Abstract
A plaque autoradiography assay to detect and quantitate thymidine kinase (TK) mutants of herpes simplex virus type 1 (HSV-1) and HSV-2 in clinical samples is described. This method utilizes the selective incorporation of [125I]iododeoxycytidine, a pyrimidine analog selectively phosphorylated by the HSV TK. Only cells infected with TK-competent virus will efficiently incorporate iododeoxycytidine and are the only cells detected by autoradiography. Furthermore, this assay discriminates between TK+ virus (TK competent) and TKA virus (TK altered or reduced). This ability to differentiate TK+ from TKA virus is enhanced when infected cells are labeled with [14C]thymidine in tandem with iododeoxycytidine labeling. Reconstruction experiments with mixtures of TK+ (HSV-1 Patton) virus and TK-deficient (TK-) (B2006) or TKA (IUDRr) mutants were performed to determine the limits of detection of this technique. Ten percent TK- or TKA virus was the lower limit for the detection of TK mutants in a mixed population, whereas 1 in 1,000 TK+ virus revertants could be detected in a TK- virus population. In reconstructed populations and 45 clinical samples, a good correlation existed between the increase in 50% inhibitory dose for acyclovir and the percent TK mutant virus present. Similarly, the results of this technique correlated well with the acyclovir phosphorylating activity of extracts from cells infected with isolates or reconstructed mixtures. Plaque autoradiography with [125I]iododeoxycytidine was able to distinguish mixed populations of TK+ and TK- virus and homogeneous populations of TKA virus. The tandem use of [125I]iododeoxycytidine and [14C]thymidine readily identified TKA virus, which appeared as TK+ virus when labeled with [14C]thymidine alone. This technique provides a sensitive screen for antiviral resistance due to alterations in the viral TK and can be used to analyze clinical samples.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Burns W. H., Saral R., Santos G. W., Laskin O. L., Lietman P. S., McLaren C., Barry D. W. Isolation and characterisation of resistant Herpes simplex virus after acyclovir therapy. Lancet. 1982 Feb 20;1(8269):421–423. doi: 10.1016/s0140-6736(82)91620-8. [DOI] [PubMed] [Google Scholar]
- Coen D. M., Furman P. A., Aschman D. P., Schaffer P. A. Mutations in the herpes simplex virus DNA polymerase gene conferring hypersensitivity to aphidicolin. Nucleic Acids Res. 1983 Aug 11;11(15):5287–5297. doi: 10.1093/nar/11.15.5287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coen D. M., Furman P. A., Gelep P. T., Schaffer P. A. Mutations in the herpes simplex virus DNA polymerase gene can confer resistance to 9-beta-D-arabinofuranosyladenine. J Virol. 1982 Mar;41(3):909–918. doi: 10.1128/jvi.41.3.909-918.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coen D. M., Schaffer P. A. Two distinct loci confer resistance to acycloguanosine in herpes simplex virus type 1. Proc Natl Acad Sci U S A. 1980 Apr;77(4):2265–2269. doi: 10.1073/pnas.77.4.2265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collins P., Bauer D. J. Relative potencies of anti-herpes compounds. Ann N Y Acad Sci. 1977 Mar 4;284:49–59. doi: 10.1111/j.1749-6632.1977.tb21936.x. [DOI] [PubMed] [Google Scholar]
- Cooper G. M. Phosphorylation of 5-bromodeoxycytidine in cells infected with herpes simplex virus. Proc Natl Acad Sci U S A. 1973 Dec;70(12):3788–3792. doi: 10.1073/pnas.70.12.3788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crumpacker C. S., Schnipper L. E., Marlowe S. I., Kowalsky P. N., Hershey B. J., Levin M. J. Resistance to antiviral drugs of herpes simplex virus isolated from a patient treated with acyclovir. N Engl J Med. 1982 Feb 11;306(6):343–346. doi: 10.1056/NEJM198202113060606. [DOI] [PubMed] [Google Scholar]
- DUBBS D. R., KIT S. MUTANT STRAINS OF HERPES SIMPLEX DEFICIENT IN THYMIDINE KINASE-INDUCING ACTIVITY. Virology. 1964 Apr;22:493–502. doi: 10.1016/0042-6822(64)90070-4. [DOI] [PubMed] [Google Scholar]
- Darby G., Field H. J., Salisbury S. A. Altered substrate specificity of herpes simplex virus thymidine kinase confers acyclovir-resistance. Nature. 1981 Jan 1;289(5793):81–83. doi: 10.1038/289081a0. [DOI] [PubMed] [Google Scholar]
- Dobersen M. J., Greer S. Herpes simplex virus type 2 induced pyrimidine nucleoside kinase: enzymatic basis for the selective antiherpetic effect of 5-halogenated analogues of deoxycytidine. Biochemistry. 1978 Mar 7;17(5):920–928. doi: 10.1021/bi00598a028. [DOI] [PubMed] [Google Scholar]
- Elion G. B., Furman P. A., Fyfe J. A., de Miranda P., Beauchamp L., Schaeffer H. J. Selectivity of action of an antiherpetic agent, 9-(2-hydroxyethoxymethyl) guanine. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5716–5720. doi: 10.1073/pnas.74.12.5716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ellis M. N., Barry D. W. Oral acyclovir therapy of genital herpes simplex virus type 2 infections in guinea pigs. Antimicrob Agents Chemother. 1985 Feb;27(2):167–171. doi: 10.1128/aac.27.2.167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Field H. J., Darby G. Pathogenicity in mice of strains of herpes simplex virus which are resistant to acyclovir in vitro and in vivo. Antimicrob Agents Chemother. 1980 Feb;17(2):209–216. doi: 10.1128/aac.17.2.209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Field H. J., Darby G., Wildy P. Isolation and characterization of acyclovir-resistant mutants of herpes simplex virus. J Gen Virol. 1980 Jul;49(1):115–124. doi: 10.1099/0022-1317-49-1-115. [DOI] [PubMed] [Google Scholar]
- Field H. J., Neden J. Isolation of bromovinyldeoxyuridine-resistant strains of herpes simplex virus and successful chemotherapy of mice infected with one such strain by using acyclovir. Antiviral Res. 1982 Oct;2(5):243–254. doi: 10.1016/0166-3542(82)90048-1. [DOI] [PubMed] [Google Scholar]
- Field H. J., Wildy P. The pathogenicity of thymidine kinase-deficient mutants of herpes simplex virus in mice. J Hyg (Lond) 1978 Oct;81(2):267–277. doi: 10.1017/s0022172400025109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Furman P. A., Coen D. M., St Clair M. H., Schaffer P. A. Acyclovir-resistant mutants of herpes simplex virus type 1 express altered DNA polymerase or reduced acyclovir phosphorylating activities. J Virol. 1981 Dec;40(3):936–941. doi: 10.1128/jvi.40.3.936-941.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Furman P. A., McGuirt P. V., Keller P. M., Fyfe J. A., Elion G. B. Inhibition by acyclovir of cell growth and DNA synthesis of cells biochemically transformed with herpesvirus genetic information. Virology. 1980 Apr 30;102(2):420–430. doi: 10.1016/0042-6822(80)90109-9. [DOI] [PubMed] [Google Scholar]
- Furman P. A., St Clair M. H., Fyfe J. A., Rideout J. L., Keller P. M., Elion G. B. Inhibition of herpes simplex virus-induced DNA polymerase activity and viral DNA replication by 9-(2-hydroxyethoxymethyl)guanine and its triphosphate. J Virol. 1979 Oct;32(1):72–77. doi: 10.1128/jvi.32.1.72-77.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Furman P. A., St Clair M. H., Spector T. Acyclovir triphosphate is a suicide inactivator of the herpes simplex virus DNA polymerase. J Biol Chem. 1984 Aug 10;259(15):9575–9579. [PubMed] [Google Scholar]
- Fyfe J. A., Keller P. M., Furman P. A., Miller R. L., Elion G. B. Thymidine kinase from herpes simplex virus phosphorylates the new antiviral compound, 9-(2-hydroxyethoxymethyl)guanine. J Biol Chem. 1978 Dec 25;253(24):8721–8727. [PubMed] [Google Scholar]
- Fyfe J. A., McKee S. A., Keller P. M. Altered thymidine-thymidylate kinases from strains of herpes simplex virus with modified drug sensitivities to acyclovir and (E)-5-(2-bromovinyl)-2'-deoxyuridine. Mol Pharmacol. 1983 Sep;24(2):316–323. [PubMed] [Google Scholar]
- Jamieson A. T., Gentry G. A., Subak-Sharpe J. H. Induction of both thymidine and deoxycytidine kinase activity by herpes viruses. J Gen Virol. 1974 Sep;24(3):465–480. doi: 10.1099/0022-1317-24-3-465. [DOI] [PubMed] [Google Scholar]
- Jamieson A. T., Subak-Sharpe J. H. Biochemical studies on the herpes simplex virus-specified deoxypyrimidine kinase activity. J Gen Virol. 1974 Sep;24(3):481–492. doi: 10.1099/0022-1317-24-3-481. [DOI] [PubMed] [Google Scholar]
- Jamieson A. T., Subak-Sharpe J. H. Interallelic complementation of mutants of herpes simplex virus deficient in deoxypyrimidine kinase activity. Virology. 1978 Mar;85(1):109–117. doi: 10.1016/0042-6822(78)90415-4. [DOI] [PubMed] [Google Scholar]
- Keller P. M., Fyfe J. A., Beauchamp L., Lubbers C. M., Furman P. A., Schaeffer H. J., Elion G. B. Enzymatic phosphorylation of acyclic nucleoside analogs and correlations with antiherpetic activities. Biochem Pharmacol. 1981 Nov 15;30(22):3071–3077. doi: 10.1016/0006-2952(81)90495-0. [DOI] [PubMed] [Google Scholar]
- Larder B. A., Cheng Y. C., Darby G. Characterization of abnormal thymidine kinases induced by drug-resistant strains of herpes simplex virus type 1. J Gen Virol. 1983 Mar;64(Pt 3):523–532. doi: 10.1099/0022-1317-64-3-523. [DOI] [PubMed] [Google Scholar]
- Larder B. A., Darby G. Properties of a novel thymidine kinase induced by an acyclovir-resistant herpes simplex virus type 1 mutant. J Virol. 1982 May;42(2):649–658. doi: 10.1128/jvi.42.2.649-658.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larder B. A., Derse D., Cheng Y. C., Darby G. Properties of purified enzymes induced by pathogenic drug-resistant mutants of herpes simplex virus. Evidence for virus variants expressing normal DNA polymerase and altered thymidine kinase. J Biol Chem. 1983 Feb 10;258(3):2027–2033. [PubMed] [Google Scholar]
- McGuirt P. V., Furman P. A. Acyclovir inhibition of viral DNA chain elongation in herpes simplex virus-infected cells. Am J Med. 1982 Jul 20;73(1A):67–71. doi: 10.1016/0002-9343(82)90066-3. [DOI] [PubMed] [Google Scholar]
- McGuirt P. V., Shaw J. E., Elion G. B., Furman P. A. Identification of small DNA fragments synthesized in herpes simplex virus-infected cells in the presence of acyclovir. Antimicrob Agents Chemother. 1984 Apr;25(4):507–509. doi: 10.1128/aac.25.4.507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLaren C., Ellis M. N., Hunter G. A. A colorimetric assay for the measurement of the sensitivity of herpes simplex viruses to antiviral agents. Antiviral Res. 1983 Nov;3(4):223–234. doi: 10.1016/0166-3542(83)90001-3. [DOI] [PubMed] [Google Scholar]
- Miller W. H., Miller R. L. Phosphorylation of acyclovir (acycloguanosine) monophosphate by GMP kinase. J Biol Chem. 1980 Aug 10;255(15):7204–7207. [PubMed] [Google Scholar]
- Miller W. H., Miller R. L. Phosphorylation of acyclovir diphosphate by cellular enzymes. Biochem Pharmacol. 1982 Dec 1;31(23):3879–3884. doi: 10.1016/0006-2952(82)90305-7. [DOI] [PubMed] [Google Scholar]
- Parris D. S., Harrington J. E. Herpes simplex virus variants restraint to high concentrations of acyclovir exist in clinical isolates. Antimicrob Agents Chemother. 1982 Jul;22(1):71–77. doi: 10.1128/aac.22.1.71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schnipper L. E., Crumpacker C. S. Resistance of herpes simplex virus to acycloguanosine: role of viral thymidine kinase and DNA polymerase loci. Proc Natl Acad Sci U S A. 1980 Apr;77(4):2270–2273. doi: 10.1073/pnas.77.4.2270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sibrack C. D., Gutman L. T., Wilfert C. M., McLaren C., St Clair M. H., Keller P. M., Barry D. W. Pathogenicity of acyclovir-resistant herpes simplex virus type 1 from an immunodeficient child. J Infect Dis. 1982 Nov;146(5):673–682. doi: 10.1093/infdis/146.5.673. [DOI] [PubMed] [Google Scholar]
- Smiley J. R., Wagner M. J., Summers W. P., Summers W. C. Genetic and physical evidence for the polarity of transcription of the thymidine kinase gene of herpes simplex virus. Virology. 1980 Apr 15;102(1):83–93. doi: 10.1016/0042-6822(80)90072-0. [DOI] [PubMed] [Google Scholar]
- Smith K. O., Kennell W. L., Poirier R. H., Lynd F. T. In vitro and in vivo resistance of herpes simplex virus to 9-(2-hydroxyethoxymethyl)guanine (acycloguanosine). Antimicrob Agents Chemother. 1980 Feb;17(2):144–150. doi: 10.1128/aac.17.2.144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Summers W. C., Summers W. P. [125I]deoxycytidine used in a rapid, sensitive, and specific assay for herpes simplex virus type 1 thymidine kinase. J Virol. 1977 Oct;24(1):314–318. doi: 10.1128/jvi.24.1.314-318.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tenser R. B., Dunstan M. E. Herpes simplex virus thymidine kinase expression in infection of the trigeminal ganglion. Virology. 1979 Dec;99(2):417–422. doi: 10.1016/0042-6822(79)90021-7. [DOI] [PubMed] [Google Scholar]
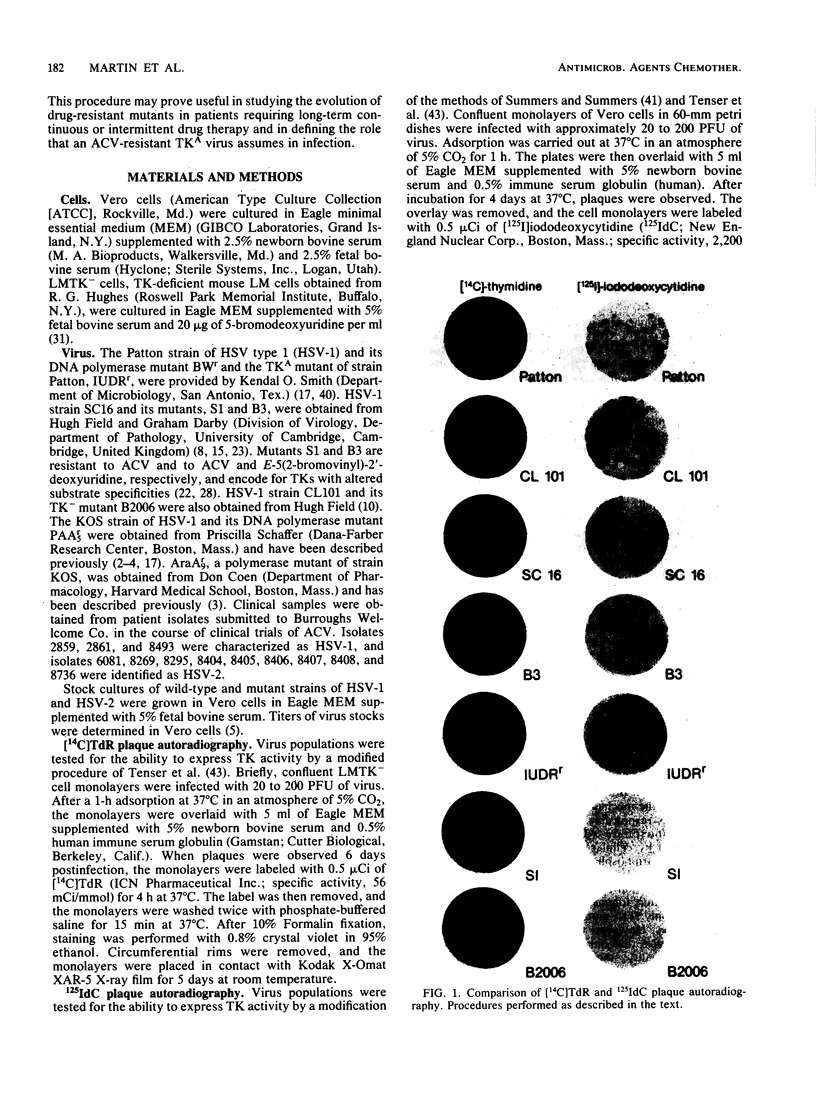
- Tenser R. B., Jones J. C., Ressel S. J., Fralish F. A. Thymidine plaque autoradiography of thymidine kinase-positive and thymidine kinase-negative herpesviruses. J Clin Microbiol. 1983 Jan;17(1):122–127. doi: 10.1128/jcm.17.1.122-127.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tenser R. B., Miller R. L., Rapp F. Trigeminal ganglion infection by thymidine kinase-negative mutants of herpes simplex virus. Science. 1979 Aug 31;205(4409):915–917. doi: 10.1126/science.224454. [DOI] [PubMed] [Google Scholar]