences in mast cell precursor frequency revisited. J. Helminthol. 2003;77:155–161. doi: 10.1079/JOH2002160. [DOI] [PubMed] [Google Scholar]
- Bundy DA, Cooper ES. Trichuris and trichuriasis in humans. Adv. Parasitol. 1989;28:107–173. doi: 10.1016/s0065-308x(08)60332-2. [DOI] [PubMed] [Google Scholar]
- Callender JE, Grantham-McGregor S, Walker S, Cooper ES. Trichuris infection and mental development in children. Lancet. 1992;339:181. doi: 10.1016/0140-6736(92)90246-y. [DOI] [PubMed] [Google Scholar]
- Chan MS. The global burden of intestinal nematode infections-fifty years on. Parasitol. Today. 1997;13:438–443. doi: 10.1016/s0169-4758(97)01144-7. [DOI] [PubMed] [Google Scholar]
- Chan MS, Medley GF, Jamison D, Bundy DA. The evaluation of potential global morbidity attributable to intestinal nematode infections. Parasitology. 1994;3(Pt 3):373–387. doi: 10.1017/s0031182000078410. [DOI] [PubMed] [Google Scholar]
- Chang NC, Hung SI, Hwa KY, Kato I, Chen JE, Liu CH, Chang AC. A macrophage protein, Ym1, transiently expressed during inflammation is a novel mammalian lectin. J. Biol. Chem. 2001;276:17497–17506. doi: 10.1074/jbc.M010417200. [DOI] [PubMed] [Google Scholar]
- Chenine AL, Buckley KA, Li PL, Rasmussen RA, Ong H, Jiang S, Wang T, Augostini P, Secor WE, Ruprecht RM. Schistosoma mansoni infection promotes SHIV clade C replication in rhesus macaques. AIDS. 2005;19:1793–1797. doi: 10.1097/01.aids.0000189857.51935.0b. [DOI] [PubMed] [Google Scholar]
- Cliffe LJ, Grencis RK. The Trichuris muris system: a paradigm of resistance and susceptibility to intestinal nematode infection. Adv. Parasitol. 2004;57:255–307. doi: 10.1016/S0065-308X(04)57004-5. [DOI] [PubMed] [Google Scholar]
- Cliffe LJ, Humphreys NE, Lane TE, Potten CS, Booth C, Grencis RK. Accelerated intestinal epithelial cell turnover: a new mechanism of parasite expulsion. Science. 2005;308:1463–1465. doi: 10.1126/science.1108661. [DOI] [PubMed] [Google Scholar]
- Colley DG, LoVerde PT, Savioli L. Infectious disease. Medical helminthology in the 21st century. Science. 2001;293:1437–1438. doi: 10.1126/science.1060733. [DOI] [PubMed] [Google Scholar]
- Cooper PJ, Chico M, Sandoval C, Espinel I, Guevara A, Levine MM, Griffin GE, Nutman TB. Human infection with Ascaris lumbricoides is associated with suppression of the interleukin-2 response to recombinant cholera toxin B subunit following vaccination with the live oral cholera vaccine CVD 103-HgR. Infect. Immun. 2001;69:1574–1580. doi: 10.1128/IAI.69.3.1574-1580.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Datta R, deSchoolmeester ML, Hedeler C, Paton NW, Brass AM, Else KJ. Identification of novel genes in intestinal tissue that are regulated after infection with an intestinal nematode parasite. Infect. Immun. 2005;73:4025–4033. doi: 10.1128/IAI.73.7.4025-4033.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- East L, Isacke CM. The mannose receptor family. Biochim. Biophys. Acta. 2002;1572:364–386. doi: 10.1016/s0304-4165(02)00319-7. [DOI] [PubMed] [Google Scholar]
- Elola MT, Chiesa ME, Alberti AF, Mordoh J, Fink NE. Galectin-1 receptors in different cell types. J. Biomed. Sci. 2005;12:13–29. doi: 10.1007/s11373-004-8169-5. [DOI] [PubMed] [Google Scholar]
- Else KJ, Finkelman FD. Intestinal nematode parasites, cytokines and effector mechanisms. Int. J. Parasitol. 1998;28:1145–1158. doi: 10.1016/s0020-7519(98)00087-3. [DOI] [PubMed] [Google Scholar]
- Ezekowitz RA, Sastry K, Bailly P, Warner A. Molecular characterization of the human macrophage mannose receptor: demonstration of multiple carbohydrate recognition-like domains and phagocytosis of yeasts in Cos-1 cells. J. Exp. Med. 1990;172:1785–1794. doi: 10.1084/jem.172.6.1785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finkelman FD, Shea-Donohue T, Goldhill J, Sullivan CA, Morris SC, Madden KB, Gause WC, Urban JF., Jr. Cytokine regulation of host defense against parasitic gastrointestinal nematodes: lessons from studies with rodent models. Annu. Rev. Immunol. 1997;15:505–533. doi: 10.1146/annurev.immunol.15.1.505. [DOI] [PubMed] [Google Scholar]
- Finkelman FD, Shea-Donohue T, Morris SC, Gildea L, Strait R, Madden KB, Schopf L, Urban JF., Jr. Interleukin-4- and interleukin-13-mediated host protection against intestinal nematode parasites. Immunol. Rev. 2004;201:139–155. doi: 10.1111/j.0105-2896.2004.00192.x. [DOI] [PubMed] [Google Scholar]
- Gerstmayer B, Kusters D, Gebel S, Muller T, Van Miert E, Hofmann K, Bosio A. Identification of RELM gamma, a novel resistin-like molecule with a distinct expression pattern. Genomics. 2003;81:588–595. doi: 10.1016/s0888-7543(03)00070-3. [DOI] [PubMed] [Google Scholar]
- Gibbons LM. General organisation. In: Lee DL, editor. The Biology of Nemotodes. Taylor & Francis; London: 2002. pp. 31–59. [Google Scholar]
- Gordon S. Alternative activation of macrophages. Nat. Rev. Immunol. 2003;3:23–35. doi: 10.1038/nri978. [DOI] [PubMed] [Google Scholar]
- Graham AL, Lamb TJ, Read AF, Allen JE. Malaria-filaria coinfection in mice makes malarial disease more severe unless filarial infection achieves patency. J. Infect. Dis. 2005;191:410–421. doi: 10.1086/426871. [DOI] [PubMed] [Google Scholar]
- Grencis RK, Cooper ES. Enterobius, Trichuris, Capillaria, and hookworm including Ancylostoma caninum. Gastroenterol. Clin. North Am. 1996;25:579–597. doi: 10.1016/s0889-8553(05)70264-8. [DOI] [PubMed] [Google Scholar]
- Gruber AD, Gandhi R, Pauli BU. The murine calcium-sensitive chloride channel (mCaCC) is widely expressed in secretory epithelia and in other select tissues. Histochem. Cell Biol. 1998;110:43–49. doi: 10.1007/s004180050263. [DOI] [PubMed] [Google Scholar]
- Gyomorey K, Garami E, Galley K, Rommens JM, Bear CE. Non-CFTR chloride channels likely contribute to secretion in the murine small intestine. Pflugers Arch. 2001;443(Suppl 1):S103–S106. doi: 10.1007/s004240100654. [DOI] [PubMed] [Google Scholar]
- Hayes KS, Bancroft AJ, Grencis RK. Immune-mediated regulation of chronic intestinal nematode infection. Immunol. Rev. 2004;201:75–88. doi: 10.1111/j.0105-2896.2004.00193.x. [DOI] [PubMed] [Google Scholar]
- He W, Wang ML, Jiang HQ, Steppan CM, Shin ME, Thurnheer MC, Cebra JJ, Lazar MA, Wu GD. Bacterial colonization leads to the colonic secretion of RELMbeta/FIZZ2, a novel goblet cell-specific protein. Gastroenterology. 2003;125:1388–1397. doi: 10.1016/j.gastro.2003.07.009. [DOI] [PubMed] [Google Scholar]
- Holcomb IN, Kabakoff RC, Chan B, Baker TW, Gurney A, Henzel W, Nelson C, Lowman HB, Wright BD, Skelton NJ, Frantz GD, Tumas DB, Peale FV, Jr.., Shelton DL, Hebert CC. FIZZ1, a novel cysteine-rich secreted protein associated with pulmonary inflammation, defines a new gene family. Embo. J. 2000;19:4046–4055. doi: 10.1093/emboj/19.15.4046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoste H. Trichostrongylus colubriformis: epithelial cell kinetics in the small intestine of infected rabbits. Exp. Parasitol. 1989;68:99–104. doi: 10.1016/0014-4894(89)90013-1. [DOI] [PubMed] [Google Scholar]
- Hoste H, Mariana JC. Trichostrongylus colubriformis: epithelial cell migration in the proximal and distal small intestine of infected rabbits. Exp. Parasitol. 1989;68:347–353. doi: 10.1016/0014-4894(89)90116-1. [DOI] [PubMed] [Google Scholar]
- Huby F, Nano JL, Mallet S, Hoste H. Effects of the excretory/secretory products of Trichostrongylus colubriformis on the growth of different cell lines. Int. J. Parasitol. 1999;29:697–702. doi: 10.1016/s0020-7519(99)00014-4. [DOI] [PubMed] [Google Scholar]
- Iwanaga S, Lee BL. Recent advances in the innate immunity of invertebrate animals. J. Biochem. Mol. Biol. 2005;38:128–150. doi: 10.5483/bmbrep.2005.38.2.128. [DOI] [PubMed] [Google Scholar]
- Jackson JA, Turner JD, Rentoul L, Faulkner H, Behnke JM, Hoyle M, Grencis RK, Else KJ, Kamgno J, Bradley JE, Boussinesq M. Cytokine response profiles predict species-specific infection patterns in human GI nematodes. Int. J. Parasitol. 2004;34:1237–1244. doi: 10.1016/j.ijpara.2004.07.009. [DOI] [PubMed] [Google Scholar]
- Jin HM, Copeland NG, Gilbert DJ, Jenkins NA, Kirkpatrick RB, Rosenberg M. Genetic characterization of the murine Ym1 gene and identification of a cluster of highly homologous genes. Genomics. 1998;54:316–322. doi: 10.1006/geno.1998.5593. [DOI] [PubMed] [Google Scholar]
- Kim KH, Lee K, Moon YS, Sul HS. A cysteine-rich adipose tissue-specific secretory factor inhibits adipocyte differentiation. J. Biol. Chem. 2001;276:11252–11256. doi: 10.1074/jbc.C100028200. [DOI] [PubMed] [Google Scholar]
- Knight PA, Wright SH, Lawrence CE, Paterson YY, Miller HR. Delayed expulsion of the nematode Trichinella spiralis in mice lacking the mucosal mast cell-specific granule chymase, mouse mast cell protease-1. J. Exp. Med. 2000;192:1849–1856. doi: 10.1084/jem.192.12.1849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knight PA, Wright SH, Brown JK, Huang X, Sheppard D, Miller HR. Enteric expression of the integrin alpha(v)beta(6) is essential for nematode-induced mucosal mast cell hyperplasia and expression of the granule chymase, mouse mast cell protease-1. Am. J. Pathol. 2002;161:771–779. doi: 10.1016/s0002-9440(10)64236-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knight PA, Pemberton AD, Robertson KA, Roy DJ, Wright SH, Miller HR. Expression profiling reveals novel innate and inflammatory responses in the jejunal epithelial compartment during infection with Trichinella spiralis. Infect. Immun. 2004;72:6076–6086. doi: 10.1128/IAI.72.10.6076-6086.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Komiya T, Tanigawa Y, Hirohashi S. Cloning of the novel gene intelectin, which is expressed in intestinal paneth cells in mice. Biochem. Biophys. Res. Commun. 1998;251:759–762. doi: 10.1006/bbrc.1998.9513. [DOI] [PubMed] [Google Scholar]
- Kuperman DA, Lewis CC, Woodruff PG, Rodriguez MW, Yang YH, Dolganov GM, Fahy JV, Erle DJ. Dissecting asthma using focused transgenic modeling and functional genomics. J. Allergy. Clin. Immunol. 2005;116:305–311. doi: 10.1016/j.jaci.2005.03.024. [DOI] [PubMed] [Google Scholar]
- Kushiyama A, Shojima N, Ogihara T, Inukai K, Sakoda H, Fujishiro M, Fukushima Y, Anai M, Ono H, Horike N, Viana AY, Uchijima Y, Nishiyama K, Shimosawa T, Fujita T, Katagiri H, Oka Y, Kurihara H, Asano T. Resistin-like molecule beta activates MAPKs, suppresses insulin signaling in hepatocytes, and induces diabetes, hyperlipidemia, and fatty liver in transgenic mice on a high fat diet. J. Biol. Chem. 2005;280:42016–42025. doi: 10.1074/jbc.M503065200. [DOI] [PubMed] [Google Scholar]
- Lee JK, Schnee J, Pang M, Wolfert M, Baum LG, Moremen KW, Pierce M. Human homologs of the Xenopus oocyte cortical granule lectin XL35. Glycobiology. 2001;11:65–73. doi: 10.1093/glycob/11.1.65. [DOI] [PubMed] [Google Scholar]
- Leverkoehne I, Gruber AD. The murine mCLCA3 (alias gob-5) protein is located in the mucin granule membranes of intestinal, respiratory, and uterine goblet cells. J. Histochem. Cytochem. 2002;50:829–838. doi: 10.1177/002215540205000609. [DOI] [PubMed] [Google Scholar]
- Li YQ, Roberts SA, Paulus U, Loeffler M, Potten CS. The crypt cycle in mouse small intestinal epithelium. J. Cell. Sci. 1994;12(Pt 12):3271–3279. doi: 10.1242/jcs.107.12.3271. [DOI] [PubMed] [Google Scholar]
- Li CK, Seth R, Gray T, Bayston R, Mahida YR, Wakelin D. Production of proinflammatory cytokines and inflammatory mediators in human intestinal epithelial cells after invasion by Trichinella spiralis. Infect. Immun. 1998;66:2200–2206. doi: 10.1128/iai.66.5.2200-2206.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu T, Dhanasekaran SM, Jin H, Hu B, Tomlins SA, Chinnaiyan AM, Phan SH. FIZZ1 stimulation of myofibroblast differentiation. Am. J. Pathol. 2004a;164:1315–1326. doi: 10.1016/S0002-9440(10)63218-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu T, Jin H, Ullenbruch M, Hu B, Hashimoto N, Moore B, McKenzie A, Lukacs NW, Phan SH. Regulation of found in inflammatory zone 1 expression in bleomycin-induced lung fibrosis: role of IL-4/IL-13 and mediation via STAT-6. J. Immunol. 2004b;173:3425–3431. doi: 10.4049/jimmunol.173.5.3425. [DOI] [PubMed] [Google Scholar]
- Lochnit G, Bongaarts R, Geyer R. Searching new targets for anthelminthic strategies: Interference with glycosphingolipid biosynthesis and phosphorylcholine metabolism affects development of Caenorhabditis elegans. Int. J. Parasitol. 2005;35:911–923. doi: 10.1016/j.ijpara.2005.02.015. [DOI] [PubMed] [Google Scholar]
- Loke P, Nair MG, Parkinson J, Guiliano D, Blaxter M, Allen JE. IL-4 dependent alternatively-activated macrophages have a distinctive in vivo gene expression phenotype. BMC Immunol. 2002;3:7. doi: 10.1186/1471-2172-3-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lowe ME. Properties and function of pancreatic lipase related protein 2. Biochimie. 2000;82:997–1004. doi: 10.1016/s0300-9084(00)01184-6. [DOI] [PubMed] [Google Scholar]
- Madden KB, Yeung KA, Zhao A, Gause WC, Finkelman FD, Katona IM, Urban JF, Jr.., Shea-Donohue T. Enteric nematodes induce stereotypic STAT6-dependent alterations in intestinal epithelial cell function. J. Immunol. 2004;172:5616–5621. doi: 10.4049/jimmunol.172.9.5616. [DOI] [PubMed] [Google Scholar]
- Maizels RM, Yazdanbakhsh M. Immune regulation by helminth parasites: cellular and molecular mechanisms. Nat. Rev. Immunol. 2003;3:733–744. doi: 10.1038/nri1183. [DOI] [PubMed] [Google Scholar]
- Maizels RM, Balic A, Gomez-Escobar N, Nair M, Taylor MD, Allen JE. Helminth parasites—masters of regulation. Immunol. Rev. 2004;201:89–116. doi: 10.1111/j.0105-2896.2004.00191.x. [DOI] [PubMed] [Google Scholar]
- Mantovani A, Sozzani S, Locati M, Allavena P, Sica A. Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol. 2002;23:549–555. doi: 10.1016/s1471-4906(02)02302-5. [DOI] [PubMed] [Google Scholar]
- Marshman E, Booth C, Potten CS. The intestinal epithelial stem cell. Bioessays. 2002;24:91–98. doi: 10.1002/bies.10028. [DOI] [PubMed] [Google Scholar]
- McDermott JR, Bartram RE, Knight PA, Miller HR, Garrod DR, Grencis RK. Mast cells disrupt epithelial barrier function during enteric nematode infection. Proc. Natl Acad. Sci. USA. 2003;100:7761–7766. doi: 10.1073/pnas.1231488100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKenzie GJ, Bancroft A, Grencis RK, McKenzie AN. A distinct role for interleukin-13 in Th2-cell-mediated immune responses. Curr. Biol. 1998;8:339–342. doi: 10.1016/s0960-9822(98)70134-4. [DOI] [PubMed] [Google Scholar]
- Nair MG, Cochrane DW, Allen JE. Macrophages in chronic type 2 inflammation have a novel phenotype characterized by the abundant expression of Ym1 and Fizz1 that can be partly replicated in vitro. Immunol. Lett. 2003;85:173–180. doi: 10.1016/s0165-2478(02)00225-0. [DOI] [PubMed] [Google Scholar]
- Nair MG, Gallagher IJ, Taylor MD, Loke P, Coulson PS, Wilson RA, Maizels RM, Allen JE. Chitinase and Fizz family members are a generalized feature of nematode infection with selective upregulation of Ym1 and Fizz1 by antigen-presenting cells. Infect. Immun. 2005;73:385–394. doi: 10.1128/IAI.73.1.385-394.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newton SE, Munn EA. The development of vaccines against gastrointestinal nematode parasites, particularly Haemonchus contortus. Parasitol. Today. 1999;15:116–122. doi: 10.1016/s0169-4758(99)01399-x. [DOI] [PubMed] [Google Scholar]
- Newton SE, Meeusen EN. Progress and new technologies for developing vaccines against gastrointestinal nematode parasites of sheep. Parasite. Immunol. 2003;25:283–296. doi: 10.1046/j.1365-3024.2003.00631.x. [DOI] [PubMed] [Google Scholar]
- Nishihara T, Wyrick RE, Working PK, Chen YH, Hedrick JL. Isolation and characterization of a lectin from the cortical granules of Xenopus laevis eggs. Biochemistry. 1986;25:6013–6020. doi: 10.1021/bi00368a027. [DOI] [PubMed] [Google Scholar]
- Noel W, Raes G, Hassanzadeh Ghassabeh G, De Baetselier P, Beschin A. Alternatively activated macrophages during parasite infections. Trends Parasitol. 2004;20:126–133. doi: 10.1016/j.pt.2004.01.004. [DOI] [PubMed] [Google Scholar]
- Nokes C, Bundy DA. Does helminth infection affect mental processing and educational achievement? Parasitol. Today. 1994;10:14–18. doi: 10.1016/0169-4758(94)90348-4. [DOI] [PubMed] [Google Scholar]
- Nokes C, Grantham-McGregor SM, Sawyer AW, Cooper ES, Robinson BA, Bundy DA. Moderate to heavy infections of Trichuris trichiura affect cognitive function in Jamaican school children. Parasitology. 1992;104(Pt 3):539–547. doi: 10.1017/s0031182000063800. [DOI] [PubMed] [Google Scholar]
- Norkina O, Kaur S, Ziemer D, De Lisle RC. Inflammation of the cystic fibrosis mouse small intestine. Am. J. Physiol. Gastrointest. Liver Physiol. 2004;286:G1032–G1041. doi: 10.1152/ajpgi.00473.2003. [DOI] [PubMed] [Google Scholar]
- Owhashi M, Arita H, Niwa A. Production of eosinophil chemotactic factor by CD8+T-cells in Toxocara canis-infected mice. Parasitol. Res. 1998;84:136–138. doi: 10.1007/s004360050370. [DOI] [PubMed] [Google Scholar]
- Owhashi M, Arita H, Hayai N. Identification of a novel eosinophil chemotactic cytokine (ECF-L) as a chitinase family protein. J. Biol. Chem. 2000;275:1279–1286. doi: 10.1074/jbc.275.2.1279. [DOI] [PubMed] [Google Scholar]
- Park-Min KH, Antoniv TT, Ivashkiv LB. Regulation of macrophage phenotype by long-term exposure to IL-10. Immunobiology. 2005;210:77–86. doi: 10.1016/j.imbio.2005.05.002. [DOI] [PubMed] [Google Scholar]
- Patel SD, Rajala MW, Rossetti L, Scherer PE, Shapiro L. Disulfide-dependent multimeric assembly of resistin family hormones. Science. 2004;304:1154–1158. doi: 10.1126/science.1093466. [DOI] [PubMed] [Google Scholar]
- Pemberton AD, Knight PA, Gamble J, Colledge WH, Lee JK, Pierce M, Miller HR. Innate BALB/c enteric epithelial responses to Trichinella spiralis: inducible expression of a novel goblet cell lectin, intelectin-2, and its natural deletion in C57BL/10 mice. J. Immunol. 2004a;173:1894–1901. doi: 10.4049/jimmunol.173.3.1894. [DOI] [PubMed] [Google Scholar]
- Pemberton AD, Knight PA, Wright SH, Miller HR. Proteomic analysis of mouse jejunal epithelium and its response to infection with the intestinal nematode, Trichinella spiralis. Proteomics. 2004b;4:1101–1108. doi: 10.1002/pmic.200300658. [DOI] [PubMed] [Google Scholar]
- Pernthaner A, Cole SA, Morrison L, Hein WR. Increased expression of interleukin-5 (IL-5), IL-13, and tumor necrosis factor alpha genes in intestinal lymph cells of sheep selected for enhanced resistance to nematodes during infection with Trichostrongylus colubriformis. Infect. Immun. 2005;73:2175–2183. doi: 10.1128/IAI.73.4.2175-2183.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Potten CS. Stem cells in gastrointestinal epithelium: numbers, characteristics and death. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 1998;353:821–830. doi: 10.1098/rstb.1998.0246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raes G, De Baetselier P, Noel W, Beschin A, Brombacher F. Differential expression of FIZZ1 and Ym1 in alternatively versus classically activated macrophages. J. Leukoc. Biol. 2002a;71:597–602. [PubMed] [Google Scholar]
- Raes G, Noel W, Beschin A, Brys L, de Baetselier P, Hassanzadeh GH. FIZZ1 and Ym as tools to discriminate between differentially activated macrophages. Dev. Immunol. 2002b;9:151–159. doi: 10.1080/1044667031000137629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rajala MW, Obici S, Scherer PE, Rossetti L. Adipose-derived resistin and gut-derived resistin-like molecule-beta selectively impair insulin action on glucose production. J. Clin. Invest. 2003;111:225–230. doi: 10.1172/JCI16521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rauh MJ, Ho V, Pereira C, Sham A, Sly LM, Lam V, Huxham L, Minchinton AI, Mui A, Krystal G. SHIP represses the generation of alternatively activated macrophages. Immunity. 2005;23:361–374. doi: 10.1016/j.immuni.2005.09.003. [DOI] [PubMed] [Google Scholar]
- Rikihisa Y, Letonja T, Pratt N, Chin Lin Y. Taenia taeniaeformis: characterization of larval metabolic products and growth of host gastric cells in vitro. Exp. Parasitol. 1984;58:230–238. doi: 10.1016/0014-4894(84)90039-0. [DOI] [PubMed] [Google Scholar]
- Sandler NG, Mentink-Kane MM, Cheever AW, Wynn TA. Global gene expression profiles during acute pathogen-induced pulmonary inflammation reveal divergent roles for Th1 and Th2 responses in tissue repair. J. Immunol. 2003;171:3655–3667. doi: 10.4049/jimmunol.171.7.3655. [DOI] [PubMed] [Google Scholar]
- Schinke T, Haberland M, Jamshidi A, Nollau P, Rueger JM, Amling M. Cloning and functional characterization of resistin-like molecule gamma. Biochem. Biophys. Res. Commun. 2004;314:356–362. doi: 10.1016/j.bbrc.2003.12.100. [DOI] [PubMed] [Google Scholar]
- Stahl PD, Ezekowitz RA. The mannose receptor is a pattern recognition receptor involved in host defense. Curr. Opin. Immunol. 1998;10:50–55. doi: 10.1016/s0952-7915(98)80031-9. [DOI] [PubMed] [Google Scholar]
- Steppan CM, Lazar MA. The current biology of resistin. J. Intern. Med. 2004;255:439–447. doi: 10.1111/j.1365-2796.2004.01306.x. [DOI] [PubMed] [Google Scholar]
- Steppan CM, Brown EJ, Wright CM, Bhat S, Banerjee RR, Dai CY, Enders GH, Silberg DG, Wen X, Wu GD, Lazar MA. A family of tissue-specific resistin-like molecules. Proc. Natl Acad. Sci. USA. 2001;98:502–506. doi: 10.1073/pnas.98.2.502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steppan CM, Wang J, Whiteman EL, Birnbaum MJ, Lazar MA. Activation of SOCS-3 by resistin. Mol. Cell. Biol. 2005;25:1569–1575. doi: 10.1128/MCB.25.4.1569-1575.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stutz AM, Pickart LA, Trifilieff A, Baumruker T, Prieschl-Strassmayr E, Woisetschlager M. The Th2 cell cytokines IL-4 and IL-13 regulate found in inflammatory zone 1/resistin-like molecule alpha gene expression by a STAT6 and CCAAT/enhancer-binding proteindependent mechanism. J. Immunol. 2003;170:1789–1796. doi: 10.4049/jimmunol.170.4.1789. [DOI] [PubMed] [Google Scholar]
- Sun YJ, Chang NC, Hung SI, Chang AC, Chou CC, Hsiao CD. The crystal structure of a novel mammalian lectin, Ym1, suggests a saccharide binding site. J. Biol. Chem. 2001;276:17507–17514. doi: 10.1074/jbc.M010416200. [DOI] [PubMed] [Google Scholar]
- Suzuki YA, Shin K, Lonnerdal B. Molecular cloning and functional expression of a human intestinal lactoferrin receptor. Biochemistry. 2001;40:15771–15779. doi: 10.1021/bi0155899. [DOI] [PubMed] [Google Scholar]
- Symons LE. Pathology of gastrointestinal helminthiases. Int. Rev. Trop. Med. 1969;3:49–100. [PubMed] [Google Scholar]
- Symons LE. Epithelia cell mitosis and morphology in worm-free regions of the intestines of the rat infected by Nippostrongylus brasiliensis. J. Parasitol. 1978;64:958–959. [PubMed] [Google Scholar]
- Tilney LG, Connelly PS, Guild GM, Vranich KA, Artis D. Adaptation of a nematode parasite to living within the mammalian epithelium. J. Exp. Zoolog. A Comp. Exp. Biol. 2005;303:927–945. doi: 10.1002/jez.a.214. [DOI] [PubMed] [Google Scholar]
- Tsuji S, Uehori J, Matsumoto M, Suzuki Y, Matsuhisa A, Toyoshima K, Seya T. Human intelectin is a novel soluble lectin that recognizes galactofuranose in carbohydrate chains of bacterial cell wall. J. Biol. Chem. 2001;276:23456–23463. doi: 10.1074/jbc.M103162200. [DOI] [PubMed] [Google Scholar]
- Urban JF, Jr.., Noben-Trauth N, Donaldson DD, Madden KB, Morris SC, Collins M, Finkelman FD. IL-13, IL-4Ralpha, and Stat6 are required for the expulsion of the gastrointestinal nematode parasite Nippostrongylus brasiliensis. Immunity. 1998;8:255–264. doi: 10.1016/s1074-7613(00)80477-x. [DOI] [PubMed] [Google Scholar]
- Urban JF, Jr.., Noben-Trauth N, Schopf L, Madden KB, Finkelman FD. Cutting edge: IL-4 receptor expression by non-bone marrow-derived cells is required to expel gastrointestinal nematode parasites. J. Immunol. 2001;167:6078–6081. doi: 10.4049/jimmunol.167.11.6078. [DOI] [PubMed] [Google Scholar]
- Wang JL, Gray RM, Haudek KC, Patterson RJ. Nucleocyto-plasmic lectins. Biochim. Biophys. Acta. 2004;1673:75–93. doi: 10.1016/j.bbagen.2004.03.013. [DOI] [PubMed] [Google Scholar]
- Wang ML, Shin ME, Knight PA, Artis D, Silberg DG, Suh E, Wu GD. Regulation of RELM/FIZZ isoform expression by Cdx2 in response to innate and adaptive immune stimulation in the intestine. Am. J. Physiol. Gastrointest. Liver Physiol. 2005;288:G1074–G1083. doi: 10.1152/ajpgi.00442.2004. [DOI] [PubMed] [Google Scholar]
- WHO . 54th World Health Assembly:schistosomiasis and soil transmitted helminth infections [item 13.3] WHO; Geneva: 2001. [Google Scholar]
- Zhao A, McDermott J, Urban JF, Jr.., Gause W, Madden KB, Yeung KA, Morris SC, Finkelman FD, Shea-Donohue T. Dependence of IL-4, IL-13, and nematode-induced alterations in murine small intestinal smooth muscle contractility on Stat6 and enteric nerves. J. Immunol. 2003;171:948–954. doi: 10.4049/jimmunol.171.2.948. [DOI] [PubMed] [Google Scholar]
- Zimmermann N, Mishra A, King NE, Fulkerson PC, Doepker MP, Nikolaidis NM, Kindinger LE, Moulton EA, Aronow BJ, Rothenberg ME. Transcript signatures in experimental asthma: identification of STAT6-dependent and -independent pathways. J. Immunol. 2004;172:1815–1824. doi: 10.4049/jimmunol.172.3.1815. [DOI] [PubMed] [Google Scholar]


