Abstract
Non-obese diabetic (NOD) mice develop autoimmunity that destroys their native beta cells causing diabetes. Their autoimmunity will also destroy syngeneic transplanted islets and transfer both autoimmunity and diabetes via spleen cells to non-diabetic mice. In this report, we studied the effects of streptozotocin (STZ) on the autoimmune diabetes in NOD mice. We transplanted NOD.SCID islets into three groups of NOD mice: (1) spontaneously diabetic NOD mice (NOD-sp.); (2) prediabetic NOD mice made diabetic by streptozotocin (NOD-stz); and (3) diabetic NOD mice also treated with streptozotocin (NOD-sp./stz). In the first group, the transplants were rejected within 3 weeks. In the second and third groups, the transplants survived indefinitely. Alloxan, a drug similar to streptozotocin, did not have the same effect as streptozotocin. The ability of streptozotocin to prevent diabetes in young NOD mice was reversed by anti-CD8 antibody treatment but not by anti-CD4 treatment. Streptozotocin also made spleen cells from diabetic NOD mice less effective transferring diabetes. These results indicate that streptozotocin treatment both prevents and reverses the islet destructive autoimmunity in NOD mice. We postulate that the effects of streptozotocin treatment may be mediated in part by regulatory T cells.
Keywords: autoimmunity, NOD mice, regulatory T cells, streptozotocin
INTRODUCTION
Autoimmune diabetes destroys insulin-producing beta cells. In humans, islet transplantation can cure diabetes, but recurrent autoimmune disease may destroy transplanted islets from an identical twin [1]. Similarly, non-diabetic (NOD) mice with autoimmune diabetes reject syngeneic islet transplants [2,3]. NOD autoimmunity may also shorten allograft survival [4]. Thus, recurrent and existing autoimmunity to islet allografts in humans could play a significant role in islet rejection even in the absence of alloimmunity. This would be especially true without long-term immunosuppression. Because some of the immunological mechanisms that cause or regulate allograft rejection are different from those that affect autoimmunity, immunosuppression targeted and tailored to mitigate allograft rejection might not necessarily reverse or suppress autoimmunity [5–7].
Numerous investigators have identified strategies and treatments that prevent the development of diabetes in NOD mice [8–19]. In at least one case, the results in NOD mice have served as the basis for initiating a clinical trial in humans, in which anti-CD3 monoclonal antibody therapy is used to delay Type I diabetes in newly diagnosed patients [20]. However, reversal of autoimmunity after mice develop diabetes is much more difficult. It is, therefore, important to identify additional ways to reverse established autoimmunity in mice in order to identify new potential targets for clinical therapy of human Type I diabetes.
Streptozotocin (STZ) is used commonly to cause experimental insulin-dependent diabetes in many species [21]. The drug concentrates preferentially in beta cells via the GLUT-2 glucose transporter [22], where it causes damage to DNA either by the generation of nitric oxide or by alkylation [23,24], or by DNA fragmentation [25]. While performing islet transplant experiments in NOD mice, we included control experiments in which NOD mice were given streptozotocin to assure that the euglycaemia was not a consequence of residual native pancreatic beta cells. Surprisingly, we found that streptozotocin prevented or suppressed NOD autoimmune diabetes. We therefore undertook a series of experiments designed to test the effectiveness of streptozotocin both in preventing and in reversing autoimmune diabetes in NOD mice.
MATERIALS AND METHODS
Mice
Pre-diabetic female NOD (NOD/LtJ) mice and NOD.SCID (NOD.CB17-Prkdcscid/J) mice were purchased from Jackson Laboratories (Bar Harbor, ME, USA) at 4 weeks of age.
Induction of diabetes
Some NOD mice were allowed to develop spontaneous autoimmune diabetes. Others were treated with a single intravenous dose of 250 mg/kg streptozotocin (Sigma, St Louis, MO, USA). Still other mice were made diabetic using alloxan at 90 mg/kg i.v. (Sigma). Mice were considered to be diabetic when non-fasting blood glucose levels were >300 mg/dl on 3 consecutive days. Blood glucose levels were also tested at the time of transplantation, then daily for the first 2 weeks, and then two to three times per week until the end of the experiment. The blood glucose levels were measured using an Accu-Check blood glucose monitor (Roche, Indianapolis, IN, USA) using whole blood collected from the tail vein. Graft failure was defined as the first day of three consecutive days of blood glucose levels >250 mg/dl. The mice were then sacrificed, and the kidney with the transplanted islets, and the pancreas were preserved in 2·5% formalin-phosphate buffered saline (PBS) for later histological examination.
Islet isolation and transplantation
NOD.SCID male mice 10–12 weeks of age were used as donors for islet transplants. Islets were isolated using a modification of the method of Gotoh et al. [26] in which the pancreatic duct is distended with collagenase P. After purification on a Histopaque gradient (HistopaqueR-1077, Sigma), islets with diameters between 75 and 250 µm were hand-picked and transplanted under the renal capsule. Each recipient received 600–800 islets. This study was performed at the Massachusetts General Hospital, where the facilities are fully accredited by the American Association for the Accreditation of Laboratory Animals Care (AAALAC).
Light microscopy
Kidneys with transplanted islets or pancreases were placed in 2·5% formalin PBS for 3 days then stored in PBS. After blocking in paraffin, serial 4 µm sections were cut through the entire graft site and every third to fifth slide containing three to five tissue sections was stained with haematoxylin–eosin for histological review. Immunoperoxidase staining for insulin and glucagon was performed on selected unstained sections [27].
In vivo T-cell depletion
Anti-CD4 (GK1·5, rat antimouse CD4) antibody was used to deplete CD4+ cells [28] and anti-CD8 antibody (116–13·1, hybridoma obtained from the American Type Culture Collection) was used to deplete CD8+ cells. All treated mice received 0·3 ml of unpurified ascites i.p. (roughly equivalent to 300 µg of purified antibody) [29] on days −1, 0, + 1 and weekly for 6 weeks. Selected mice were tested for T cell subset depletion by FACS analysis. Streptozotocin treatment had no effect on the numbers of splenic T cells. Anti-CD4 treatment depleted more than 95% of the CD4+ population for up to 3 weeks after the last dose of monoclonal antibody (MoAb), and anti-CD8 treatment depleted more than 98% of CD8+ cells for up to 5 weeks after the last dose of the antibody (data not shown).
Adoptive transfer experiments
Splenocytes, 5 × 107, prepared in PBS-5% fetal calf serum (FCS) were injected i.v. into the recipient's tail vein in a volume of 0·3 ml of PBS.
RESULTS
The effect of streptozotocin treatment on the development of autoimmunity
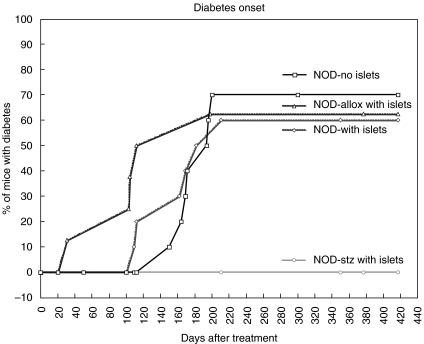
Female NOD mice develop diabetes starting about 90 days of age until approximately 70–80% are diabetic by 250 days of age. To test if streptozotocin affects the development of autoimmune diabetes in 6–8-week-old female NOD mice, we transplanted syngeneic islets into streptozotocin-treated non-diabetic NOD mice. As shown in Fig. 1, in a control group NOD mice developed diabetes starting at 100 days. Similarly, NOD mice that received an islet transplant from NOD.SCID donors, without any other treatment, developed diabetes at almost exactly the same time. In contrast, 6–8-week-old NOD mice that were treated with streptozotocin and transplanted with NOD.SCID islets never became diabetic during more than a year of observation. NOD mice in a fourth group were made diabetic at 6–8 weeks of age using alloxan and given NOD.SCID islet transplants. They developed diabetes faster than the mice in the control group, although the difference was not statistically significant.
Fig. 1.
The effect of islet transplantation, streptozotocin and alloxan treatment on the development of autoimmunity in NOD mice. Young NOD mice (6–8 weeks) were divided into four groups to study the effect of islet transplantation on the development of diabetes under various conditions. Group 1 NOD mice did not receive islet transplantation (control group) (n = 10) (□); Group 2 animals received islets without any other treatment (n = 10) (⋄); Group 3 NOD mice were treated with STZ and given NOD.SCID islets (n = 22) (○) and Group 4 mice were treated with alloxan and then given NOD.SCID islets (n = 8) (▵). The individual mice in Group 3 are shown in Table 2. Some mice in this group were removed for further experimental manipulation and others were sacrificed at various times for histology.
The effect of streptozotocin treatment on established autoimmune diabetes in NOD mice
We then tested whether or not streptozotocin treatment would affect the autoimmunity in NOD mice with established autoimmune diabetes. As shown in Table 1, NOD mice with diabetes rapidly destroyed their islet transplants derived from NOD.SCID mice (Group 1). In contrast, mice that had been treated with streptozotocin after they developed spontaneous autoimmune diabetes did not reject islets from NOD.SCID donors for as long as we observed them in this experiment (Group 2). Diabetic NOD mice were also treated with anti-CD4 at a dosing schedule known to cause indefinite allograft survival [30]. This dosing schedule delayed only modestly the loss of syngeneic islets, with all animals becoming diabetic within 21 days (Table 1, Group 3).
Table 1.
NOD mice were transplanted with NOD.SCID islets shortly after they had developed spontaneous autoimmune diabetes. Some mice were also treated with anti-CD4 antibody or streptozotocin as described in Materials and methods
| Group | Donor (islets) | Recipients | Treatment | Survival (days) |
|---|---|---|---|---|
| 1 | NOD.SCID | NOD-sp. | None | 7, 8, 10, 12, 21 |
| 2 | NOD.SCID | NOD-sp. | Streptozotocin | >28*, >39*, >100*, >150*, >152*, >173*, >460 |
| 3 | NOD.SCID | NOD-sp. | Anti-CD4 | 14–21 (n = 10) |
Normoglycaemic mice sacrificed at different times for histology.
To confirm that the islet grafts, rather than the native pancreas, were responsible for euglycaemia, three mice were unilaterally nephrectomized to remove their islet grafts. These nephrectomized animals became diabetic within 12 h.
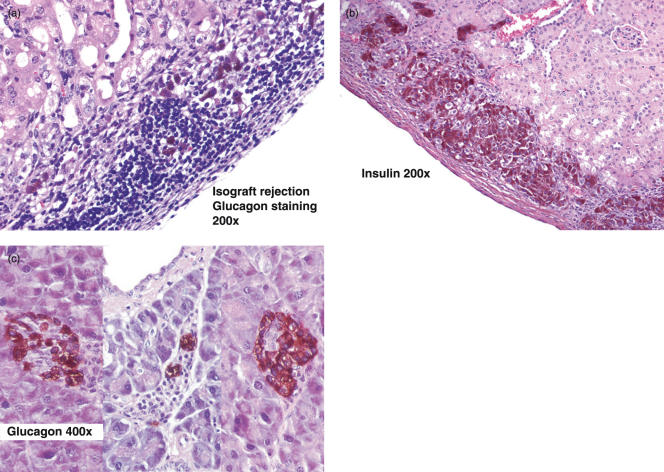
Histological examination of islet transplants and native pancreases were performed at the time of graft loss (recurrent diabetes) or at intervals after transplant in the mice that did not develop diabetes. As illustrated in Fig. 2a, the islet transplants in the untreated group showed a destructive lymphocytic infiltrate without insulin but with positive glucagon staining. The islets in native NOD pancreases showed moderate to severe intra-islet destructive lymphocytic inflammation (isletitis) and negative to severely reduced insulin staining (data not shown) [31]. In contrast, histological analysis of the islet transplants in mice that had been treated with streptozotocin showed intact islets with strong insulin and glucagon staining and no intra-islet lymphocytic destruction (isletitis) (Fig. 2b). The islets in the native pancreases of these streptozotocin treated mice were atrophic with negative insulin but positive glucagon staining (Fig. 2c) [32].
Fig. 2.
(a) Destruction of islet isografts in NOD mice. Light micrograph of NOD.SCID islets transplanted into an NOD-sp. mouse. The specimen was obtained at the time of recurrent diabetes. The graft site shows destructive lymphocytic infiltration of the islet graft (insulin negative; glucagon positive, 200×). (b) Prevention of islet isograft destruction. Light micrograph of NOD.SCID islets transplanted into an NOD-sp./stz mouse. Graft site showing well-preserved intact islets with minimal peri-islet inflammation (insulin-positive cells, 200×). (c) Host pancreas of a diabetic NOD-sp./stz mouse. Light micrograph of streptozotocin-treated pancreas, showing atrophic islets negative for insulin but positive for glucagon. Composite of three representative islets (400×).
Mechanistic studies of the streptozotocin effect
We next performed a series of experiments designed to explore how streptozotocin might alter NOD autoimmunity. As shown in Table 2, these experiments used NOD mice that were treated at 6–8 weeks of age before they developed spontaneous diabetes. The first group in Table 2 represents the same group of streptozotocin-treated islet transplanted mice that were illustrated in Fig. 1. None of these mice ever developed diabetes. Various mice in this group were used at the times indicated for some of the experiments described below.
Table 2.
Six-to-eight-week-old NOD mice that were not diabetic were treated with streptozotocin and then transplanted with NOD.SCID islets. The survival of the islet transplants in Group 1 is illustrated in Fig. 1
| Donor (islets) | Recipients | Treatment | Survival (days) |
|---|---|---|---|
| 1 NOD.SCID | NOD-stz | None | >74, >90, >85, >108, 117,>131, >162†, >162†, >204†,>218†, >218†, 218†, >230†,>230†, >233†, >315†, >315†,>327†, >364*, >364*,>392†, >417* |
| 2 None | NOD-SCID | Spl cells NOD sp. | 10, 11, 14 |
| 3 None | NOD.SCID | Spl cells NOD sp./stz | 22, 43, 29, 37, 37, 45, 45, 50 |
| 4 NOD.SCID | NOD-stz | Anti-CD4 (D-0) | 47, 55, >306*, >306*, >223, >223,>223, >223 |
| 5 NOD.SCID | NOD-stz | Anti-CD4 (D-233, 315, 315, 327, 392) | >25*, >27*, >61*, >55*, >37* |
| 6 NOD.SCID | NOD-stz | Anti-CD8 (D-0) | 11, 21, 13, 13 |
| 7 NOD.SCID | NOD-stz | Anti-CD8 (D- 201, 223, 223, 223, 223) | 13, >56*, >56*, >98, >112 |
Euglycaemic mice used for further manipulations as shown in Groups 2, 3 and 5.
Euglycaemic mice sacrificed at different times for histology. Anti-CD4 or anti-CD8 MoAb was given on 3 consecutive days starting as the day indicated (0·3 mg per day). D = day on which additional treatment was performed.
We asked if streptozotocin affects the ability of 5 × 107 splenocytes from NOD mice with established diabetes to transfer diabetes to non-diabetic animals. In Group 3, we transferred spleen cells from diabetic NOD mice after streptozotocin treatment. These spleen cells were less effective at transferring autoimmune diabetes to non-diabetic NOD.SCID recipients compared to the positive control in Group 2.
To verify that the spleen cells from NOD mice with established autoimmunity can transfer diabetes, three NOD.SCID recipient mice were given 50 million spleen cells from diabetic NOD mice. All became diabetic by 14 days, showing that these spleen cells can transfer diabetes (Table 2, Group 2) [33].
Finally, we asked whether a particular subset of T cells might be responsible for the development and maintenance of resistance to autoimmunity in streptozotocin-treated mice. Eight non-diabetic NOD mice were treated with streptozotocin at 6–8 weeks of age. In addition, they were treated with three daily doses of a depleting anti-CD4 antibody starting on the day of their NOD.SCID islet transplant. As shown in Group 4, two of these mice developed diabetes within 7–8 weeks, but the remaining six mice remained normoglycaemic for prolonged periods. Five additional streptozotocin-treated NOD mice were treated with anti-CD4 antibody roughly a year after their NOD.SCID islet transplants. As shown Group 5, Table 2, all these mice have remained euglycaemic. This dosing schedule for the administration of anti-CD4 antibody causes permanent acceptance of allografts [34,35]. In contrast to these results with anti-CD4 treatment, four mice (shown in Group 6, Table 2) treated with a depleting anti-CD8 antibody at the time of streptozotocin-treatment and NOD.SCID islet transplantation developed diabetes within 2 weeks. As shown in Group 7, one of five streptozotocin-treated mice that underwent CD8-depletion more than half a year after NOD.SCID islet transplantation became diabetic within 2 weeks. The other four mice have remained euglycaemic for a prolonged time.
To test if streptozotocin is markedly immunosuppressive, streptozotocin treated Balb/c (n = 4) and NOD mice (n = 5) received allogeneic B6 islets. NOD mice rejected their allogeneic grafts in 12–20 days, and the Balb/c mice rejected their allografts in 10–18 days. In addition, B6 allogeneic hearts were transplanted heterotopically into streptozotocin-treated NOD and Balb/c mice. These hearts were all rejected by 14 days, a time interval similar to non-streptozotocin-treated animals (data not shown). These data suggest that streptozotocin is minimally immunosuppressive, at least with respect to allograft immunity.
DISCUSSION
The NOD mouse strain provides an excellent in vivo model for testing strategies that might modulate the autoimmunity responsible for Type I diabetes [8,9]. Transplants of syngeneic islets into diabetic NOD mice reverse hyperglycaemia transiently until rejected by existing autoimmunity, thereby confirming that transplanted islets are susceptible to autoimmune destruction. This report demonstrates that streptozotocin both prevents and suppresses the antibeta cell autoimmunity in NOD mice as defined as the survival of syngeneic islets.
Syngeneic islets transplanted into streptozotocin-treated prediabetic or diabetic NOD enjoy prolonged or indefinite survival (Fig. 1; Table 1, Group 2; Table 2, Group 1). The streptozotocin effect appears complex. Spleen cells from diabetic NOD mice after streptozotocin appear less potent transferring diabetes (Table 2, Group 3). The euglycaemia in the streptozotocin-treated and islet-transplanted animals is clearly attributable to the islet graft because the native pancreas is depleted of beta cells, the islet graft shows strong insulin staining, and nephrectomy of the islet graft rapidly causes diabetes.
The streptozotocin effect observed in these experiments is not merely immunosuppressive. Transplanted allogeneic islets are readily rejected within 2 weeks in a variety of different strains of streptozotocin treated mice [5,6,36–38]. Allogeneic islets are also rejected in streptozotocin-treated NOD mice within 14 days [6,39,40] (see Results). Other immunosuppressive therapies commonly used in mice, such as anti-CD4 treatment and co-stimulatory blockade, cause indefinite survival of allogeneic islets but are marginally effective suppressing autoimmunity in NOD mice [40,41]. Our results also indicate that streptozotocin prevents the development of destructive autoimmunity in NOD mice, whereas alloxan does not, so that the streptozotocin effect is not mediated simply through the death of beta cells.
Many other investigators have described treatments that prevent the development of destructive autoimmunity in NOD mice [8,9]. These treatments have frequently involved manipulations that shift the balance from Th1 to Th2 cytokine production or that restore peripheral regulatory T cell function. These treatments may affect autoimmunity but have little effect on allotransplantation rejection [5]. Streptozotocin is one of the treatments that has been reported previously to prevent autoimmunity. Hugues et al. found that a single low-dose injection of streptozotocin (40 or 60 mg/kg) caused impaired T cell responses to islet antigens and that the mice were protected from developing spontaneous diabetes [42]. Their experiments suggested that the effect was due to the apoptosis of islet cells because it was not seen in mice expressing a caspase inhibitor and that it was associated with the development of regulatory T cells. Our results are consistent with these findings, except that Hugues et al. found evidence for CD4+ regulatory T cells, whereas our results suggested an important role for CD8+ cells.
In contrast to the numerous ways of preventing diabetes in NOD mice, few strategies exist that reverse the autoimmunity of NOD mice with established diabetes [5–14]. Thus, the effectiveness of streptozotocin treatment in allowing the long-term survival of syngeneic islets transplanted into diabetic NOD recipients indicates that this strategy is unusually powerful.
Takayama et al. previously reported that streptozotocin treatment of diabetic NOD mice allowed survival of syngeneic islets for 2–84 days in eight recipients with one other recipient maintaining graft survival for over 200 days [39]. In our experiments, all the recipients maintained indefinite graft survival with follow-up for more than 1 year. One difference between our experiments and those previously reported is that we used NOD.SCID islets for our transplants, whereas the previous study used islets from young NOD mice treated with antilymphocyte serum to suppress the inflammation in the donor pancreas. These transplants may have transferred some remaining donor lymphocytes along with the islet transplants. Takayama et al. considered the possibility that the streptozotocin effect was due to enhancement of suppressor activity [39]. However, they thought that the most likely explanation was that streptozotocin was a mild immunosuppressive agent.
However, numerous studies of both islet allotransplantation and xeno-transplantation after streptozotocin treatment have shown rapid rejection of these grafts [3,5,27,36,37,43,44]. Thus, it is unlikely that streptozotocin prevented the onset of autoimmunity and reversed it in our studies simply on the basis of its mild immunosuppressive properties. We found that depletion of a CD8 population altered the effect of streptozotocin treatment, and that spleen cells from diabetic NOD mice also treated with streptozotocin were less potent transferring diabetes. Thus, the immunoregulatory effects of streptozotocin are therefore likely complex. Our results, like those of Hugues et al., suggest that streptozotocin may prevent autoimmunity by inducing in part a regulatory T cell population. Hugues et al. suggested that the regulatory cells were induced by the limited apoptosis of beta cells induced by the low dose of streptozotocin they used for treatment. If this is true, our results suggest that streptozotocin can also be effective when full doses are used that induce complete beta cell destruction.
Depleting CD8 T cells at the time of streptozotocin treatment, rather than the elimination of CD4+ cells, blocked the effectiveness of the streptozotocin therapy (Table 2, Group 6). While most studies of the regulatory cells that modulate autoimmunity have suggested that CD4+ cells are primarily involved, our results indicated that some type of CD8+ cells are involved in the streptozotocin-induced suppression of NOD diabetic autoimmunity. Later depletion of either CD4+ cells or CD8+ cells had a small and inconsistent effect on autoimmunity. Thus, we could not determine at this time what balance might exist between various autoimmune effector and regulatory populations after streptozotocin treatment.
Understanding how streptozotocin affects autoimmunity in NOD mice will provide insights that might help to develop new strategies for the treatment of patients with Type I diabetes. This requires identification of the mechanism by which streptozotocin affects autoimmunity in NOD mice.
Acknowledgments
We thank Jack O’Neil from the Joslin Diabetes Center for providing NOD.SCID islets and Luba Zachachin for expert assistance with tissue processing. This work was funded in part by the Juvenile Diabetes Research Foundation Center for Islet Transplantation at Harvard Medical School and by NIH PO1 DK53087.
REFERENCES
- 1.Sibley RK, Sutherland DE, Goetz F, Michael AF. Recurrent diabetes mellitus in the pancreas iso – and allograft. A light and electron microscopic and immunohistochemical analysis of four cases. Laboratory Invest. 1985;53:132–44. [PubMed] [Google Scholar]
- 2.Drage M, Zaccone P, Phillips JM, Nicoletti F, Dawson J, Andrew Bradley J, Cooke A. Nondepleting anti-CD4 and soluble interleukin-1 receptor prevent autoimmune destruction of syngeneic islet grafts in diabetic NOD mice. Transplantation. 2002;74:611–9. doi: 10.1097/00007890-200209150-00005. [DOI] [PubMed] [Google Scholar]
- 3.Koulmanda M, Qipo A, Smith RN, Auchincloss H., Jr Pig islet xenografts are resistant to autoimmune destruction by NOD recipients after anti-CD4 treatment. Xenotransplantation. 2002;9:1. doi: 10.1034/j.1399-3089.2003.02040.x. [DOI] [PubMed] [Google Scholar]
- 4.Okitsu T, Bartlett ST, Hadley GA, Drachenberg CB, Farney AC. Recurrent autoimmunity accelerates destruction of minor and major histoincompatible islet grafts in nonobese diabetic (NOD) mice. Am J Transplant. 2001;1:138–45. [PubMed] [Google Scholar]
- 5.Li XC, Zand MS, Li Y, Zheng XX, Strom TB. On histocompatibility barriers, Th1 to Th2 immune deviation, and the nature of the allograft responses. J Immunol. 1998;161:2241–7. [PMC free article] [PubMed] [Google Scholar]
- 6.Makhlouf L, Kishimoto K, Smith RN, Abdi R, Koulmanda M, Winn HJ, Auchincloss H, Jr, Sayegh MH. The Role of Autoimmunity in Islet Allograft Destruction. Major Histocompatibility Complex Class II Matching Is Necessary for Autoimmune Destruction of Allogeneic Islet Transplants After T-Cell Costimulatory Blockade. Diabetes. 2002;51:3202–10. doi: 10.2337/diabetes.51.11.3202. [DOI] [PubMed] [Google Scholar]
- 7.Salomon B, Bluestone JA. Complexities of CD28/B7: CTLA-4 costimulatory pathways in autoimmunity and transplantation. Annu Rev Immunol. 2001;19:225–52. doi: 10.1146/annurev.immunol.19.1.225. [DOI] [PubMed] [Google Scholar]
- 8.Atkinson MA, Leiter EH. The NOD mouse model of Type I diabetes: as good as it gets? Nat Med. 1999;5:601–4. doi: 10.1038/9442. [DOI] [PubMed] [Google Scholar]
- 9.Delovitch TL, Singh B. The nonobese diabetic mouse as a model of autoimmune diabetes: immune dysregulation gets the NOD. Immunity. 1997;7:727–38. doi: 10.1016/s1074-7613(00)80392-1. [DOI] [PubMed] [Google Scholar]
- 10.Lakey JR, Singh B, Warnock GL, Rajotte RV. BCG immunotherapy prevents recurrence of diabetes in islet grafts transplanted into spontaneously diabetic NOD mice. Transplantation. 1994;57:1213–7. doi: 10.1097/00007890-199404270-00013. [DOI] [PubMed] [Google Scholar]
- 11.Wang T, Singh B, Warnock GL, Rajotte RV. Prevention of recurrence of IDDM in islet-transplanted diabetic NOD mice by adjuvant immunotherapy. Diabetes. 1992;41:114–7. doi: 10.2337/diab.41.1.114. [DOI] [PubMed] [Google Scholar]
- 12.Chatenoud L, Primo J, Bach JF. CD3 antibody-induced dominant self tolerance in overtly diabetic NOD mice. J Immunol. 1997;158:2947–54. [PubMed] [Google Scholar]
- 13.Sandberg JO, Eizirik DL, Sandler S. IL-1 receptor antagonist inhibits recurrence of disease after syngeneic pancreatic islet transplantation to spontaneously diabetic non-obese diabetic (NOD) mice. Clin Exp Immunol. 1997;108:314–7. doi: 10.1046/j.1365-2249.1997.3771275.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ryu S, Kodama S, Ryu K, Schoenfeld DA, Faustman DL. Reversal of established autoimmune diabetes by restoration of endogenous beta cell function. J Clin Invest. 2001;108:63–72. doi: 10.1172/JCI12335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wu Q, Salomon B, Chen M, Wang Y, Hoffman LM, Bluestone JA, Fu YX. Reversal of spontaneous autoimmune insulitis in nonobese diabetic mice by soluble lymphotoxin receptor. J Exp Med. 2001;193:1327–32. doi: 10.1084/jem.193.11.1327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Rabinovitch A, Suarez-Pinzon WL, Shapiro AM, Rajotte RV, Power R. Combination therapy with sirolimus and interleukin-2 prevents spontaneous and recurrent autoimmune diabetes in NOD mice. Diabetes. 2002;51:638–45. doi: 10.2337/diabetes.51.3.638. [DOI] [PubMed] [Google Scholar]
- 17.Gregori S, Giarratana N, Smiroldo S, Uskokovic M, Adorini LA. 1alpha,25-dihydroxyvitamin D (3) analog enhances regulatory T-cells and arrests autoimmune diabetes in NOD mice. Diabetes. 2002;51:1367–74. doi: 10.2337/diabetes.51.5.1367. [DOI] [PubMed] [Google Scholar]
- 18.Bertry-Coussot L, Lucas B, Danel C, Halbwachs-Mecarelli L, Bach JF, Chatenoud L, Lemarchand P. Long-term reversal of established autoimmunity upon transient blockade of the LFA-1/intercellular adhesion molecule-1 pathway. J Immunol. 2002;168:3641–8. doi: 10.4049/jimmunol.168.7.3641. [DOI] [PubMed] [Google Scholar]
- 19.Rayat GR, Singh B, Korbutt GS, Rajotte RV. Single injection of insulin delays the recurrence of diabetes in syngeneic islet-transplanted diabetic NOD mice. Transplantation. 2000;70:976–9. doi: 10.1097/00007890-200009270-00016. [DOI] [PubMed] [Google Scholar]
- 20.Herold KC, Hagopian W, Auger JA, et al. Anti-CD3 monoclonal antibody in new-onset Type I diabetes mellitus. N Engl J Med. 2002;346:1692–8. doi: 10.1056/NEJMoa012864. [DOI] [PubMed] [Google Scholar]
- 21.Rossini AA, Like AA, Dulin WE, Cahill GF., Jr Pancreatic beta cell toxicity by streptozotocin anomers. Diabetes. 1977;26:1120–4. doi: 10.2337/diab.26.12.1120. [DOI] [PubMed] [Google Scholar]
- 22.Schnedl WJ, Ferber S, Johnson JH, Newgard CB. STZ transport and cytotoxicity. Specific enhancement in GLUT2-expressing cells. Diabetes. 1994;43:1326–33. doi: 10.2337/diab.43.11.1326. [DOI] [PubMed] [Google Scholar]
- 23.Turk J, Corbett JA, Ramanadham S, Bohrer A, McDaniel ML. Biochemical evidence for nitric oxide formation from streptozotocin in isolated pancreatic islets. Biochem Biophys Res Commun. 1993;197:1458–64. doi: 10.1006/bbrc.1993.2641. [DOI] [PubMed] [Google Scholar]
- 24.Kroncke KD, Fehsel K, Sommer A, Rodriguez ML, Kolb-Bachofen V. Nitric oxide generation during cellular metabolization of the diabetogenic N-methyl-N-nitroso-urea streptozotozin contributes to islet cell DNA damage. Biol Chem Hoppe Seyler. 1995;376:179–85. doi: 10.1515/bchm3.1995.376.3.179. [DOI] [PubMed] [Google Scholar]
- 25.Yamamoto H, Uchigata Y, Okamoto H. Streptozotocin and alloxan induce DNA strand breaks and poly (ADP-ribose) synthetase in pancreatic islets. Nature. 1981;294:284–6. doi: 10.1038/294284a0. [DOI] [PubMed] [Google Scholar]
- 26.Gotoh M, Maki T, Kiyoizumi T, Satomi S, Monaco AP. An improved method for isolation of mouse pancreatic islets. Transplantation. 1985;40:437–8. doi: 10.1097/00007890-198510000-00018. [DOI] [PubMed] [Google Scholar]
- 27.Friedman T, Smith RN, Colvin RB, Iacomini J. A critical role for human CD4+ T-cells in rejection of porcine islet cell xenografts. Diabetes. 1999;48:2340–8. doi: 10.2337/diabetes.48.12.2340. [DOI] [PubMed] [Google Scholar]
- 28.Dialynas DP, Quan ZS, Wall KA, Pierres A, Quintans J, Loken MR, Pierres M, Fitch FW. Characterization of the murine T cell surface molecule, designated L3T4, identified by monoclonal antibody GK1.5. similarity of L3T4 to the human Leu-3/T4 molecule. J Immunol. 1983;131:2445–51. [PubMed] [Google Scholar]
- 29.Ghobrial RR, Boublik M, Winn HJ, Auchincloss H., Jr In vivo use of monoclonal antibodies against murine T cell antigens. Clin Immunol Immunopathol. 1989;52:486–506. doi: 10.1016/0090-1229(89)90162-1. [DOI] [PubMed] [Google Scholar]
- 30.Barbara JA, Turvey SE, Kingsley CI, Spriewald BM, Hara M, Witzke O, Morris PJ, Wood KJ. Islet allograft rejection can be mediated by CD4+, alloantigen experienced, direct pathway T cells of TH1 and TH2 cytokine phenotype. Transplantation. 2000;70:1641–9. doi: 10.1097/00007890-200012150-00017. [DOI] [PubMed] [Google Scholar]
- 31.Yoon JW, Yoon CS, Lim HW, et al. Control of autoimmune diabetes in NOD mice by GAD expression or suppression in beta cells. Science. 1999;284:1183–7. doi: 10.1126/science.284.5417.1183. [DOI] [PubMed] [Google Scholar]
- 32.Koulmanda M, Qipo A, Chebrolu S, O'Neil J, Auchincloss H, Smith RN. The effect of low versus high dose of streptozotocin in cynomolgus monkeys (Macaca fascicularis) Am J Transplant. 2003;3:267–72. doi: 10.1034/j.1600-6143.2003.00040.x. [DOI] [PubMed] [Google Scholar]
- 33.Christianson SW, Shultz LD, Leiter EH. Adoptive transfer of diabetes into immunodeficient NOD-scid/scid mice. Relative contributions of CD4+ and CD8+ T-cells from diabetic versus prediabetic NOD.NON-Thy-1a donors. Diabetes. 1993;42:44–55. doi: 10.2337/diab.42.1.44. [DOI] [PubMed] [Google Scholar]
- 34.Balasa B, Krahl T, Patstone G, Lee J, Tisch R, McDevitt HO, Sarvetnick N. CD40 ligand–CD40 interactions are necessary for the initiation of insulitis and diabetes in nonobese diabetic mice. J Immunol. 1997;159:4620–7. [PubMed] [Google Scholar]
- 35.Herbelin A, Gombert JM, Lepault F, Bach JF, Chatenoud L. Mature mainstream TCR alpha beta+CD4+ thymocytes expressing 1-selectin mediate ‘active tolerance’ in the nonobese diabetic mouse. J Immunol. 1998;161:2620–8. [PubMed] [Google Scholar]
- 36.Mandel TE, Hoffman L, Collier S, Carter WM, Koulmanda M. Organ culture of fetal mouse and fetal human pancreatic islets for allografting. Diabetes. 1982;31:39–47. doi: 10.2337/diab.31.4.s39. [DOI] [PubMed] [Google Scholar]
- 37.Mandel TE, Koulmanda M. Effect of culture conditions on fetal mouse pancreas in vitro and after transplantation in syngeneic and allogeneic recipients. Diabetes. 1985;34:1082–7. doi: 10.2337/diab.34.11.1082. [DOI] [PubMed] [Google Scholar]
- 38.Abdi R, Smith RN, Makhlouf L, Najafian N, Luster AD, Auchincloss H, Jr, Sayegh MH. The role of CC chemokine receptor 5 (CCR5) in islet allograft rejection. Diabetes. 2002;51:2489–95. doi: 10.2337/diabetes.51.8.2489. [DOI] [PubMed] [Google Scholar]
- 39.Takayama Y, Ichikawa T, Maki T. Effect of STZ administration on islet isograft and allograft survival in NOD mice. Diabetes. 1993;42:324–9. doi: 10.2337/diab.42.2.324. [DOI] [PubMed] [Google Scholar]
- 40.Guo Z, Wu T, Kirchhof N, Mital D, Williams JW, Azuma M. Sutherland DE, Hering BJ. Immunotherapy with nondepleting anti-CD4 monoclonal antibodies but not CD28 antagonists protects islet graft in spontaneously diabetic nod mice from autoimmune destruction and allogeneic and xenogeneic graft rejection. Transplantation. 2001;71:1656–65. doi: 10.1097/00007890-200106150-00027. [DOI] [PubMed] [Google Scholar]
- 41.Molano RD, Berney T, Li H, et al. Prolonged islet graft survival in NOD mice by blockade of the CD40-CD154 pathway of T-cell costimulation. Diabetes. 2001;50:270–6. doi: 10.2337/diabetes.50.2.270. [DOI] [PubMed] [Google Scholar]
- 42.Hugues S, Mougneau E, Ferlin W, et al. Tolerance to islet antigens and prevention from diabetes induced by limited apoptosis of pancreatic beta cells. Immunity. 2002;16:169–81. doi: 10.1016/s1074-7613(02)00273-x. [DOI] [PubMed] [Google Scholar]
- 43.Ferrari-Lacraz S, Zheng XX, Kim YS, Li Y, Maslinski W, Li XC, Strom TB. An antagonist IL-15/Fc protein prevents costimulation blockade-resistant rejection. J Immunol. 2001;167:3478–85. doi: 10.4049/jimmunol.167.6.3478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Mandel TE. Transplantation of organ cultured fetal pancreas: Experimental studies and potential clinical application in diabetes mellitus. World J Surg. 1984;8:158–68. doi: 10.1007/BF01655131. [DOI] [PubMed] [Google Scholar]