Abstract
Tuberculosis (TB) remains a leading infectious cause of mortality worldwide. While the overall prevalence of TB in the United States has declined in the general population, certain groups remain at high risk, including the homeless, those who are HIV seropositive, individuals with a history of alcohol or drug abuse, and immigrants from a country in which TB is endemic. Many recipients of psychiatric services possess 1 or more of these risk factors, and consequently TB may be overrepresented in this population. Conversely, psychiatric illness may develop subsequent to TB infection. Mood disorders seem to be particularly common in TB patients compared with those with other medical diagnoses. It is important that primary care physicians understand the high prevalence of mental illness in TB patients so that proper treatment provisions can be implemented. Likewise, it is important for psychiatrists to understand the clinical manifestations of TB so that when a patient presents with symptoms of TB proper precautions can be taken and appropriate referrals can be made. This article integrates information concerning mental illness in TB patients with diagnostic and treatment guidelines for TB. Brief suggestions are offered for the treatment of TB patients with comorbid mental illness.
Tuberculosis (TB) is the leading infectious cause of mortality in people over 5 years of age, accounting for 6% of deaths worldwide.1 The World Health Organization reported that one third of the world population is latently infected, and there are 8 to 10 million new active cases each year.2 In the United States, 4% to 6% of the population (10–15 million people) have latent infections.2 In more industrialized nations, the disease has declined in the general population, predominantly affecting specific subgroups, including patients receiving psychiatric services.
TB is particularly common among individuals with mood disorders. In 1 study,3 71 patients in a psychiatric day program were given tuberculin skin tests. Of the 20 patients with mood disorders, 6 patients (30%) had positive purified protein derivative of tuberculin (PPD) results, accounting for half of all positive findings in the total group. Comparatively, only 6 (14%) of 41 patients with psychotic disorders demonstrated a positive PPD result. A survey of 100 hospitalized TB patients in South Africa indicated that 68% had some degree of clinical depression: 22 patients were mildly depressed, 38 patients were moderately depressed, and 8 patients were severely depressed.4 Other studies5,6 have indicated that between 24% and 36% of medically ill people meet the criteria for depression. These findings seem to indicate that the link between depression and TB is stronger than that between depression and other medical illnesses.
Because of the frequent comorbidity of TB and mood disorders, it is important for primary care physicians who treat TB patients to be mindful of the clinical manifestations of depression. Also, because of the highly infectious nature of TB, psychiatrists should employ systematic screening methods and be aware of diagnostic and treatment considerations for this disease. As a reference for psychiatrists, issues in the diagnosis and management of TB are described below. Considerations for the treatment of comorbid TB and depression are specifically emphasized, providing clinically relevant information to primary care physicians as well as psychiatrists.
PREVALENCE OF TB AND MENTAL ILLNESS
While TB is considered to be more common among individuals with mood disorders as opposed to those with other psychiatric diagnoses, most studies do not provide specific information about psychiatric diagnoses. Thus, the majority of epidemiologic information is presented in general terms. In an analysis of 43 consecutive psychiatric admissions to San Francisco General Hospital (San Francisco, Calif.), 8 (19%) displayed positive PPD results.7 Conversely, in a study8 of 121 TB patients seen at Columbia Presbyterian Medical Center (New York, N.Y.), 27 (22%) required psychiatric consultation. Psychiatric patients are more prone to TB infection, since they are more likely to be exposed to risk factors for the disease.7 Many psychiatric patients are homeless or have unstable housing. These individuals may spend time in heavily populated shelters where TB infection is common and often untreated. Even when TB is diagnosed, homeless people frequently fail to comply with treatment, exacerbating the spread of the illness.8 In a study9 of mentally ill men in a New York City homeless shelter, 33 individuals (36.7%) indicated positive PPD status, 6 of whom were diagnosed with active TB. In the same study, all 12 men who were human immunodeficiency virus (HIV)-seropositive also showed positive PPD results. This indicated that individuals who are HIV-seropositive might be more susceptible to TB.
Another risk factor for TB is emigration from a country in which TB is endemic. In a study by McQuistion et al.3 that examined the prevalence of TB in psychiatric patients, 7 of 13 immigrants showed positive PPD results compared with 5 of 58 native-born Americans. Other factors commonly associated with TB infection include age over 32 years, male gender, prior psychiatric hospitalizations, injection drug use, alcohol abuse, known exposure to a person with active tubercular disease, and concurrent illness.3,10 Sanchez et al.10 devised a screening questionnaire to assess the likelihood of TB infection based on exposure to risk factors. Of a random sample of 187 patients seen by a psychiatric emergency service, 23 patients demonstrated abnormalities on chest radiographs consistent with pulmonary TB. Of these patients, 83% were older than 32 years, 61% were male, 48% were immigrants, 44% had prior psychiatric hospitalizations, 35% abused alcohol, 35% were homeless, and 17% admitted to intravenous drug use. While these figures did not reach statistical significance, there appeared to be a trend between various risk factors and TB infection.
PATHOGENESIS
The etiologic agent responsible for TB is a small, non–spore-forming bacillus known as Mycobacterium tuberculosis. M tuberculosis is an obligate aerobe; the tissues it affects are characterized by high oxygen density.
The effects of M tuberculosis directly stem from its actions on the immune system. During the immune response to TB, products are formed that lead to inflammatory illness and tissue destruction. The immune response is initiated when alveolar macrophages release T lymphocytes after engulfing tubercle bacilli. In hosts with more competent immunity, TB tends to be isolated in the lungs or other single sites. If the immune system is less robust, the disease tends to proliferate to extrapulmonary sites including kidneys, bones, or meninges.2 These sites serve as foci for subsequent reactivation of TB.
Transmission of TB occurs almost exclusively by the aerosolization of contaminated respiratory secretions and the subsequent inhalation of TB, causing nuclei to enter the lungs. Less frequently, TB is transmitted through aerosols generated by other means, such as soft tissue agitation.1,2 For infection to occur, the bacilli must travel to the alveoli and be taken in by macrophages.2 After reaching the lungs, some organisms may survive and spread to the lymph nodes, where cell-mediated immunity is further activated. This results in the formation of tubercles. In most cases, the bacilli are contained within the tubercles, although they may spread throughout the body via the thoracic duct.1 In extrapulmonary TB, involved tissues usually show giant cell granulomas with caseating necrosis.2
DIAGNOSIS
The symptoms most commonly associated with TB include fatigue, fever (generally in the range of 40°C–41°C [104.0–105.8°F]), weight loss, night sweats, and cough (initially dry, but later productive of sputum and blood).2,11 There are several laboratory tests used in the diagnosis of TB. Definitive diagnosis depends on the recovery of M tuberculosis from cultures or smears, or identification by a DNA probe. The presence of acid-fast bacilli (AFB) on a smear of sputum or a sputum culture positive for mycobacteria is insufficient to make a positive diagnosis. Fiberoptic bronchoscopy can be used for verification in patients suspected to have TB despite negative sputum smears. Another option is using DNA fingerprinting to identify individual strains of TB.11
The standard test used to screen patients for TB is the Mantoux test, which involves the intracutaneous injection of 0.1 mL of a PPD from filtrates of M tuberculosis into the forearm.12 After 48 to 72 hours, any induration, a firm elevated area, that appears at the skin test site should be measured. A width ≥ 5 mm is considered a positive result for patients with HIV, close contact with a TB-positive individual, or an abnormal chest radiograph suggestive of prior TB. A width of ≥ 10 mm is a positive result for patients not meeting the above criteria who are intravenous drug users or who are in high-prevalence groups (immigrants, long-term care facility residents, and persons in locally high-risk areas). An induration ≥ 15 mm is considered a positive result in all others.11 Diagnosis of newly infected persons in screening programs is determined by an increase in induration of ≥ 10 mm for people under 35 years of age and ≥ 15 mm for people 35 or older over the course of a 2-year period.1
Imaging techniques are also utilized in the diagnosis of TB. The chest radiograph is used to screen patients suspected to have active TB, including those with positive PPD results. In primary TB, parenchymal infiltrates may be found in any area of the lung. Specific abnormalities associated with primary TB include hilar and paratracheal lymph node enlargement and segmental atelectasis. Pleural effusion is common in adults. Ghon and Ranke complexes are indicative of healed primary TB.1 Ghon complexes are calcifications seen in pulmonary parenchyma and hilar nodes, wedge-shaped depressions on the mediastinal surface of the lung. Ranke complexes are small calcified nodules with an associated calcified lymph node.13 Classic radiographic abnormalities among reactivation patients include cavitary or noncavitary lesions in the upper lobe or superior segment of the lower lobe of the lungs, nodules, and pneumonic infiltrates.1,11
In HIV-infected patients, radiographic features tend to vary with the progression of the disease. In the early stages of HIV infection, radiographic features are similar to those of patients without HIV. Patients with late-stage HIV infection often display lower lung zone, diffuse, or miliary infiltrates; pleural effusions; and involvement of hilar and mediastinal lymph nodes.11
TREATMENT
The pharmacologic treatment of TB is well established, and patient adherence to drug regimens is critical in reducing the risk of resistance. However, compliance to treatment is sometimes lacking among patients, particularly those with comorbid psychiatric disorders. Patients with personality disorders may be particularly unlikely to see the necessity of treatment for their well-being as well as others'. Residential treatment and isolation are sometimes used for TB-positive psychiatric patients to limit the spread of the disease. While these practices may increase compliance, they may induce further emotional stress. Thus, the establishment of a support system and a stable housing situation is essential.4 Another common strategy is directly observed therapy (DOT). DOT involves the monitoring of a patient's adherence to a drug regimen by a health care worker. Using DOT has yielded a treatment completion rate of 90% as opposed to 78.6% to 87.6% in less intensive programs.14
The use of at least 2 drugs is recommended for the treatment of TB, since resistant mutants are formed at a rate of 1 in 106 bacillus by spontaneous unlinked chromosomal mutation.11,14,15 There are 5 standard first-line drugs generally prescribed for the treatment of TB (Table 1). Isoniazid and rifampin are commonly used as central treatment agents because of their superior bactericidal activity and low toxicity. Pyrazinamide is particularly effective in the rapid early reduction of the bacillary burden. Ethambutol is generally used to protect against drug resistance. Streptomycin currently has a limited role in TB, as it is administered by injection and is used only in severe cases.11
Table 1.
First-Line Agents Used in the Treatment of Tuberculosisa
There are several multidrug treatment regimens used for TB, among which 3 pharmacologic combinations are particularly popular. In 1 regimen, a combination of isoniazid, rifampin, pyrazinamide, and either ethambutol or streptomycin is used. Therapy may be administered daily or 2 to 3 times per week under direct observation. Ethambutol is generally favored over streptomycin, which penetrates poorly and acts mainly on extracellular sites. Streptomycin takes significantly longer to act, since at any moment 90% of tubercle bacilli are intracellular. Ethambutol or streptomycin can be discontinued if susceptibility to isoniazid or rifampin is documented. Pyrazinamide can be eliminated from the regimen after 8 weeks. For patients with HIV or patients receiving a protease inhibitor, rifabutin (150 mg/day) should be used instead of rifampin. The duration of therapy is a minimum of 6 months, and at least 3 months after sputum cultures convert to negative. The second option involves the daily usage of isoniazid, rifampin, pyrazinamide, and streptomycin or ethambutol for 2 weeks. Then, the same drugs are administered twice weekly, under direct supervision, for 6 weeks. Isoniazid and rifampin are then administered twice weekly for 16 weeks if susceptibility to these agents is documented. The third primary option involves directly observed thrice-weekly administration of isoniazid, rifampin, pyrazinamide, and ethambutol or streptomycin for 6 months.11,15
Additional treatment provisions may be necessary for certain subpopulations. HIV patients can use any of the 3 standard regimens, but should continue treatment for a minimum of 9 months and at least 6 months after culture conversion. Extending treatment to 9 months is also suggested for patients with extrapulmonary TB. If any of the regimens are failing, at least 2 new drugs should be added. Patients resistant to isoniazid or rifampin should be prescribed 5 or 6 drug regimens initially, which should be continued for 12 to 24 months after a negative sputum culture.11
Treatment of multidrug-resistant TB (MDRTB) has been particularly problematic. MDRTB generally refers to resistance to at least isoniazid and rifampin. Risk factors for MDRTB include previous treatment for TB, being born in high-incidence areas, having contact with MDRTB, or having cavitary lung disease. The effective treatment of MDRTB requires prompt recognition of the disease, rapid susceptibility results, and early administration of individualized retreatment regimens. Such regimens are usually based on a quinolone and an injectable agent such as aminoglycoside or capreomycin, which can be supplemented by other second-line drugs. Therapy sometimes lasts as long as 24 months. Initial treatment regimens, which typically include 3 to 4 agents, must be prescribed while awaiting susceptibility results. Weekly sputum examinations are generally the best indicator of whether the regimen has been successful. Some experts suggest withdrawing the weaker and more toxic drugs from the regimen when sputum conversion is obtained. The patient is then to continue using the 2 to 3 active, well-tolerated drugs for another 18 to 24 months.14 Others suggest continuing with the entire regimen to increase the chances of effectiveness.14
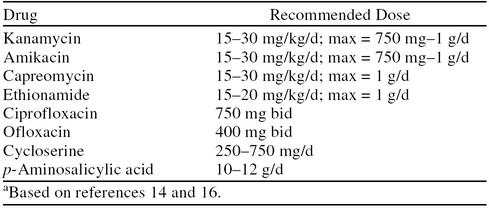
There are several second-line agents used in the treatment of MDRTB (Table 2). Kanamycin, amikacin, and capreomycin are injected intramuscularly and are effective against mycobacteria in concentrations available in vivo.14,16 Ethionamide exerts a bactericidal effect by inhibiting mycolic acid synthesis.14 Quinolones exhibit a mycobactericidal effect by inhibiting DNA synthesis. Newer broad-based quinolones tend to be more efficacious than narrow-spectrum fluoroquinolones. However, the fluoroquinolones ciprofloxacin and ofloxacin have been commonly prescribed.14 p-Aminosalicylic acid is effective in inhibiting most tubercle bacilli.14,16 Cycloserine competitively blocks the enzymes necessary for the synthesis of a dipeptide that is essential for the bacterial cell wall. The dosage is initially 250 mg/day for a few days, then 250 mg twice a day, and ultimately 750 mg/day.14
Table 2.
Second-Line Agents Used in the Treatment of Multidrug-Resistant Tuberculosisa
If intensive pharmacologic treatment of TB does not exert an effect after 4 to 8 months, it can probably be deemed unsuccessful. In some cases, resectional surgery has proved effective for certain patients who have failed in pharmacologic treatment. One study performed by Iseman and colleagues17 indicated that 23 of 29 patients who had received the procedure remained sputum culture negative for 6 to 69 months. However, the use of surgical procedures as a treatment for TB remains rare.
Preventative therapy (chemoprophylaxis) is sometimes administered to individuals at increased risk for TB. Among the groups who commonly receive chemoprophylaxis are persons with HIV, close contacts of persons with newly diagnosed infectious TB, recent skin test converters, and persons with medical conditions that increase the risk of TB. Preventative therapy is commonly administered in the form of isoniazid (300 mg/day for adults and 10–14 mg/kg/day for children for 6–12 months). Isoniazid can also be administered twice weekly in doses of 900 mg. If continued for 12 months, chemoprophylaxis may reduce expected incidence of TB by 93%. Another option in TB prevention is using 600 mg of rifampin daily or twice weekly.11 In some countries, bacille Calmette-Guérin (BCG) vaccination is used to prevent TB. The performance of BCG has ranged from 80% successful to having detrimental effects.2 Since BCG works only for patients who have not been previously infected with TB, it is not appropriate for widespread use in the United States.2 Moreover, BCG makes interpretation of the tuberculin skin test difficult, since BCG often produces results indicative of exposure. While BCG vaccination may cause PPD reactivity indicative of TB exposure for an extended period of time, this effect subsides within 10 years of vaccination in most patients.12,15
Emergency room management has been a major issue in the treatment of TB, particularly in the late 1980s and early 1990s, when TB increased dramatically in prevalence. In 1992, New York City had one of the largest populations of TB patients in the nation, with 3811 cases reported.18 A study by Stricof et al.18 examined the effectiveness of emergency TB treatment in 22 New York City facilities. The study indicated that the proportion of TB patients placed in AFB isolation increased from 75% in 1992 to 84% in 1994. The proportion of TB patients given a minimum of 4 anti-TB drugs increased from 88% in 1992 to 94% in 1994. Also, the proportion of medically discharged patients with 3 negative AFB smears increased significantly from 26% to 48%. Generally, the study indicated improvements in the quality of emergency treatment of TB offered in New York City. Although this improvement is not necessarily reflective of treatment in other facilities throughout the nation, it indicates the increased importance placed on isolation, treatment with multiple drugs, and follow-up therapy after negative test results.
The prognosis for TB patients is generally favorable, provided that patients do not have complications such as drug resistance or HIV. Most patients treated with 4-drug regimens test negative within 3 months of treatment initiation.19 Patients who have developed MDRTB have a less favorable prognosis. In trials conducted by the British Medical Research Council, of 8 patients who had MDRTB, 5 failed treatment and 2 relapsed. A subsequent study by Goble et al.20 indicated a 44% treatment failure rate among 134 evaluable patients. These results were contradicted in a study by Telzak et al.19 in which 24 of 25 MDRTB patients showed a clinical response to treatment. The difference between Telzak and colleagues' study and the previous experiments was that the subjects in Telzak and colleagues' study were earlier in the progression of TB. The key to successful treatment of MDRTB appears to be initiating therapy before physical deterioration and resistance to other drugs ensue.14
The prognosis for HIV patients with MDRTB is particularly bleak. MDRTB mortality rates have been estimated to be as high as 90% for HIV patients.14 A study by Fischl et al.21 compared 62 HIV-seropositive patients with MDRTB and 55 HIV-seropositive patients with single drug–resistant or susceptible TB. HIV-seropositive MDRTB patients had a median survival time of 2.1 months, while controls had a median survival time of 14.6 months.
TREATMENT CONCERNS FOR PATIENTS WITH COMORBID DEPRESSION AND TB
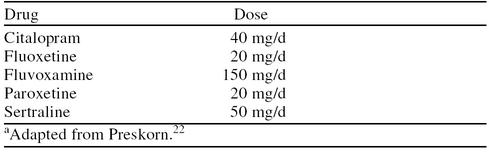
Currently, selective serotonin reuptake inhibitors (SSRIs) are recommended as the first-line treatment for depression (Table 3). SSRIs tend to be favored over other pharmacologic treatments for depression such as tricyclic antidepressants (TCAs) and monoamine oxidase inhibitors (MAOIs) because of their relatively benign side effect profiles. However, in patients comorbid for TB, concerns have been raised over the potential for drug interactions between various SSRIs and isoniazid. This concern is based on the ability of isoniazid to inhibit monoamine oxidase in plasma.23 Generally, the combination of SSRIs or TCAs with a drug that inhibits monoamine oxidase is contraindicated because of the potential to induce serotonin syndrome, characterized by excitation, diaphoresis, hyperthermia, myoclonus, rigidity, and hypertension.24 No reports of serotonin syndrome induced by combining SSRIs and isoniazid are published.
Table 3.
Standard Doses of Selective Serotonin Reuptake Inhibitorsa
Malek-Ahmadi et al.25 reported on 2 patients who received isoniazid in conjunction with antidepressants. The first patient was prescribed sertraline (150 mg/day) in combination with isoniazid (300 mg/day). The second patient received nefazodone (400 mg/day) and buspirone (10 mg/day) in conjunction with isoniazid (300 mg/day). Neither patient reported adverse effects. Judd et al.26 examined the combination of isoniazid and antidepressants in 3 HIV-seropositive male patients. Two of the 3 patients experienced symptoms including nausea, diarrhea, and fever while receiving treatment. However, the authors did not feel that these effects definitively resulted from an interaction between isoniazid and antidepressants. Judd et al. suggested that these symptoms more likely resulted solely from the antidepressant.
Evans and Kortas24 referred to accounts of people who consumed foods containing large amounts of tyramine, such as cheese, while taking isoniazid. In some cases, these patients reported flushing, headaches, and palpitations.27 These reactions were attributed to inhibition of monoamine oxidases and used as support for the possibility of adverse interactions between isoniazid and antidepressants.24 As Stockley28 indicated, there are other, more feasible explanations for this phenomenon, such as a histamine reaction resulting from the inhibition of histaminase by isoniazid. Currently, there is insufficient clinical evidence to definitively establish the potential for an adverse interaction between isoniazid and antidepressants.
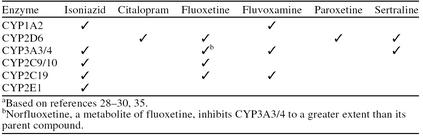
At the molecular level, there is evidence that isoniazid and SSRIs are metabolized by similar mechanisms. Hepatic cytochrome P450 (CYP) enzymes are largely responsible for metabolism of isoniazid, citalopram, fluoxetine, fluvoxamine, paroxetine, and sertraline. While it has not been definitively established which isoenzymes are implicated in the metabolism of isoniazid, CYP2E1, CYP1A2, CYP2C9, CYP2C19, and CYP3A are inhibited to varying degrees by isoniazid (Table 4).29,30 In a study employing human liver microsomes, Desta et al.30 indicated that CYP2C19 and CYP3A were inhibited potently by isoniazid in a concentration-dependent manner. Both enzymes were inhibited approximately 40% by doses in the therapeutic range. Desta et al. felt that inhibition of 1 or both of these enzymes slowed the elimination of coadministered drugs. This study also indicated that isoniazid induced competitive inhibition of CYP2D6 and weak noncompetitive inhibition of CYP2E1.
Table 4.
Cytochrome P450 (CYP) Enzymes Reported to Be Inhibited by Isoniazid and Selective Serotonin Reuptake Inhibitorsa
All SSRIs appear to be metabolized by cytochrome P450 enzymes; however, the pharmacokinetic interactions of each drug are variable (Table 4). As in the case of isoniazid, the metabolic pathways of SSRIs have not been firmly established. In vivo data have indicated that CYP2C19 is the major isoenzyme involved in the first step of citalopram metabolism,31,32 while an in vitro study implicated CYP3A4 as the primary isoenzyme with CYP2C19 having a secondary role.33 This discrepancy was accounted for by genetic differences in the populations studied. CYP2D6, which appears to be implicated in the second metabolic step, is mildly inhibited by citalopram. This was indicated in a study34 in which administration of levomepromazine, a potent inhibitor of CYP2D6, increased plasma levels of norcitalopram. While the primary isoenzyme responsible for metabolizing fluvoxamine has not been established, it is known to potently inhibit CYP1A2. Fluvoxamine also causes clinically meaningful inhibition of CYP2C19 and CYP3A3/4. The principal enzyme responsible for the metabolism of fluoxetine is unclear, but CYP2D6 and CYP3A3/4 are believed to be involved. Fluoxetine inhibits CYP2D6 and probably CYP2C9/10 significantly, and CYP3A3/4 and CYP2C19 to a lesser extent. The fluoxetine metabolite norfluoxetine is a more potent inhibitor of CYP3A3/4 than its parent compound and is approximately equal in magnitude for CYP2D6. Norfluoxetine also has an unusually long half-life, which increases the chances that it will be involved in drug interactions. Sertraline produces mild inhibition of CYP2D6, but seems to have little inhibitory effect on the other isoenzymes. However, in vitro conversion of sertraline to N-desmethylsertraline correlates more with CYP3A3/4 activity, although inhibition of this isoenzyme by sertraline appears to be weak. Paroxetine is principally metabolized by CYP2D6 as well as another cytochrome enzyme, which has not yet been specified. Paroxetine has a CYP2D6 inhibitory effect that is 2 to 4 times more potent than that of fluoxetine, 5 to 20 times more potent than that of sertraline, and also significantly more potent than those of citalopram or fluvoxamine.22
To date, the precise metabolic mechanisms of isoniazid and SSRIs have not been definitively established. However, the available literature indicates that there are some similarities in the metabolism of isoniazid and certain SSRIs.22,29,30,35 Although no substantial clinical evidence indicates that the combination of isoniazid and SSRIs is harmful, available evidence indicates that some SSRIs might be a better choice than others for concurrent treatment. Citalopram appears to be metabolized primarily by CYP2C19 and/or CYP3A4, both of which are inhibited by isoniazid, and thus it might not be the best choice for a patient taking isoniazid. Fluvoxamine is known to inhibit CYP1A2, CYP2C19, and possibly CYP3A3/4, all of which are also inhibited by isoniazid. Fluoxetine itself does not metabolically overlap isoniazid as much as other SSRIs; however, its metabolite norfluoxetine inhibits CYP3A3/4.22,35 Moreover, norfluoxetine has a half-life of 7 to 15 days, while the half-lives of other SSRIs are on the order of 1 day.22 Since fluoxetine and its metabolite remain in the system for a greater span of time, they have an increased potential for drug interactions. Sertraline might be a reasonable choice; however, there is still some question as to whether it inhibits CYP3A, which is implicated in the metabolism of isoniazid. According to available evidence, paroxetine appears to be the safest SSRI for use in conjunction with isoniazid. Paroxetine is metabolized primarily by CYP2D6, which is affected negligibly by isoniazid.22,35 Thus, the potential for drug interactions would appear to be minimal.
There have been historical instances in which drugs used to treat TB have been found to have effects on mental illness. For example, MAOIs, which were at one point a popular treatment for depression, were discovered during efforts to develop anti-TB drugs. There were various accounts of euphoria among patients receiving iproniazid after its introduction as a TB drug in 1952.36,37 Zeller et al.38 determined that iproniazid was an inhibitor of monoamine oxidase. Iproniazid was ultimately removed from the market because it commonly induced hepatotoxicity, but it led to the development of other MAOIs for antidepressant use.36 Later, attempts were made to reintroduce iproniazid as an antidepressant. Despite the improvement it induced in depressive symptoms and energy levels, iproniazid caused adverse effects including overactivity, insomnia, agitation, and paranoia trends.39 Ultimately, iproniazid was again abandoned due to its toxicity.37
More recently, cycloserine, a second-line agent in the treatment of TB, has received attention as a potential therapeutic agent for the negative symptoms of schizophrenia. Two placebo-controlled studies40,41 have indicated that cycloserine improves the negative symptoms of schizophrenia when used alone or with traditional neuroleptics. This improvement is thought to occur by the action of cycloserine on the glycine modulatory site of the N-methyl-d-aspartate receptors. The use of cycloserine alone does not appear to be a viable clinical option, since cycloserine is not believed to improve the positive symptoms of psychosis.41 Preliminary evidence has indicated that the appropriate dose of cycloserine for the treatment of schizophrenia is in the range of 50 to 100 mg/day.40–42 Larger doses of 250 mg/day and 1 g/day have been associated with anxiety, irritability, and depression.43,44 Cycloserine may be contraindicated in combination with clozapine. Two placebo-controlled trials45,46 in which cycloserine was used as an adjunct to clozapine indicated a worsening of negative symptoms when the 2 drugs were used in unison. Although further study is required, cycloserine appears to offer a moderate improvement in negative symptoms when used in a narrow dose range and with the appropriate antipsychotic agents.
CONCLUSION
In recent years, concerns have been raised about the concurrent treatment of TB and depression. Isoniazid remains a cornerstone in the treatment of TB, while SSRIs are widely regarded as the first-line treatment for depression. If these 2 types of drugs were to have adverse interactions, this would be a significant finding, since the co-occurrence of TB and depression is relatively common. To date, no clinical evidence has indicated that drug interactions exist between isoniazid and any SSRI. However, cytochrome P450 enzymes in the liver metabolize both drugs. Thus, it might be prudent for a clinician to be selective in prescribing an SSRI for a patient who is concurrently taking isoniazid. According to the evidence available, paroxetine appears to have the metabolic pathway that is the least similar to that of isoniazid and may be the best choice for the treatment of depression in a patient concurrently taking isoniazid.
Because of the frequent comorbidity of TB and depression, it is probable that primary care physicians treating TB will encounter undiagnosed cases of depression. Likewise, psychiatrists are likely to encounter TB among the mentally ill. Both primary care physicians and psychiatrists need to be familiar with the clinical features of each disease so that a proper diagnosis can be made and appropriate treatment and referrals will ensue.
Drug names: amikacin (Amikin), ciprofloxacin (Cipro), citalopram (Celexa), clozapine (Clozaril and others), ethambutol (Myambutol), ethionamide (Trecator-SC), fluoxetine (Prozac and others), fluvoxamine (Luvox and others), kanamycin (Kantrex), nefazodone (Serzone), ofloxacin (Floxin), paroxetine (Paxil), rifabutin (Mycobutin), rifampin (Rifadin), rifampin and isoniazid (Rifamate), rifampin, isoniazid, and pyrazinamide (Rifater), sertraline (Zoloft).
Footnotes
Mr. Trenton and Dr. Currier report no affiliation or relationship relevant to the subject matter in this article.
REFERENCES
- Poponick JM, Moll J. Tuberculosis. In: Tintinalli JE, Kelen GD, Stapczynski JS, eds. Emergency Medicine. New York, NY: McGraw-Hill. 2000 466–471. [Google Scholar]
- Iseman MD. Tuberculosis. In: Goldman LG, Bennett JC, eds. Cecil Textbook of Medicine. Philadelphia, Pa: WB Saunders Co. 2000 1723–1731. [Google Scholar]
- McQuistion HL, Colson P, Yankowitz R, et al. Tuberculosis infection among people with severe mental illness. Psychiatr Serv. 1997;48:833–835. doi: 10.1176/ps.48.6.833. [DOI] [PubMed] [Google Scholar]
- Westaway MS, Wolmarans L. Depression and self-esteem: rapid screening for depression in black, low literacy, hospitalized tuberculosis patients. Soc Sci Med. 1992;35:1311–1315. doi: 10.1016/0277-9536(92)90184-r. [DOI] [PubMed] [Google Scholar]
- Moffic HS, Paykel ES. Depression in medical in-patients. Br J Psychiatry. 1975;126:346–353. doi: 10.1192/bjp.126.4.346. [DOI] [PubMed] [Google Scholar]
- Cavanaugh SV. The prevalence of emotional and cognitive dysfunction in a general medical population: using the MMSE, GHQ, and BDI. Gen Hosp Psychiatry. 1983;5:15–24. doi: 10.1016/0163-8343(83)90038-5. [DOI] [PubMed] [Google Scholar]
- Lopez AG. Tuberculosis and the severely mentally ill [letter] Am J Psychiatry. 1994;151:151–152. doi: 10.1176/ajp.151.1.151b. [DOI] [PubMed] [Google Scholar]
- Fullilove MT, Young R, Panzer PG, et al. Psychosocial issues in the management of patients with tuberculosis. J Law Med Ethics. 1993;21:324–331. doi: 10.1111/j.1748-720x.1993.tb01257.x. [DOI] [PubMed] [Google Scholar]
- Saez H, Valencia E, Conover S, et al. Tuberculosis and HIV among mentally ill men in a New York City shelter. Am J Pub Health. 1996;86:1318–1319. doi: 10.2105/ajph.86.9.1318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanchez M, Nicholls T, Currier G. Risk factors for tuberculosis in the psychiatric emergency department. Emerg Psychiatry. 1998;4:33–34. [Google Scholar]
- Chesnutt MS, Prendergast TJ. Lung. In: Tierney LM, McPhee SJ, Papadakis MA, eds. Current Medical Diagnosis and Treatment. New York, NY: Lange Medical Books/McGraw-Hill. 2000 301–307. [Google Scholar]
- Friedman LN. Skin testing and chemoprophylaxis. In: Friedman LN, ed. Tuberculosis: Current Concepts and Treatment. 2nd ed. New York, NY: CRC Press. 2001 377–411. [Google Scholar]
- Curtis AM. Radiology of mycobacterial disease. In: Friedman LN, ed. Tuberculosis: Current Concepts and Treatment. 2nd ed. New York, NY: CRC Press. 2001 271–300. [Google Scholar]
- Bastian I, Colebunders R. Treatment and prevention of multidrug-resistant tuberculosis. Drugs. 1999;58:633–661. doi: 10.2165/00003495-199958040-00005. [DOI] [PubMed] [Google Scholar]
- Chambers HF. Mycobacterium tuberculosis infections. In: Tierney LM, McPhee SJ, Papadakis MA, eds. Current Medical Diagnosis and Treatment. New York, NY: Lange Medical Books/McGraw-Hill. 2000 1384–1386. [Google Scholar]
- Jacobs RA, Guglielmo J. Anti-infective chemotherapeutic and antibiotic agents. In: Tierney LM, McPhee SJ, Papadakis MA, eds. Current Medical Diagnosis and Treatment. New York, NY: Lange Medical Books/McGraw-Hill. 2000 1518–1521. [Google Scholar]
- Iseman MD, Madsen L, Goble M, et al. Surgical intervention in the treatment of pulmonary disease caused by drug-resistant mycobacterium tuberculosis. Am Rev Respir Dis. 1990;141:623–625. doi: 10.1164/ajrccm/141.3.623. [DOI] [PubMed] [Google Scholar]
- Stricof RL, DiFerdinando GT Jr, Osten WM, et al. Tuberculosis control in New York City hospitals. Am J Infect Control. 1998;26:270–276. doi: 10.1016/s0196-6553(98)80012-6. [DOI] [PubMed] [Google Scholar]
- Telzak EE, Sepkowitz K, Alpert P, et al. Multidrug-resistant tuberculosis in patients without HIV infection. N Engl J Med. 1995;333:907–911. doi: 10.1056/NEJM199510053331404. [DOI] [PubMed] [Google Scholar]
- Goble M, Iseman MD, Madsen LA, et al. Treatment of 171 patients with pulmonary tuberculosis resistant to isoniazid and rifampin. N Engl J Med. 1993;328:527–532. doi: 10.1056/NEJM199302253280802. [DOI] [PubMed] [Google Scholar]
- Fischl MA, Daikos GL, Uttamchandani RB, et al. Clinical presentation and outcome of patients with HIV infection and tuberculosis caused by multiple-drug-resistant bacilli. Ann Intern Med. 1992;117:184–190. doi: 10.7326/0003-4819-117-3-184. [DOI] [PubMed] [Google Scholar]
- Preskorn SH. Clinically relevant pharmacology of selective serotonin reuptake inhibitors. Clin Pharmacokinet. 1997;32(suppl 1):1–21. doi: 10.2165/00003088-199700321-00003. [DOI] [PubMed] [Google Scholar]
- Robinson DS, Lovenberg W, Keiser H, et al. Effects of drugs on human blood platelet and plasma amine oxidase activity in vitro and in vivo. Biochem Pharmacol. 1968;17:109–119. doi: 10.1016/0006-2952(68)90163-9. [DOI] [PubMed] [Google Scholar]
- Evans ME, Kortas KJ. Potential interaction between isoniazid and selective serotonin reuptake inhibitors. Am J Health Syst Pharm. 1995;52:2135–2136. doi: 10.1093/ajhp/52.19.2135. [DOI] [PubMed] [Google Scholar]
- Malek-Ahmadi P, Chavez M, Contreras SA. Coadministration of isoniazid and antidepressant drugs [letter] J Clin Psychiatry. 1996;57:550. doi: 10.4088/jcp.v57n1109g. [DOI] [PubMed] [Google Scholar]
- Judd FK, Mijch AM, Cockram A, et al. Isoniazid and antidepressants: is there cause for concern? Int Clin Psychopharmacol. 1994;9:123–125. [PubMed] [Google Scholar]
- Smith CK, Durack DT. Isoniazid and reaction to cheese. Ann Intern Med. 1978;88:520–521. doi: 10.7326/0003-4819-88-4-520. [DOI] [PubMed] [Google Scholar]
- Stockley IH. Lack of clinical evidence for potential interaction between isoniazid and selective serotonin-reuptake inhibitors [letter] Am J Health Syst Pharm. 1996;53:2217. doi: 10.1093/ajhp/53.18.2217. [DOI] [PubMed] [Google Scholar]
- Tseng A. AIDS/HIV: drugs for opportunistic infections. In: Piscitelli SC, Rodvold KA, eds. Drug Interactions in Infectious Diseases. Totowa, NJ: Humana Press. 2001 61–107. [Google Scholar]
- Desta Z, Soukhova NV, Flockhart DA. Inhibition of cytochrome P450 (CYP450) isoforms by isoniazid: potent inhibition of CYP2C19 and CYP3A. Antimicrob Agents Chemother. 2001;45:382–392. doi: 10.1128/AAC.45.2.382-392.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sindrup SH, Brosen K, Hansen MGJ, et al. Pharmacokinetics of citalopram in relation to the sparteine and the mephenytoin oxidation polymorphisms. Ther Drug Monit. 1993;15:11–17. doi: 10.1097/00007691-199302000-00002. [DOI] [PubMed] [Google Scholar]
- Baumann P, Nil R, Souche A, et al. A double-blind placebo-controlled study of citalopram with and without lithium in the treatment of therapy-resistant depressive patients: a clinical, pharamacokinetic, and pharmacogenic investigation. J Clin Psychopharmacol. 1996;16:307–314. doi: 10.1097/00004714-199608000-00006. [DOI] [PubMed] [Google Scholar]
- Kobayashi K, Chiba K, Yagi T, et al. Identification of cytochrome P450 isoforms involved in citalopram N-desmethylation by human liver microsomes. J Pharmacol Exp Ther. 1997;280:927–933. [PubMed] [Google Scholar]
- Gram LF, Hansen MGJ, Sindrup SH, et al. Citalopram: interaction studies with levopromazine, imipramine, and lithium. Ther Drug Monit. 1993;15:18–24. [PubMed] [Google Scholar]
- Caccia S. Metabolism of the newer antidepressants. Clin Pharmacokinet. 1998;34:281–302. doi: 10.2165/00003088-199834040-00002. [DOI] [PubMed] [Google Scholar]
- Rudorfer MV. Monoamine oxidase inhibitors: reversible and irreversible. Psychopharmacol Bull. 1992;28:45–57. [PubMed] [Google Scholar]
- Jacobsen E. The early history of psychotherapeutic drugs. Psychopharmacology (Berl) 1986;89:138–144. doi: 10.1007/BF00310617. [DOI] [PubMed] [Google Scholar]
- Zeller EA, Barsky J, Fouts JR, et al. Influence of isonicotinic acid hydrazide (INH) and 1-isonicotinyl-2-isopropyl hydrazide (IIH) on bacterial and mammalian enzymes. Experientia. 1952;8:349–350. [Google Scholar]
- Crane GE. Further studies on iproniazid phosphate. J Nerv Ment Dis. 1956;124:322–331. doi: 10.1097/00005053-195609000-00014. [DOI] [PubMed] [Google Scholar]
- Goff DC, Tsai G, Levitt J, et al. A placebo controlled trial of d-cycloserine added to conventional neuroleptics in patients with schizophrenia. Arch Gen Psychiatry. 1999;56:21–27. doi: 10.1001/archpsyc.56.1.21. [DOI] [PubMed] [Google Scholar]
- van Berckel BNM, Hijman R, van der Linden JA. Efficacy and tolerance of d-cycloserine in drug-free schizophrenic patients. Biol Psychiatry. 1996;40:1298–1300. doi: 10.1016/S0006-3223(96)00311-3. [DOI] [PubMed] [Google Scholar]
- Cascella NG, Macciardi F, Cavallini C, et al. d-cycloserine adjuvant therapy to conventional neuroleptic treatment in schizophrenia: an open-label study. J Neural Transm. 1993;95:105–111. doi: 10.1007/BF01276429. [DOI] [PubMed] [Google Scholar]
- Goff DC, Tsai G, Manoach DS, et al. Dose-finding trial of d-cycloserine added to neuroleptics for negative symptoms in schizophrenia. Am J Psychiatry. 1995;152:1213–1215. doi: 10.1176/ajp.152.8.1213. [DOI] [PubMed] [Google Scholar]
- Pasargiklian M, Biondi L. Neurologic and behavioural reactions of tuberculous patients treated with cycloserine. Scand J Respir Dis Suppl. 1970;71:201–208. [PubMed] [Google Scholar]
- Goff DC, Henderson DC, Evins AE, et al. A placebo-controlled crossover trial of d-cycloserine added to clozapine in patients with schizophrenia. Biol Psychiatry. 1999;45:512–514. doi: 10.1016/s0006-3223(98)00367-9. [DOI] [PubMed] [Google Scholar]
- Goff DC, Tsai G, Manoach DS, et al. d-cycloserine added to clozapine for patients with schizophrenia. Am J Psychiatry. 1996;153:1628–1630. doi: 10.1176/ajp.153.12.1628. [DOI] [PubMed] [Google Scholar]