Abstract
We reported recently that exposure of human T cells to soluble HIV-1 envelope glycoprotein gp120 induced biologically active tumour necrosis factor (TNF)-α-related cytokine receptor of activated NF-κB ligand (RANKL), the primary drive to osteoclast differentiation and bone resorption. Furthermore, certain anti-HIV protease inhibitors linked clinically to accelerated bone loss in HIV disease blocked the physiological control of RANKL activity by interferon (IFN)-γthrough inhibition of degradation of the RANKL nuclear adapter signalling protein, TNF receptor associated protein 6 (TRAF6). We now report a series of reciprocal interactions among HIV-1, RANKL and IFN-γ. RANKL augmented HIV replication in acutely and chronically infected cells of T lymphocyte and monocyte lineage, effects which occurred at a transcriptional level in conjunction with activation of NF-κB. TNF-α and RANKL were markedly synergistic in induction of HIV. Low pharmacological levels of IFN-γ (0·75–3 ng/ml) suppressed RANKL-driven enhancement of HIV replication, as did L-T6DP-1, a cell-permeable peptide inhibitor of TRAF6. In contrast, HIV replication induced by TNF-α and phorbol ester were not inhibited, and in some cases augmented, by IFN-γ. We conclude that a positive feedback loop exists between RANKL production and HIV replication, which may be relevant to both the pathophysiology of HIV-linked osteopenia and control of HIV growth. This pathway appears distinct from those of other cytokine activators of HIV, with respect to its utilization of TRAF6 and its suppression by IFN-γ. These data raise the possibility that TRAF-specific inhibitory peptides, alone or in conjunction with IFN-γ, could be used to regulate HIV activation in vivo.
Keywords: cytokines, HIV, interferon-γ, RANKL, TNF-α, TRAF6
INTRODUCTION
Receptor of activated NF-κB ligand (RANKL) is essential to the regulation of bone resorption through facilitation of osteoclast differentiation [1]. We documented the induction of RANKL production by CD4+ T cells infected with HIV-1 or exposed to soluble recombinant HIV-1 envelope glycoprotein gp120 [2], and showed that peripheral blood mononuclear cells (PBMC) from HIV-1 seropositive patients spontaneously overproduced biologically active RANKL [3]. This was paralleled by elevated levels of RANKL in the sera of untreated and highly active antiretroviral therapy (HAART)-treated HIV+ patients [3]. These effects may underlie the high prevalence of osteopenia in HIV infected adults and children, ranging up to 89% [4,5]. In addition, we found that two anti-HIV protease inhibitors (PI) linked clinically to enhanced bone mineral resorption, ritonavir and saquinavir, blocked the physiological suppression, by interferon (IFN)-γ, of RANKL activity via tumour necrosis factor (TNF) receptor associated factor 6 (TRAF6) signalling [2]. We now sought to define potential interactions among RANKL, IFN-γ and TRAF6 in active and chronic HIV infection.
As another T cell activation product, TNF-α, has a profound effect on HIV replication in T cells and monocytes, acting via NF-κB [6], and RANKL can activate osteoclast precursors via this same transcription factor [7], we hypothesized that RANKL would up-regulate HIV replication and HIV gene transcription via NFκB enhancer elements in the HIV long-terminal repeat (LTR). We also postulated that RANKL would synergize with TNF-α in this effect, as while TNF-α regulates a variety of cellular processes via nuclear adapter proteins TRAF1 and TRAF2, RANKL signalling is dependent on TRAF6 [8,9]. Taking advantage of the fact that the unique biological function of TRAF6 is determined by its TRAF-C domain, which does not interact with motifs recognized by TRAF 1, -2, -3 or -5, we used a cell-permeable peptide prepared from the Pro-X-Glu-X-X binding motif of TRAF6 [9] in an attempt to block RANKL effects on HIV replication. Finally, the effects of low pharmacological levels of IFN-γ, capable of overcoming HIV protease inhibitor-mediated blocks to TRAF6 degradation, on induction of HIV by RANKL versus other cytokines were examined. Our data are consistent with a model by which HIV-1 facilitates osteoclastogenesis and bone loss via up-regulation of RANKL and, in a positive feedback loop, RANKL activates production of HIV-1. They also illuminate a distinct role for RANKL in up-regulation of HIV, as IFN-γ blocked RANKL-induced viral replication but did not inhibit TNF-α or phorbol myristate acetate (PMA)-mediated viral induction.
MATERIALS AND METHODS
Cell isolation and culture
CD4+ U937 human promonocytic cells and CD4+ U1 cells, representing U937 cells chronically infected with two integrated copies of pol defective HIV-1 [10], were grown in RPMI-1640 plus 10% heat-inactivated fetal bovine serum (FBS). PBMCs were derived from heparinized venous blood by density gradient centrifugation using Ficol-paque (Amersham Bioscience, Piscataway, NJ, USA) [11], and cultured in RPMI-1640 plus 10% heat-inactivated FBS. For in vitro HIV infections, PBMCs were preactivated with 1 µg/ml phytohaemagglutinin (PHA) and 32 U/ml interleukin (IL)-2 for 2–3 days prior to exposure to HIV-1. Mature CD3+ T cells were prepared from PHA-activated PBMCs by incubation with Dynabeads M-450 CD3 at a ratio of 5 beads/target cell, as per the manufacturer's directions (Dyanal, Oslo, Norway). This resulted in a CD3+ T cell population >95–99% pure, as evaluated by fluorescence-activated flow cytometry.
Reagents
Recombinant, endotoxin-free RANKL and TNF-α were purchased from Peprotech (Rocky Hill, NJ, USA). Recombinant RANK-Fc protein, recombinant osteoprotegerin (OPG), a RANK receptor decoy, and recombinant human IFN-γ were purchased from R&D Systems (Minneapolis, MN, USA).
TRAF6 decoy peptides
Two cell-permeable TRAF6 decoy peptides, L-T6DP-1 and L-T6DP-2, derived from the TRAF6 interacting motif (TRAF-C domain, residues 346–504) of RANK-1 and -2, respectively, were fused with the hydrophobic sequence from the Kaposi fibroblast growth factor signal sequence. Their synthesis and purification have been described previously [9]. L-T6DP-1 (AAVALLPAVL LALLAPRKIPTEDEYTDRPSQPST) inhibits endogenous RANK-mediated NF-κB activation upon RANKL stimulation, while L-T6DP-2 (AAVALLPAVLLALLAPIPPFQEPLEV GEND) does not [9]. This is consistent with the much lower affinity of RANK-2 for TRAF6 [9].
In vitro HIV-1 infection
HIV-1 infections were performed as described previously [11]. Briefly, 2·5 × 105 target cells (cell lines or PHA-activated PBMCs) were exposed to stock virus (500 pg of HIV-1 p24 antigen) for 2 h at 37°C, washed twice with phosphate buffered saline (PBS), and replated with fresh medium. One half of the culture supernatants was removed from each well every 3–4 days and replaced with fresh medium. At various times after viral inoculation, HIV-1 activity was determined by antigen capture ELISA (NEN) for HIV-1 p24 Gag protein in Triton-X 100 solubilized culture supernatants, as per the manufacturer's directions. The three HIV-1 isolates used, the laboratory-adapted strains IIIB and Bal, and the primary patient isolate p13, were obtained from the NIH AIDS Research and Reference Reagent Program.
Plasmid constructs, transfections and chloramphenical acetyl transferase (CAT) reporter assays
Two reporter plasmids containing sequences for bacterial CAT and the HIV-1 LTR were used, one with intact κB enhanced elements and one with mutated κB sites (p-κB-HIV-CAT) [12]. For transfections, cells were washed with serum-free RPMI-1640, and 2 × 106 cells per condition were resuspended in 1 ml of Optimum media (Invitrogen, Carlsbad, CA, USA) along with 2–6 µg plasmid DNA and DMRIE-C transfection reagent (Invitrogen). Cells were incubated at 37°C for 5 h, and fresh RPMI-1640 containing 10% FBS added. 36 h after transfection, select samples were treated with RANKL or PMA, a known inducer of the HIV-LTR. CAT assays were performed using an ELISA kit (Roche, Indianapolis, IN, USA), as per the manufacturer's directions. Assays for each transfection condition were repeated at least four times, using several different plasmid preparations.
Electrophoretic mobility shift assay (EMSA)
Nuclear extracts were prepared from PBMCs using a nuclear extract kit (Sigma). Ten µg of nuclear extract were dissolved in a buffer containing 1 ng of 32P-5′-end-labelled κB probe, 1 µg of poly(dI-dC), 50 ng of sonicated salmon sperm DNA, 10 mm MgCl2, 25 mm KCl, 1 mm DTT, 12·5 mm HEPES pH 7·8, 10% glycerol and 0·05% nonidet p-40. Mixtures were incubated for 15 min at 4°C and protein bound DNA complexes were analysed by electrophoresis on a 6% polyacrylamide gel. For competition assays, unlabelled κB oligonucleotide was added in 50-fold excess at the same time as the probe.
Statistical analysis
Differences between means of paired samples were determined using Student's t-test (Microsoft Excel). Error bars represent the mean ± standard deviation.
RESULTS
RANKL facilitates HIV replication in acute infection and induces its replication in chronically infected cells
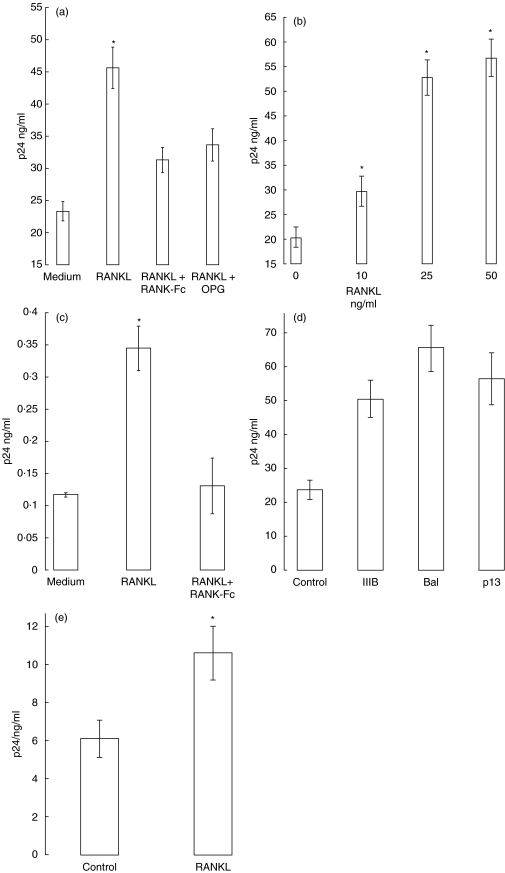
Many of the biological effects of RANKL on osteoclasts [7] and T cells [13] are mediated through post-receptor activation of NF-κB. As most cytokines and other activators of HIV replication are NF-κB-dependent [14], and NF-κB response elements are present in two copies in the HIV-1 LTR, we hypothesized that RANKL would also up-regulate HIV replication. We first investigated the effect of RANKL on HIV-1 production by actively infected PBMCs. 12 h post-inoculation of PHA-PBMC cells with HIV-1 IIIB, cells were treated with varying concentrations of recombinant, endotoxin-free RANKL and HIV p24 Gag antigen assessed in detergent-solubilized culture supernatants. RANKL exposure led to enhancement of p24 Gag production at each time-point monitored (Fig. 1a; P < 0·004 for control versus RANKL-treated cultures, day 7 post-HIV exposure). This effect was RANKL dose-dependent (Fig. 1b), and specificity for RANKL was further documented by the ability of both RANK-Fc (100 ng/ml), and the RANKL decoy protein OPG (10 ng/ml) to inhibit RANKL-mediated acceleration of HIV replication (Fig. 1a; P < 0·01 for RANKL plus RANK-Fc and P < 0·02 for RANKL versus OPG).
Fig. 1.
RANKL stimulates HIV-1 replication in acutely infected PHA-activated PBMCs and chronically infected U1 monocytic cells. (a) PHA-activated PBMCs were exposed to HIV-1 isolate IIIB for 2 h, washed, and cultured; 12 h later they were treated with RANKL (25 ng/ml). Production of HIV p24 Gag antigen in the culture supernatants was evaluated on day 7. RANKL augmented HIV replication (*P < 0·004 for controlversus RANKL-treated cultures), an effect blocked by RANK-Fc (100 ng/ml) and OPG (10 ng/ml) (P < 0·01 and < 0·02 for RANKL versus RANKL + RANK-Fc or OPG, respectively). (b) Enhancement of HIV replication in acutely infected PBMCs was dose-dependent, with HIV p24 antigen measured on day 7 post-HIV exposure. Similar results were obtained in three independent experiments (*indicates statistically significant differences at P < 0·05 between control and RANKL treated cultures). (c) U1 cells (2 × 106/ml), chronically infected with two integrated copies of HIV-1, were incubated in the presence or absence of RANKL (25 ng/ml) and the p24 Gag concentration of culture supernatants determined 48 h later. RANK-Fc (100 ng/ml) was used to block RANKL-specific effects. (*P < 0·03 for control versus RANKL treated cultures.) (d) RANKL induction of HIV replication in PHA-PBMCs acutely infected with CXCR4 (IIIB) or CCR5 (Bal and p13) co-receptor utilizing HIV-1 isolates. (e) RANKL up-regulates HIV replication in purified CD3+ primary T cells. CD3+ cells were positively selected from PHA-PBMC then infected with HIV-1 IIIB as described in (a). (*P < 0·015 for control versus RANKL cultures). Each bar depicts the mean ± s.d. of quadruplicate samples.
We then examined the effects of RANKL on a chronically HIV-infected monocytic cell, U1. When cultured at low density in the absence of mitogen it produces little basal HIV p24 antigen, but activation via protein kinase C and related pathways markedly induces HIV replication [11]. U1 cells were exposed to RANKL (25 ng/ml) for 24 h and p24 antigen production assessed in culture supernatants at 48 h. RANKL up-regulated HIV expression by threefold (Fig. 1c; P < 0·03), an effect completely blocked by RANK-Fc (Fig. 1c).
Effector cells in the enhancement of HIV replication by RANKL
RANK receptors can be induced on resting T cells by a variety of mediators, including lectins, anti-CD3 antibody, and IL-4 plus transformimg growth factor (TGF)-β [8,15]. In addition, monocytes constitutively express RANK(data not shown). It is therefore possible that RANKL can act directly on T cells in PHA-activated PBMC cultures or indirectly, via cytokines secreted by RANKL-activated monocytes. To explore these possibilities, we first examined the ability of RANKL to modulate replication of CXCR4 coreceptor utilizing, T cell tropic versus CCR5 coreceptor utilizing, T and monocyte-tropic strains of HIV. As shown in Fig. 1d, RANKL induced replication of all three viral types to equivalent levels. In addition, RANKL augmented replication of HIV-1 IIIB in PHA-activated, purified CD3+ T cells, in the absence of a detectable monocyte population (Fig. 1e).
Synergy between RANKL and IL-2 in enhancement of HIV replication
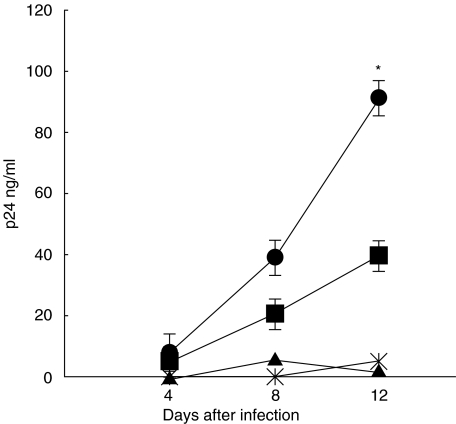
Given divergent signalling pathways for RANKL and IL-2 we explored the possibility that these cytokines would synergize in augmentation of HIV replication. HIV infects resting T cells but, due to incomplete reverse transcription and a block to proviral DNA integration, cannot replicate in such cells [16]. As shown in Fig. 2, HIV replication was not detected in unactivated PBMC exposed to RANKL (25 ng/ml) alone, consistent with the paucity of RANK receptors on these cells. In contrast, resting PBMCs exposed to IL-2 showed marked replication of HIV, and this was enhanced synergistically by RANKL (Fig. 2).
Fig. 2.
RANKL synergizes with IL-2 in the activation of HIV replication. Resting PBMCs were exposed to culture medium alone or with RANKL (25 ng/ml), IL-2 (32 U/ml), or RANKL plus IL-2 for 48 h, followed by a 2 h exposure to HIV-1 IIIB. Cells were washed and cultured for 12 days, with HIV p24 Gag antigen measured on days 4, 8 and 12 post viral exposure. Points represent means ± s.d. of triplicate measurements (*P < 0·007 for IL-2 versus IL-2 + RANKL-treated cultures) (• IL2 + RANKL; ▪ IL-2 alone; ▴ unstimulated;  RANKL alone).
RANKL alone).
RANKL activates the HIV-1 LTR via NF-κB response elements
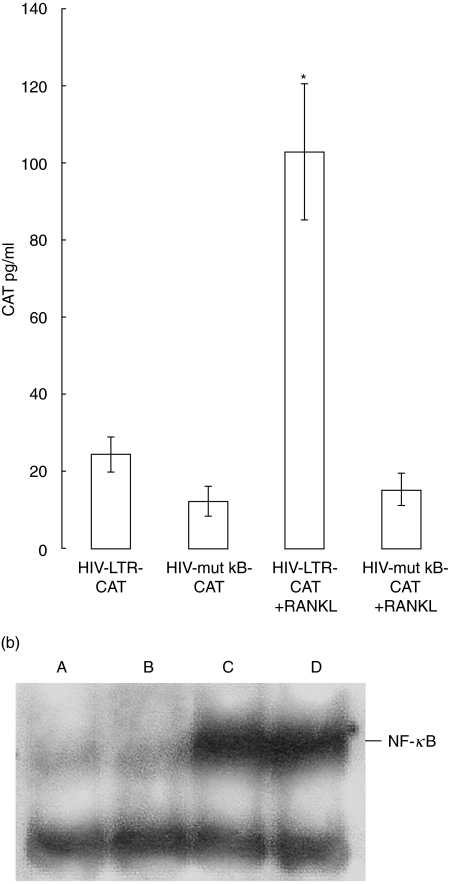
RANKL (25 ng/ml) was added to U937 monocytic cells 36 h post-transfection with LTR-CAT or p-κB plasmids, and CAT expression assessed 24 h later. As shown in Fig. 3a, RANKL increased CAT expression by a mean of 4·5-fold over control levels (P < 0·004) in conjunction with an intact HIV-LTR, but it had no effect on transcription driven by an HIV-LTR with mutated κB sites.
Fig. 3.
RANKL induces HIV-1 LTR-driven reporter gene transcription in T cells via NF-κB activation. (a) CD4+ U937 monocytic cells were transfected with HIV-1 LTR-CAT reporter plasmids containing intact or mutated κB sites, as described in Materials and methods; 36 h post-transfection, cells were untreated or exposed to RANKL (25 ng/ml). Cell extracts were prepared 24 h later and CAT assays performed by ELISA. (*P < 0·004 for control versus RANKL-treated cultures transfected with intact HIV-LTR). (b) EMSAs were carried out with nuclear extracts prepared from resting PBMCs treated with either medium (lane A), RANKL (10 ng/ml, lanes B and C), or PMA (10 ng/ml, lane D) for 30 min. A competition experiment was performed with a 50 molar excess of unlabelled κB oligonucleotide (lane B).
To examine whether RANKL could directly affect NF-κB activity, we performed EMSA experiments with nuclear extracts from resting PBMCs treated for 30 min with soluble RANKL (25 ng/ml) or PMA (10 ng/ml), a known inducer of HIV and NF-κB [10]. RANKL induced a large increase in DNA-protein complexes, equivalent to those seen in PMA-treated cells (Fig. 3b). These complexes were specific for NF-κB as they were competed out by a 50-fold excess of unlabelled κB oligonucleotide (Fig. 3b for RANKL).
Blockade of RANKL-mediated induction of HIV by TRAF6 peptides
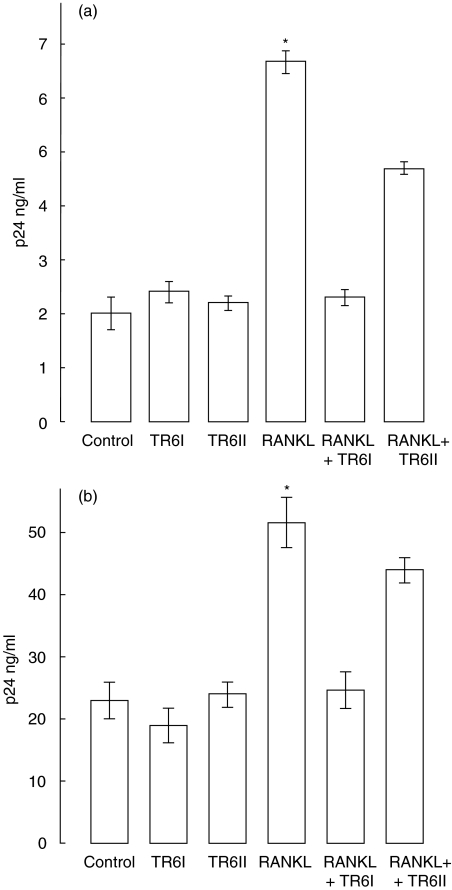
Chronically HIV infected U1 monocytic cells (2 × 106) were incubated with buffer or two peptides prepared from the TRAF-C binding domain of TRAF6, either alone or in the presence of RANKL (25 ng/ml). 48 h later HIV p24 antigen was measured in the culture supernatants. As shown in Fig. 4a, neither L-T6DP1, capable of inhibiting TRAF6 signalling and NFκB activation in murine osteoclasts [9], nor L-T6DP2, which does not have this activity, altered basal production of HIV-1 at the maximum concentration tested, 30 µm. L-T6DP2 (30 µm) similarly had no effect on HIV induced by RANKL. In contrast, L-T6DP1 (30 µm) completely abrogated RANKL-mediated virus induction. This suppressive effect of the L-T6DP1 peptide was replicated in acutely HIV infected PHA-PBMCs (Fig. 4b).
Fig. 4.
Anti-TRAF6 peptide blocks RANKL-mediated induction of HIV from chronically and acutely infected cells. Chronically HIV-1 infected U1 cells (2 × 106) (a) or PHA-activated PBMCs inoculated with HIV-1 IIIB for 2 h (b) were exposed to anti-TRAF6 peptide L-T6DP1 (30 µm) (TR6I), capable of inhibiting TRAF6 signalling and NFκB activation in murine osteoclasts, or anti-TRAF6 peptide L-T6DP2 (30 µm) (TR6II), which does not have this capacity, in the presence and absence of RANKL (25 ng/ml). HIV-1 p24 Gag antigen was assessed in U1 cultures at 48 h and in PBMC cultures at 7 days. (*P < 0·04 and *P < 0·0047 for RANKL versus RANKL + TR6I peptide in U1 and PBMC cultures, respectively.)
Synergy between RANKL and TNF-α in induction of HIV replication
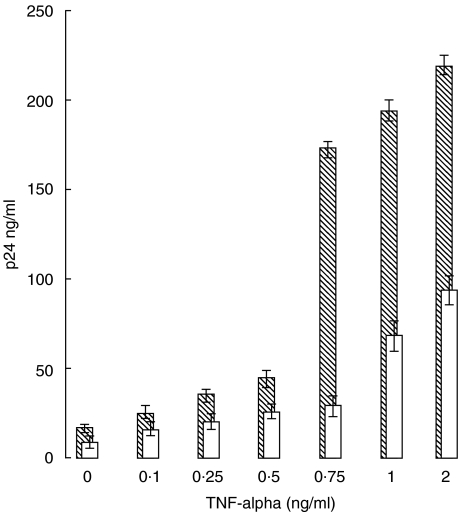
In vitro, TNF-α can synergize with RANKL in the support of osteoclast differentiation in murine [17] and human [2] systems. We sought to examine whether similar co-operation exists in terms of HIV-1 replication. HIV p24 Gag levels were determined on day 7 post-infection in culture supernatants of PBMCs exposed to a fixed concentration of RANKL, 25 ng/ml, and varying concentrations of TNF-α. An additive effect was noted up to 500 pg/ml TNF-α, while higher levels were markedly synergistic (Fig. 5).
Fig. 5.
RANKL synergizes with TNF-α in enhancement of HIV-1 replication. PHA-activated PBMCs were exposed to HIV-1 for 2 h, washed, then cultured with increasing concentrations of TNF-α alone (open bars) or in the presence of 25 ng/ml RANKL (hatched bars). After 6 d, HIV p24 Gag levels were determined. Data are expressed as the mean ± s.d. of three independent samples. Synergy was observed at >500 pg/ml of TNF-α.
IFN-γ suppresses HIV replication induced by RANKL but not by other positive regulators of HIV
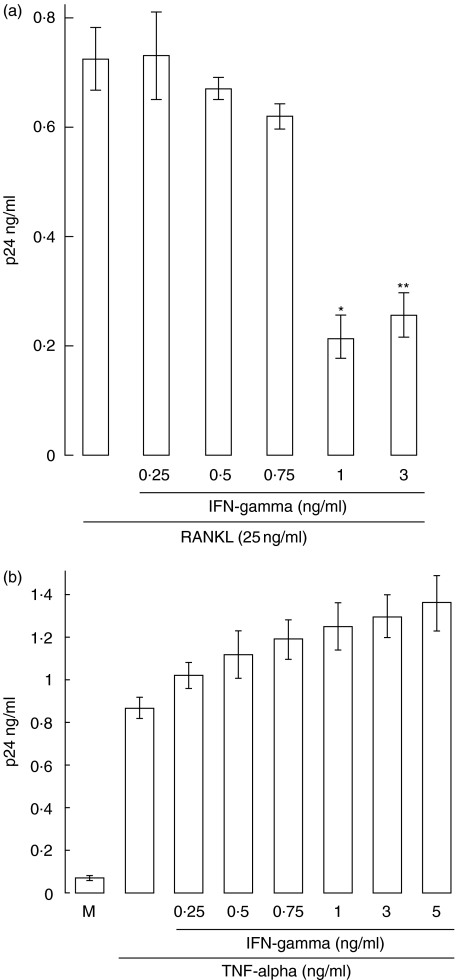
Many cytokines up-regulate HIV replication [14]. We sought to determine whether RANKL acted in an unique manner, examining its interaction with IFN-γ, the physiological regulator of RANKL signal protein TRAF6, and contrasting these effects with those for TNF-α and PMA. As shown in Fig. 6a, low pharmacological doses of IFN-γ (0·75–3 ng/ml; 1 U = 0·1 ng) blocked RANKL-mediated up-regulation of HIV in U1 monocytic cells, while it did not suppress, but rather significantly enhanced the activity of TNF-α (Fig. 6b) and PMA (data not shown). Viability of the RANKL/IFN-γ and PMA/IFN-γ cultures was nearly 100%, as assessed by Trypan blue dye exclusion. In contrast, viability of the TNF-α/IFN-γ cultures was <20% at 48 h, consistent with the findings of others utilizing this U1 system [18].
Fig. 6.
Effects of IFN-γ on induction of HIV from chronically infected monocytic cells mediated by RANKL versus TNF-α. U1 cells (2 × 106) were exposed to RANKL (25 ng/ml) (a) or TNF-α (500 ng/ml) (b), either alone or in the presence of varying concentrations of IFN-γ. HIV-1 p24 Gag antigen production was measured in culture supernatants 48 h later (a: *P < 0·01; **P < 0·04; b: P < 0·01 for all concentrations tested).
DISCUSSION
Cytokines produced by the host in response to an ongoing HIV infection play several critical roles in virus–host interactions, in terms of regulation of viral replication and a variety of metabolic complications of HIV and its therapy. We examined interactions between RANKL, implicated in bone loss characteristic of both HIV disease and its treatment [2–5], and TNF-α, a related T cell activation cytokine up-regulated in HIV infection in the presence and absence of HAART [19], in terms of HIV replication and its control by IFN-γ. We uncovered a positive feedback between RANKL production and HIV replication in T cells and monocytes, which was enhanced synergistically by TNF-α, and suppressed by IFN-γ. These effects have implications for certain metabolic abnormalities linked to HIV as well as HIV immune pathogenesis.
In terms of metabolic complications, we had demonstrated previously that HIV-1 infection of CD4+ T cells, or their exposure to soluble HIV-1 envelope gp120, up-regulates production of biologically active RANKL [2]. We also showed that certain HAART regimens enhance RANKL activity in the osteoclast via another cytokine dysregulated in HIV disease [2]. Specifically, IFN-γ provides a physiological block to RANKL signalling through its enhancement of TRAF6 degradation by proteasomes [20], but IFN-γ production is markedly down-regulated in advanced HIV infection [21]. This should limit its capacity to suppress RANKL, resulting in sustained osteoclast activation and bone resorption. In addition, two anti-HIV protease inhibitors, ritonavir and saquinavir, associated with accelerated bone loss in uncontrolled clinical studies [22], blocked IFN-γ-mediated degradation of TRAF6, further increasing the biologic activity of RANKL, at least in vitro [2]. This was not seen with two other anti-HIV protease inhibitors, indinavir and nelfinavir [2], which are not associated clinically with bone loss [22].
Focusing on HIV replication, our observation of HIV-LTR activation by RANKL via NF-κB response elements suggests that, like many cytokines, RANKL augments HIV-1 replication via HIV-LTR-driven gene transcription. RANKL itself is up-regulated by proinflammatory cytokines induced by HIV infection, including IL-1, IL-6, IL-7 and TNF-α [15]. However, an unique pathway for RANKL in induction of HIV is suggested by the fact that this effect was suppressed by IFN-γ, while IFN-γ enhanced HIV induction by PMA and TNF-α.
These latter results are consistent with the work of others showing that TNF-α [18] and PMA [23] synegize with IFN-γ in chronically infected cells to promote HIV replication. They also parallel work with other proinflammatory cytokines and chemokines, including IL-8, showing synergy with IFN-γ in up-regulation of HIV [24], rather than the suppressive effects we observe with RANKL. This differential response may relate to differences in signal adapter mechanisms used by the various activators of HIV. For example, degradation of the RANKL signalling adapter TRAF6 is facilitated in the proteasome by IFN-γ [20], while IFN-γ has no effect on the nuclear signal adapters for TNF-α, TRAF1 and TRAF2, nor on protein kinase C, utilized by PMA.
Our work has additional implications for the pathophysiology and potential treatment of HIV infection. RANKL is an important survival factor for dendritic cells (DC) [25] and cross-presentation by DC of HIV antigens, captured from viable or apoptotic HIV-infected cells, to uninfected T cells in the induction of cytolytic T cells and maintenance of anti-HIV memory responses is extremely efficient [26]. HIV could exploit this process as the same cytokine serving to promote DC growth, RANKL, also up-regulates HIV replication. Alternatively, the DC-HIV presentation pathway might be subverted to help eradicate latently infected reservoirs, normally unrecognized by the immune system [26], if HIV expression were to be transiently induced in vivo, utilizing exogenous RANKL or RANKL plus IL-2, followed by intensified HAART. Our results also suggest that attempts to regulate HIV replication or induction from a chronic/latent state must consider a variety of signalling pathways, including those related to IFN-γ, for this key lymphokine is not only decreased in advanced HIV disease, but it interacts very differently with divergent HIV activators.
Acknowledgments
This work was supported by NIH grant HL55646 (to J. L.) and a Scholar Award (RF02652-27) from the American Foundation for AIDS Research (amfAR) (to J. M. F.). We thank Hao Wu PhD, Department of Biochemistry, Weill Medical College of Cornell University, New York and Bryant G. Darnay PhD, Department of Bioimmunotherapy, The University of Texas M. D. Anderson Cancer Center, Texas for providing the TRAF6 decoy peptides.
REFERENCES
- 1.Kong YY, Feige U, Sarosi I, et al. Activated T cells regulate bone loss and joint destruction adjuvant arthritis through osteoprotegerin ligand. Nature. 1999;402:304–9. doi: 10.1038/46303. [DOI] [PubMed] [Google Scholar]
- 2.Fakruddin JM, Laurence J. HIV envelope gp120-mediated regulation of osteoclastogenesis via receptor activator of nuclear factor κB ligand (RANKL) secretion and its modulation by HIV protease inhibitors through interferon-γ/RANKL cross-talk. J Biol Chem. 2003;278:48251–8. doi: 10.1074/jbc.M304676200. [DOI] [PubMed] [Google Scholar]
- 3.Fakruddin JM, Laurence J. Regulation of RANKL expression in HIV infection and its therapy: relationship to HIV-linked osteopenia; XV International AIDS Conference, Bankok, Thailand. 2004. [Abstract WePpB2063]. [Google Scholar]
- 4.Knobel H, Guelar A, Vallecillo G, Nogues A. Osteopenia in HIV-infected patients: is it the disease or its treatment? AIDS. 2001;15:807–8. doi: 10.1097/00002030-200104130-00022. [DOI] [PubMed] [Google Scholar]
- 5.Thomas J, Doherty SM. HIV infection − a risk factor for osteoporosis. J Acquir Immune Defic Syndr. 2003;33:281–91. doi: 10.1097/00126334-200307010-00001. [DOI] [PubMed] [Google Scholar]
- 6.Yang X, Chen Y, Gabuzda D. ERK MAP kinase links cytokine signals to activation of latent HIV-1 infection by stimulating a cooperative interaction of AP-1 and NF-kappaB. J Biol Chem. 1999;274:27981–8. doi: 10.1074/jbc.274.39.27981. [DOI] [PubMed] [Google Scholar]
- 7.Lee ZH, Kim H-H. Signal transduction by receptor activator of nuclear factor kappaB in osteoclasts. Biochem Biophys Res Commun. 2003;305:211–4. doi: 10.1016/s0006-291x(03)00695-8. [DOI] [PubMed] [Google Scholar]
- 8.Theill LE, Boyle WJ, Penninger JM. RANK-L and RANK: T cells, bone loss, and mammalian evolution. Annu Rev Immunol. 2002;20:795–823. doi: 10.1146/annurev.immunol.20.100301.064753. [DOI] [PubMed] [Google Scholar]
- 9.Yu H, Arron JR, Lamothe B, et al. Distinct molecular mechanism for initiating TRAF6 signalling. Nature. 2002;418:443–7. doi: 10.1038/nature00888. [DOI] [PubMed] [Google Scholar]
- 10.Laurence J, Sikder SK, Jhivari S, Salmon JE. Phorbol ester-mediated induction of HIV-1 from a chronically infected promonocyte clone: blockade by protein kinase inhibitors and relationship to tat-associated trans-activation. Biochem Biophys Res Commun. 1990;166:349–57. doi: 10.1016/0006-291x(90)91952-o. [DOI] [PubMed] [Google Scholar]
- 11.Crombie R, Silverstein RL, MacLow C, Pearce SFA, Nachman RL, Laurence J. Identification of a CD36-related thrombospondin 1-binding domain in HIV-1 envelope glycoprotein gp120: relationship to HIV-1-specific inhibitory factors in saliva. J Exp Med. 1998;187:25–35. doi: 10.1084/jem.187.1.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Nabel G, Baltimore D. An inducible transcription factor activates expression of human immunodeficiency virus in T cells. Nature. 1987;326:711–3. doi: 10.1038/326711a0. [DOI] [PubMed] [Google Scholar]
- 13.Wong BR, Besser D, Kim N, et al. TRANCE, a TNF family member, activates Akt/PKB through a signaling complex involving TRAF6 and c-Src. Mol Cell Biol. 1999;4:1041–9. doi: 10.1016/s1097-2765(00)80232-4. [DOI] [PubMed] [Google Scholar]
- 14.Osborn L, Kunkel S, Nabel GJ. Tumor necrosis factor alpha and interleukin 1 stimulate the human immunodeficiency virus enhancer by activation of the nuclear factor kappa B. Proc Natl Acad Sci USA. 1989;86:2336–41. doi: 10.1073/pnas.86.7.2336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Josien R, Wong BR, Li HL, Steinman RM, Choi Y. TRANCE, a TNF family member, is differentially expressed on T cell subsets and induces cytokine production in dendritic cells. J Immunol. 1999;162:2562–8. [PubMed] [Google Scholar]
- 16.Muthumani K, Wadsworth SA, Dayes NS, et al. Suppression of HIV-1 viral replication and cellular pathogenesis by a novel p38/JNK kinase inhibitor. AIDS. 2004;18:739–48. doi: 10.1097/00002030-200403260-00004. [DOI] [PubMed] [Google Scholar]
- 17.Lam J, Takeshita S, Barker JE, Kamagawa O, Ross FP, Teitelbaum SL. TNF-α induces osteoclastogenesis by direct stimulation of macrophages exposed to permissive levels of RANK ligand. J Clin Invest. 2000;106:1481–8. doi: 10.1172/JCI11176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Han X, Becker K, Degen HJ, Jablonowski H, Strohmeyer G. Synergistic stimulatory effects of tumor necrosis factor α and interferon γ on replication of human immunodeficiency virus type 1 and on apoptosis of HIV-1-infected host cells. Eur J Clin Invest. 1996;26:286–92. doi: 10.1046/j.1365-2362.1996.116271.x. [DOI] [PubMed] [Google Scholar]
- 19.Aukrust P, Muller F, Lien E, et al. Tumor necrosis factor (TNF) system levels in human immunodeficiency virus-infected patients during highly active antiretroviral therapy: persistent TNF activation is associated with virologic and immunologic treatment failure. J Infect Dis. 1999;169:74–82. doi: 10.1086/314572. [DOI] [PubMed] [Google Scholar]
- 20.Takayanagi H, Ogasawara K, Hida S, et al. T-cell-mediated regulation of osteoclastogenesis by signalling cross-talk between RANKL and IFN-γ. Nature. 2000;408:600–5. doi: 10.1038/35046102. [DOI] [PubMed] [Google Scholar]
- 21.Maggi E, Macchia D, Parronchi O, et al. Reduced production of interleukin 2 and interferon-gamma and enhanced helper activity for IgG synthesis by cloned CD4+ T cells from patients with AIDS. Eur J Immunol. 1987;17:1685–90. doi: 10.1002/eji.1830171202. [DOI] [PubMed] [Google Scholar]
- 22.Nolan D, Upton R, McKinnon E, et al. Stable or increasing bone mineral density in HIV-infected patients treated with nelfinavir or indinavir. AIDS. 2001;15:1275–80. doi: 10.1097/00002030-200107060-00009. [DOI] [PubMed] [Google Scholar]
- 23.Biswas P, Poli G, Kinter AL, et al. Interferon γ induces the expression of human immunodeficiency virus in persistently infected promonocytic cells (U1) and redirects the production of virions to intracytoplasmic vaculoes in phorbol myristate acetate-differentiated U1 cells. J Exp Med. 1992;176:739–50. doi: 10.1084/jem.176.3.739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tiemessen CT, Kilroe B, Martin DJ. Interleukin-8 fails to induce human immunodeficiency virus-1 expression in chronically infected promonocytic U1 cells but differently modulates induction by proinflammatory cytokines. Immunology. 2000;101:140–6. doi: 10.1046/j.1365-2567.2000.00100.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kong YY, Boyle WJ, Penninger JM. Osteoprotegerin ligand: a common link between osteoclastogenesis, lymph node formation and lymphocyte development. Immunol Cell Biol. 1999;77:188–93. doi: 10.1046/j.1440-1711.1999.00815.x. [DOI] [PubMed] [Google Scholar]
- 26.Msaranon C, Desoutter J-F, Hoeffel G, Cohen W, Hanau D, Hosmalin A. Dendritic cells cross-present HIV antigens from live as well as apoptotic infected CD4+ T lymphocytes. Proc Natl Acad Sci USA. 2004;101:6092–7. doi: 10.1073/pnas.0304860101. [DOI] [PMC free article] [PubMed] [Google Scholar]