Abstract
Summary
The Toll-like receptor (TLR) family provide key components of mammalian immunity and are part of the earliest surveillance mechanisms responding to infection. Their activation triggers the innate immune response, and is crucial to the successful induction of Th1/Th2-phenotyped adaptive immunity. Innate immunity was long considered to be non-specific and somewhat simple compared to adaptive immunity, mediated via the engulfment and lysis of microbial pathogens by phagocytic cells such as macrophages and neutrophils, and involving no complex protein–protein interactions. The emergence of the TLR field has contributed to a revision of our understanding, and innate immunity is now viewed as a highly complex process, in line with adaptive immunity. This review will give a brief overview of our current knowledge of TLR biology, and will focus on TLRs as key components in complex networks that activate, integrate and select the appropriate innate and adaptive immune responses in the face of immunological danger.
Keywords: inflammation, innate immunity, TLR
An evolutionary history of Toll-like receptors (TLRs)
TLRs are products of an evolutionary process that began prior to the separation of plants and animals, and are identified by a highly conserved intracellular Toll–interleukin (IL)-1 receptor (TIR) motif [1]. In plants, homologues of these proteins are represented by plant disease resistance genes encoding TIR-bearing proteins, although their precise mechanisms of action are still unknown [2]. In insects, notably Drosophila, the founding member of the TLR family, Toll, was shown in 1996 to be responsible for anti-fungal responses in the adult fly [3]. This discovery led the way to identification of a mammalian TLR [4], and a crucial link to immune function was elucidated using naturally occurring mouse strains that respond poorly to endotoxin, with meticulous genetic analysis revealing mutations in the gene encoding TLR4 [5–7]. Mammalian TLRs are now known to comprise a family of a minimum of 10 proteins [8] (an eleventh functional receptor has been identified in mice but not man [9]).
TLR activation
A diverse range of endogenous (host-derived) and exogenous (pathogen-derived) putative TLR ligands have now been identified (reviewed in [10,11]). Ligand recognition is thought to be determined by leucine-rich repeat (LRR) motifs within the TLR extracellular domains [12,13]. There has been a relative lack of experimental evidence confirming direct binding or physical association of TLRs and their ligands; however, recent work has identified the structure of the TLR3 ectodomain [12,13], and located the TLR3 ligand binding site using mutational analysis [14]. Some TLRs probably recognize their ligands in a complex association of other TLRs or co-molecules forming large activation clusters at the membrane [15,16], which may account for the delay in showing direct ligand binding. Of note, some caution is necessary when investigating TLR responses due to the capacity for small amounts of contaminants within the individual preparations to cause spurious responses (reviewed in [10,17]).
TLRs frequently expressed on the cell surface (1, 2, 4, 5, 6 and 10) recognize predominantly, but not exclusively, bacterial products. TLR2 acts as a heterodimer in concert with either TLR1 or TLR6, to mediate responses to moieties including lipoproteins and lipoteichoic acids (LTA) from Gram-positive bacteria [18] and rare lipopolysaccharide (LPS) species [19], and lipoarabinomannan from mycobacteria [20]. TLR10 was the orphan member of the family, but recent work has demonstrated its ability to form homodimers and also heterodimers with TLR1 and TLR2, although specific ligands for these combinations have yet to be identified [21]. TLR4 acts essentially as a homodimer (although there is some evidence for heterodimerization with TLR1 [22] and TLR5 [23]), and it also recognizes an extensive range of agonists. The involvement of accessory proteins at the cell membrane may help to confer ligand specificity and responses (such as CD14, MD2 and CD11b/CD18 [24]). TLR4 is crucial for effective responses to the Gram-negative bacterial component LPS [5–7], but also recognizes viral proteins [25,26]. In contrast, only a single agonist group has been identified so far for TLR5 comprising the flagellins, an essential protein component of bacterial flagella found in both Gram-positive and Gram-negative bacteria [27,28].
TLRs expressed primarily intracellularly (3, 7, 8 and 9) specialize in viral detection or the recognition of nucleic acids. TLR3 recognizes double-stranded RNA [29], while TLR7 and/or 8 mediate responses to single-stranded RNA, also derived from viruses [30,31]. TLR7 and 8 also respond to potent immunomodulatory nucleoside or nucleoside-like drugs, such as loxoribine and the imidazoquinolines [30,32], and appear to be responsible for the potent actions of the imidazoquinolines in the treatment of skin tumours [33]. TLR9 recognizes unmethylated CG dinucleotides (CpG motifs), mediating responses to bacterial DNA and some viruses [34,35]. It should be noted that receptor localization is not definitive and can also depend on cell type; for example, dendritic cells and airway smooth muscle cells express TLR3 intracellularly, while fibroblasts and epithelial cells have the potential to express TLR3 both on the cell surface and within the cell [36–39]. Furthermore, in phagocytic cells there is evidence that cell surface receptors can be recruited to the phagosome upon ligand-driven activation [40–42].
There is increasing evidence that ‘endogenous’ ligands may also activate TLRs; these include products derived from host cells, for example mRNA [43] (recognized by TLR3), and anti-microbial molecules such as defensins [44] and reactive oxygen species (TLR2) [45]. In addition, products derived from damaged or dying cells can act as agonists, and proposed members of this category of TLR4-activators include heat shock protein B8 [46], fibrinogen [47], surfactant protein A [48] and the tissue matrix breakdown products fibronectin extra domain A [49] and hyaluronic acid oligosaccharides [50]. Finally, high mobility group box 1 (HMGB1) protein can act as a ligand for TLR2 and TLR4 [51].
TLR signalling
TLR signalling events have been the subject of intense investigation and are reviewed extensively elsewhere [52–54]. The tailoring of individual responses to pathogens is mediated partly through a family of adapter molecules comprising MyD88, Mal (MyD88 adapter-like)/TIRAP (TIR-domain-containing adapter protein), TRIF (Toll-receptor-associated activator of interferon) and TRAM (TRIF-related adapter molecule). A further adapter, SARM (sterile α and armadillo motifs) [55], is a member of the same family but does not yet have a clearly identified signalling role. The prototypic pathway is the MyD88-dependent pathway. MyD88 is utilized by all the TLRs, except TLR3, and requires the bridging adapter Mal/TIRAP for TLR2 and TLR4 signalling. Upon activation MyD88 recruits IRAK-4, allowing association and phosphorylation of IRAK-1, which in turn recruits TRAF6. TRAF6 activates the TAK-1/TAB1/2/3 complex, resulting in downstream cascades that lead ultimately to activation of nuclear factor (NF)-κB [56], and also initiation of distinct parallel signalling pathways leading to mitogen-activated protein (MAP) kinase [57–59] and phosphoinositide 3-kinase (PI3K) [60,61] activation. These separate pathways all regulate the transcription, mRNA stability and translation of a myriad of proinflammatory cytokine genes (see TLRs and leucocytes). In addition, TLR7 and TLR9 signalling via MyD88 can induce the type I interferons (IFNs) in a cascade also involving TRAF3 [62,63] and IFN regulatory factor (IRF)-7, which translocates to the nucleus and induces the IFN-α and IFN-β promoters [64,65]. Critical roles for other IRF family members have also been identified. IRF-5 co-localizes and associates with MyD88 and TRAF-6 and is required for inflammatory cytokine, but not IFN-α/β, production upon stimulation with TLR3, 4, 5, 7/8 and 9 ligands [66]. IRF-4 is a negative regulator of the signalling pathway, competing with IRF-5 for association with MyD88 [67]. IRF-3 is involved in the MyD88-independent pathway, which utilizes the TRIF adapter [68,69]. TRIF is recruited selectively by TLR3 and TLR4, although TLR4 requires a fourth adapter molecule, TRAM, to link it to TRIF [70]. This pathway again utilizes TRAF3, which interacts with TBK-1 and IKKε to mediate activation of IRF-3 and finally induction of the IFN-β promoter and several other target genes, for example CXCL10 [71,72]. TRIF also directly interacts with TRAF6, leading to NF-κB and MAP kinase activation, and with receptor-interacting protein (RIP) 1, a kinase thought to play a critical role in NF-κB activation [73].
Control of innate immunity by TLRs
TLRs and leucocytes
TLRs are often considered to be the starting point of immunity, continuously sampling the extracellular environment, informing the cell to respond to infection, and facilitating these cellular responses via signalling pathways culminating in new gene transcription. One of the earliest phagocytes to respond to infection is the tissue macrophage, which originates as monocytes in the peripheral blood system. Monocytes (and other blood mononuclear cells) are highly responsive to TLR agonists, expressing a substantial complement of the TLR repertoire [74–76]. Indeed, TLR stimulation activates a myriad of genes in human monocytes, leading to the release of cytokines, chemokines and growth factors that may exert potent autocrine and paracrine inflammatory responses [77–79]. Cytokines such as tumour necrosis factor (TNF)-α, interleukin (IL)-1β and IL-6 are often held to be principal exemplars of the panel of proinflammatory mediators produced in response to TLR stimuli; however, chemokine genes actually show the strongest gene induction [77,80]. This has important implications for the inflammatory phenotype produced during microbial insults, specifically the recruitment and infiltration of other cell types to the site of infection. CCL2, CCL3 and CCL4 are the principal CC chemokine family members produced [77], and are chemotactic mainly for monocytes and T cells, but also basophils, dendritic and natural killer (NK) cells [81]. CXCL8 and CXCL2/3 are the main CXC family members released in response to TLR activation [77], and are chemotactic primarily for neutrophils augmenting their degranulation, adhesion and microbicidal activity [82]; however, they can also influence T cell, basophil, eosinophil, NK cell and endothelial cell chemotaxis [81]. Importantly, tissue macrophages can profoundly alter their phenotype as they differentiate in tissues. For example, alveolar macrophages show poor TLR9 responses compared to macrophages from other tissues [83]. While alveolar macrophages are important in the induction of inflammation in response to inhaled LPS [84], intestinal macrophages are non-responsive to LPS from early life − indeed, even before they are ever exposed to LPS [85]. Much work therefore remains to be conducted in developing a complete understanding of the initiation and control of inflammation by macrophages from specific tissues.
As well as cell recruitment, cell–cell communication is a vital part of innate immunity. It is now clear that granulocytes require co-operation from monocytic cells in order to perform their full range of TLR-mediated functions. While LPS is an effective direct activator of neutrophils [86–88], neutrophil survival in response to LPS is dependent upon survival signals released from small numbers of monocytes that commonly contaminate neutrophil cell preparations [89,90]. A similar role for monocytes in mediating LPS-induced eosinophil survival has also been described [91]. Recruitment of neutrophils into the lung after inhaled LPS challenge [84], or recruitment of eosinophils into the pleural space after local LPS administration, requires resident macrophages and, for eosinophil recruitment, lymphocytes [92,93], further corroborating the importance of innate immune cell communication in the context of TLR signalling and pathogen responsiveness.
TLRs and tissue cells
Although the expression of TLRs is thought typically to be a feature of immune cells, tissue cells can also respond to inflammatory stimuli via TLRs. An increasing number of tissue cell types have been shown to express TLRs, which may contribute to protection against infection; these include epithelial cells at potential sites of entry, including the skin [94,95], respiratory [78,96,97], intestinal [98] and genitourinary [99] tracts, all of which act as a protective barrier against invading pathogens. If pathogens gain entry to the host successfully, endothelial cells [39,100–102] and smooth muscle cells [39,103,104] can also play a role in their detection via TLRs.
The role of TLRs on tissue cells is highly complex, with effective inflammatory responses to some TLRs occurring directly, while others are dependent upon co-operative interactions between leucocytes and tissue cells. For example, studies from our group have shown that airway smooth muscle cells (ASMCs) express restricted members of the TLR family, which can be modulated by proinflammatory stimuli [39,104]. TLR3 activation of ASMCs leads directly to the release of chemokines and cytokines, resulting in a markedly proinflammatory phenotype [39]. One of the major chemokines released is CXCL10, a Th1-associated chemokine with the potential to recruit T cells to the lung [105] and mast cells to airway smooth muscle bundles [106,107], a specific feature of asthmatic airways [108]. ASMCs also express TLR4 mRNA, but in our hands they exhibit no or minimal responses to LPS [104], although an effective lack of an LPS response in these cells is not a universal finding [109]. If ASMCs are cultured with even low numbers of mononuclear cells, LPS is able to cause marked cytokine production from the tissue cells, an indirect response that is abrogated substantially by IL-1 receptor antagonist (IL-1ra) [39,104], and dominates any response observed to LPS in the absence of leucocytes. Similarly, ASMC are negative for TLR7/8 expression and responses to TLR7/8 agonists are, not surprisingly, also dependent upon the presence of mononuclear cells [39]. Thus, complex networks are formed in which patterns of TLR expression and co-localization of tissue cells and leucocytes are critical for both cell–cell communication and the development of effective immunity. The role of TLRs in anti-viral immunity is explored in a further paper in this mini-series.
TLRs link innate and adaptive immunity
Again, the subject of a separate paper in this mini-series, TLRs play a crucial role in linking innate and adaptive immunity through actions on T cells [110–112] and particularly via actions on dendritic cells (DCs) (reviewed in [113–115]). DCs start out as immature cells with high endocytic activity and low T cell activation potential, and function to detect, capture and phagocytose pathogens, leading to TLR activation. Upon signal transduction through TLRs, DCs undergo a complex differentiation programme termed collectively DC maturation, characterized by up-regulation of cell surface MHC molecules containing pathogen-derived peptide fragments and co-receptors (CD40, CD80 and CD86) that enhance the ability of DCs to activate T cells [116].
Stimulation of many cell types with TLR agonists, bacteria and viruses results in type 1 interferon (IFN-α/β) production [117–119], now thought to be part of the first line of defence against infection and a central modulator of adaptive immunity [120]. The classical pathway of IFN production via TLRs is described above (see TLR signalling). Although thought to function mainly in an anti-viral capacity, IFNs have diverse functions in the development of adaptive immunity, including proliferation of memory T cells, inhibition of T cell apoptosis, enhanced IFN-γ secretion, B cell isotype switching and differentiation into plasma cells and NK cell activation [114].
TLR networks: enhancing inflammation and disease
Excessive proinflammatory cytokine production coupled with enhanced cell activation can lead to inflammatory diseases affecting a broad range of tissues and organs. Accordingly, amplification of immune responses by TLRs, while crucial in bringing together multiple aspects of the immune system, may also contribute to disease.
Asthma is one of many obvious examples of such, where it is clear that TLRs play a role in controlling bacterial and/or viral respiratory infections, which are important modulators of allergic airway inflammation [121,122]. There are numerous opportunities for multiple TLR agonists to interact directly and/or indirectly (via leucocytes) with respiratory tissue cells. Inhaled endotoxin is present routinely and can play a fundamental role in the development and progression of asthma [123,124] through differential actions on TLR4, determined by specific cell-dependent mechanisms. For example, it was demonstrated recently that inhaled endotoxin-induced bronchoconstriction is dependent upon resident lung cells, most probably epithelial cells, while recruited haematopoietic cells mediate cytokine secretion into the alveolar space in response to the same insult [125]. As well as forming a barrier against infection, epithelial cells are dynamic produces of anti-microbial peptides in response to infection [126–128] and to growth factors released when the physical barrier is penetrated [129], with subsequent tissue damage also resulting in activation of innate immunity and TLR signalling. The release of chemokines and adhesion molecules from these resident tissue cells also controls neutrophil infiltration and activation, resulting in the further release of mediators of tissue injury and repair processes and supporting the anti-microbial potential of neutrophils themselves, but also of the resident cells such as epithelia and kerotinocytes [130]. Recruited haematopoietic cells and alveolar macrophages have also been shown to play a crucial role in initiating LPS-induced neutrophil recruitment to the lung [84], while other work identifies monocytes as key players in alveolar neutrophil recruitment, with direct co-operation between monocytes and neutrophils contributing to acute lung inflammation [131]. Monocytes can also act indirectly to amplify epithelial cell production of anti-microbial peptides in response to LPS through an IL-1-mediated pathway [132,133]. Finally, monocytes directly activate endothelial cells and smooth muscle cells to support further adhesion and recruitment of leucocytes, potentially creating a self-perpetuating cycle of inflammation [104,134].
Viruses such as respiratory syncytial virus (RSV) and influenza A cause a myriad of respiratory diseases, including exacerbation of asthma, through preferential infection of host airway epithelial cells. Viral infections enhance responses to LPS and other TLR agonists by triggering additional TLR signalling pathways through presentation of agonists for TLR3, TLR4, TLR7, TLR8 and TLR9. For example, a downstream effect of viral infection is sensitization of the airway cells to subsequent exposure to viral and bacterial products; this occurs via up-regulation of TLR3 and TLR4, respectively, leading to increased signalling activity and proinflammatory cytokine production [78,135–137]. Interestingly, this can be a selective effect, as RSV infection does not enhance airway epithelial cell responses to ligands acting on the viral receptors TLR7/8 or TLR9 [135]; in contrast, the synthetic viral mimic poly(I:C), which activates TLR3, augments expression of TLR1 and TLR2, receptors that mediate bacterial responses [78]. Accordingly, the profound immune and inflammatory response triggered during diseases such as asthma will be dependent upon the complicated patterns of TLR cross-talk that occur between each individual cell type, and co-operative responses of a range of tissue cells and professional immune cells to TLR activation.
Concluding remarks
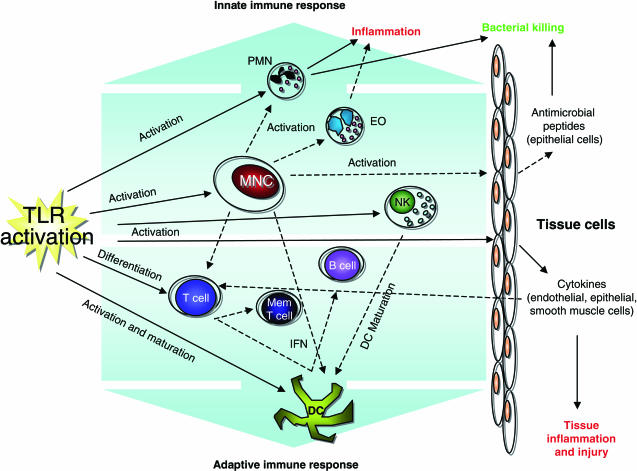
As our depth of knowledge about TLRs continues to expand, it is becoming ever more clear that an inflammatory model whereby a single microbial ligand targets a lone receptor is insufficient to describe the vast diversity of the host's response to attack. A more useful approach may be to think of inflammatory networks with microbes activating multiple receptors, resulting in the initiation of numerous signalling pathways to integrate and evaluate the threat and determine the appropriate activation status of the host cells and the ultimate inflammatory response (Fig. 1). This approach highlights the complexity of disease pathophysiology and the difficulties faced when trying to delineate single causes or factors, with a given disease dependent upon the network of cells and TLRs that are activated. Thus, to facilitate our ability to intervene when inflammatory responses to disease are inadequate, or indeed excessive, models of inflammatory cell networks must continue to be developed to determine the most appropriate and effective response to disease.
Fig. 1.
Immune pathways activated by Toll-like receptor (TLR) signalling. TLR signalling activates a number of apical pathways that result in the stimulation of both the innate and adaptive immune responses. Mononuclear cells (MNC) in particular serve to amplify TLR activation by the production of cytokines and growth factors. These mediators facilitate communication between different cell types, co-ordinating the recruitment and activation of the immune response network as a whole. Solid arrows illustrate effects of direct TLR activation, dashed arrows represent paracrine actions resulting from TLR activation of an intermediary cell. MNC; mononuclear cells, PMN; polymorphonuclear cells, EO; eosinophils, NK; natural killer cells, DC; dendritic cells, Mem T cell; memory T cell, IFN; interferon.
References
- 1.Beutler B, Rehli M. Evolution of the TIR, tolls and TLRs: functional inferences from computational biology. Curr Top Microbiol Immunol. 2002;270:1–21. doi: 10.1007/978-3-642-59430-4_1. [DOI] [PubMed] [Google Scholar]
- 2.Dangl JL, Jones JD. Plant pathogens and integrated defence responses to infection. Nature. 2001;411:826–33. doi: 10.1038/35081161. [DOI] [PubMed] [Google Scholar]
- 3.Lemaitre B, Nicolas E, Michaut L, Reichhart JM, Hoffmann JA. The dorsoventral regulatory gene cassette spatzle/Toll/cactus controls the potent antifungal response in Drosophila adults. Cell. 1996;86:973–83. doi: 10.1016/s0092-8674(00)80172-5. [DOI] [PubMed] [Google Scholar]
- 4.Medzhitov R, Preston-Hurlburt P, Janeway CA., Jr A human homologue of the Drosophila Toll protein signals activation of adaptive immunity. Nature. 1997;388:394–7. doi: 10.1038/41131. [DOI] [PubMed] [Google Scholar]
- 5.Poltorak A, He X, Smirnova I, et al. Defective LPS signaling in C3H/HeJ and C57BL/10ScCr mice: mutations in Tlr4 gene. Science. 1998;282:2085–8. doi: 10.1126/science.282.5396.2085. [DOI] [PubMed] [Google Scholar]
- 6.Qureshi ST, Lariviere L, Leveque G, et al. Endotoxin-tolerant mice have mutations in Toll-like receptor 4 (Tlr4) J Exp Med. 1999;189:615–25. doi: 10.1084/jem.189.4.615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hoshino K, Takeuchi O, Kawai T, et al. Cutting edge: Toll-like receptor 4 (TLR4)-deficient mice are hyporesponsive to lipopolysaccharide: evidence for TLR4 as the Lps gene product. J Immunol. 1999;162:3749–52. [PubMed] [Google Scholar]
- 8.Akira S. Mammalian Toll-like receptors. Curr Opin Immunol. 2003;15:5–11. doi: 10.1016/s0952-7915(02)00013-4. [DOI] [PubMed] [Google Scholar]
- 9.Zhang D, Zhang G, Hayden MS, et al. A Toll-like receptor that prevents infection by uropathogenic bacteria. Science. 2004;303:1522–6. doi: 10.1126/science.1094351. [DOI] [PubMed] [Google Scholar]
- 10.Tsan MF, Gao B. Endogenous ligands of Toll-like receptors. J Leukoc Biol. 2004;76:514–19. doi: 10.1189/jlb.0304127. [DOI] [PubMed] [Google Scholar]
- 11.Ishii KJ, Coban C, Akira S. Manifold mechanisms of Toll-like receptor-ligand recognition. J Clin Immunol. 2005;25:511–21. doi: 10.1007/s10875-005-7829-1. [DOI] [PubMed] [Google Scholar]
- 12.Bell JK, Botos I, Hall PR, et al. The molecular structure of the Toll-like receptor 3 ligand-binding domain. Proc Natl Acad Sci USA. 2005;102:10976–80. doi: 10.1073/pnas.0505077102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Choe J, Kelker MS, Wilson IA. Crystal structure of human Toll-like receptor 3 (TLR3) ectodomain. Science. 2005;309:581–5. doi: 10.1126/science.1115253. [DOI] [PubMed] [Google Scholar]
- 14.Bell JK, Askins J, Hall PR, Davies DR, Segal DM. The dsRNA binding site of human Toll-like receptor 3. Proc Natl Acad Sci USA. 2006;103:8792–7. doi: 10.1073/pnas.0603245103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Underhill DM. Toll-like receptors: networking for success. Eur J Immunol. 2003;33:1767–75. doi: 10.1002/eji.200324037. [DOI] [PubMed] [Google Scholar]
- 16.Triantafilou M, Brandenburg K, Kusumoto S, et al. Combinational clustering of receptors following stimulation by bacterial products determines lipopolysaccharide responses. Biochem J. 2004;381:527–36. doi: 10.1042/BJ20040172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wakelin SJ, Sabroe I, Gregory CD, et al. ‘Dirty little secrets’ − endotoxin contamination of recombinant proteins. Immunol Lett. 2006;106:1–7. doi: 10.1016/j.imlet.2006.04.007. [DOI] [PubMed] [Google Scholar]
- 18.Knapp S, Wieland CW, van't Veer C, et al. Toll-like receptor 2 plays a role in the early inflammatory response to murine pneumococcal pneumonia but does not contribute to antibacterial defense. J Immunol. 2004;172:3132–8. doi: 10.4049/jimmunol.172.5.3132. [DOI] [PubMed] [Google Scholar]
- 19.Martin M, Katz J, Vogel SN, Michalek SM. Differential induction of endotoxin tolerance by lipopolysaccharides derived from Porphyromonas gingivalis and Escherichia coli. J Immunol. 2001;167:5278–85. doi: 10.4049/jimmunol.167.9.5278. [DOI] [PubMed] [Google Scholar]
- 20.Tapping RI, Tobias PS. Mycobacterial lipoarabinomannan mediates physical interactions between TLR1 and TLR2 to induce signaling. J Endotoxin Res. 2003;9:264–8. doi: 10.1179/096805103225001477. [DOI] [PubMed] [Google Scholar]
- 21.Hasan U, Chaffois C, Gaillard C, et al. Human TLR10 is a functional receptor, expressed by B cells and plasmacytoid dendritic cells, which activates gene transcription through MyD88. J Immunol. 2005;174:2942–50. doi: 10.4049/jimmunol.174.5.2942. [DOI] [PubMed] [Google Scholar]
- 22.Spitzer JH, Visintin A, Mazzoni A, Kennedy MN, Segal DM. Toll-like receptor 1 inhibits Toll-like receptor 4 signaling in endothelial cells. Eur J Immunol. 2002;32:1182–7. doi: 10.1002/1521-4141(200204)32:4<1182::AID-IMMU1182>3.0.CO;2-9. [DOI] [PubMed] [Google Scholar]
- 23.Mizel SB, Honko AN, Moors MA, Smith PS, West AP. Induction of macrophage nitric oxide production by Gram-negative flagellin involves signaling via heteromeric Toll-like receptor 5/Toll-like receptor 4 complexes. J Immunol. 2003;170:6217–23. doi: 10.4049/jimmunol.170.12.6217. [DOI] [PubMed] [Google Scholar]
- 24.Perera PY, Mayadas TN, Takeuchi O, et al. CD11b/CD18 acts in concert with CD14 and Toll-like receptor (TLR) 4 to elicit full lipopolysaccharide and taxol-inducible gene expression. J Immunol. 2001;166:574–81. doi: 10.4049/jimmunol.166.1.574. [DOI] [PubMed] [Google Scholar]
- 25.Kurt-Jones EA, Popova L, Kwinn L, et al. Pattern recognition receptors TLR4 and CD14 mediate response to respiratory syncytial virus. Nat Immunol. 2000;1:398–401. doi: 10.1038/80833. [DOI] [PubMed] [Google Scholar]
- 26.Rassa JC, Meyers JL, Zhang Y, Kudaravalli R, Ross SR. Murine retroviruses activate B cells via interaction with Toll-like receptor 4. Proc Natl Acad Sci USA. 2002;99:2281–6. doi: 10.1073/pnas.042355399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hayashi F, Smith KD, Ozinsky A, et al. The innate immune response to bacterial flagellin is mediated by Toll-like receptor 5. Nature. 2001;410:1099–103. doi: 10.1038/35074106. [DOI] [PubMed] [Google Scholar]
- 28.Smith KD, Andersen-Nissen E, Hayashi F, et al. Toll-like receptor 5 recognizes a conserved site on flagellin required for protofilament formation and bacterial motility. Nat Immunol. 2003;4:1247–53. doi: 10.1038/ni1011. [DOI] [PubMed] [Google Scholar]
- 29.Alexopoulou L, Holt AC, Medzhitov R, Flavell RA. Recognition of double-stranded RNA and activation of NF-kappaB by Toll-like receptor 3. Nature. 2001;413:732–8. doi: 10.1038/35099560. [DOI] [PubMed] [Google Scholar]
- 30.Heil F, Ahmad-Nejad P, Hemmi H, et al. The Toll-like receptor 7 (TLR7)-specific stimulus loxoribine uncovers a strong relationship within the TLR7, 8 and 9 subfamily. Eur J Immunol. 2003;33:2987–97. doi: 10.1002/eji.200324238. [DOI] [PubMed] [Google Scholar]
- 31.Diebold SS, Kaisho T, Hemmi H, Akira S, Reis e Sousa C. Innate antiviral responses by means of TLR7-mediated recognition of single-stranded RNA. Science. 2004;303:1529–31. doi: 10.1126/science.1093616. [DOI] [PubMed] [Google Scholar]
- 32.Stanley MA. Imiquimod and the imidazoquinolones: mechanism of action and therapeutic potential. Clin Exp Dermatol. 2002;27:571–7. doi: 10.1046/j.1365-2230.2002.01151.x. [DOI] [PubMed] [Google Scholar]
- 33.Spaner DE, Miller RL, Mena J, Grossman L, Sorrenti V, Shi Y. Regression of lymphomatous skin deposits in a chronic lymphocytic leukemia patient treated with the Toll-like receptor-7/8 agonist, imiquimod. Leuk Lymph. 2005;46:935–9. doi: 10.1080/10428190500054426. [DOI] [PubMed] [Google Scholar]
- 34.Krug A, Luker GD, Barchet W, Leib DA, Akira S, Colonna M. Herpes simplex virus type 1 activates murine natural interferon-producing cells through toll-like receptor 9. Blood. 2004;103:1433–7. doi: 10.1182/blood-2003-08-2674. [DOI] [PubMed] [Google Scholar]
- 35.Lund J, Sato A, Akira S, Medzhitov R, Iwasaki A. Toll-like receptor 9-mediated recognition of Herpes simplex virus-2 by plasmacytoid dendritic cells. J Exp Med. 2003;198:513–20. doi: 10.1084/jem.20030162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Matsumoto M, Kikkawa S, Kohase M, Miyake K, Seya T. Establishment of a monoclonal antibody against human Toll-like receptor 3 that blocks double-stranded RNA-mediated signaling. Biochem Biophys Res Commun. 2002;293:1364–9. doi: 10.1016/S0006-291X(02)00380-7. [DOI] [PubMed] [Google Scholar]
- 37.Matsumoto M, Funami K, Tanabe M, et al. Subcellular localization of Toll-like receptor 3 in human dendritic cells. J Immunol. 2003;171:3154–62. doi: 10.4049/jimmunol.171.6.3154. [DOI] [PubMed] [Google Scholar]
- 38.Sha Q, Truong-Tran AQ, Plitt JR, Beck LA, Schleimer RP. Activation of airway epithelial cells by toll-like receptor agonists. Am J Respir Cell Mol Biol. 2004;31:358–64. doi: 10.1165/rcmb.2003-0388OC. [DOI] [PubMed] [Google Scholar]
- 39.Morris GE, Parker LC, Ward JR, et al. Cooperative molecular and cellular networks regulate TLR-dependent inflammatory responses. FASEB J. doi: 10.1096/fj.06-5910fje. in press. [DOI] [PubMed] [Google Scholar]
- 40.Underhill DM, Ozinsky A. Phagocytosis of microbes: complexity in action. Annu Rev Immunol 2002. 2006;20:825–52. doi: 10.1146/annurev.immunol.20.103001.114744. [DOI] [PubMed] [Google Scholar]
- 41.Underhill DM, Ozinsky A, Hajjar AM, et al. The Toll-like receptor 2 is recruited to macrophage phagosomes and discriminates between pathogens. Nature. 1999;401:811–15. doi: 10.1038/44605. [DOI] [PubMed] [Google Scholar]
- 42.Ozinsky A, Underhill DM, Fontenot JD, et al. The repertoire for pattern recognition of pathogens by the innate immune system is defined by cooperation between Toll-like receptors. Proc Natl Acad Sci USA. 2000;97:13766–71. doi: 10.1073/pnas.250476497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kariko K, Ni H, Capodici J, Lamphier M, Weissman D. mRNA is an endogenous ligand for Toll-like receptor 3. J Biol Chem. 2004;279:12542–50. doi: 10.1074/jbc.M310175200. [DOI] [PubMed] [Google Scholar]
- 44.Biragyn A, Ruffini PA, Leifer CA, et al. Toll-like receptor 4-dependent activation of dendritic cells by beta-defensin 2. Science. 2002;298:1025–9. doi: 10.1126/science.1075565. [DOI] [PubMed] [Google Scholar]
- 45.Frantz S, Kelly RA, Bourcier T. Role of TLR-2 in the activation of nuclear factor kappaB by oxidative stress in cardiac myocytes. J Biol Chem. 2001;276:5197–203. doi: 10.1074/jbc.M009160200. [DOI] [PubMed] [Google Scholar]
- 46.Roelofs MF, Boelens WC, Joosten LA, et al. Identification of small heat shock protein B8 (HSP22) as a novel TLR4 ligand and potential involvement in the pathogenesis of rheumatoid arthritis. J Immunol. 2006;176:7021–7. doi: 10.4049/jimmunol.176.11.7021. [DOI] [PubMed] [Google Scholar]
- 47.Smiley ST, King JA, Hancock WW. Fibrinogen stimulates macrophage chemokine secretion through toll-like receptor 4. J Immunol. 2001;167:2887–94. doi: 10.4049/jimmunol.167.5.2887. [DOI] [PubMed] [Google Scholar]
- 48.Guillot L, Balloy V, McCormack FX, Golenbock DT, Chignard M, Si-Tahar M. Cutting edge: the immunostimulatory activity of the lung surfactant protein-A involves Toll-like receptor 4. J Immunol. 2002;168:5989–92. doi: 10.4049/jimmunol.168.12.5989. [DOI] [PubMed] [Google Scholar]
- 49.Okamura Y, Watari M, Jerud ES, et al. The extra domain A of fibronectin activates Toll-like receptor 4. J Biol Chem. 2001;276:10229–33. doi: 10.1074/jbc.M100099200. [DOI] [PubMed] [Google Scholar]
- 50.Johnson GB, Brunn GJ, Kodaira Y, Platt JL. Receptor-mediated monitoring of tissue well-being via detection of soluble heparan sulfate by Toll-like receptor 4. J Immunol. 2002;168:5233–9. doi: 10.4049/jimmunol.168.10.5233. [DOI] [PubMed] [Google Scholar]
- 51.Park JS, Svetkauskaite D, He Q, et al. Involvement of toll-like receptors 2 and 4 in cellular activation by high mobility group box 1 protein. J Biol Chem. 2004;279:7370–7. doi: 10.1074/jbc.M306793200. [DOI] [PubMed] [Google Scholar]
- 52.O'Neill LA. How Toll-like receptors signal: what we know and what we don't know. Curr Opin Immunol. 2006;18:3–9. doi: 10.1016/j.coi.2005.11.012. [DOI] [PubMed] [Google Scholar]
- 53.Kawai T, Akira S. TLR signaling. Cell Death Differ. 2006;13:816–25. doi: 10.1038/sj.cdd.4401850. [DOI] [PubMed] [Google Scholar]
- 54.Moynagh PN. TLR signalling and activation of IRFs: revisiting old friends from the NF-kappaB pathway. Trends Immunol. 2005;26:469–76. doi: 10.1016/j.it.2005.06.009. [DOI] [PubMed] [Google Scholar]
- 55.O'Neill LA, Fitzgerald KA, Bowie AG. The Toll-IL-1 receptor adaptor family grows to five members. Trends Immunol. 2003;24:286–90. doi: 10.1016/s1471-4906(03)00115-7. [DOI] [PubMed] [Google Scholar]
- 56.Zhang FX, Kirschning CJ, Mancinelli R, et al. Bacterial lipopolysaccharide activates nuclear factor-kappaB through interleukin-1 signaling mediators in cultured human dermal endothelial cells and mononuclear phagocytes. J Biol Chem. 1999;274:7611–14. doi: 10.1074/jbc.274.12.7611. [DOI] [PubMed] [Google Scholar]
- 57.Hacker H, Mischak H, Hacker G, et al. Cell type-specific activation of mitogen-activated protein kinases by CpG-DNA controls interleukin-12 release from antigen-presenting cells. EMBO J. 1999;18:6973–82. doi: 10.1093/emboj/18.24.6973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Cario E, Rosenberg IM, Brandwein SL, Beck PL, Reinecker HC, Podolsky DK. Lipopolysaccharide activates distinct signaling pathways in intestinal epithelial cell lines expressing Toll-like receptors. J Immunol. 2000;164:966–72. doi: 10.4049/jimmunol.164.2.966. [DOI] [PubMed] [Google Scholar]
- 59.Ropert C, Almeida IC, Closel M, et al. Requirement of mitogen-activated protein kinases and I kappa B phosphorylation for induction of proinflammatory cytokines synthesis by macrophages indicates functional similarity of receptors triggered by glycosylphosphatidylinositol anchors from parasitic protozoa and bacterial lipopolysaccharide. J Immunol. 2001;166:3423–31. doi: 10.4049/jimmunol.166.5.3423. [DOI] [PubMed] [Google Scholar]
- 60.Arndt PG, Suzuki N, Avdi NJ, Malcolm KC, Worthen GS. Lipopolysaccharide-induced c-Jun NH2-terminal kinase activation in human neutrophils: role of phosphatidylinositol 3-kinase and Syk-mediated pathways. J Biol Chem. 2004;279:10883–91. doi: 10.1074/jbc.M309901200. [DOI] [PubMed] [Google Scholar]
- 61.Strassheim D, Asehnoune K, Park JS, et al. Phosphoinositide 3-kinase and Akt occupy central roles in inflammatory responses of Toll-like receptor 2-stimulated neutrophils. J Immunol. 2004;172:5727–33. doi: 10.4049/jimmunol.172.9.5727. [DOI] [PubMed] [Google Scholar]
- 62.Hacker H, Redecke V, Blagoev B, et al. Specificity in Toll-like receptor signalling through distinct effector functions of TRAF3 and TRAF6. Nature. 2006;439:204–7. doi: 10.1038/nature04369. [DOI] [PubMed] [Google Scholar]
- 63.Oganesyan G, Saha SK, Guo B, et al. Critical role of TRAF3 in the Toll-like receptor-dependent and -independent antiviral response. Nature. 2006;43:208–11. doi: 10.1038/nature04374. [DOI] [PubMed] [Google Scholar]
- 64.Kawai T, Sato S, Ishii KJ, et al. Interferon-alpha induction through Toll-like receptors involves a direct interaction of IRF7 with MyD88 and TRAF6. Nat Immunol. 2004;5:1061–8. doi: 10.1038/ni1118. [DOI] [PubMed] [Google Scholar]
- 65.Honda K, Yanai H, Mizutani T, et al. Role of a transductional-transcriptional processor complex involving MyD88 and IRF-7 in Toll-like receptor signaling. Proc Natl Acad Sci USA. 2004;101:15416–21. doi: 10.1073/pnas.0406933101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Takaoka A, Yanai H, Kondo S, et al. Integral role of IRF-5 in the gene induction programme activated by Toll-like receptors. Nature. 2005;434:243–9. doi: 10.1038/nature03308. [DOI] [PubMed] [Google Scholar]
- 67.Negishi H, Ohba Y, Yanai H, et al. Negative regulation of Toll-like-receptor signaling by IRF-4. Proc Natl Acad Sci USA. 2005;102:15989–94. doi: 10.1073/pnas.0508327102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Yamamoto M, Sato S, Hemmi H, et al. Role of adaptor TRIF in the MyD88-independent Toll-like receptor signaling pathway. Science. 2003;301:640–3. doi: 10.1126/science.1087262. [DOI] [PubMed] [Google Scholar]
- 69.Yamamoto M, Sato S, Hemmi H, et al. Essential role for TIRAP in activation of the signalling cascade shared by TLR2 and TLR4. Nature. 2002;420:324–9. doi: 10.1038/nature01182. [DOI] [PubMed] [Google Scholar]
- 70.Yamamoto M, Sato S, Hemmi H, et al. TRAM is specifically involved in the Toll-like receptor 4-mediated MyD88-independent signaling pathway. Nat Immunol. 2003;4:1144–50. doi: 10.1038/ni986. [DOI] [PubMed] [Google Scholar]
- 71.Hoebe K, Beutler B. TRAF3: a new component of the TLR-signaling apparatus. Trends Mol Med. 2006;12:187–9. doi: 10.1016/j.molmed.2006.03.008. [DOI] [PubMed] [Google Scholar]
- 72.Kawai T, Takeuchi O, Fujita T, et al. Lipopolysaccharide stimulates the MyD88-independent pathway and results in activation of IFN-regulatory factor 3 and the expression of a subset of lipopolysaccharide-inducible genes. J Immunol. 2001;167:5887–94. doi: 10.4049/jimmunol.167.10.5887. [DOI] [PubMed] [Google Scholar]
- 73.Meylan E, Burns K, Hofmann K, et al. RIP1 is an essential mediator of Toll-like receptor 3-induced NF-kappa B activation. Nat Immunol. 2004;5:503–7. doi: 10.1038/ni1061. [DOI] [PubMed] [Google Scholar]
- 74.Zarember KA, Godowski PJ. Tissue expression of human Toll-like receptors and differential regulation of Toll-like receptor mRNAs in leukocytes in response to microbes, their products, and cytokines. J Immunol. 2002;168:554–61. doi: 10.4049/jimmunol.168.2.554. [DOI] [PubMed] [Google Scholar]
- 75.Hornung V, Rothenfusser S, Britsch S, et al. Quantitative expression of Toll-like receptor 1–10 mRNA in cellular subsets of human peripheral blood mononuclear cells and sensitivity to CpG oligodeoxynucleotides. J Immunol. 2002;168:4531–7. doi: 10.4049/jimmunol.168.9.4531. [DOI] [PubMed] [Google Scholar]
- 76.Muzio M, Bosisio D, Polentarutti N, et al. Differential expression and regulation of Toll-like receptors (TLR) in human leukocytes: selective expression of TLR3 in dendritic cells. J Immunol. 2000;164:5998–6004. doi: 10.4049/jimmunol.164.11.5998. [DOI] [PubMed] [Google Scholar]
- 77.Wang ZM, Liu C, Dziarski R. Chemokines are the main proinflammatory mediators in human monocytes activated by Staphylococcus aureus, peptidoglycan, and endotoxin. J Biol Chem. 2000;275:20260–7. doi: 10.1074/jbc.M909168199. [DOI] [PubMed] [Google Scholar]
- 78.Ritter M, Mennerich D, Weith A, Seither P. Characterization of Toll-like receptors in primary lung epithelial cells: strong impact of the TLR3 ligand poly(I:C) on the regulation of Toll-like receptors, adaptor proteins and inflammatory response. J Inflamm. 2005;2:16. doi: 10.1186/1476-9255-2-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Kopydlowski KM, Salkowski CA, Cody MJ, et al. Regulation of macrophage chemokine expression by lipopolysaccharide in vitro and in vivo. J Immunol. 1999;163:1537–44. [PubMed] [Google Scholar]
- 80.Salkowski CA, Detore G, Franks A, Falk MC, Vogel SN. Pulmonary and hepatic gene expression following cecal ligation and puncture: monophosphoryl lipid A prophylaxis attenuates sepsis-induced cytokine and chemokine expression and neutrophil infiltration. Infect Immun. 1998;66:3569–78. doi: 10.1128/iai.66.8.3569-3578.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Rollins BJ. Chemokines. Blood. 1997;90:909–28. [PubMed] [Google Scholar]
- 82.Standiford TJ, Kunkel SL, Greenberger MJ, Laichalk LL, Strieter RM. Expression and regulation of chemokines in bacterial pneumonia. J Leukoc Biol. 1996;59:24–8. doi: 10.1002/jlb.59.1.24. [DOI] [PubMed] [Google Scholar]
- 83.Suzuki K, Suda T, Naito T, Ide K, Chida K, Nakamura H. Impaired toll-like receptor 9 expression in alveolar macrophages with no sensitivity to CpG DNA. Am J Respir Crit Care Med. 2005;171:707–13. doi: 10.1164/rccm.200408-1078OC. [DOI] [PubMed] [Google Scholar]
- 84.Hollingsworth JW, Chen BJ, Brass DM, et al. The critical role of hematopoietic cells in lipopolysaccharide-induced airway inflammation. Am J Respir Crit Care Med. 2005;171:806–13. doi: 10.1164/rccm.200407-953OC. [DOI] [PubMed] [Google Scholar]
- 85.Lotz M, Gutle D, Walther S, Menard S, Bogdan C, Hornef MW. Postnatal acquisition of endotoxin tolerance in intestinal epithelial cells. J Exp Med. 2006;203:973–84. doi: 10.1084/jem.20050625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Sabroe I, Prince LR, Dower SK, Walmsley SR, Chilvers ER, Whyte MK. What can we learn from highly purified neutrophils? Biochem Soc Trans. 2004;32:468–9. doi: 10.1042/BST0320468. [DOI] [PubMed] [Google Scholar]
- 87.Sabroe I, Prince LR, Jones EC, et al. Selective roles for Toll-like receptor (TLR) 2 and TLR4 in the regulation of neutrophil activation and life span. J Immunol. 2003;170:5268–75. doi: 10.4049/jimmunol.170.10.5268. [DOI] [PubMed] [Google Scholar]
- 88.Sabroe I, Jones EC, Whyte MK, Dower SK. Regulation of human neutrophil chemokine receptor expression and function by activation of Toll-like receptors 2 and 4. Immunology. 2005;115:90–8. doi: 10.1111/j.1365-2567.2005.02133.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Prince LR, Allen L, Jones EC, et al. The role of interleukin-1{beta} in direct and Toll-like receptor 4-mediated neutrophil activation and survival. Am J Pathol. 2004;165:1819–26. doi: 10.1016/s0002-9440(10)63437-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Sabroe I, Jones EC, Usher LR, Whyte MK, Dower SK. Toll-like receptor (TLR) 2 and TLR4 in human peripheral blood granulocytes: a critical role for monocytes in leukocyte lipopolysaccharide responses. J Immunol. 2002;168:4701–10. doi: 10.4049/jimmunol.168.9.4701. [DOI] [PubMed] [Google Scholar]
- 91.Meerschaert J, Busse WW, Bertics PJ, Mosher DF. CD14(+) cells are necessary for increased survival of eosinophils in response to lipopolysaccharide. Am J Respir Cell Mol Biol. 2000;23:780–7. doi: 10.1165/ajrcmb.23.6.4171. [DOI] [PubMed] [Google Scholar]
- 92.Bozza PT, Castro-Faria-Neto HC, Penido C, et al. Requirement for lymphocytes and resident macrophages in LPS-induced pleural eosinophil accumulation. J Leukoc Biol. 1994;56:151–8. doi: 10.1002/jlb.56.2.151. [DOI] [PubMed] [Google Scholar]
- 93.Penido C, Castro-Faria-Neto HC, Larangeira AP, et al. The role of gammadelta T lymphocytes in lipopolysaccharide-induced eosinophil accumulation into the mouse pleural cavity. J Immunol. 1997;159:853–60. [PubMed] [Google Scholar]
- 94.Pivarcsi A, Bodai L, Rethi B, et al. Expression and function of Toll-like receptors 2 and 4 in human keratinocytes. Int Immunol. 2003;15:721–30. doi: 10.1093/intimm/dxg068. [DOI] [PubMed] [Google Scholar]
- 95.Kollisch G, Kalali BN, Voelcker V, et al. Various members of the Toll-like receptor family contribute to the innate immune response of human epidermal keratinocytes. Immunology. 2005;114:531–41. doi: 10.1111/j.1365-2567.2005.02122.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Droemann D, Goldmann T, Branscheid D, et al. Toll-like receptor 2 is expressed by alveolar epithelial cells type II and macrophages in the human lung. Histochem Cell Biol. 2003;119:103–8. doi: 10.1007/s00418-003-0497-4. [DOI] [PubMed] [Google Scholar]
- 97.Guillot L, Medjane S, Le-Barillec K, et al. Response of human pulmonary epithelial cells to lipopolysaccharide involves Toll-like receptor 4 (TLR4)-dependent signaling pathways: evidence for an intracellular compartmentalization of TLR4. J Biol Chem. 2004;279:2712–18. doi: 10.1074/jbc.M305790200. [DOI] [PubMed] [Google Scholar]
- 98.Abreu MT, Fukata M, Arditi M. TLR signaling in the gut in health and disease. J Immunol. 2005;174:4453–60. doi: 10.4049/jimmunol.174.8.4453. [DOI] [PubMed] [Google Scholar]
- 99.Fazeli A, Bruce C, Anumba DO. Characterization of Toll-like receptors in the female reproductive tract in humans. Hum Reprod. 2005;20:1372–8. doi: 10.1093/humrep/deh775. [DOI] [PubMed] [Google Scholar]
- 100.Faure E, Equils O, Sieling PA, et al. Bacterial lipopolysaccharide activates NF-kappaB through Toll-like receptor 4 (TLR-4) in cultured human dermal endothelial cells. Differential expression of TLR-4 and TLR-2 in endothelial cells. J Biol Chem. 2000;275:11058–63. doi: 10.1074/jbc.275.15.11058. [DOI] [PubMed] [Google Scholar]
- 101.Tissari J, Siren J, Meri S, Julkunen I, Matikainen S. IFN-alpha enhances TLR3-mediated antiviral cytokine expression in human endothelial and epithelial cells by up-regulating TLR3 expression. J Immunol. 2005;174:4289–94. doi: 10.4049/jimmunol.174.7.4289. [DOI] [PubMed] [Google Scholar]
- 102.Li J, Ma Z, Tang ZL, Stevens T, Pitt B, Li S. CpG DNA-mediated immune response in pulmonary endothelial cells. Am J Physiol Lung Cell Mol Physiol. 2004;287:L552–8. doi: 10.1152/ajplung.00436.2003. [DOI] [PubMed] [Google Scholar]
- 103.Yang X, Coriolan D, Murthy V, Schultz K, Golenbock DT, Beasley D. Proinflammatory phenotype of vascular smooth muscle cells: role of efficient Toll-like receptor 4 signaling. Am J Physiol Heart Circ Physiol. 2005;289:H1069–76. doi: 10.1152/ajpheart.00143.2005. [DOI] [PubMed] [Google Scholar]
- 104.Morris GE, Whyte MK, Martin GF, Jose PJ, Dower SK, Sabroe I. Agonists of Toll-like receptors 2 and 4 activate airway smooth muscle via mononuclear leukocytes. Am J Respir Crit Care Med. 2005;171:814–22. doi: 10.1164/rccm.200403-406OC. [DOI] [PubMed] [Google Scholar]
- 105.Sauty A, Dziejman M, Taha RA, et al. The T cell-specific CXC chemokines IP-10, Mig, and I-TAC are expressed by activated human bronchial epithelial cells. J Immunol. 1999;162:3549–58. [PubMed] [Google Scholar]
- 106.Brightling CE, Kaur D, Berger P, Morgan AJ, Wardlaw AJ, Bradding P. Differential expression of CCR3 and CXCR3 by human lung and bone marrow-derived mast cells: implications for tissue mast cell migration. J Leukoc Biol. 2005;77:759–66. doi: 10.1189/jlb.0904511. [DOI] [PubMed] [Google Scholar]
- 107.Brightling CE, Ammit AJ, Kaur D, et al. The CXCL10/CXCR3 axis mediates human lung mast cell migration to asthmatic airway smooth muscle. Am J Respir Crit Care Med. 2005;171:1103–8. doi: 10.1164/rccm.200409-1220OC. [DOI] [PubMed] [Google Scholar]
- 108.Brightling CE, Bradding P, Symon FA, Holgate ST, Wardlaw AJ, Pavord ID. Mast-cell infiltration of airway smooth muscle in asthma. N Engl J Med. 2002;346:1699–705. doi: 10.1056/NEJMoa012705. [DOI] [PubMed] [Google Scholar]
- 109.Shan X, Hu A, Veler H, et al. Regulation of Toll-like receptor 4-induced pro-asthmatic changes in airway smooth muscle function by opposing actions of ERK1/2 and p38 MAPK signaling. Am J Physiol Lung Cell Mol Physiol. 291:L324–33. doi: 10.1152/ajplung.00056.2006. [DOI] [PubMed] [Google Scholar]
- 110.Liew FY, Komai-Koma M, Xu D. A toll for T cell costimulation. Ann Rheum Dis. 2004;63(Suppl. 2):ii76–8. doi: 10.1136/ard.2004.028308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Liu H, Komai-Koma M, Xu D, Liew FY. Toll-like receptor 2 signaling modulates the functions of CD4+ CD25+ regulatory T cells. Proc Natl Acad Sci USA. 2006;103:7048–53. doi: 10.1073/pnas.0601554103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Xu D, Komai-Koma M, Liew FY. Expression and function of Toll-like receptor on T cells. Cell Immunol. 2005;233:85–9. doi: 10.1016/j.cellimm.2005.04.019. [DOI] [PubMed] [Google Scholar]
- 113.Pasare C, Medzhitov R. Toll-like receptors and acquired immunity. Semin Immunol. 2004;16:23–6. doi: 10.1016/j.smim.2003.10.006. [DOI] [PubMed] [Google Scholar]
- 114.Iwasaki A, Medzhitov R. Toll-like receptor control of the adaptive immune responses. Nat Immunol. 2004;5:987–95. doi: 10.1038/ni1112. [DOI] [PubMed] [Google Scholar]
- 115.Hemmi H, Akira S. TLR signalling and the function of dendritic cells. Chem Immunol Allergy. 2005;86:120–35. doi: 10.1159/000086657. [DOI] [PubMed] [Google Scholar]
- 116.Banchereau J, Steinman RM. Dendritic cells and the control of immunity. Nature. 1998;392:245–52. doi: 10.1038/32588. [DOI] [PubMed] [Google Scholar]
- 117.Hoebe K, Janssen EM, Kim SO, et al. Upregulation of costimulatory molecules induced by lipopolysaccharide and double-stranded RNA occurs by Trif-dependent and Trif-independent pathways. Nat Immunol. 2003;4:1223–9. doi: 10.1038/ni1010. [DOI] [PubMed] [Google Scholar]
- 118.Honda K, Sakaguchi S, Nakajima C, et al. Selective contribution of IFN-alpha/beta signaling to the maturation of dendritic cells induced by double-stranded RNA or viral infection. Proc Natl Acad Sci USA. 2003;100:10872–7. doi: 10.1073/pnas.1934678100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Hoshino K, Kaisho T, Iwabe T, Takeuchi O, Akira S. Differential involvement of IFN-beta in Toll-like receptor-stimulated dendritic cell activation. Int Immunol. 2002;14:1225–31. doi: 10.1093/intimm/dxf089. [DOI] [PubMed] [Google Scholar]
- 120.Biron CA. Interferons alpha and beta as immune regulators − a new look. Immunity. 2001;14:661–4. doi: 10.1016/s1074-7613(01)00154-6. [DOI] [PubMed] [Google Scholar]
- 121.Chaudhuri N, Dower SK, Whyte MK, Sabroe I. Toll-like receptors and chronic lung disease. Clin Sci. 2005;109:125–33. doi: 10.1042/CS20050044. [DOI] [PubMed] [Google Scholar]
- 122.Oliver BG, Johnston SL, Baraket M, et al. Increased proinflammatory responses from asthmatic human airway smooth muscle cells in response to rhinovirus infection. Respir Res. 2006;7:71. doi: 10.1186/1465-9921-7-71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Liu AH. Something old, something new: indoor endotoxin, allergens and asthma. Paediatr Respir Rev. 2004;5(Suppl. A):S65–71. doi: 10.1016/s1526-0542(04)90013-9. [DOI] [PubMed] [Google Scholar]
- 124.Michel O, Kips J, Duchateau J, et al. Severity of asthma is related to endotoxin in house dust. Am J Respir Crit Care Med. 1996;154:1641–6. doi: 10.1164/ajrccm.154.6.8970348. [DOI] [PubMed] [Google Scholar]
- 125.Noulin N, Quesniaux VF, Schnyder-Candrian S, et al. Both hemopoietic and resident cells are required for MyD88-dependent pulmonary inflammatory response to inhaled endotoxin. J Immunol. 2005;175:6861–9. doi: 10.4049/jimmunol.175.10.6861. [DOI] [PubMed] [Google Scholar]
- 126.Borregaard N, Theilgaard-Monch K, Cowland JB, Stahle M, Sorensen OE. Neutrophils and keratinocytes in innate immunity − cooperative actions to provide antimicrobial defense at the right time and place. J Leukoc Biol. 2005;77:439–43. doi: 10.1189/jlb.0704381. [DOI] [PubMed] [Google Scholar]
- 127.Kagnoff MF, Eckmann L. Epithelial cells as sensors for microbial infection. J Clin Invest. 1997;100:6–10. doi: 10.1172/JCI119522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Hertz CJ, Wu Q, Porter EM, et al. Activation of Toll-like receptor 2 on human tracheobronchial epithelial cells induces the antimicrobial peptide human beta defensin-2. J Immunol. 2003;171:6820–6. doi: 10.4049/jimmunol.171.12.6820. [DOI] [PubMed] [Google Scholar]
- 129.Sorensen OE, Cowland JB, Theilgaard-Monch K, Liu L, Ganz T, Borregaard N. Wound healing and expression of antimicrobial peptides/polypeptides in human keratinocytes, a consequence of common growth factors. J Immunol. 2003;170:5583–9. doi: 10.4049/jimmunol.170.11.5583. [DOI] [PubMed] [Google Scholar]
- 130.van Wetering S, Tjabringa GS, Hiemstra PS. Interactions between neutrophil-derived antimicrobial peptides and airway epithelial cells. J Leukoc Biol. 2005;77:444–50. doi: 10.1189/jlb.0604367. [DOI] [PubMed] [Google Scholar]
- 131.Maus UA, Waelsch K, Kuziel WA, et al. Monocytes are potent facilitators of alveolar neutrophil emigration during lung inflammation: role of the CCL2-CCR2 axis. J Immunol. 2003;170:3273–8. doi: 10.4049/jimmunol.170.6.3273. [DOI] [PubMed] [Google Scholar]
- 132.Liu L, Roberts AA, Ganz T. By IL-1 signaling, monocyte-derived cells dramatically enhance the epidermal antimicrobial response to lipopolysaccharide. J Immunol. 2003;170:575–80. doi: 10.4049/jimmunol.170.1.575. [DOI] [PubMed] [Google Scholar]
- 133.Pioli PA, Weaver LK, Schaefer TM, Wright JA, Wira CR, Guyre PM. Lipopolysaccharide-induced IL-1beta production by human uterine macrophages up-regulates uterine epithelial cell expression of human beta-defensin 2. J Immunol. 2006;176:6647–55. doi: 10.4049/jimmunol.176.11.6647. [DOI] [PubMed] [Google Scholar]
- 134.Tsouknos A, Nash GB, Rainger GE. Monocytes initiate a cycle of leukocyte recruitment when cocultured with endothelial cells. Atherosclerosis. 2003;170:49–58. doi: 10.1016/s0021-9150(03)00288-0. [DOI] [PubMed] [Google Scholar]
- 135.Groskreutz DJ, Monick MM, Powers LS, Yarovinsky TO, Look DC, Hunninghake GW. Respiratory syncytial virus induces TLR3 protein and protein kinase R, leading to increased double-stranded RNA responsiveness in airway epithelial cells. J Immunol. 2006;176:1733–40. doi: 10.4049/jimmunol.176.3.1733. [DOI] [PubMed] [Google Scholar]
- 136.Monick MM, Yarovinsky TO, Powers LS, et al. Respiratory syncytial virus up-regulates TLR4 and sensitizes airway epithelial cells to endotoxin. J Biol Chem. 2003;278:53035–44. doi: 10.1074/jbc.M308093200. [DOI] [PubMed] [Google Scholar]
- 137.Guillot L, Le Goffic R, Bloch S, et al. Involvement of toll-like receptor 3 in the immune response of lung epithelial cells to double-stranded RNA and influenza A virus. J Biol Chem. 2005;280:5571–80. doi: 10.1074/jbc.M410592200. [DOI] [PubMed] [Google Scholar]