Abstract
Respiratory syncytial virus (RSV) infection may have an effect on the development of T cell memory responses. RSV bronchiolitis in infants is associated with a transient decline in circulating lymphocytes. We hypothesized that the mechanism underlying this lymphopenia is apoptosis. Blood was taken from 32 infants during primary RSV bronchiolitis and three months later. Using flow cytometry, we found that absolute numbers of both CD3+/CD4+ T-helper lymphocytes (P = 0·029) and CD3+/CD8+ cytotoxic lymphocytes (CTL) (P = 0·043) were significantly reduced during acute infection. Up-regulated expression both of Fas (P < 0·001) and tumour necrosis factor-related apoptosis-inducing ligand (TRAIL) receptor (P < 0·001) was found during acute illness on both CD3+/CD4+ and CD3+/CD8+ lymphocytes, when compared with convalescent samples. Expression of Fas on CD4+ lymphocytes was inversely related to CD4+ number (P = 0·03). Plasma levels of soluble Fas ligand (P = 0·028) and caspase-1 (P = 0·037), determined by enzyme-linked immunosorbent assay, were increased during bronchiolitis. Plasma interleukin-18, a product of caspase-1 activity, was not raised. Taken together, these data suggest that in acute RSV infection, CD4+ helper lymphocytes and CD8+ cytotoxic lymphocytes are primed to undergo apoptosis. This is a mechanism through which lymphopenia may occur and T cell memory may be altered.
Keywords: apoptosis, bronchiolitis, lymphocyte, respiratory syncytial virus, TRAIL receptor, virus
INTRODUCTION
Respiratory syncytial virus (RSV), a human paramyxovirus, is the most important cause of viral lower respiratory tract disease in infants and children world-wide. Primary RSV bronchiolitis causes substantial morbidity, mortality and cost with hospital admission rates of about 2·5% [1]. A high proportion of infants will develop recurrent episodes of wheeze, cough and asthma-like symptoms after recovery from bronchiolitis [2]. Interest has therefore focused on the role of the host immune response in the pathogenesis of bronchiolitis and its clinical sequelae.
The immunopathogenesis of RSV bronchiolitis plays an important part in the clinical manifestations of the primary infection. Evidence from human and mouse studies suggest that lymphocytes clear RSV during infection. Children with cell-mediated immunodeficiencies take longer to terminate infection with RSV [3]. In the mouse model, Graham et al. [4] have studied the effect of depletion of CD4+ or CD8+ lymphocytes during acute RSV infection. CD4+ and CD8+ lymphocyte subsets are involved in terminating primary infection and if both T lymphocyte subsets are depleted RSV shedding is prolonged. However, depletion of lymphocytes in mice also reduces clinical symptoms and lung injury. Research into the increased pathology in recipients of the formalin-inactivated RSV vaccine in the 1960s has implicated aberrant CD4 lymphocytes as the mediators [5]. These data suggest that lymphocytes may also play a role in causing lung pathology in RSV infection.
Previous reviews have commented that the white cell count in RSV bronchiolitis is varied and there is a preponderance of neutrophils [6,7]. The most striking change in circulating white cell count is a reduction in lymphocyte numbers and this effect is more pronounced in sicker children [8]. It has been suggested that this reduction in lymphocyte count be due to redistribution to the lung [9]. However, lymphopenia is also known to occur in other viral illnesses, including measles, severe acute respiratory syndrome (SARS) and ebola [10–12]. There are data to suggest that apoptosis of lymphocytes occurs in both these illnesses and may offer a mechanism for this lymphocyte decline.
Apoptosis is programmed cell death via a number of regulated, energy-dependent pathways. Two key apoptotic cascades have so far been identified as important in the removal of mature, circulating lymphocytes; death receptor mediated and mitochondrial-dependent [13]. Cell surface death receptors, including Fas (CD95) and tumour necrosis factor-related apoptosis-inducing ligand (TRAIL) receptor, are activated by the binding of specific ligands: Fas ligand and TRAIL. Both pathways lead to the activation of a cascade of specific proteolytic enzymes, caspases, causing the cell to die. Fas ligand may be membrane bound or soluble (sFas) ligand and the soluble form may mediate Fas-dependent apoptosis of bystander cells [14].
The pathways of apoptosis and inflammation are intertwined. Caspase-1 converts the inactive molecules, pro-interleukin-1 and pro-interleukin-18, into the active cytokines, interleukin-1 (IL-1) and interleukin-18 (IL-18), respectively. Interleukin-18 activates the immune system, stimulating chemokine release and the development of a Th1 or Th2 response and IL-18 affects apoptosis through the up-regulation of Fas [15].
In this study of infants admitted to hospital with RSV bronchiolitis we undertook to find evidence to support or refute the hypothesis that circulating CD3+/CD4+ and CD3+/CD8+ lymphocytes may be undergoing apoptosis. Our results suggest that RSV disease may be associated with lymphocyte apoptosis and offers a novel mechanism through which RSV may alter long-term immune responses.
METHODS
Subjects
The study protocol was approved by the Addenbrooke's Hospital Cambridge Local and Regional Ethics Committee. Infants were included in the study if admitted to hospital with a diagnosis of bronchiolitis at 9 months of age or younger; they were RSV positive by routine nasopharyngeal aspirate immunofluorescence and if parents gave written informed consent. For the purposes of this study, the clinical diagnosis of bronchiolitis also included a requirement for supplemental oxygen to maintain arterial saturations greater than 92%. All blood samples were taken within 48 h of admission. It is known that parental recollection of day that their child's symptoms began is unreliable and so we chose not state which day of the illness blood was sampled on. However, the day of admission to hospital is usually close to the peak of clinical symptoms and we attempted to obtain blood samples as close to this time as possible. Children with a suspected or known immunodeficiency were excluded from the study. Parents were asked to return with their child for convalescent blood sampling 3 months following the acute illness. All samples were taken immediately to the laboratory for analysis for processing after being taken.
Blood samples
Blood samples were heparinized. An EDTA sample was taken for a full blood count and white cell differential. Plasma was separated from the heparinized sample and frozen at −80°C. The remaining cellular components were resuspended in phosphate-buffered saline (pH 7·2) supplemented with 0·45% human albumin (PBSA). Lymphocytes were obtained by density gradient separation (Histopaque: Sigma, Poole, UK) and resuspended in PBSA to a concentration of 10–20 × 109/l.
Flow cytometric acquisition and analysis
1 × 106 lymphocytes were incubated with monoclonal antibodies to CD45-ECD (Beckman Coulter), CD3- RPE Cy5 (DakoCytomation, UK), CD4-FITC or PE (CaltagMedsystems, UK), CD8-Texas Red (Caltag), CD95-FITC (DakoCytomation), and TRAIL R-PE (Pharmingen) in four colour combinations. All incubations were in the dark at room temperature for 15 min. The stained cells were then washed twice in PBSA and analysed on a Beckman Coulter Epics XL flow cytometer using System II software. A standard lymphocyte gate was set using low forward and side scatter properties. The position of the positive regions were set with isotype matched controls. CD45 expression allowed the extent of red cell contamination in the gated region to be assessed. Results were corrected accordingly. Results were expressed as a percentage and as the mean fluorescence intensity. Flow cytometric data was analysed using WinMDI version 2·8 software.
Enzyme-linked immunosorbent assays
Plasma levels of Soluble Fas Ligand, Caspase-1 and Interleukin-18 Assays were measured with commercially available enzyme-linked immunosorbent assay kits: soluble Fas Ligand (Diaclone IDS Ltd. UK), Caspase-1 (R & D Systems Europe Ltd, UK), IL-18 (Alexis Corporation (UK) Ltd).
Statistical analysis
Values are reported as median with interquartile range (IQR) and 95% confidence limits for the median using box and whisker plots. The data was analysed using Mann–Whitney test for nonparametric data. A P-value < 0·05 was considered to be significant.
RESULTS
Patients and outcomes
Thirty-two infants (9 girls; 23 boys) were enrolled in the study; their characteristics are summarized in Table 1. Thirteen (40%) of these children required endotracheal intubation and mechanical ventilation for respiratory failure and were admitted to the paediatric intensive care unit (PICU) during part of their admission. Primary apnoea was not the indication for ventilation in any of these patients. Nineteen (60%) children only required paediatric ward admission for supplemental oxygen therapy and fluid management.
Table 1.
Characteristics of study population*
| Acute | |||||
|---|---|---|---|---|---|
| Acute | Convalescent | PICU | Ward | Comparison of PICU v Ward | |
| Number | 32 | 23 | 13 | 19 | |
| Age (weeks) | 8·8 (5·2–14·6) | 24·0 (19·9–38·7) | 6·4 (5·3–9·0) | 11·7 (4·9–26·7) | P = 0·045 |
| Weight (kg) | 4·66 (3·44–6·08) | 7·51 (6·60–9·95) | 3·40 (2·65–4·80) | 5·40 (4·32–7·60) | P = 0·004 |
| Gestational age (weeks) | 38·5 (36·0–40·0) | 37·0 (30·5–39·0) | 40·0 (37·0–40·0) | P = 0·013 | |
data expressed as median and interquartile range.
Overall the median (IQR) for the group were 4·66 kg (3·44–6·08) for weight, 8·8 weeks (5·2–14·6) for age at admission and 38·5 weeks (36–40) for gestation at birth. In common with previous studies of children those admitted to the PICU were younger (P = 0·045), weighed less (P = 0·004) and had a lower gestational age at birth (P = 0·013). All children survived their illnesses and were discharged home.
From the acute cohort, 23 (72%) infants returned for a convalescent blood samples at a mean of 15 weeks after the acute illness. In this follow-up cohort, there were 7 girls and 16 boys and 8 of these children had required PICU admission. Due to the design of the study, the follow-up infants are, on average, 3 months older and correspondingly heavier than the acute group. In one patient from each cohort, a white cell differential count was not obtained.
T lymphocytes and subsets during RSV infection
Absolute numbers of CD3+ lymphocytes were reduced in RSV bronchiolitis (P = 0·02). Both CD3+/CD4+ lymphocytes (P = 0·029) and CD3+/CD8+ (P = 0·043) lymphocytes were significantly reduced during illness (Table 2).
Table 2.
Peripheral blood lymphocyte populations in acute RSV infected and convalescent infants*
| Acute | Convalescent | P-value | |
|---|---|---|---|
| Total white cell count (×109/l) | 10·9 (8·30–14·2) | 11·6 (9·20–13·8) | 0·222 |
| Lymphocytes (×109/l) | 5·55 (3·25–7·43) | 7·25 (6·03–9·43) | 0·010 |
| CD3+ T -lymphocytes (×109/l) | 3·25 (1·94–4·16) | 3·75 (3·06–4·72) | 0·020 |
| CD4+ T-helper lymphocytes (×109/l) | 2·09 (1·08–3·25) | 2·80 (2·09–3·61) | 0·029 |
| CD8+ Cytotoxic T-lymphocytes (×109/l) | 0·50 (0·26–0·79) | 0·79 (0·41–1·15) | 0·043 |
data expressed as median and interquartile range.
Fas and TRAIL–R on CD4+ and CD8+ lymphocytes during RSV infection
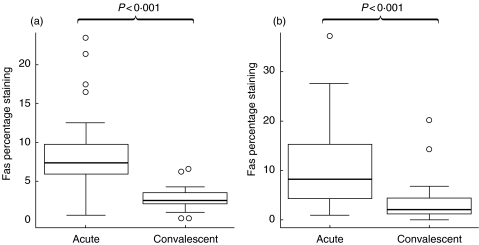
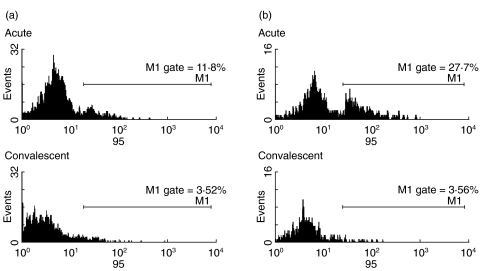
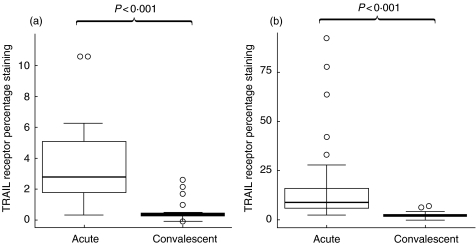
In acute illness CD3+/CD4+ lymphocytes had up-regulation of Fas (CD95) compared with samples taken at convalescence (P < 0·001); both the proportion staining high for Fas and the median fluorescent intensity were increased (Figs 1a and 2a). CD3+/CD8+ lymphocytes also expressed higher levels of surface Fas (P < 0·001) compared with convalescent samples (Figs 1b and 2b). TRAIL receptor was also up regulated on the CD3+/CD4+ population (P < 0·001) and on the CD3+/CD8+ population (P < 0·001) (Fig. 3a,b).
Fig. 1.
Fas (CD95) staining on (a) CD4+ lymphocytes and (b) CD8+ lymphocytes from acute RSV-infected and convalescent infants. Data presented as medians and interquartile ranges with outliers.
Fig. 2.
Examples of Acute and Convalescent Fas (CD95) staining on (a) CD4+ and (b) CD8+ lymphocytes. Acute and Convalescent plots are taken from same patient.
Fig. 3.
TRAIL receptor staining on (a) CD4+ lymphocytes and (b) CD8+ lymphocytes from acute RSV-infected and convalescent infants. Data presented as medians and interquartile ranges with outliers.
Correlation between Fas and lymphocyte subsets
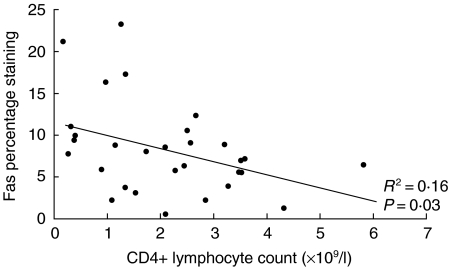
To support the hypothesis that increased expression of death receptors on lymphocytes is associated with lymphopenia, we performed a linear regression analysis. Levels of Fas expression correlated with CD4+ count (R2 = 0·16, P = 0·03) (Fig. 4).
Fig. 4.
Correlation of Fas percentage staining on CD4+ lymphocytes with CD4+ lymphocyte count.
Levels of soluble Fas ligand in plasma during RSV infection
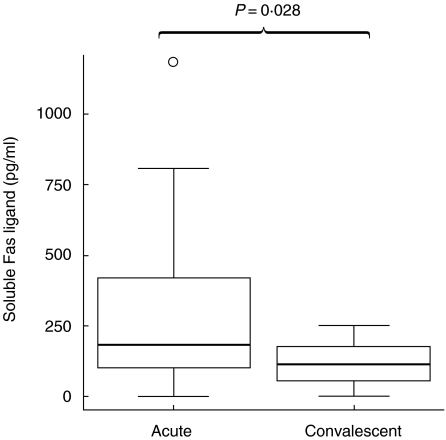
Plasma levels of sFas ligand were significantly higher in the acute samples compared with the convalescent samples (P = 0·028) (Fig. 5).
Fig. 5.
Plasma levels of soluble Fas ligand from acute RSV-infected and convalescent infants. Data presented as medians and interquartile ranges with outliers.
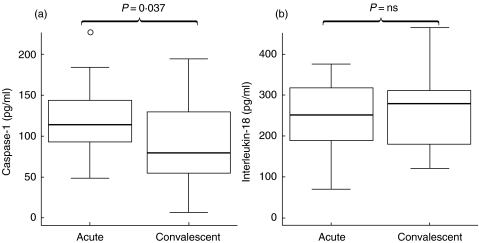
Levels of Caspase-1 and Interleukin-18 in plasma during RSV infection
Plasma levels of Caspase-1 were significantly higher in the acute samples than in convalescent samples (P = 0·037). There was no statistically significant difference between plasma levels of IL-18 from acute and convalescent samples and both medians were within the normal reported range for IL-18 in healthy adults (Fig. 6).
Fig. 6.
Plasma levels of (a) caspase-1 and (b) Interleukin-18 from acute RSV-infected and convalescent infants. Data presented as medians and interquartile ranges with outliers.
Effect of age on lymphocyte counts
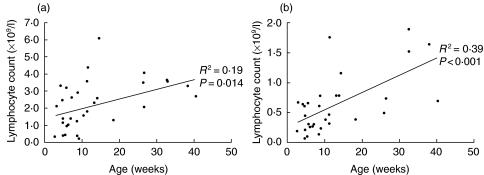
Lymphocyte subsets in acute illness were analysed with respect to age. Absolute lymphocyte counts (R2 = 0·25, P = 0·004), CD3+ (R2 = 0·27, p = 0·003), CD3+/CD4+ (R2 = 0·19, P = 0·014) and CD3+/CD8+ (R2 = 0·39, P < 0·001) counts were all inversely related to age (Fig. 7). There was no relationship between age and the same lymphocyte counts in convalescent samples.
Fig. 7.
Correlation of (a) CD4+ lymphocyte count and (b) CD8+ lymphocyte count with age of infant with acute RSV infection.
DISCUSSION
In this study we have looked for evidence that lymphocyte apoptosis may be a mechanism for RSV-induced lymphopenia. We have found that absolute counts of CD4+ helper T cells and CD8+ cytotoxic lymphocytes are reduced in RSV bronchiolitis. We have also demonstrated up-regulated expression of cell surface receptors (Fas and TRAIL receptor) on CD4+ and CD8+ lymphocytes and increased plasma levels of sFas ligand in the acute illness. Increased Fas expression correlated with lymphopenia. Direct evidence of intravascular apoptosis was not sought since the small quantity of blood that can be obtained from a critically ill baby precluded the use of direct assays such as DNA laddering. Taken together these findings suggest that death receptor-mediated apoptosis is involved in the reduction of lymphocyte numbers in this disease. Our data show that there is an inverse correlation with age suggesting there may be a critical period of infancy during which these effects are most pronounced.
We demonstrated elevation of plasma caspase-1 but not IL-18 in acute illness. IL-18 is important in determining lymphocyte phenotype and has been linked to the modulation of apoptosis. Our results for caspase-1 and IL-18 may also have implications for lymphocyte functioning and the regulation of apoptosis.
We have already reported that lymphopenia is more pronounced in sicker children [8]. de Weerd et al. [9] studied 18 children under the age of 2 years with acute RSV bronchiolitis and showed a significant reduction in CD8+ cells in all infected infants and reductions of all lymphocyte subsets in those ventilated. Their data also found nonsignificant reductions of total lymphocyte, CD3+ and CD4+ counts for all RSV positive patients. Roman et al. [16] found nonsignificant reductions in total and all T-lymphocyte subsets. The larger size of our study group and inclusion of more severely affected infants may help to explain our clearer findings.
RSV predominantly infects respiratory epithelial cells. It has been shown that in vitro infection of type 2 pneumocytes with RSV leads to apoptosis. Increased levels of Fas and Fas-ligand expression can be demonstrated on infected cells and this is through RSV mediated up-regulation of the nuclear transcription factor NF-IL-6 [17]. Reverse-transcriptase polymerase chain reaction (RT-PCR) has been used to show that RSV viral and mRNA can be detected in PBMCs from a small number of infants with acute bronchiolitis [18,19]. However an extensive decrease in the number of lymphocytes is unlikely to be explained by directly infected cells which are at very low levels suggesting bystander apoptosis may be important.
In measles, apoptosis of uninfected lymphocytes is also associated with up-regulated cell surface expression of Fas and TRAIL-R [20]. Lymphocyte apoptosis occurs in infection with ebola virus [21] which may be closely genetically linked to RSV [22]. Bystander lymphocyte apoptosis occurs in severe sepsis [23]. In a mouse model of severe sepsis blocking apoptosis with specific caspases inhibitors is associated with improved outcome. Although these models are very different to natural infection, the data from mice suggests possible future clinical therapeutic interventions [24].
In studies of the coronavirus associated with the severe acute respiratory syndrome (SARS), lymphopenia has been described as a hallmark laboratory finding [25]. The reasons for this lymphopenia are unknown but Panesar [26] has offered the suggestion that the use of steroids or loss of vascular integrity may have contributed. Our data would indicate that lymphocyte apoptosis might be a target for future study in this disease [27].
Different subsets of T cells are believed to have different susceptibility to apoptosis. In atopic dermatitis, there is data to suggest that apoptotic cell death may remove Th1 cells allowing Th2 responses to become dominant [28]. In this way lymphocyte apoptosis may offer a mechanism through which the generation of long-term immune memory may be altered.
The debate about the causes of long-term cough and wheeze after bronchiolitis has lead to a polarization of views. Children hospitalized with RSV bronchiolitis are at increased risk for allergic sensitization compared with controls [29]. There is evidence from animal models that RSV can be associated with both the allergic sensitization and the maintenance airways hyperresponsiveness [30,31]. Early allergic sensitization has been identified as a key risk factor for persistent asthma but the role of viruses in early life remains controversial [32].
Cell mediated immunity including proliferation, cytotoxicity, memory and cytokine production may all be affected by RSV infection. Preston et al. [33] have shown that T cell proliferative responses may be inhibited by RSV. RSV inhibits CD8+ T cells infiltrating the lung and the development of CD8+ T-cell memory [34]. In the mouse model different proteins of RSV have been shown to prime specific lymphocyte subsets. The surface glycoprotein G is associated with the induction of type 2 T cells, and lung eosinophilia during RSV infection [35,36].
Interleukin-18 is an important early cytokine that, together with IL-12, regulates interferon-γ (IFN-γ) production and promotes the development of the type 1 T-helper response [37]. In contrast, in the absence of IL-12, IL-18 may stimulate the development of type 2 immune responses. Our data suggest that IL-18 levels are not increased in acute bronchiolitis. Legg et al. [38] have shown that LPS stimulated PBMCs from children with acute bronchiolitis have reduced IL-18 mRNA expression when compared to those from children with RSV-negative upper respiratory tract infections. In contrast a study of nasal secretions from infants with RSV suggests that IL-18 production by epithelial cells may be elevated [39].
In summary, our data support the hypothesis that the lymphopenia seen in RSV bronchiolitis is associated with apoptosis. The effect is most pronounced in younger infants suggesting there is an important time in infancy when the immune system may be vulnerable. The reduction in lymphocytes is related to the degree of cellular expression of the receptors for apoptosis. Our findings mirror those in severe sepsis where data indicates that inhibition of apoptosis may ameliorate illness. Future dissection of this novel mechanism may help to delineate the delayed effects of RSV infection and offer new therapeutic avenues.
Acknowledgments
We thank Dr R. Patel for help taking some of the blood samples, the nurses and doctors in paediatrics and virology for helping to identify children with RSV bronchiolitis. We are grateful to Miss Eleanor Pinto for assistance in the study and statistical design. We would particularly like to thank all the parents and children who made the study possible. We are grateful to the H.C. Roscoe Fellowship, British Medical Association, 2002 for the financial support for this project.
References
- 1.Leader S, Kohlhase K. Respiratory syncytial virus-coded pediatric hospitalizations 1997 to 1999. Pediatr Infect Dis J. 2002;21:629–32. doi: 10.1097/00006454-200207000-00005. [DOI] [PubMed] [Google Scholar]
- 2.Martinez FD. Respiratory syncytial virus bronchiolitis and the pathogenesis of childhood asthma. Pediatr Infect Dis J. 2003;22:S76–82. doi: 10.1097/01.inf.0000053889.39392.a7. [DOI] [PubMed] [Google Scholar]
- 3.Hall CB, Powell KR, MacDonald NE, Gala CL, Menegus ME, Suffin SC, Cohen HJ. Respiratory syncytial viral infection in children with compromised immune function. N Engl J Med. 1986;315:77–81. doi: 10.1056/NEJM198607103150201. [DOI] [PubMed] [Google Scholar]
- 4.Graham BS, Bunton LA, Wright PF, Karzon DT. Role of T lymphocyte subsets in the pathogenesis of primary infection and rechallenge with respiratory syncytial virus in mice. J Clin Invest. 1991;88:1026–33. doi: 10.1172/JCI115362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Openshaw PJ, Culley FJ, Olszewska W. Immunopathogenesis of vaccine-enhanced RSV disease. Vaccine. 2001;20(Suppl. 1):S27–31. doi: 10.1016/s0264-410x(01)00301-2. [DOI] [PubMed] [Google Scholar]
- 6.Hall CB. Respiratory Syncytial Virus. In: Feigin RD, Cherry JD, editors. Textbook of Pediatric Infectious Diseases. Philadelphia: W.B. Saunders Co; 1998. pp. 2084–111. [Google Scholar]
- 7.Wohl MEB. Bronchiolitis. In: Chernick V, Kendig EL, editors. Kendig's Disorders of the Respiratory Tract in Children. Philadelphia: W.B. Saunders Co; 1998. pp. 473–85. [Google Scholar]
- 8.O'Donnell DR, Carrington D. Peripheral blood lymphopenia and neutrophilia in children with severe respiratory syncytial virus disease. Pediatr Pulmonol. 2002;34:128–30. doi: 10.1002/ppul.10140. [DOI] [PubMed] [Google Scholar]
- 9.de Weerd W, Twilhaar WN, Kimpen JLT. cell subset analysis in peripheral blood of children with RSV bronchiolitis. Scand J Infect Dis. 1998;30:77–80. doi: 10.1080/003655498750002349. [DOI] [PubMed] [Google Scholar]
- 10.Coovadia HM, Wesley A, Brain P, Henderson LG, Hallett AF, Vos GH. Immunoparesis and outcome in measles. Lancet. 1977;1:619–21. doi: 10.1016/s0140-6736(77)92056-6. [DOI] [PubMed] [Google Scholar]
- 11.Wong RS, Wu A, To KF, et al. Haematological manifestations in patients with severe acute respiratory syndrome: retrospective analysis. Br Med J. 2003;326:1358–62. doi: 10.1136/bmj.326.7403.1358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Geisbert TW, Hensley LE, Gibb TR, Steele KE, Jaax NK, Jahrling PB. Apoptosis induced in vitro and in vivo during infection by Ebola and Marburg viruses. Lab Invest. 2000;80:171–86. doi: 10.1038/labinvest.3780021. [DOI] [PubMed] [Google Scholar]
- 13.Budd RC. Activation-induced cell death. Curr Opin Immunol. 2001;13:356–62. doi: 10.1016/s0952-7915(00)00227-2. [DOI] [PubMed] [Google Scholar]
- 14.Zarozinski CC, McNally JM, Lohman BL, Daniels KA, Welsh RM. Bystander sensitization to activation-induced cell death as a mechanism of virus-induced immune suppression. J Virol. 2000;74:3650–8. doi: 10.1128/jvi.74.8.3650-3658.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Reddy P, Teshima T, Kukuruga M, Ordemann R, Liu C, Lowler K, Ferrara JL. Interleukin-18 Regulates Acute Graft-Versus-Host Disease by Enhancing Fas-mediated Donor T Cell Apoptosis. J Exp Med. 2001;194:1433–40. doi: 10.1084/jem.194.10.1433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Roman M, Calhoun WJ, Hinton KL, Avendano LF, Simon V, Escobar AM, Gaggero A, Diaz PV. Respiratory syncytial virus infection in infants is associated with predominant Th-2-like response. Am. J Respir Crit Care Med. 1997;156:190–5. doi: 10.1164/ajrccm.156.1.9611050. [DOI] [PubMed] [Google Scholar]
- 17.O'Donnell DR, Milligan L, Stark JM. Induction of CD95 (Fas) and apoptosis in respiratory epithelial cell cultures following respiratory syncytial virus infection. Virology. 1999;257:198–207. doi: 10.1006/viro.1999.9650. [DOI] [PubMed] [Google Scholar]
- 18.Rohwedder A, Keminer O, Forster J, Schneider K, Schneider E, Werchau H. Detection of respiratory syncytial virus RNA in blood of neonates by polymerase chain reaction. J Med Virol. 1998;54:320–7. doi: 10.1002/(sici)1096-9071(199804)54:4<320::aid-jmv13>3.0.co;2-j. [DOI] [PubMed] [Google Scholar]
- 19.O'Donnell DR, McGarvey MJ, Tully JM, Balfour-Lynn IM, Openshaw PJ. Respiratory syncytial virus RNA in cells from the peripheral blood during acute infection. J Pediatr. 1998;133:272–4. doi: 10.1016/s0022-3476(98)70234-3. [DOI] [PubMed] [Google Scholar]
- 20.Okada H, Kobune F, Sato TA, et al. Extensive lymphopenia due to apoptosis of uninfected lymphocytes in acute measles patients. Arch Virol. 2000;145:905–20. doi: 10.1007/s007050050683. [DOI] [PubMed] [Google Scholar]
- 21.Baize S, Leroy EM, Georges-Courbot MC, et al. Defective humoral responses and extensive intravascular apoptosis are associated with fatal outcome in Ebola virus-infected patients. Nat Med. 1999;5:423–6. doi: 10.1038/7422. [DOI] [PubMed] [Google Scholar]
- 22.Barr J, Chambers P, Pringle CR, Easton AJ. Sequence of the major nucleocapsid protein gene of pneumonia virus of mice: sequence comparisons suggest structural homology between nucleocapsid proteins of pneumoviruses, paramyxoviruses, rhabdoviruses and filoviruses. J Gen Virol. 1991;72:677–85. doi: 10.1099/0022-1317-72-3-677. [DOI] [PubMed] [Google Scholar]
- 23.Hotchkiss RS, Tinsley KW, Swanson PE, et al. Sepsis-induced apoptosis causes progressive profound depletion of B and CD4+ T lymphocytes in humans. J Immunol. 2001;166:6952–63. doi: 10.4049/jimmunol.166.11.6952. [DOI] [PubMed] [Google Scholar]
- 24.Hotchkiss RS, Chang KC, Swanson PE, et al. Caspase inhibitors improve survival in sepsis: a critical role of the lymphocyte. Nat Immunol. 2000;1:496–501. doi: 10.1038/82741. [DOI] [PubMed] [Google Scholar]
- 25.Lee N, Hui D, Wu A, et al. A major outbreak of severe acute respiratory syndrome in Hong Kong. N Engl J Med. 2003;348:1986–94. doi: 10.1056/NEJMoa030685. [DOI] [PubMed] [Google Scholar]
- 26.Panesar NS. Lymphopenia in SARS. Lancet. 2003;361:1985. doi: 10.1016/S0140-6736(03)13557-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.O'Donnell R, Tasker RC, Roe MFE. SARS: understanding the coronavirus: Apoptosis may explain lymphopenia of SARS. Br Med J. 2003;327:620–62b. doi: 10.1136/bmj.327.7415.620-b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Akdis M, Trautmann A, Klunker S, et al. T helper (Th) 2 predominance in atopic diseases is due to preferential apoptosis of circulating memory/effector Th1 cells. FASEB J. 2003;17:1026–35. doi: 10.1096/fj.02-1070com. [DOI] [PubMed] [Google Scholar]
- 29.Sigurs N, Bjarnason R, Sigurbergsson F, Kjellman B. Respiratory syncytial virus bronchiolitis in infancy is an important risk factor for asthma and allergy at age 7. Am J Respir Crit Care Med. 2000;161:1501–7. doi: 10.1164/ajrccm.161.5.9906076. [DOI] [PubMed] [Google Scholar]
- 30.O'Donnell DR, Openshaw PJM. Anaphylactic sensitization to aeroantigen during respiratory virus infection. Clin Exp Allergy. 1998;28:1501–8. doi: 10.1046/j.1365-2222.1998.00438.x. [DOI] [PubMed] [Google Scholar]
- 31.Peebles RSJ, Sheller JR, Johnson JE, Mitchell DB, Graham BS. Respiratory syncytial virus infection prolongs methacholine-induced airway hyperresponsiveness in ovalbumin-sensitized mice. J Med Virol. 1999;57:186–92. doi: 10.1002/(sici)1096-9071(199902)57:2<186::aid-jmv17>3.0.co;2-q. [DOI] [PubMed] [Google Scholar]
- 32.Martinez FD. Viruses and atopic sensitization in the first years of life. Am. J Respir Crit Care Med. 2000;162:S95–S99. doi: 10.1164/ajrccm.162.supplement_2.ras-8. [DOI] [PubMed] [Google Scholar]
- 33.Preston FM, Beier PL, Pope JH. Infectious respiratory syncytial virus (RSV) effectively inhibits the proliferative T cell response to inactivated RSV in vitro. J Infect Dis. 1992;165:819–25. doi: 10.1093/infdis/165.5.819. [DOI] [PubMed] [Google Scholar]
- 34.Chang J, Braciale TJ. Respiratory syncytial virus infection suppresses lung CD8+ T-cell effector activity and peripheral CD8+ T-cell memory in the respiratory tract. Nat Med. 2002;8:54–60. doi: 10.1038/nm0102-54. [DOI] [PubMed] [Google Scholar]
- 35.Alwan WH, Kozlowska WJ, Openshaw PJ. Distinct types of lung disease caused by functional subsets of antiviral T cells. J Exp Med. 1994;179:81–9. doi: 10.1084/jem.179.1.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Openshaw PJ. Immunity and immunopathology to respiratory syncytial virus. The mouse model. Am J Respir Crit Care Med. 1995;152:S59–S62. doi: 10.1164/ajrccm/152.4_Pt_2.S59. [DOI] [PubMed] [Google Scholar]
- 37.Okamura H, Kashiwamura S, Tsutsui H, Yoshimoto T, Nakanishi K. Regulation of interferon-gamma production by IL-12 and IL-18. Curr Opin Immunol. 1998;10:259–64. doi: 10.1016/s0952-7915(98)80163-5. [DOI] [PubMed] [Google Scholar]
- 38.Legg JP, Hussain IR, Warner JA, Johnston SL, Warner JO. Type 1 and type 2 cytokine imbalance in acute respiratory syncytial virus bronchiolitis. Am J Respir Crit Care Med. 2003;168:633–9. doi: 10.1164/rccm.200210-1148OC. [DOI] [PubMed] [Google Scholar]
- 39.van Benten IJ, van Drunen CM, Koopman LP, et al. RSV-induced bronchiolitis but not upper respiratory tract infection is accompanied by an increased nasal IL-18 response. J Med Virol. 2003;71:290–7. doi: 10.1002/jmv.10482. [DOI] [PMC free article] [PubMed] [Google Scholar]