Abstract
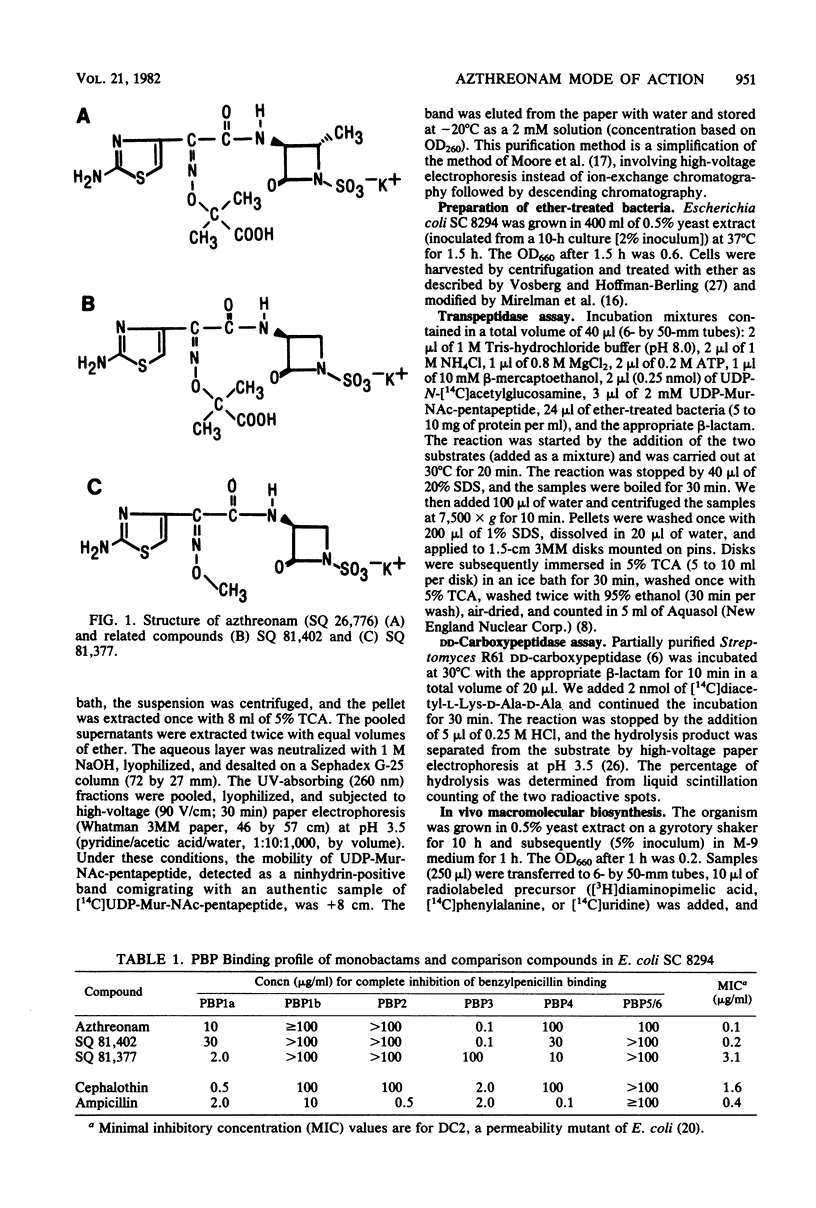
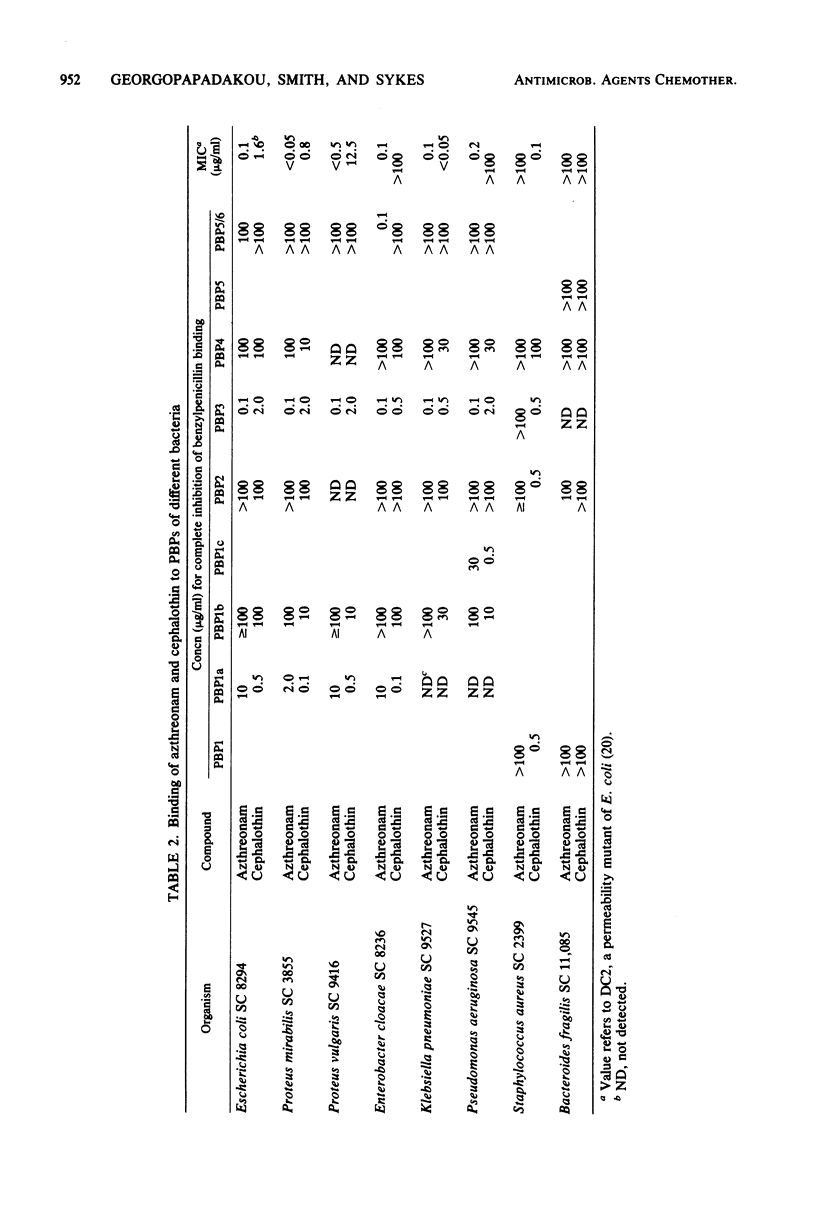
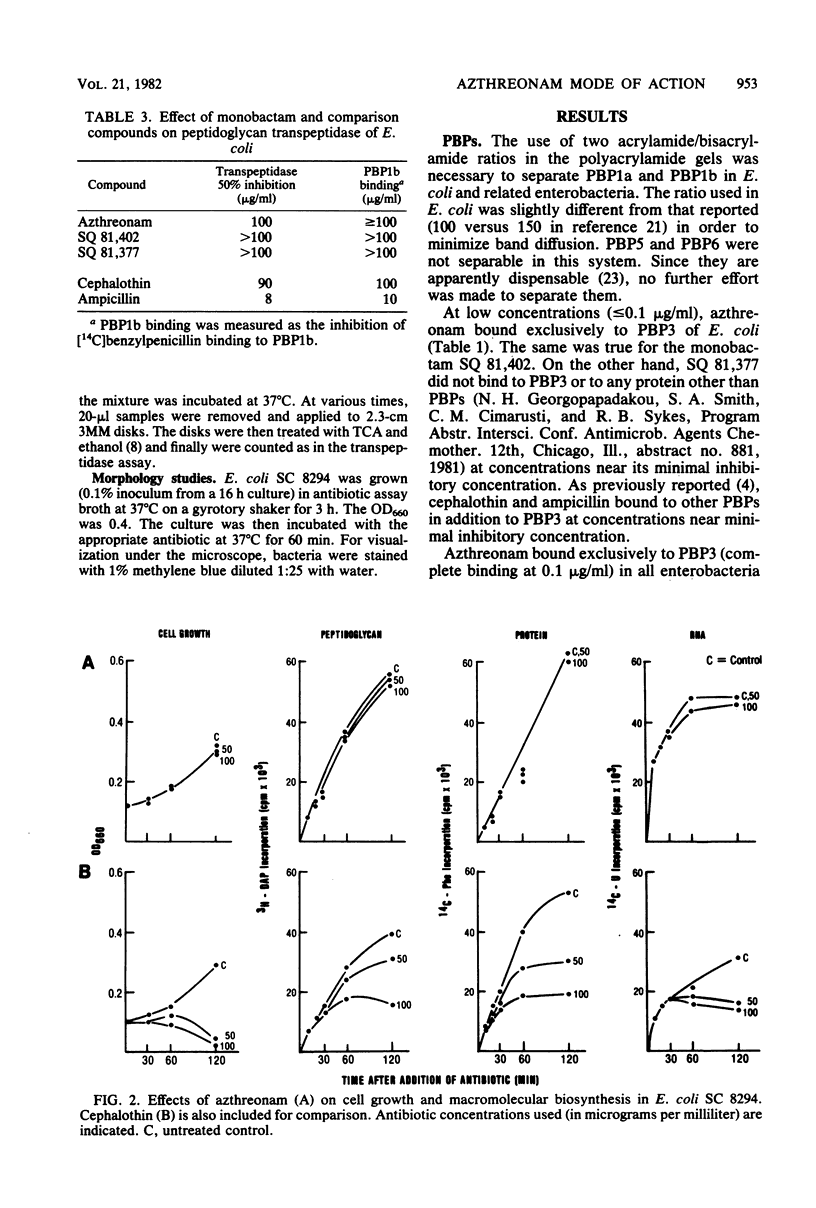
Azthreonam (SQ 26,776) is a member of a new class of monocyclic beta-lactam antibiotics. In Escherichia coli, azthreonam caused filamentation at its lowest effective concentration (0.2 microgram/ml), a morphological effect identical to that observed with cephalothin. The penicillin-binding protein (PBP) profile indicated a very high affinity for PBP3 (complete binding at 0.1 microgram/ml), a moderate affinity for PBP1a (complete binding at 10 micrograms/ml), and poor affinities for PBP1b, PBP2, PBP4, and PBP5/6 (complete binding at greater than or equal to 100 micrograms/ml). Accordingly, azthreonam had poor activity against Streptomyces R61 DD-carboxypeptidase (50% inhibition, greater than 100 micrograms/ml) and E. coli peptidoglycan transpeptidase (50% inhibition, 100 micrograms/ml). Azthreonam also showed very high affinity for PBP3 (complete binding at 0.1 microgram/ml) in Proteus vulgaris, Enterobacter cloacae, Klebsiella pneumoniae, and Pseudomonas aeruginosa. In all four organisms, its PBP profile was similar to that observed in E. coli. It is concluded that azthreonam, although of novel structure, has a mode of action similar to that of cephalosporins, affecting specifically septation in E. coli and most likely other gram-negative bacteria.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bonner D. P., Whitney R. R., Baughn C. O., Miller B. H., Olsen S. J., Sykes R. B. In-vivo properties of SQ 26,776. J Antimicrob Chemother. 1981 Dec;8 (Suppl E):123–130. doi: 10.1093/jac/8.suppl_e.123. [DOI] [PubMed] [Google Scholar]
- Breuer H., Cimarusti C. M., Denzel T., Koster W. H., Slusarchyk W. A., Treuner U. D. Monobactams--structure-activity relationships leading to SQ 26,776. J Antimicrob Chemother. 1981 Dec;8 (Suppl E):21–28. doi: 10.1093/jac/8.suppl_e.21. [DOI] [PubMed] [Google Scholar]
- Chase H. A., Fuller C., Reynolds P. E. The role of penicillin-proteins in the action of cephalosporins against Escherichia coli and Salmonella typhimurium. Eur J Biochem. 1981 Jul;117(2):301–310. doi: 10.1111/j.1432-1033.1981.tb06337.x. [DOI] [PubMed] [Google Scholar]
- Curtis N. A., Orr D., Ross G. W., Boulton M. G. Affinities of penicillins and cephalosporins for the penicillin-binding proteins of Escherichia coli K-12 and their antibacterial activity. Antimicrob Agents Chemother. 1979 Nov;16(5):533–539. doi: 10.1128/aac.16.5.533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frère J. M., Ghuysen J. M., Perkins H. R., Nieto M. Molecular weight and amino acid composition of the exocellular DD-carboxypeptidase-transpeptidase of Streptomyces R61. Biochem J. 1973 Nov;135(3):463–468. doi: 10.1042/bj1350463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Georgopapadakou N. H., Liu F. Y. Penicillin-binding proteins in bacteria. Antimicrob Agents Chemother. 1980 Jul;18(1):148–157. doi: 10.1128/aac.18.1.148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishino F., Matsuhashi M. Peptidoglycan synthetic enzyme activities of highly purified penicillin-binding protein 3 in Escherichia coli: a septum-forming reaction sequence. Biochem Biophys Res Commun. 1981 Aug 14;101(3):905–911. doi: 10.1016/0006-291x(81)91835-0. [DOI] [PubMed] [Google Scholar]
- Ishino F., Mitsui K., Tamaki S., Matsuhashi M. Dual enzyme activities of cell wall peptidoglycan synthesis, peptidoglycan transglycosylase and penicillin-sensitive transpeptidase, in purified preparations of Escherichia coli penicillin-binding protein 1A. Biochem Biophys Res Commun. 1980 Nov 17;97(1):287–293. doi: 10.1016/s0006-291x(80)80166-5. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laskey R. A., Mills A. D. Quantitative film detection of 3H and 14C in polyacrylamide gels by fluorography. Eur J Biochem. 1975 Aug 15;56(2):335–341. doi: 10.1111/j.1432-1033.1975.tb02238.x. [DOI] [PubMed] [Google Scholar]
- Livermore D. M., Williams J. D. In-vitro activity of the monobactam, SQ 26,776, against Gram-negative bacteria and its stability to their beta-lactamases. J Antimicrob Chemother. 1981 Dec;8 (Suppl E):29–37. doi: 10.1093/jac/8.suppl_e.29. [DOI] [PubMed] [Google Scholar]
- Lugtenberg E. J., v Schijndel-van Dam A., van Bellegem T. H. In vivo and in vitro action of new antibiotics interfering with the utilization of N-acetyl-glucosamine-N-acetyl-muramyl-pentapeptide. J Bacteriol. 1971 Oct;108(1):20–29. doi: 10.1128/jb.108.1.20-29.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mirelman D., Yashouv-Gan Y., Schwarz U. Peptidoglycan biosynthesis in a thermosensitive division mutant of Escherichia coli. Biochemistry. 1976 May 4;15(9):1781–1790. doi: 10.1021/bi00654a001. [DOI] [PubMed] [Google Scholar]
- Moore B. A., Jevons S., Brammer K. W. Peptidoglycan transpeptidase inhibition in Pseudomonas aeruginosa and Escherichia coli by Penicillins and Cephalosporins. Antimicrob Agents Chemother. 1979 Apr;15(4):513–517. doi: 10.1128/aac.15.4.513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Norrby R., Friberg K., Holm S. E. In-vitro antibacterial activity of SQ 26,776. J Antimicrob Chemother. 1981 Dec;8 (Suppl E):69–76. doi: 10.1093/jac/8.suppl_e.69. [DOI] [PubMed] [Google Scholar]
- Perkins H. R., Nieto M., Frére J. M., Leyh-Bouille M., Ghuysen J. M. Streptomyces DD-carboxypeptidases as transpeptidases. The specificity for amino compounds acting as carboxyl acceptors. Biochem J. 1973 Apr;131(4):707–718. doi: 10.1042/bj1310707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richmond M. H., Clark D. C., Wotton S. Indirect method for assessing the penetration of beta-lactamase-nonsusceptible penicillins and cephalosporins in Escherichia coli strains. Antimicrob Agents Chemother. 1976 Aug;10(2):215–218. doi: 10.1128/aac.10.2.215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shah P. M., Losert-Brüggner B., Stille W. Bactericidal activity of SQ 26776. J Antimicrob Chemother. 1981 Dec;8 (Suppl E):77–80. doi: 10.1093/jac/8.suppl_e.77. [DOI] [PubMed] [Google Scholar]
- Spratt B. G., Jobanputra V. Mutants of Escherichia coli which lack a component of penicillin-binding protein 1 are viable. FEBS Lett. 1977 Jul 15;79(2):374–378. doi: 10.1016/0014-5793(77)80824-7. [DOI] [PubMed] [Google Scholar]
- Suzuki H., Nishimura Y., Hirota Y. On the process of cellular division in Escherichia coli: a series of mutants of E. coli altered in the penicillin-binding proteins. Proc Natl Acad Sci U S A. 1978 Feb;75(2):664–668. doi: 10.1073/pnas.75.2.664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sykes R. B., Bonner D. P., Bush K., Georgopapadakou N. H. Azthreonam (SQ 26,776), a synthetic monobactam specifically active against aerobic gram-negative bacteria. Antimicrob Agents Chemother. 1982 Jan;21(1):85–92. doi: 10.1128/aac.21.1.85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sykes R. B., Cimarusti C. M., Bonner D. P., Bush K., Floyd D. M., Georgopapadakou N. H., Koster W. M., Liu W. C., Parker W. L., Principe P. A. Monocyclic beta-lactam antibiotics produced by bacteria. Nature. 1981 Jun 11;291(5815):489–491. doi: 10.1038/291489a0. [DOI] [PubMed] [Google Scholar]
- Tamura T., Imae Y., Strominger J. L. Purification to homogeneity and properties of two D-alanine carboxypeptidases I From Escherichia coli. J Biol Chem. 1976 Jan 25;251(2):414–423. [PubMed] [Google Scholar]
- Vosberg H. P., Hoffmann-Berling H. DNA synthesis in nucleotide-permeable Escherichia coli cells. I. Preparation and properties of ether-treated cells. J Mol Biol. 1971 Jun 28;58(3):739–753. doi: 10.1016/0022-2836(71)90037-4. [DOI] [PubMed] [Google Scholar]