Abstract
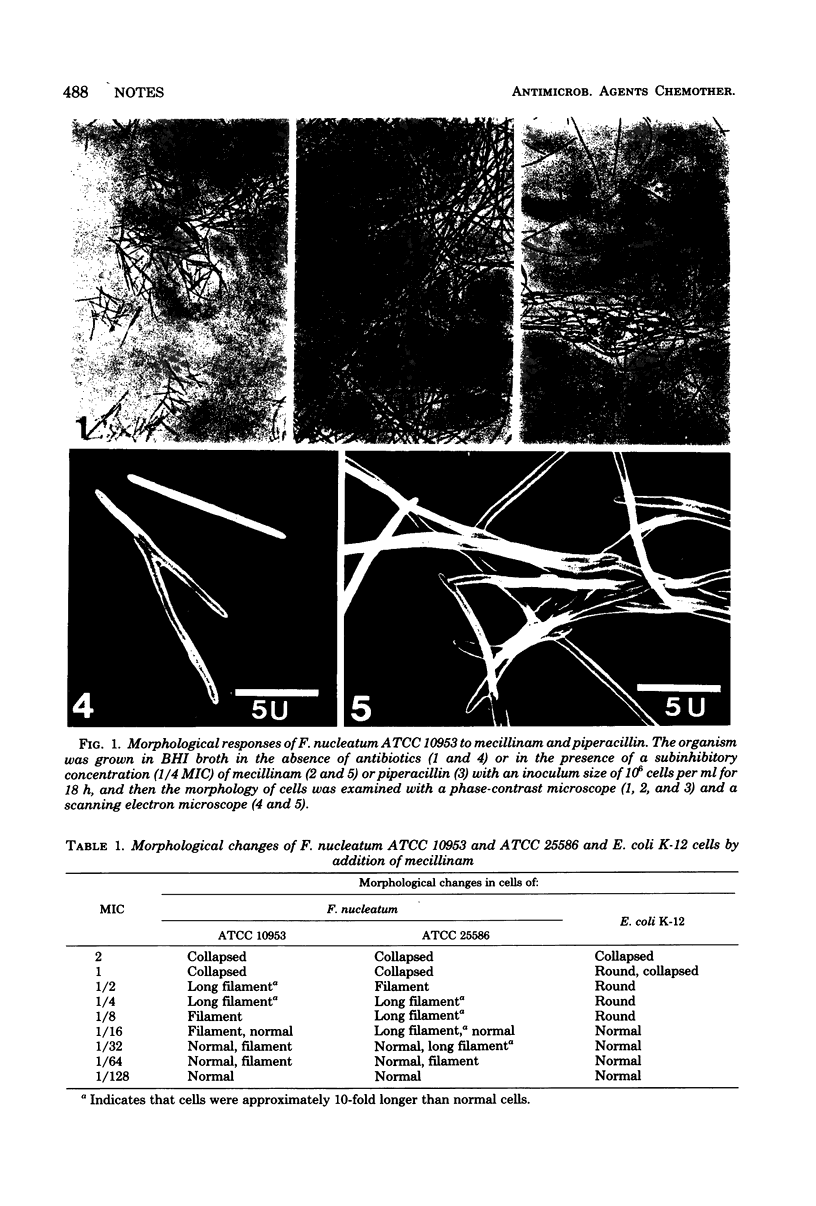
Subinhibitory concentrations of mecillinam transformed Fusobacterium nucleatum cells into a marked filament form, quite different from a spherical form demonstrated in Escherichia coli.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Chase H. A., Reynolds P. E., Ward J. B. Purification and characterization of the penicillin-binding protein that is the lethal target of penicillin in Bacillus megaterium and Bacillus licheniformis. Protein exchange and complex stability. Eur J Biochem. 1978 Jul 17;88(1):275–285. doi: 10.1111/j.1432-1033.1978.tb12448.x. [DOI] [PubMed] [Google Scholar]
- Coyette J., Ghuysen J. M., Fontana R. Solubilization and isolation of the membrane-bound DD-carboxypeptidase of Streptococcus faecalis ATCC9790. Properties of the purified enzyme. Eur J Biochem. 1978 Jul 17;88(1):297–305. doi: 10.1111/j.1432-1033.1978.tb12450.x. [DOI] [PubMed] [Google Scholar]
- Curtis N. A., Orr D., Ross G. W., Boulton M. G. Competition of beta-lactam antibiotics for the penicillin-binding proteins of Pseudomonas aeruginosa, Enterobacter cloacae, Klebsiella aerogenes, Proteus rettgeri, and Escherichia coli: comparison with antibacterial activity and effects upon bacterial morphology. Antimicrob Agents Chemother. 1979 Sep;16(3):325–328. doi: 10.1128/aac.16.3.325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greenwood D., O'Grady F. Antibiotic-induced surface changes in microorganisms demonstrated by scanning electron microscopy. Science. 1969 Mar 7;163(3871):1076–1078. doi: 10.1126/science.163.3871.1076. [DOI] [PubMed] [Google Scholar]
- Greenwood D., O'Grady F. FL 1060: a new beta-lactam antibiotic with novel properties. J Clin Pathol. 1973 Jan;26(1):1–6. doi: 10.1136/jcp.26.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iida K., Hirata S., Nakamuta S., Koike M. Inhibition of cell division of Escherichia coli by a new synthetic penicillin, piperacillin. Antimicrob Agents Chemother. 1978 Aug;14(2):257–266. doi: 10.1128/aac.14.2.257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kozarich J. W., Strominger J. L. A membrane enzyme from Staphylococcus aureus which catalyzes transpeptidase, carboxypeptidase, and penicillinase activities. J Biol Chem. 1978 Feb 25;253(4):1272–1278. [PubMed] [Google Scholar]
- Lederberg J. BACTERIAL PROTOPLASTS INDUCED BY PENICILLIN. Proc Natl Acad Sci U S A. 1956 Sep;42(9):574–577. doi: 10.1073/pnas.42.9.574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melchior N. H., Blom J., Tybring L., Birch-Andersen A. Light and electron microscopy of the early response of Escherichia coli to a 6beta-amidinopenicillanic acid (FL 1060). Acta Pathol Microbiol Scand B Microbiol Immunol. 1973 Aug;81(4):393–407. doi: 10.1111/j.1699-0463.1973.tb02222.x. [DOI] [PubMed] [Google Scholar]
- Muggleton P. W., O'Callaghan C. H., Foord R. D., Kirby S. M., Ryan D. M. Laoratory appraisal of cephalexin. Antimicrob Agents Chemother (Bethesda) 1968;8:353–360. [PubMed] [Google Scholar]
- Noguchi H., Matsuhashi M., Mitsuhashi S. Comparative studies of penicillin-binding proteins in Pseudomonas aeruginosa and Escherichia coli. Eur J Biochem. 1979 Oct;100(1):41–49. doi: 10.1111/j.1432-1033.1979.tb02031.x. [DOI] [PubMed] [Google Scholar]
- Ohya S., Yamazaki M., Sugawara S., Matsuhashi M. Penicillin-binding proteins in Proteus species. J Bacteriol. 1979 Jan;137(1):474–479. doi: 10.1128/jb.137.1.474-479.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reeves D. S., Wise R., Bywater M. J. A laboratory evaluation of a novel (beta-lactam antibiotic mecillinam. J Antimicrob Chemother. 1975 Sep;1(3):337–344. doi: 10.1093/jac/1.3.337. [DOI] [PubMed] [Google Scholar]
- Rodriguez W. J., Saz A. K. Differential binding of penicillin by membrane fractions from penicillin-susceptible and -resistant gonococci. Antimicrob Agents Chemother. 1978 Apr;13(4):589–597. doi: 10.1128/aac.13.4.589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shepherd S. T., Chase H. A., Reynolds P. E. The separation and properties of two penicillin-binding proteins from Salmonella typhimurium. Eur J Biochem. 1977 Sep;78(2):521–523. doi: 10.1111/j.1432-1033.1977.tb11765.x. [DOI] [PubMed] [Google Scholar]
- Spratt B. G. Distinct penicillin binding proteins involved in the division, elongation, and shape of Escherichia coli K12. Proc Natl Acad Sci U S A. 1975 Aug;72(8):2999–3003. doi: 10.1073/pnas.72.8.2999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suginaka H., Blumberg P. M., Strominger J. L. Multiple penicillin-binding components in Bacillus subtilis, Bacillus cereus, Staphylococcus aureus, and Escherichia coli. J Biol Chem. 1972 Sep 10;247(17):5279–5288. [PubMed] [Google Scholar]



