Abstract
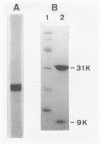
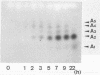
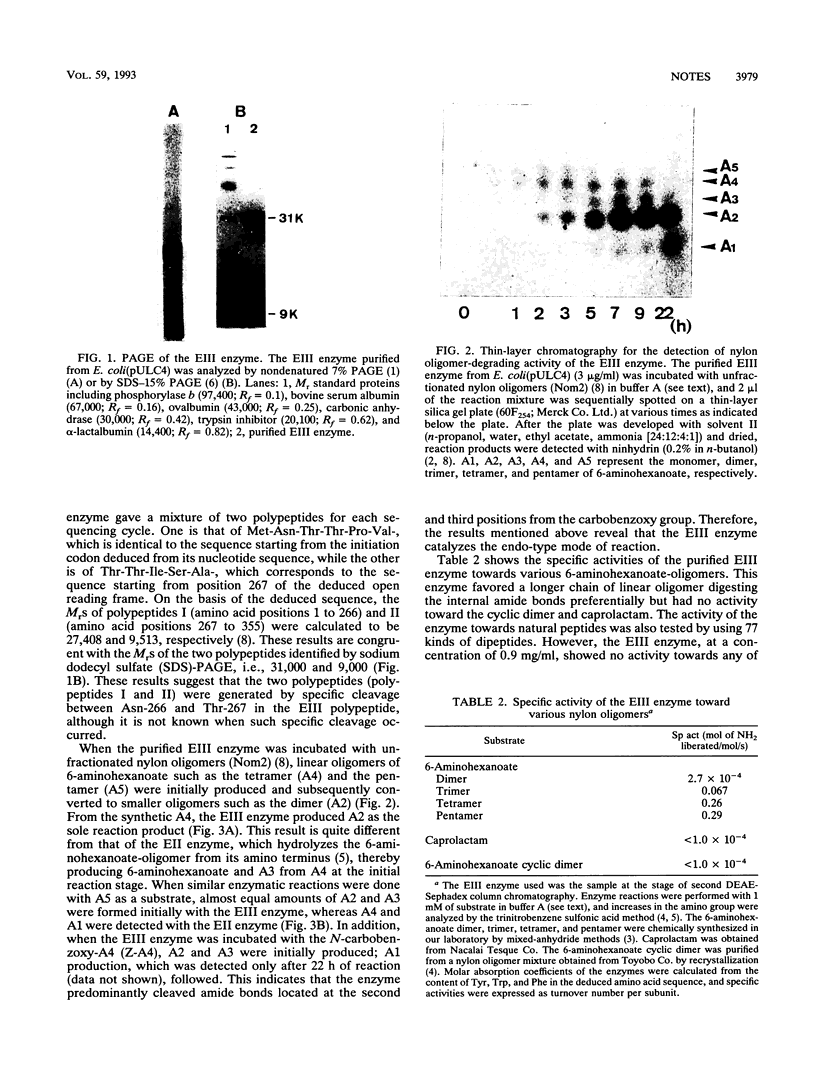
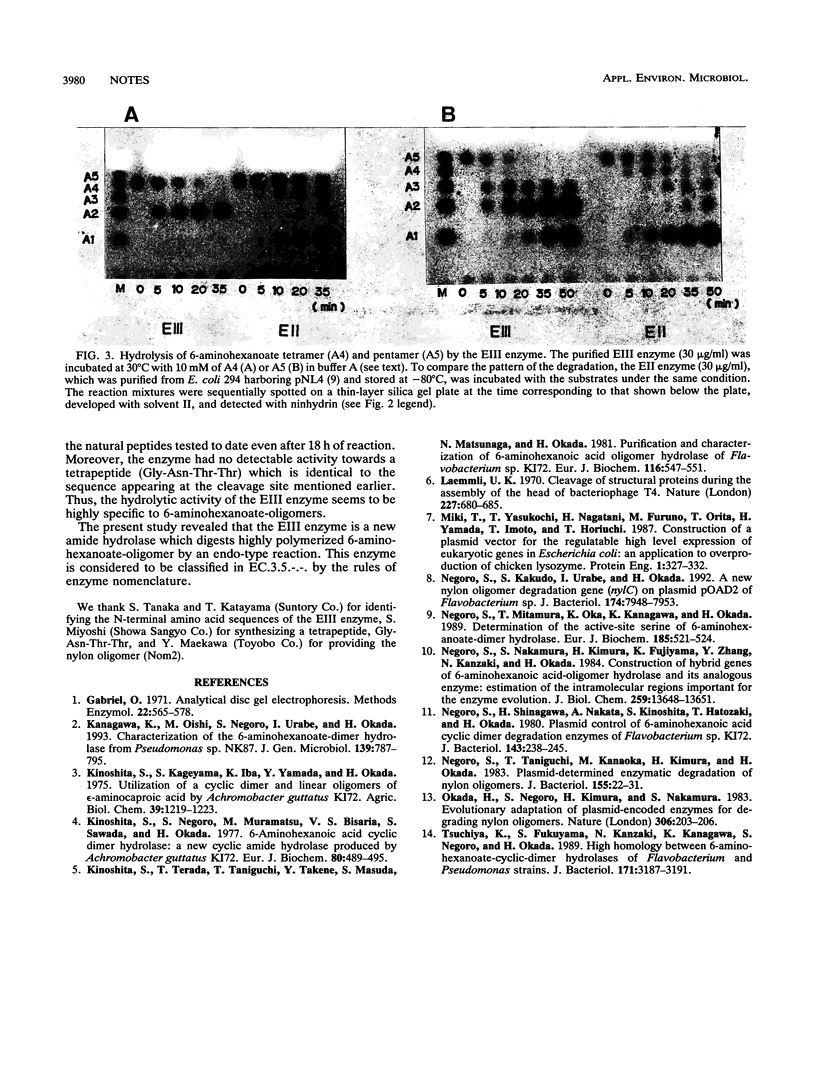
A new type of nylon oligomer degradation enzyme (EIII) was purified from an Escherichia coli clone harboring the EIII gene (nylC). This enzyme hydrolyzed the linear trimer, tetramer, and pentamer of 6-aminohexanoate by an endo-type reaction, and this specificity is different from that of the EI (nylA gene product) and EII (nylB gene product). Amino acid sequencing and sodium dodecyl sulfate-polyacrylamide gel electrophoresis of the purified EIII demonstrated that the enzyme is made of two polypeptide chains arising from an internal cleavage between amino acid residues 266 and 267.
Full text
PDF
Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Kanagawa K., Oishi M., Negoro S., Urabe I., Okada H. Characterization of the 6-aminohexanoate-dimer hydrolase from Pseudomonas sp. NK87. J Gen Microbiol. 1993 Apr;139(4):787–795. doi: 10.1099/00221287-139-4-787. [DOI] [PubMed] [Google Scholar]
- Kinoshita S., Negoro S., Muramatsu M., Bisaria V. S., Sawada S., Okada H. 6-Aminohexanoic acid cyclic dimer hydrolase. A new cyclic amide hydrolase produced by Achromobacter guttatus KI74. Eur J Biochem. 1977 Nov 1;80(2):489–495. doi: 10.1111/j.1432-1033.1977.tb11904.x. [DOI] [PubMed] [Google Scholar]
- Kinoshita S., Terada T., Taniguchi T., Takene Y., Masuda S., Matsunaga N., Okada H. Purification and characterization of 6-aminohexanoic-acid-oligomer hydrolase of Flavobacterium sp. Ki72. Eur J Biochem. 1981 Jun 1;116(3):547–551. doi: 10.1111/j.1432-1033.1981.tb05371.x. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Miki T., Yasukochi T., Nagatani H., Furuno M., Orita T., Yamada H., Imoto T., Horiuchi T. Construction of a plasmid vector for the regulatable high level expression of eukaryotic genes in Escherichia coli: an application to overproduction of chicken lysozyme. Protein Eng. 1987 Aug-Sep;1(4):327–332. doi: 10.1093/protein/1.4.327. [DOI] [PubMed] [Google Scholar]
- Negoro S., Kakudo S., Urabe I., Okada H. A new nylon oligomer degradation gene (nylC) on plasmid pOAD2 from a Flavobacterium sp. J Bacteriol. 1992 Dec;174(24):7948–7953. doi: 10.1128/jb.174.24.7948-7953.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Negoro S., Mitamura T., Oka K., Kanagawa K., Okada H. Determination of the active-site serine of 6-aminohexanoate-dimer hydrolase. Eur J Biochem. 1989 Nov 20;185(3):521–524. doi: 10.1111/j.1432-1033.1989.tb15144.x. [DOI] [PubMed] [Google Scholar]
- Negoro S., Nakamura S., Kimura H., Fujiyama K., Zhang Y. Z., Kanzaki N., Okada H. Construction of hybrid genes of 6-aminohexanoic acid-oligomer hydrolase and its analogous enzyme. Estimation of the intramolecular regions important for the enzyme evolution. J Biol Chem. 1984 Nov 25;259(22):13648–13651. [PubMed] [Google Scholar]
- Negoro S., Shinagawa H., Nakata A., Kinoshita S., Hatozaki T., Okada H. Plasmid control of 6-aminohexanoic acid cyclic dimer degradation enzymes of Flavobacterium sp. KI72. J Bacteriol. 1980 Jul;143(1):238–245. doi: 10.1128/jb.143.1.238-245.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Negoro S., Taniguchi T., Kanaoka M., Kimura H., Okada H. Plasmid-determined enzymatic degradation of nylon oligomers. J Bacteriol. 1983 Jul;155(1):22–31. doi: 10.1128/jb.155.1.22-31.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okada H., Negoro S., Kimura H., Nakamura S. Evolutionary adaptation of plasmid-encoded enzymes for degrading nylon oligomers. Nature. 1983 Nov 10;306(5939):203–206. doi: 10.1038/306203a0. [DOI] [PubMed] [Google Scholar]
- Tsuchiya K., Fukuyama S., Kanzaki N., Kanagawa K., Negoro S., Okada H. High homology between 6-aminohexanoate-cyclic-dimer hydrolases of Flavobacterium and Pseudomonas strains. J Bacteriol. 1989 Jun;171(6):3187–3191. doi: 10.1128/jb.171.6.3187-3191.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]