Abstract
Systemic lupus erythematosus (SLE) is a chronic, systemic autoimmune disease characterized by the loss of tolerance to self-antigen. Because it is currently not known if regulatory T (Treg) cells are involved in the pathogenesis, we determined the frequency of CD4+CD25+ T cells and assayed the related gene expression levels in CD4+CD25+ T cells isolated from both lupus mice (NZB/NZW F1) and normal control mice (DBA2/NZW F1). The results showed that the frequency of CD4+CD25+ T cells in lupus mice was lower than that of normal mice. Except for the high expression level of interleukin (IL)-10 mRNA, CD4+CD25+ T cells from lupus mice expressed normal forkhead box P3 (Foxp3) and transforming growth factor (TGF)-β mRNA, and exerted suppressive functions. Furthermore, we depleted CD25+ Treg cells of non-autoimmune mice with anti-CD25 antibody and broke their tolerance with apoptotic cell-pulsed dendritic cells for the follow-up of autoantibody levels. The mice in the CD25+ cell-depleted group had higher titres of anti-double-strand/single-strand DNA antibodies than those of the isotype control antibody-treated group. These findings indicated that CD4+CD25+ T cells might be involved in the regulatory mechanism of autoantibody production.
Keywords: autoantibodies, lupus/systemic lupus erythematosus, regulatory T cells
Introduction
The primary mechanism that sustains self-tolerance is thymic deletion of autoreactive T cells, but this process might be incomplete. During development, self-reactive T cells that escape thymic deletion and exit into the peripheral circulation are controlled by mechanisms responsible for peripheral tolerance. These mechanisms include T cell anergy, ignorance, phenotypic skewing and apoptosis [1]. Recent studies have suggested that CD4+CD25+ regulatory T (Treg) cells exhibit immune suppressive activity and also play a critical role in the maintenance of self-tolerance [2–5]. Nude mice transferred with CD4+ T cells depleted of CD25+ cells developed various organ-specific autoimmune diseases. Reconstitution of CD4+CD25+ cells within a limited period after transfer of CD4+CD25− cells prevented the onset of these diseases [6]. The forkhead box P3 (Foxp3) gene is shown to be central in the development and function of CD25+ Treg cells [7–9]. Although there is a marked contrast with regard to the importance of immunosuppressive cytokines in vivo compared to CD25+ Treg cell suppression in vitro, several cytokines have been implicated as mediators of inhibition, such as interleukin (IL)-10 and transforming growth factor (TGF)-β [10].
Systemic lupus erythematosus (SLE) is a chronic, systemic autoimmune disease characterized by the loss of tolerance to self-antigen and the production of autoantibodies against components of the cell nucleus [11]. Many factors are involved in the pathogenesis of SLE, including genetic factors, environmental factors, hormone, hyperactivated B cells and T cells, abnormal phagocytic functions and abnormal immunoregulation [12–14]. Female NZB × NZW F1 (BWF1) mice develop lupus resembling human SLE spontaneously, with polyclonal B cell activation leading to the production of autoantibodies and immune complex-mediated glomerulonephritis. To examine if CD4+CD25+ Treg cells are also involved in the pathogenesis of SLE, recent studies have revealed a deficiency of Treg cell numbers in two murine models, BWF1 and SNF1 [15] and in human patients [16–18].
Moreover, to determine if there are any defects in CD4+CD25+ T cells of BWF1 mice, we compared their frequencies and functions with CD4+CD25+ T cells of DBA-2 × NZW F1 (D2WF1) mice. We attempted to investigate whether a deficiency of CD4+CD25+ T cell numbers could cause lupus-like diseases in normal mice. In our previous studies, we have successfully broken the self-tolerance of normal mice with apoptotic cells. D2WF1 mice produced high titres of anti-DNA antibodies after immunization with apoptotic cell-pulsed dendritic cells (DCs) and then received a boost of apoptotic cells [19]. The approach of this study was the depletion of CD25+ T cells before breaking the tolerance of normal mice. We found that mice lacking CD4+CD25+ T cells had more severe autoimmune responses than mice with normal Treg cell numbers.
Materials and methods
Mice
Female NZB × NZW F1 (BWF1) mice were purchased from Jackson Laboratories (Bar Harbor, ME, USA). Female DBA-2 × NZW F1 (D2WF1) mice of non-autoimmune strain were obtained from and maintained by the Animal Centre of the College of Medicine of the National Taiwan University in a pathogen-free facility. At 6–8 weeks of age, female D2WF1 mice were used as the source of bone marrow-derived dendritic cells (BMDCs). Mice of both strains have identical major histocompatibility complex class II (H-2d/u). The experimental protocol was approved by the Animal Committee of the College of Medicine of the National Taiwan University.
Isolation of cells
The CD4+CD25+ and CD4+CD25− T cells were purified with auto-magnetic affinity cell sorting (MACS) using the CD4+CD25+ regulatory T cell isolation kit (Miltenyi Biotec, Sunnyvale, CA, USA). First, CD4+ T cells were enriched from splenocytes stained with CD4+CD25+ regulatory T cell biotin–antibody cocktail and anti-biotin microbeads. CD4+CD25+ and CD4+CD25− T cell magnetic separations were performed by staining with CD25-phycoerythrin (PE), followed by staining with anti-PE microbeads. Antigen-presenting cells (APC) were selected negatively from splenocytes by depleting Thy-1·2+ T cells. DCs were positively selected with anti-CD11c-coated magnetic microbeads (Miltenyi Biotec).
Flow cytometry
After the red blood cells (RBC) were depleted using ACK lysis buffer (0·15M NH4Cl, 10mM KHCO3, 0·1mM Na2 EDTA, pH7·2), splenocytes or peripheral blood were incubated with fluorescein isothiocyanate (FITC)-conjugated anti-CD3 monoclonal antibody (MoAb) (145–2C11; eBioscience, San Diego, CA, USA), PE-conjugated anti-CD25 MoAb (PC61; eBioscience), cychrome-conjugated anti-CD4 Moab (L3T4; eBioscience) or relevant isotype controls (eBR2a; eBioscience) for 30 min at 4°C. The cells were then washed with fluorescence activated cell sorter (FACScan) buffer and analysed on FACScan (BD Biosciences) using CellQuest software.
Analysis of gene expression
Total cellular mRNA was extracted from 1 × 106 isolated cells using GenesStrip™ hybridization tubes (RNATure, West Irvine, CA, USA) and reverse transcribed into cDNA by using SuperScript™ II reverse transcriptase (Invitrogen, Carlsbad, CA, USA) and random hexamer primer (RNATure). Gene expression was quantified by real-time polymerase chain reaction (PCR) using Assays-on-Demand™ gene expression products [forkhead box P3 (Foxp3), IL-10, TGF-β, IL-4 and hypoxanthine phosphoribosyltransferase (HPRT)], Assays-on-Design™ Gene Expression Products (IFN-γ) and the ABI/PRISM 7700 sequence detection system (Applied Biosystems Inc., Foster City, CA, USA). Samples were run in triplicate. The amount of gene expression was calculated from the standard curve (a serial dilution of D2WF1 CD4+CD25+ cDNA) and the relative expression of the target gene was determined by dividing the target gene value by the HPRT value.
Suppression assay
Isolated CD4+CD25− T cells (1 × 105 cells/well) were stimulated in triplicate with autologous T-depleted, irradiated splenocytes (1 × 105 cells/well) in the presence of 1 µg/ml anti-CD3 MoAb (145–2C11; eBioscience). Isolated CD4+CD25+ T cells were used as Treg cells and added to the wells at a different ratio (CD4+CD25− T cells: CD4+CD25+ T cells = 1 : 0·25, 1 : 0·5, 1 : 1). Cells were co-cultured in a final volume of 200 µl of RPMI-1640 supplemented with 10% fetal calf serum (FCS) in 96-well round-bottomed plates for 3 days. The wells were pulsed with 1 µCi of [3H]-thymidine 18 h before harvesting and the cells were collected onto glass fibre filters using an automated multi-sample harvester, and the amount of incorporated [3H]-thymidine was then measured with a dry scintillation counter (Packard, Meridan, CT, USA).
Generation of dendritic cells and apoptotic cells
BMDCs were prepared as described previously [20]. In brief, erythrocytes-depleted bone marrow cells were cultured in a medium supplemented with recombinant murine granulocyte–macrophage colony-stimulating factor (GM-CSF) (750 U/ml) and IL-4 (1000 U/ml) (Pepro Tech Inc. Rocky Hill, NJ, USA). Apoptotic cells were generated by treating single-cell suspensions of thymocytes with 1·2 × 10−6 M dexamethasone for 15 h. On day 4 of culture, apoptotic cells were added to BMDC cultures at the ratio of 10 : 1. After 48 h, cells were collected and DCs were enriched by anti-CD11c-coated magnetic microbeads.
Immunization of mice
D2WF1 mice were divided into three groups and given an intravenous injection of phosphate-buffered saline (PBS), isotype control antibody (250 µg/mouse) (A110-1, PharMingen, San Diego, CA, USA) or anti-CD25 antibody (250 µg/mouse) (PC61, PharMingen) on day 0. The mice were then immunized with PBS or dendritic cells pulsed with apoptotic cells (1·5 × 105 DCs/mouse) on day 4 and boosted with PBS or apoptotic cells (1 × 107 cells/mouse) on day 9. All the mice were bled at indicated time-points. Sera were collected for autoantibody detection.
Enzyme-linked immunosorbent assay
Antibodies to double-strand (ds) DNA and single-strand (ss) DNA in the serum samples were measured by enzyme-linked immunosorbent assay (ELISA). The level of anti-DNA IgG was presented as ELISA units (EU/ml) compared with the MoAb 10F10, which is specific for either dsDNA or ssDNA. The absorbance value generated by 74 ng/ml of 10F10 antibody was defined as 1 EU/ml.
Statistical analysis
The significant differences between the experimental and control groups were analysed with the Mann–Whitney U-test by using GraphPad Prism version 4·00 (GraphPad Software, San Diego, CA, USA). A P-value of < 0·05 was considered to be statistically significant.
Results
Percentage of CD4+CD25+ T cells in BWF1 and D2WF1 mice
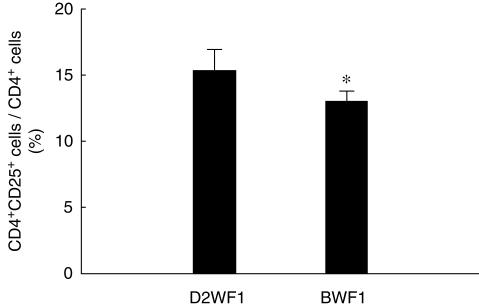
In order to examine whether lupus mice have a deficiency in CD4+CD25+ T cells, we determined the percentage of these cells in the splenocytes of lupus and normal mice. Because D2WF1 mice have identical major histocompatibility complex class II (H-2d/u) with BWF1, we used D2WF1 mice as the normal control mice. The percentage of CD4+CD25+ T cells in splenic CD4+ T cells of BWF1 mice was significantly lower than that of D2WF1 mice, and the mean percentages of BWF1 mice and D2WF1 mice were 13·0% and 15·2%, respectively (Fig. 1). We also measured the levels of peripheral CD4+CD25+ T cells in autoimmune and normal mice. However, the frequency of peripheral CD4+CD25+ T cells of BWF1 mice was similar to that of D2WF1 mice (data not show).
Fig 1.
The relative percentage of splenic CD4+CD25+ T cell in DBA-2 × NZW F1 (D2WF1) mice and NZB × NZW F1 (BWF1) mice. The CD3+ T cells were gated and the percentage of CD25+ cells in CD4+ T cells was shown above. Bars show mean ± s.d. *P < 0·05 (D2WF1, n = 4; BWF1, n = 5).
Gene expression profile of CD4+CD25+ T cells in D2WF1 and BWF1 mice
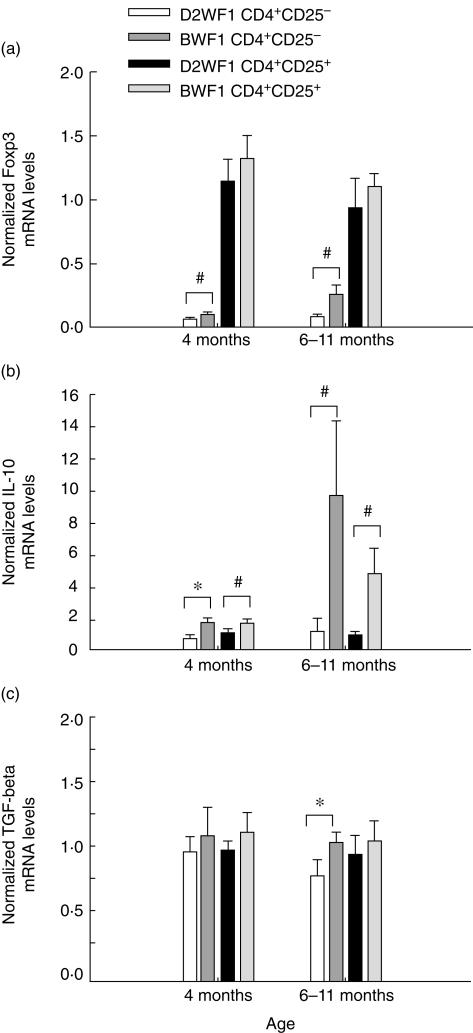
We further analysed the expression of some specific genes which are relative to the development and the function of Treg cells, such as Foxp3, IL-10 and TGF-β. Cells were isolated from splenocytes of both mice at different ages. The purity of CD4+CD25+ T cells was greater than 90%, and that of CD4+CD25− T cells was greater than 95% (data not shown). The mRNA expression of freshly isolated cells was quantified by real-time PCR. We found that the mRNA expression of Foxp3 was predominant in CD4+CD25+ T cells. Foxp3 expression in BWF1 CD4+CD25+ T cells was similar to that in D2WF1 CD4+CD25+ T cells. The expression level of Foxp3 in CD4+CD25− T cells was used as a negative control. However, we found that Foxp3 expression in BWF1 CD4+CD25− T cells was higher than that in D2WF1 CD4+CD25− T cells (Fig. 2a).
Fig 2.
Gene expression profiles of CD4+CD25+ cells and CD4+CD25− cells isolated from DBA-2 × NZW F1 (D2WF1) mice or NZB × NZW F1 (BWF1) mice at different ages. The relative mRNA expression of forkhead box P3 (Foxp3) (a), interleukin (IL)-10 (b) and transforming growth factor (TGF)-β (c) was determined by normalizing the expression of each target gene to hypoxanthine phosphoribosyltransferase (HPRT). Bars show mean ± s.d. *P < 0·05, ♯P < 0·01 (n = 5).
Moreover, TGF-β expression in BWF1 CD4+CD25+ T cells was also normal (Fig. 2c). Both BWF1 CD4+CD25+ and CD4+CD25− T cells expressed higher levels of IL-10 than D2WF1 CD4+CD25+ and CD4+CD25− T cells, and this phenomenon was more significant in old BWF1 mice at 6–11 months of age (Fig. 2b).
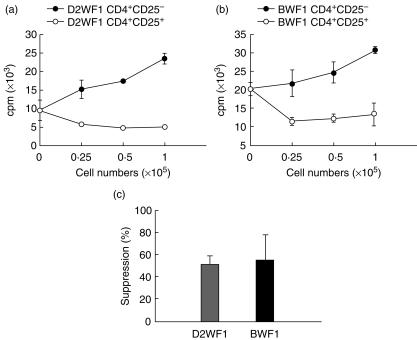
The suppressive function of CD4+CD25+ T cells from D2WF1 and BWF1 mice
To determine the suppressive function of CD4+CD25+ T cells isolated from both mice, different numbers of D2WF1 CD4+CD25+ or CD4+CD25− T cells were added to the culture of CD4+CD25− T cells to suppress their proliferation. It was found that CD4+CD25+ T cells from both D2WF1 (Fig. 3a) and BWF1 mice (Fig. 3b) inhibited the proliferation of autologous CD4+CD25− T cells at a ratio of 0·25 : 1, but CD4+CD25− T cells did not. The 1 : 1 ratio of CD25+ cells to CD25− cells was then used to compare the activities of D2WF1 CD4+CD25+ T cells with BWF1 CD4+CD25+ T cells. Data showed that CD4+CD25+ T cells have similar suppressive capability and the mean inhibition of D2WF1 and BWF1 mice were 50·8% and 54·8%, respectively (Fig. 3c).
Fig 3.
The suppressive function of CD4+CD25+ cells isolated from DBA-2 × NZW F1 (D2WF1) mice and NZB × NZW F1 (BWF1) mice. Different numbers of CD4+CD25+ T cells or CD4+CD25− cells from D2WF1 mice (a) or BWF1 mice (b) were added to the culture of CD4+CD25− T cells stimulated with autologous T-depleted splenocytes and anti-CD3 monoclonal antibody. (c) CD4+CD25+ T cells from D2WF1F1 mice and BWF1 mice suppressed the proliferation of autologous CD4+CD25− cells at the ratio of 1 : 1. Bars show mean ± s.d. (n = 3).
CD4+CD25+ T cells play a role in controlling autoantibody production
The data above indicate that CD4+CD25+ T cells in BWF1 mice had a deficiency in cell numbers, but had a normal expression level of Foxp3 and normal suppressive functions. To investigate further if the deficiency of Treg cell numbers in BWF1 mice causes the development of lupus-like disease, we depleted CD25+ cells in normal mice and then broke down their tolerance. The depleted efficiency in mouse peripheral blood was followed after mice received an intravenous injection of anti-CD25 MoAb. Data showed that the percentage of CD4+CD25+ T cells in peripheral CD4+ T cells decreased from 4% to less than 1% and was maintained for more than 20 days (Fig. 4).
Fig 4.
Depletion of CD4+CD25+ cells in DBA-2 × NZW F1 (D2WF1) mice by administration of anti-CD25 monoclonal antibody (MoAb). D2WF1 mice received an intravenously injection of phosphate-buffered saline or anti-CD25 antibody (250 µg/mouse) on day 0. All mice were bled before immunization and at each time-point. The CD3+ T cells were gated and the relative percentage of CD4+CD25+ T cell in CD4+ T cells was determined. Values are mean ± s.d. (n = 4).
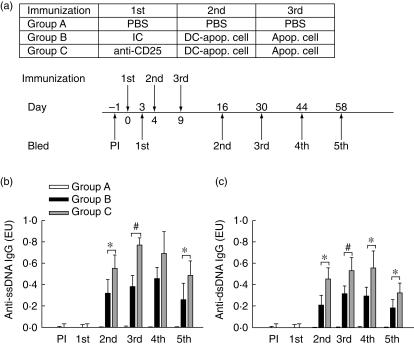
Our previous experiments showed that after adoptive transfer of apoptotic cell-pulsed DCs into D2WF1 mice, it initiated an autoimmune response in normal mice, such as the production of anti-dsDNA antibody and anti-ssDNA antibody [19]. The effect of depletion of CD25+ cells 4 days before D2WF1 mice were immunized with PBS or apoptotic cell-pulsed DCs was studied. Mice further received a boost of PBS or apoptotic cells to enhance their immune response on day 9 and were bled at indicated time-points, as shown in Fig. 5a. The ELISA data of mouse serum showed that mice treated with apoptotic cell-pulsed DCs and a boost of apoptotic cells produced anti-ssDNA and anti-dsDNA antibodies (Fig. 5b,c). In addition, mice in the CD25+ cell-depleted group developed a more severe autoimmune response than mice in the isotype control antibody-treated groups.
Fig 5.
IgG autoantibodies levels in the serum of DBA-2 × NZW F1 (D2WF1) mice. (a) Schematic depiction of immunization protocol as described in Materials and methods. D2WF1 mice were divided into three groups and received an intravenously injection of phosphate-buffered saline (PBS) (group A), 250 µg isotype control antibody (group B) or 250 µg anti-CD25 antibody (group C) on day 0. Then, the mice were immunized with PBS (group A) or 1·5 × 105 dendritic cells pulsed with apoptotic cells (groups B, C) on day 4 and boosted with PBS (group A) or 1 × 107 apoptotic cells (groups B, C) on day 9. All mice were bled 1 day (PI) before and 3 (1st), 16 (2nd), 30 (3rd), 44 (4th) and 58 (5th) days after first injection. Serums were collected for anti-ssDNA antibody (b) and anti-dsDNA antibody (c) detection. Bars show the mean ± s.d. *P < 0·05, ♯P < 0·01 (n = 5 for each group).
Discussion
SLE is a prototype of systemic autoimmune disease. Multiple defects in the immune system of SLE patients have been described, including B and T cell hyperactivity and aberrant cytokine production. Some studies concerning the role of CD4+CD25+ T cells in lupus reveal decreased frequencies of these cells in SLE patients [16–18] and lupus mice [15]. However, it remains unclear if the suppressive function of Treg cells in SLE is impaired. From our findings, splenic Treg cells in lupus mice show deceased frequency, but Treg cells of lupus mice have no significant defect in their suppressive activity. Similar results have been noted in lupus-prone MRL/Mp mice. Their CD4+CD25+ T cells have normal frequencies and display only subtle abnormalities of function [21].
Foxp3 is a critical molecule for the development and function of CD4+CD25+ T cells [7–9]. At both mRNA and protein levels, Foxp3 expression in peripheral CD4+ T cells was predominant in the CD4+CD25+ subset. Scurfy mice with spontaneous Foxp3 mutation have a fatal lymphoproliferative disease associated with multi-organ infiltration and early death at 3–4 weeks of age in hemizygous (sf/Y) males [22]. Moreover, mutations in human FOXP3 have also been identified in patients with a severe fatal autoimmune/allergic syndrome, known as IPEX (immune dysregulation, polyendocrinopathy, enteropathy, X–linked syndrome) [23–25].
An indispensable study for Foxp3 in Treg cell development showed directly that CD4+CD25+ T cells in the periphery of scurfy or Foxp3-deficient mice were neither anergic nor suppressive in vitro [8]. In BWF1 mice, Foxp3 expression seemed normal in CD4+CD25+ T cells and these cells had normal suppressive function. We therefore excluded the possibility of Foxp3 deficiency in BWF1 mice.
The CD4+CD25+ T cells have diverse T cell receptor (TCR) repertoires [26,27], suggesting that they are capable of responding to a wide spectrum of antigens. Some studies of human CD4+CD25+ T cells have shown that they suppress proliferation and cytokine production to both self- and foreign antigens. In our study, although the suppressive function of lupus CD4+CD25+ T cells to TCR stimulation (anti-CD3) is normal, we do not know whether they fail to suppress the response to self-antigen. Recent findings have indicated that CD25+ T cells from New Zealand mixed 2328 mice had defects in suppressing lupus glomerulonephritis and sialoadenitis [28]. Defective antigen-specific Treg cells may thus contribute to the pathogenesis of lupus. Therefore, the antigen-specific suppression of lupus Treg cells merits being investigated further.
IL-10 is an important immunoregulatory cytokine, mainly by inhibiting the production of proinflammatory cytokines and by regulating the differentiation and proliferation of several immune cells, such as T cells, B cells and APC [29]. In some experimental systems, the effect of Treg cells is also mediated by IL-10 [30]. However, high amounts of IL-10 have been found in SLE patients and most of the IL-10 have been secreted by monocytes and B lymphocytes, with a small contribution from T lymphocytes [31].
In our study, both CD4+CD25+ T and CD4+CD25− T cells isolated from BWF1 mice with severe lupus disease produced higher levels of IL-10 mRNA than those of normal mice. It is still unknown whether this represents the CD4+CD25+ T cells exerting their suppressive function through IL-10, or if lupus mice have an unusual genetic transcription of IL-10.
The major source of autoantigens in SLE might be the apoptotic cells. Our previous study indicated that DCs treated with apoptotic cells could initiate the response of autoreactive T cells and the production of autoantibodies [19]. DCs presenting self-antigen can initiate autoantibody response, indicating that autoreactive cells exist in the normal body under the control of peripheral tolerance. When the mechanism of peripheral tolerance is broken, such as depletion of Treg cells, autoreactive cells would cause autoimmune diseases. Recent data also show that CD4+CD25+ T cells down-regulate the maturation and function of the DCs [32] and inhibit the maturation, rather than initiation, of autoantibody responses [33]. In our experiment, the depletion of Treg cells combined with accumulated autoantigens caused severe autoimmune responses in normal animal model.
A recent study demonstrated that eliminating CD4+CD25+ T cells induced an increase in anti-nuclear antibodies and accelerated the development of glomerulonephritis during the pre-active phase in BWF1 mice [34]. In our in vivo experiment, D2WF1 mice whose CD25+ cells were depleted produced higher titres of autoantibodies after being immunized with apoptotic cells-pulsed DCs. However, the production of autoantibodies was inhibited further after day 44 (Fig. 5). The reason may be that the haematopoietic system could reconstitute CD4+CD25+ T cells to control autoimmune response in normal mice.
Moreover, high titres of anti-DNA antibodies were not accompanied by the development of kidney disease in our animal experiment. Other key factors may be involved in the mechanism of end-organ damage. In lupus-prone NZM2328 mice, a locus Cgnz1 on chromosome 1 was linked to chronic glomerulonephritis and severe proteinuria in females. The study indicated that breaking tolerance to dsDNA and chromatin is not required for the pathogenesis of lupus nephritis [35].
The pathogenesis of SLE is complex. Several studies suggest that the causes of SLE were defective clearance of autoantigens and abnormal immunoregulation. Our study suggested that the defect of Treg cells in BWF1 mice may also contribute to ineffective inhibition of autoantibody production and subsequent pathological damage.
References
- 1.Walker LS, Abbas AK. The enemy within: keeping self-reactive T cells at bay in the periphery. Nat Rev Immunol. 2002;2:11–9. doi: 10.1038/nri701. [DOI] [PubMed] [Google Scholar]
- 2.Shevach EM. CD4+ CD25+ suppressor T cells: more questions than answers. Nat Rev Immunol. 2002;2:389–400. doi: 10.1038/nri821. [DOI] [PubMed] [Google Scholar]
- 3.Takahashi T, Kuniyasu Y, Toda M, et al. Immunologic self-tolerance maintained by CD25+CD4+ naturally anergic and suppressive T cells: induction of autoimmune disease by breaking their anergic/suppressive state. Int Immunol. 1998;10:1969–80. doi: 10.1093/intimm/10.12.1969. [DOI] [PubMed] [Google Scholar]
- 4.Itoh M, Takahashi T, Sakaguchi N, et al. Thymus and autoimmunity. production of CD25+CD4+ naturally anergic and suppressive T cells as a key function of the thymus in maintaining immunologic self-tolerance. J Immunol. 1999;162:5317–26. [PubMed] [Google Scholar]
- 5.Kuniyasu Y, Takahashi T, Itoh M, Shimizu J, Toda G, Sakaguchi S. Naturally anergic and suppressive CD25(+) CD4(+) T cells as a functionally and phenotypically distinct immunoregulatory T cell subpopulation. Int Immunol. 2000;12:1145–55. doi: 10.1093/intimm/12.8.1145. [DOI] [PubMed] [Google Scholar]
- 6.Sakaguchi S, Sakaguchi N, Asano M, Itoh M, Toda M. Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J Immunol. 1995;155:1151–64. [PubMed] [Google Scholar]
- 7.Hori S, Nomura T, Sakaguchi S. Control of regulatory T cell development by the transcription factor Foxp3. Science. 2003;299:1057–61. doi: 10.1126/science.1079490. [DOI] [PubMed] [Google Scholar]
- 8.Fontenot JD, Gavin MA, Rudensky AY. Foxp3 programs the development and function of CD4+CD25+ regulatory T cells. Nat Immunol. 2003;4:330–6. doi: 10.1038/ni904. [DOI] [PubMed] [Google Scholar]
- 9.Khattri R, Cox T, Yasayko SA, Ramsdell F. An essential role for Scurfin in CD4+CD25+ T regulatory cells. Nat Immunol. 2003;4:337–42. doi: 10.1038/ni909. [DOI] [PubMed] [Google Scholar]
- 10.von Boehmer H. Mechanisms of suppression by suppressor T cells. Nat Immunol. 2005;6:338–44. doi: 10.1038/ni1180. [DOI] [PubMed] [Google Scholar]
- 11.Maddison PJ. Is it SLE? Best Pract Res Clin Rheumatol. 2002;16:167–80. doi: 10.1053/berh.2001.0219. [DOI] [PubMed] [Google Scholar]
- 12.Kotzin BL. Systemic lupus erythematosus. Cell. 1996;85:303–6. doi: 10.1016/s0092-8674(00)81108-3. [DOI] [PubMed] [Google Scholar]
- 13.Robson MG, Walport MJ. Pathogenesis of systemic lupus erythematosus (SLE) Clin Exp Allergy. 2001;31:678–85. doi: 10.1046/j.1365-2222.2001.01147.x. [DOI] [PubMed] [Google Scholar]
- 14.Mok CC, Lau CS. Pathogenesis of systemic lupus erythematosus. J Clin Pathol. 2003;56:481–90. doi: 10.1136/jcp.56.7.481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wu HY, Staines NA. A deficiency of CD4+CD25+ T cells permits the development of spontaneous lupus-like disease in mice, and can be reversed by induction of mucosal tolerance to histone peptide autoantigen. Lupus. 2004;13:192–200. doi: 10.1191/0961203303lu1002oa. [DOI] [PubMed] [Google Scholar]
- 16.Crispin JC, Martinez A, Alcocer-Varela J. Quantification of regulatory T cells in patients with systemic lupus erythematosus. J Autoimmun. 2003;21:273–6. doi: 10.1016/s0896-8411(03)00121-5. [DOI] [PubMed] [Google Scholar]
- 17.Liu MF, Wang CR, Fung LL, Wu CR. Decreased CD4+CD25+ T cells in peripheral blood of patients with systemic lupus erythematosus. Scand J Immunol. 2004;59:198–202. doi: 10.1111/j.0300-9475.2004.01370.x. [DOI] [PubMed] [Google Scholar]
- 18.Lee JH, Wang LC, Lin YT, Yang YH, Lin DT, Chiang BL. Inverse correlation between CD4+ regulatory T-cell population and autoantibody levels in paediatric patients with systemic lupus erythematosus. Immunology. 2006;117:280–6. doi: 10.1111/j.1365-2567.2005.02306.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tzeng TC, Suen JL, Chiang BL. Dendritic cells pulsed with apoptotic cells activate self-reactive T cells of lupus mice both in vitro and in vivo. Rheumatology. 2006 doi: 10.1093/rheumatology/kel106. in press. [DOI] [PubMed] [Google Scholar]
- 20.Inaba K, Inaba M, Romani N, et al. Generation of large numbers of dendritic cells from mouse bone marrow cultures supplemented with granulocyte/macrophage colony-stimulating factor. J Exp Med. 1992;176:1693–702. doi: 10.1084/jem.176.6.1693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Monk CR, Spachidou M, Rovis F, et al. MRL/Mp CD4+, CD25- T cells show reduced sensitivity to suppression by CD4+, CD25+ regulatory T cells in vitro: a novel defect of T cell regulation in systemic lupus erythematosus. Arthritis Rheum. 2005;52:1180–4. doi: 10.1002/art.20976. [DOI] [PubMed] [Google Scholar]
- 22.Brunkow ME, Jeffery EW, Hjerrild KA, et al. Disruption of a new forkhead/winged-helix protein, scurfin, results in the fatal lymphoproliferative disorder of the scurfy mouse. Nat Genet. 2001;27:68–73. doi: 10.1038/83784. [DOI] [PubMed] [Google Scholar]
- 23.Wildin RS, Ramsdell F, Peake J, et al. X-linked neonatal diabetes mellitus, enteropathy and endocrinopathy syndrome is the human equivalent of mouse scurfy. Nat Genet. 2001;27:18–20. doi: 10.1038/83707. [DOI] [PubMed] [Google Scholar]
- 24.Bennett CL, Brunkow ME, Ramsdell F, et al. A rare polyadenylation signal mutation of the FOXP3 gene (AAUAAA → AAUGAA) leads to the IPEX syndrome. Immunogenetics. 2001;53:435–9. doi: 10.1007/s002510100358. [DOI] [PubMed] [Google Scholar]
- 25.Bennett CL, Christie J, Ramsdell F, et al. The immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) is caused by mutations of FOXP3. Nat Genet. 2001;27:20–1. doi: 10.1038/83713. [DOI] [PubMed] [Google Scholar]
- 26.Taams LS, Vukmanovic-Stejic M, Smith J, et al. Antigen-specific T cell suppression by human CD4+CD25+ regulatory T cells. Eur J Immunol. 2002;32:1621–30. doi: 10.1002/1521-4141(200206)32:6<1621::AID-IMMU1621>3.0.CO;2-Q. [DOI] [PubMed] [Google Scholar]
- 27.Kasow KA, Chen X, Knowles J, Wichlan D, Handgretinger R, Riberdy JM. Human CD4+CD25+ regulatory T cells share equally complex and comparable repertoires with CD4+CD25− counterparts. J Immunol. 2004;172:6123–8. doi: 10.4049/jimmunol.172.10.6123. [DOI] [PubMed] [Google Scholar]
- 28.Bagavant H, Tung KS. Failure of CD25+ T cells from lupus-prone mice to suppress lupus glomerulonephritis and sialoadenitis. J Immunol. 2005;175:944–50. doi: 10.4049/jimmunol.175.2.944. [DOI] [PubMed] [Google Scholar]
- 29.Asadullah K, Sterry W, Volk HD. Interleukin-10 therapy – review of a new approach. Pharmacol Rev. 2003;55:241–69. doi: 10.1124/pr.55.2.4. [DOI] [PubMed] [Google Scholar]
- 30.Liu H, Hu B, Xu D, Liew FY. CD4+CD25+ regulatory T cells cure murine colitis: the role of IL-10, TGF-beta, and CTLA4. J Immunol. 2003;171:5012–7. doi: 10.4049/jimmunol.171.10.5012. [DOI] [PubMed] [Google Scholar]
- 31.Beebe AM, Cua DJ, de Waal Malefyt R. The role of interleukin-10 in autoimmune disease: systemic lupus erythematosus (SLE) and multiple sclerosis (MS) Cytokine Growth Factor Rev. 2002;13:403–12. doi: 10.1016/s1359-6101(02)00025-4. [DOI] [PubMed] [Google Scholar]
- 32.Misra N, Bayry J, Lacroix-Desmazes S, Kazatchkine MD, Kaveri SV. Cutting edge: human CD4+CD25+ T cells restrain the maturation and antigen-presenting function of dendritic cells. J Immunol. 2004;172:4676–80. doi: 10.4049/jimmunol.172.8.4676. [DOI] [PubMed] [Google Scholar]
- 33.Fields ML, Hondowicz BD, Metzgar MH, et al. CD4+ CD25+ regulatory T cells inhibit the maturation but not the initiation of an autoantibody response. J Immunol. 2005;175:4255–64. doi: 10.4049/jimmunol.175.7.4255. [DOI] [PubMed] [Google Scholar]
- 34.Hayashi T, Hasegawa K, Adachi C. Elimination of CD4(+) CD25(+) T cell accelerates the development of glomerulonephritis during the preactive phase in autoimmune-prone female NZB × NZW F mice. Int J Exp Pathol. 2005;86:289–96. doi: 10.1111/j.0959-9673.2005.00438.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Waters ST, McDuffie M, Bagavant H, et al. Breaking tolerance to double stranded DNA, nucleosome, and other nuclear antigens is not required for the pathogenesis of lupus glomerulonephritis. J Exp Med. 2004;199:255–64. doi: 10.1084/jem.20031519. [DOI] [PMC free article] [PubMed] [Google Scholar]