Abstract
The genetic structures (ca. 10-kb DNA fragment) surrounding the plasmid-borne extended-spectrum β-lactamase blaCTX-M-19 gene in a Klebsiella pneumoniae clinical isolate were determined. This β-lactamase gene was part of a 4,797-bp transposon inserted inside orf1 of Tn1721. Inside this transposon, blaCTX-M-19 was bracketed upstream and downstream by insertion sequences ISE cp1B and IS903D, respectively, and further downstream by a truncated gene encoding an outer membrane protein for iron transport. The single-copy ISEcp1B element was probably involved alone in the mobilization process that led to a 5-bp duplication at the target site of the transposed fragment. This mobilization event probably involved one inverted repeat of ISE cp1B and a second sequence farther away, resembling its second inverted repeat. Additionally, ISEcp1B provided −35 and −10 promoter sequences, contributing to the high-level expression of the blaCTX-M-19 gene. Southern blot analysis failed to identify a reservoir of ISEcp1-like sequences among a series of gram-negative and gram-positive bacterial species usually found in the skin and intestinal human floras. The ability of ISEcp1-like elements to mobilize and to promote the expression of β-lactamase genes may explain, in part, the current spread of CTX-M-type enzymes worldwide.
The clavulanic acid-inhibited Ambler class A extended-spectrum β-lactamases (ESBLs) of the CTX-M type are increasingly reported worldwide in members of the Enterobacteriaceae (2, 5, 9, 10, 16-18, 25, 39, 42). They may be classified according to their amino acid identity in four groups: CTX-M-1 (CTX-M-1, CTX-M-3, CTX-M-15,…), CTX-M-2 (CTX-M-2, CTX-M-4, …), CTX-M-8, and CTX-M-9 (CTX-M-9, CTX-M-14/-18, CTX-M-16, CTX-M-17, CTX-M-19, …) (42). It is known now that the progenitors of several plasmid-located CTX-M-type genes may be enterobacterial species such as Kluyvera cryocrescens (14), Kluyvera georgiana (36), and Kluyvera ascorbata (24), which are rarely a source of human infections and belong to the human intestinal flora. A recent spread of these plasmid-located ESBL genes may threaten further the clinical efficacy of expanded-spectrum cephalosporins and raises the question of which genetic background(s) may have contributed to their selection in recent years.
Although rare ESBL genes are associated with class 1 integrons (that enhance their expression [37, 38]), integron location of CTX-M genes has not been reported. Insertion sequences (IS) are also an important source of genetic plasticity in prokaryotes. In the case of β-lactamase genes, association with IS elements and transposons has been found mostly in Enterobacteriaceae and Pseudomonas aeruginosa. Tn1/Tn3 and Tn21 derivatives are associated with genes encoding narrow-spectrum β-lactamases such as TEM-1/TEM-2 and oxacillinases, respectively (4, 28) whereas IS26 and IS1-like elements are associated with genes encoding the ESBLs SHV-2a, TEM-3, and TEM-6 (19, 33).
We have published part of the sequence of an IS element (ISEcp1-like element) that was associated with blaCTX-M-15 identified in several enterobacterial isolates from India (27). Subsequently, several reports identified ISEcp1-like sequences upstream of other blaCTX-M-type genes (5, 9, 10). These results raise the question of the role of ISEcp1-like sequences in the spread and expression of blaCTX-M genes.
Recently, we have reported another plasmid-borne CTX-M-type gene, blaCTX-M-19, from a Klebsiella pneumoniae isolate from Vietnam (39). CTX-M-19 is a point mutant derivative of CTX-M-14/-18 that is able to hydrolyze ceftazidime to a significant degree (39). In the present study, we have investigated the genetic environment of the blaCTX-M-19 gene and described the surrounding sequences that included two IS elements, ISEcp1B and IS903D. One of these IS elements, ISEcp1B, may have mobilized the β-lactamase gene through a transposition mechanism and may drive the expression of the β-lactamase at a high level. We failed to identify the reservoir of ISEcp1-like elements among many gram-positive and gram-negative species screened.
MATERIALS AND METHODS
Bacterial strains, plasmids, and susceptibility testing.
K. pneumoniae clinical isolate ILT-3, which produces CTX-M-19 β-lactamase, has been reported previously (39). Escherichia coli reference strains and phagemids used in this study are referenced in Table 1. Antibiotic-containing disks (Bio-Rad, Marnes-la-Coquette, France) on Mueller-Hinton agar plates were used for routine antibiograms (www.sfm.asso.fr). A series of bacterial species were screened for the presence of ISEcp1-like sequences; they were representative (type strains in each case) of several bacterial species usually isolated from the skin and intestinal human flora. These include, among the Enterobacteriaceae, Citrobacter freundii (two strains), Citrobacter koseri (two strains), Citrobacter braakii, Citrobacter werkmanii, Citrobacter murliniae, Citrobacter gillenii, Edwarsiella hoshinae, Erwinia carotovora, Erwinia amylovora, Erwinia chrysanthemi, Erwinia quecina, E. coli (six strains), Enterobacter cloacae, Enterobacter aerogenes, Enterobacter gergoviae, Klebsiella pneumoniae, Kluyvera ascorbata (four strains), Kluyvera cryocrescens (two strains), Pantoea spp., Proteus mirabilis, Proteus vulgaris, Proteus penneri, Providencia stuartii, Rahnella aquatilis (two strains), Salmonella enterica (four strains), Serratia plymuthica, Serratia rubidaea, Serratia odorifera, and Shigella spp. (nine strains); among the gram-negative rods, Acinetobacter baumannii, Acinetobacter johnsonii, Aeromonas hydrophila, Alcaligenes xylosoxydans, Alcalinogenes denitrificans, Bordetella bronchiseptica, Brevium diminuta, Comamonas acidovorans, Pseudomonas aeruginosa, Pseudomonas fluorescens, Pseudomonas putida, and Stenotrophomonas maltophilia; among the gram-positive rods and anaerobes, Bifidobacterium spp., Clostridium difficile (two strains), Clostridium perfringens (two strains), and Propionibacterium spp.; among the gram-positive cocci and aerobes, Enterococcus faecium (three strains), Enterococcus faecalis (two strains), Staphylococcus aureus (two strains), coagulase-negative Staphylococcus spp. (three strains), Streptococcus equinus, Streptococcus pneumoniae (two strains), and viridans streptococci (two strains); and among anaerobic gram-positive cocci, Peptostreptococcus spp., and Campylobacter spp. (five strains).
TABLE 1.
Reference strains and plasmids used in this study
| Strain or plasmid | Relevant genotype or phenotype | Source or reference |
|---|---|---|
| Strains | ||
| E. coli JM109 | endA1 gyrA96 hsdR17 Δ(lac proA) relA recA1 supE44 thi F (lacIqZΔM15 proAB+traD36) | Gibco BRL-Life Technologies |
| E. coli DH10B | araD139 Δ(ara leu)7697 deoR endA1 galK1 galU nupG recA1-rpsL F′ mrcA Δ(mrr-hsdRMS-mrcBC)Φ80dlacZΔM15 ΔlacX74 | Stratagene |
| Plasmids | ||
| pPCRScript Amp SK(+) | Ampicillin resistant | Stratagene |
| pACYC184 | Chloramphenicol resistant, low copy number | 11 |
| pBK-CMV | Kanamycin resistant, high copy number | Stratagene |
| pILT-3 | Natural ca. 50-kb plasmid of K. pneumoniae ILT-3 producing CTX-M-19 | 39 |
| β-lactamase | ||
| pA-1 | Recombinant pPCR-Script Amp with a 6.5-kb PCR fragment from whole-cell DNA of K. pneumoniae ILT-3 containing ISEcp1B and a truncated blaCTX-M-19 gene | This study |
| pB-1 | Recombinant pPCR-Script Cam with a 1.1-kb PCR fragment from whole-cell DNA of K. pneumoniae ILT-3 containing ISEcp1B promoter sequences and the entire blaCTX-M-19 gene | This study |
| pB-2 | Recombinant pPCR-Script Cam with a 1-kb PCR fragment from whole-cell DNA of K. pneumoniae ILT-3 lacking ISEcp1B-promoting sequences and containing the entire blaCTX-M-19 gene | This study |
Cloning experiments, recombinant plasmid analysis, and DNA sequencing.
Whole-cell DNAs of K. pneumoniae ILT-3 and of bacterial strains tested in screening for ISEcp1-like presence were extracted as described previously (38). Partially digested Sau3AI fragments of whole-cell DNA of K. pneumoniae ILT-3 were ligated into BamHI-restricted phagemid pBK-CMV (Stratagene, Amsterdam, The Netherlands) (38). Ligation was performed at 4°C for 18 h at a 1:3 vector/insert ratio, at a final concentration of 1 μg of DNA in a ligation mixture containing 1 U of T4 DNA ligase. Recombinant plasmids were transformed by electroporation (Gene Pulser II; Bio-Rad, Ivry-sur-Seine, France) into electrocompetent E. coli DH10B cells.
Then, cloning experiments were performed with PCR-generated fragments, using a series of primers (Table 2) at the SrfI site of pPCRScript Amp SK(+) or at the SmaI site of the high-copy-number plasmid pBK-CMV or the EcoRV site of the low-copy-number vector pACYC184 (Tables 1 and 2). Long-range PCRs were performed under specific conditions (extension step, 72°C for 6 min; 6 U of Taq polymerase). Cloning of the INT2F/CTX-MB PCR fragment into pPCRScript Amp to identify integron features gave recombinant plasmid pA-1 (Fig. 1). Cloning of the PROM+/PRECTX-M3B and PROM−/PRECTX-M3B fragments containing the entire blaCTX-M-19 gene with or without the ISEcp1B-mediated promoter sequences into pACYC184 gave recombinant plasmids pB-1 and pB-2, respectively (Fig. 1). Antibiotic-resistant colonies were selected onto Trypticase soy agar plates containing 50 μg of amoxicillin per ml, except for E. coli harboring recombinant pA-1, which was subjected to a classical blue-white selection (β-galactosidase test). Recombinant plasmids were purified using Qiagen (Courtaboeuf, France) columns. Inserts of recombinant plasmids were sequenced on both strands by using an ABI 377 sequencer (Applied Biosystems, Foster City, Calif.). The nucleotide and the deduced protein sequences were analyzed with software available over the internet at the National Center of Biotechnology Information website (http://www.ncbi.nlm.nih.gov), and at the Institut Pasteur website (http://www.bioweb.pasteur.fr/seqanal/interfaces/clustalw-simple.html).
TABLE 2.
Sequences of primers used in this study
| Primer | Sequence (5′→3′) | Location | No. as shown in Fig. 1 |
|---|---|---|---|
| ISEcp1A | GCAGGTCTTTTTCTGCTCC | ISEcp1B transposase | 1 |
| ISEcp1B | TTTCCGCAGCACCGTTTGC | ISEcp1B transposase, reverse primer | 2 |
| PRECTX-M-3B | CCGTTTCCGCTATTACAAAC | 3′ end of blaCTX-M, reverse primer | 3 |
| PROM+ | TGCTCTGTGGATAACTTGC | Right part of ISEcp1B including −35 and −10 promoter sequences | 4 |
| PROM− | GCAGTCTAAATTCTTCGTG | Right part of ISEcp1B lacking −35 and −10 promoter sequences | 5 |
| CTX-M-REV | CCGCGAACATCATCCGTTGC | 5′ end of blaCTX-M-19, reverse primer for primer extension experiments | 6 |
| IS903-A | CATATGAAATCATCTGCGC | IS903D transposase | 7 |
| IS903-B | CCGTAGCGGGTTGTGTTTTC | IS903D transposase, reverse primer | 8 |
| INT2F | TCTCGGGTAACATCAAGGCCC | 3′ end of the int11 integrase gene | 9 |
| 3′-CS | AAGCAGACTTGACCTGA | 5′ end of the qacEΔ1 gene, reverse primer | 10 |
| CTX-MA | ACCGCGATATCGTTGGT | blaCTX-M-19 gene | 11 |
| CTX-MB | CGCTTTGCGATGTGCAG | blaCTX-M-19 gene, reverse primer | 12 |
| ORF1B | ATACTCTTGCTCATATGGGG | 5′ end of ORF1 of Tn1721, reverse primer | 13 |
FIG. 1.
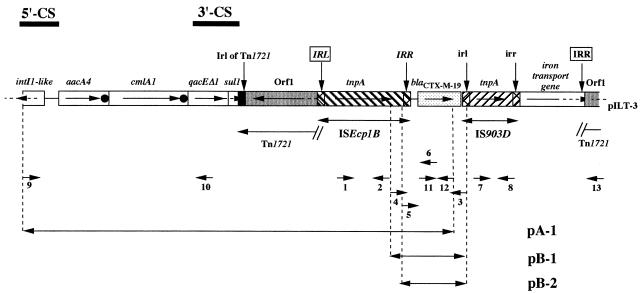
Schematic map of a 9,590-bp DNA fragment of the natural plasmid pILT-3 from K. pneumoniae ILT-3 containing ISEcp1B, IS903D, and the blaCTX-M-19 gene. The 5′ and 3′ conserved sequences (5′-CS and 3′-CS) of class 1 integrons are indicated in the upper left part of the figure. ORFs and genes are shown as boxes with an arrow indicating the transcription orientation. Black dots are for 59-be sequences. IRL and IRR motifs are indicated by vertical arrows. The IRL and IRR of the ISEcp1B-mobilized DNA fragment are boxed, those of the ISEcp1B element are italicized, and those of IS903D are in lowercase; the inverted repeat left of Tn1721 (Irl) is also shown. The cloned sequences of recombinant plasmids are indicated by arrows at both ends, with the corresponding plasmid names indicated on the right.
Hybridization experiments.
A 527-bp PCR-obtained internal fragment of the ISEcp1B transposase gene was generated using primers ISEcp1A and ISEcp1B, and a 472-bp PCR-obtained internal fragment of the IS903D transposase was generated with primers IS903A and IS903B (Fig. 1; Table 2) and with BamHI- and PvuI-restricted whole-cell DNA of K. pneumoniae ILT-3 and E. coli recombinant clones as templates. These PCR fragments were end labeled using the ECL nonradioactive labeling kit (Amersham Pharmacia Biotech). Hybridizations with these PCR products used as probes were performed as previously described (37). Similarly, Southern experiments were performed with ApaI- and HindIII-restricted DNA of strains of a series of bacterial species with the internal probe of ISEcp1B.
β-Lactamase activity.
Specific β-lactamase activities were determined with cultures of E. coli DH10B harboring recombinant plasmids pB-1 and pB-2 derived from pACYC184 (Fig. 1), which does not possess strong promoter sequences near its EcoRV cloning site. Overnight cultures were performed in 10 ml of Trypicase soy broth containing 50 μg of amoxicillin per ml and 30 μg of chloramphenicol per ml. Bacterial cultures were harvested by centrifugation for 15 min at 5,000 × g, and the bacterial pellets were resuspended in 500 μl of 50 mM phosphate buffer (pH 7) at 4°C, disrupted by sonication (1 min at 4 W), and centrifuged (30 min at 10,000 × g and 4°C), and the supernatants were analyzed. Hydrolytic activities were measured spectrophotometrically with 100 μM cephalothin as the substrate, as described previously (39). The protein content was measured by the Bio-Rad DC protein assay.
RNA extraction and primer extension analysis.
Total RNAs of E. coli DH10B (pA-1) and E. coli DH10B(pB-1) were extracted using the Qiagen RNeasy maxi-kit. Samples were treated with 1 U of DNase (Amersham Pharmacia Biotech) at 37°C for 30 min. Primer extension experiments were performed with the primer extension system avian myeloblastosis virus reverse transcriptase kit (Promega, Charbonnières, France) as specified by the manufacturer. The cDNA was generated from the CTX-M-REV primer (Table 2), which had been previously 5′ end labeled with [γ-32P]ATP and polynucleotide kinase.
Nucleotide sequence accession number.
The nucleotide sequence data for the entire ISEcp1B-mobilized DNA segment have been submitted to the GenBank nucleotide sequence database under accession number AF458080.
RESULTS
Identification and characterization of the genetic structures surrounding the β-lactamase gene.
Cloning of Sau3AI partially digested fragments of whole-cell DNA of K. pneumoniae ILT-3 into kanamycin-resistant pBK-CMV followed by selection on ampicillin-containing plates was performed. Despite several attempts, E. coli DH10B clones harboring recombinant plasmids were not obtained but ampicillin-resistant and kanamycin-susceptible E. coli DH10B clones were isolated. These clones displayed a β-lactam-resistant phenotype consistent with the expression of the blaCTX-M-19 gene. In these cases, the blaCTX-M-19 gene could have been integrated into the chromosome of these recombinant strains.
Since several ESBL genes may be transposon or integron borne (23, 37, 38, 40, 41), the surrounding sequences of the blaCTX-M-19 gene were explored further to identify a class 1 integron that may contain this gene (22). This has been done using PCR amplification experiments and the natural plasmid pILT-3 carrying the blaCTX-M-19 gene as the template (39).
Plasmid DNA of E. coli DH10B(pILT-3) was used as a template in long-range PCR experiments using primers INT2F and 3′-CS (located in the 5 and 3′ conserved sequences [5′-CS and 3′-CS] of class 1 integrons, respectively) in combination with primers CTX-MA and CTX-MB (located at the 5′ and 3′ ends of the blaCTX-M-19 gene, respectively) (Fig. 1; Table 2). A positive result was not obtained with primers CTX-MA and 3′-CS whereas a ca. 6.5-kb PCR product was obtained using primers INT2F and CTX-MB. This fragment, which contained a truncated blaCTX-M-19 β-lactamase gene, was cloned into ampicillin-resistant plasmid pPCRScript Amp SK(+), giving rise to pA-1 (Fig. 1). In addition to ampicillin resistance, plasmid pA-1 conferred resistance to chloramphenicol, kanamycin, gentamicin, netilmicin, and tobramycin. Sequencing of the insert of this recombinant plasmid identified several open reading frames (ORFs) (Fig. 1 and 2).
FIG. 2.
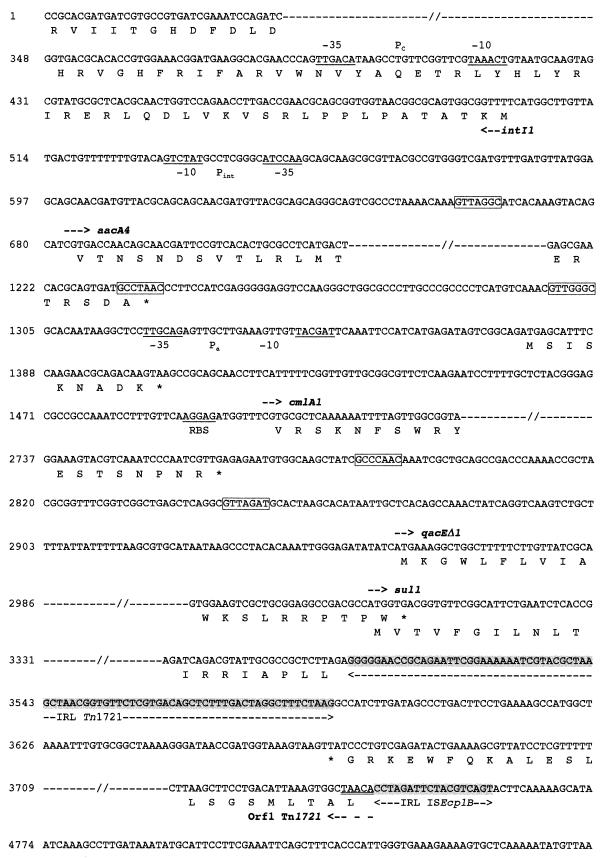
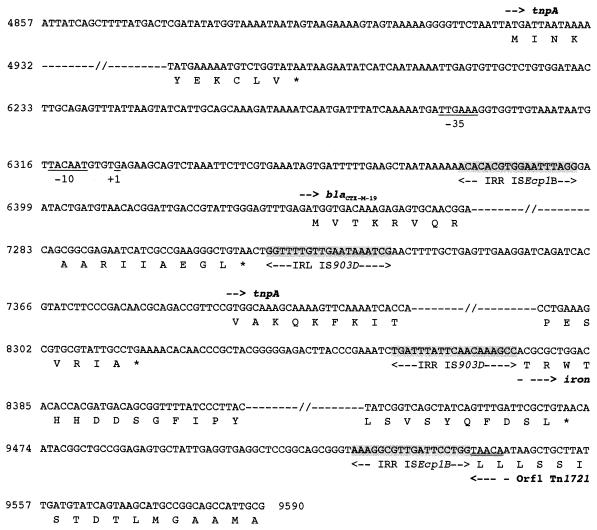
Nucleotide sequence of a 9,590-bp DNA fragment of the natural pILT-3 plasmid of K. pneumoniae ILT-3 containing ISEcp1B, β-lactamase CTX-M-19, and IS903D coding sequences. The deduced amino acid sequence is indicated in single-letter code below the nucleotide sequence. The start codons of the ORFs are indicated by horizontal arrows, and stop codons are indicated by asterisks. The −35 and −10 promoter sequences of the Pc, Pint, and Pa promoters are underlined, as well as the +1 position of the transcriptional start of the blaCTX-M-19 site. The 5-bp duplicated target sites of the putative insertion site for the DNA fragment resulting from an ISEcp1B-mediated transposition process are doubly underlined. RBS indicates the putative ribosome binding site for the cmlA1-like gene, and the 9-amino-acid leader peptide sequence of the variant of cmlA1 cassette is indicated. Core and inverse core sites located at each cassette boundary are boxed. TnpA, transposase gene; iron, gene encoding a putative outer membrane lipoprotein for iron uptake; and Orf1, the gene encoding the putative methyl-accepting chemotaxis protein of Tn1721.
Part of an intI1-like integrase gene was found, which contained typical Pc promoter features consisting of −35 (TTGACA) and −10 (TAAACT) sequences able to promote the expression of gene cassettes, whereas the secondary P2 promoter was in its inactive form (12, 27) (Fig. 1 and 2). An aacA4 gene cassette encoding an aminoglycoside 6′-N-acetyltransferase (26) and conferring resistance to gentamicin and kanamycin was found immediately upstream of the integrase gene (Fig. 1 and 2). A second gene cassette that carried a cmlA1 variant gene conferring resistance to chloramphenicol was identified downstream of the aacA4 cassette (Fig. 1 and 2) (41). The deduced 419-amino-acid sequence contained a protein that differred from CMLA1 by five amino acid substitutions. This gene was preceded by its own putative promoter sequences, Pa (−35 [TTGCAG] and −10 [TACGAT]) (Fig. 2), that may drive the expression of the chloramphenicol resistance determinant (41) independently of the promoter sequences located in the integrase sequence. The 59-be that was associated with this chloramphenicol resistance gene differed from that of the cmlA1 gene cassette by only a 7-bp substitution (41), suggesting a common origin.
Downstream of these gene cassettes, typical features of a 3′-CS of class 1 integrons were found, with a qacEΔ1 gene fused with a sul1-type gene (22) (Fig. 1 and 2). The sul1-type gene was interrupted by insertion of the inverted repeat left (IRL) of Tn1721 (1), thus explaining the susceptibility to sulfonamides of E. coli DH10B harboring natural plasmid pILT-3. The putative site of insertion of Tn1721 consisted of an AT-rich 5-bp target site (TTAGA), as reported previously (1) (Fig. 2, bp 3501 to 3505). On the right side of the IRL of Tn1721, the 3′ end of the so-called ORF1 of Tn1721 encoding a putative methyl-accepting chemotaxis protein was identified (Fig. 1 and 2). This coding sequence was truncated at its 5′ end by insertion of an ISEcp1-like element designated ISEcp1B (Fig. 1 and 2).
ISEcp1B (1,655 bp long) differred from ISEcp1 (GenBank accession no. AJ242809) by three nucleotide substitutions and was weakly related to other IS elements, with the amino acid sequence of its transposase having only 25% identity to that of transposases of the IS1380 family elements (29). ISEcp1B contained two imperfect 18-bp inverted-repeat sequences (four mismatches) surrounding a tnpA gene that encoded a putative transposase with one amino acid change (Q179R) compared to the ISEcp1 transposase. The same “DDE” motif found in most phosphotransferases (29) was present in the transposase sequence of ISEcp1B. The blaCTX-M-19 gene was located on the right side of ISEcp1B (Fig. 1 and 2), with its ATG start codon being located 42 bp downstream of the inverted repeat right (IRR) of ISEcp1B (Fig. 2).
To identify further DNA sequences surrounding the blaCTX-M-19 gene, several long-range PCR experiments were performed using a series of primers (Fig. 1 and Table 2). The CTX-MA primer annealing to the 5′ internal part of the blaCTX-M-19 gene was used in combination with primers annealing to (i) the ISEcp1B-IRR or ISEcp1B-IRL sequences in order to search for a putative insertion of a second ISEcp1B element downstream of blaCTX-M-19, (ii) the 5′ end of ORF1 of Tn1721, or (iii) the 3′ end of class 1 integrons (Table 2). Only PCR experiments with primer located in the 5′ end of ORF1 and primer CTX-MA gave positive results, suggesting that the 3′ end of a class 1 integron or a second ISEcp1B insertion sequence (able to constitute a composite transposon) was not present in the immediate vicinity of blaCTX-M-19. Sequencing of this ca. 3-kb PCR product revealed that an IS903-like insertion sequence was located downstream of the blaCTX-M-19 gene.
The 1,056-bp IS element, designated IS903D, was bracketed by two imperfect 19-bp inverted-repeat sequences (one mismatch) and contained a transposase-coding sequence. The sequence of this transposase differed by 7 of the 467 residues of IS903 (20) and by 3 residues from that of IS903B (V105I, N156K, A189S) (31). No significant identity was found between the DNA sequences of the IRs of ISEcp1B and IS903D. Furthermore, target site duplications on each side of IS903D were not identified (15). On the right side of IS903D, a DNA sequence was found that likely encodes the carboxy-terminal 365 of 700 amino acids of a protein with 63% amino acid identity to an outer membrane lipoprotein involved in iron transport and identified in E. coli K-12 (8).
Inside this lipoprotein gene, a second 18-bp sequence with consistent nucleotide identity to the IRR of ISEcp1B was identified at the place where truncation has occurred and was followed by a TAACA sequence identical to that found on the left side of ISEcp1B-IRL. This 5-bp duplication was probably the signature of an insertion generated by transposition (Fig. 2). Thus, the 4,797-bp fragment, bracketed on one side by the IRL of ISEcp1B and on the other side by this 18-bp sequence used as the IRR, probably constituted a transposon.
Finally, another part of ORF1 of Tn1721 was identified on the right side of the second IRR of ISEcp1B (Fig. 2). No deletion had occurred in the ORF1 sequence resulting from the insertion of the DNA fragment that contained ISEcp1B, blaCTX-M-19, IS903D, and the truncated gene encoding the outer membrane lipoprotein (Fig. 1 and 2).
ISEcp1B as an enhancer of blaCTX-M-19 gene expression.
No putative promoter sequences were found in the 42-bp sequence that separated the IRR of ISEcp1B from the ATG site of the blaCTX-M-19 gene. This suggested a role of ISEcp1B in β-lactamase expression. Indeed, as we have reported previously (27), upstream of another blaCTX-M gene (blaCTX-M-15), −35 (TTGAAA) and −10 (TACAAT) regions corresponding to a putative promoter were identified in ISEcp1B near its IRR (Fig. 2). To determine the role of ISEcp1B in blaCTX-M-19 expression, β-lactamase activities were determined with cell cultures of E. coli DH10B harboring several recombinant plasmids, and primer extension experiments were performed.
Two recombinant plasmids containing the blaCTX-M-19 gene were constructed with or without the putative promoter sequences located next to the IRR of ISEcp1B; these were recombinant plasmids pB-1 and pB-2, respectively (Fig. 1). Specific β-lactamase activities with cultures of E. coli DH10B harboring pB-1 and pB-2 were 35 ± 4 and 2 ± 0.8 U per mg of protein, respectively, corresponding to a ca. 17-fold difference. Thus, the role of ISEcp1B sequences in blaCTX-M-19 expression was demonstrated.
Primer extension experiments were performed with RNAs extracted from cultures of E. coli DH10B(pA-1) and E. coli DH10B(pB-1) by using extension primer CTX-M-REV (Table 2). The +1 signal was identified 110 bp upstream of the ATG start codon of the blaCTX-M-19 gene, and the deduced −35 and −10 promoter sequences were located at the end of ISEcp1B, as expected (Fig. 2).
Copy number of ISEcp1B.
To determine the copy number of ISEcp1B in K. pneumoniae ILT-3 and E. coli DH10B clones resulting from preliminary Sau3AI cloning experiments, whole-cell DNAs of these strains were digested with BamHI and PvuI, which did not cut inside the ISEcp1B and IS903D DNA sequences. These restricted fragments were then hybridized with internal probes for ISEcp1B transposase and IS903D transposase genes. A single copy of ISEcp1B and IS903D was found in all strains (data not shown). Since blaCTX-M-19 was plasmid borne and was associated with ISEcp1B in K. pneumoniae ILT-3 (37), this result indicated that the ISEcp1B-transposed fragment was not chromosome located in K. pneumoniae ILT-3. Positive hybridization results obtained with E. coli DH10B clones resulting from preliminary Sau3AI cloning experiments indicated a likely chromosomal integration of at least ISEcp1B and blaCTX-M-19 in those cases.
Screening for progenitor of ISEcp1.
Since the blaCTX-M genes have spread worldwide, we hypothesized that the reservoir of ISEcp1-like elements could have been either the progenitor of blaCTX-M genes (Kluyvera spp.) or bacterial species that may have been in close contact with those enterobacterial species. Thus, selected bacterial species that are common components of human intestinal and skin floras were studied.
ApaI- and HindIII-restricted whole-cell DNA of strains of these bacterial species were subjected to a Southern blot analysis. Hybridizations with an ISEcp1B-specific probe did not give positive signals (data not shown), except for a single Salmonella enterica serotype Blockley strain. This result was confirmed by PCR analysis with primers specific for the transposase gene of ISEcp1B (data not shown). Sequence analysis revealed perfect DNA identity to ISEcp1B. Analysis of the antibiotic susceptibility of that Salmonella isolate showed that it remained susceptible to β-lactams, and PCR experiments failed to detect any blaCTX-M genes. Other S. enterica strains belonging to the same serotype were ISEcp1B negative (data not shown).
DISCUSSION
This work showed that the ISEcp1B element seems to act as a key factor in the dissemination of CTX-M-type β-lactamase genes. ISEcp1B also acts as a strong positive factor for blaCTX-M-19 gene expression. The −35 and −10 promoter sequences for blaCTX-M-19 expression are located at the end of ISEcp1B near its IRR, as described for the −35 and −10 promoter sequences driving the expression of the carbapenem-hydrolyzing cfiA gene of Bacteroides fragilis, which are located in the IS1186 element (35). Other IS elements may enhance the expression of β-lactamase genes, but, in most of these cases, the −35 promoter sequences are located in the IS inverted repeats whereas the −10 promoter sequences are part of the downstream β-lactamase genes (19, 30, 33).
The mechanism of mobilization generated by ISEcp1B seems to correspond to a normal transposition mechanism and not to a one-ended transposition mechanism as suggested previously (P. D. Stapleton, Program Abstr. 39th Intersci. Conf. Antimicrob. Agents Chemother, abstr. 1457, 1999). Indeed, one-ended transposition requires only a single copy of an IS element but does not use specific inverted repeat sequences (32). Transposition usually requires IR sequences located at both ends of a transposon that are recognized by a transposon-encoded transposase (13). It is likely that the ISEcp1B transposase may recognize structurally related IR sequences and use them as ends in regular transposition (21). As observed for IS1247, which is a also a member of the IS1380 family, transposition of the insertion element together with adjacent sequences is possible without the requirement for a second IS element (43).
While this work was in progress, Cao et al. detailed the genetic structure of the 7-kb natural pIP843 plasmid of a K. pneumoniae isolate encoding β-lactamase CTX-M-17, which differed from CTX-M-19 by two amino acid substitutions (9). This K. pneumoniae isolate was from Vietnam, whereas K. pneumoniae strain ILT-3, analyzed in the present study, was isolated in Paris (France) but from a Vietnamese patient. A very similar structure was found bracketing the blaCTX-M-17 gene, with an upstream ISEcp1-like element (five amino acid substitutions in the transposase sequence compared to that of ISEcp1B) and a downstream IS903C element (two amino acid substitutions in that transposase compared to that of IS903D). Additionally, a truncated gene encoding an outer membrane lipoprotein was identified in both cases, and these genes differed by a few nucleotide substitutions. In fact, a similar transposon was present compared to that in K. pneumoniae ILT-3, containing an ISEcp1-like sequence, blaCTX-M-17, IS903C, and a truncated outer membrane lipoprotein gene. These transposons have 99% nucleotide identity, differing by 19 of 4,797 bp. In addition, a careful analysis of the published sequence led us to identify a 6-bp duplication consisting of the motif ATAATA on each side of the transposon carrying the blaCTX-M-17 gene. However, in the case of the blaCTX-M-17 gene, the fragment was not bracketed by DNA structure related to Tn1721 and was inserted in a smaller plasmid of 7 kb.
Additionally, Chanawong et al. (10) reported clinical E. coli isolates from Southern China (near Vietnam) that harbored a structurally related blaCTX-M gene, blaCTX-M-14 (a point mutant analogue of blaCTX-M-19), which is bracketed by the ends of an ISEcp1-like element and of an IS903-like element. This latter result suggests dissemination of similar genetic structures carrying structurally related blaCTX-M genes in that part of the world.
Cao et al. have shown that the −35 and −10 promoter sequences at the end of the ISEcp1-like element near its IRR were located 110 bp upstream of the ATG site of the blaCTX-M-17 gene and drove the expression of the β-lactamase exactly as for the expression of blaCTX-M-19 (9). In other cases, the ATG sites of blaCTX-M-3 and of its point mutant derivative blaCTX-M-15 from Polish isolates were located 128 and 48 bp downstream of the −10 promoter sequences of the ISEcp1-like sequences, respectively (5, 25). This result indicates the variety of genetic events that have enabled associations between ISEcp1-like sequences and several blaCTX-M genes in clinical strains of different geographic origins.
We hypothesized that blaCTX-M genes and ISEcp1-like sequences must have been in close contact somewhere to enable the formation these hybrid genetic structures. The blaCTX-M genes may possess low-strength promoter sequences in Kluyvera spp. that do not allow a high level of their expression. In recipient species such as E. coli, K. pneumoniae, and S. enterica serovar Typhimurium, ISEcp1-like sequences may provide a higher level of expression of the plasmid-located blaCTX-M genes. Although it has been shown that several enterobacterial species of the Kluyvera genus are a natural reservoir of blaCTX-M-like genes (24, 36), we did not identify the reservoir of ISEcp1-like sequences. Future studies should look for bacterial species in animal floras, since blaCTX-M genes have been identified in animal isolates, especially in Salmonella spp. that are a source of anthropozoonotic infections.
Additionally, two recent reports describe genetic structures surrounding a blaCTX-M-2 gene, described as a complex sul1-type integron, similar to In6 and In7 (2, 16). These structures possess an ORF for a putative transposase that is not related to an ISEcp1-like transposase and that may enhance blaCTX-M-2 mobilization. Thus, genetic plasticity linked to blaCTX-M genes may be more complex than expected.
Finally, ISEcp1-like sequences may be more general mobilizing and expression elements for β-lactamase genes since analysis of GenBank databases also identified them upstream of plasmid-located cephalosporinase genes (6, 7, 34).
Acknowledgments
We thank Daniel Aubert for help with the primer extension experiments.
This work was financed by a grant from the Ministère de l'Education Nationale et de la Recherche (grant UPRES-EA3539), Université Paris XI, Paris, France.
REFERENCES
- 1.Allmeier, H., B. Cresnar, M. Greck, and R. Schmitt. 1992. Complete nucleotide sequence of Tn1721: gene organization and a novel gene product with features of a chemotaxis protein. Gene 111:11-20. [DOI] [PubMed] [Google Scholar]
- 2.Arduino, S. M., P. H. Roy, G. A. Jacoby, B. E. Orman, S. A. Pineiro, and D. Centron. 2002. blaCTX-M-2 is located in an unusual class 1 integron (In35) which includes Orf513. Antimicrob. Agents Chemother. 46:2303-2306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Avila, P., J. Grinsted, and F. de la Cruz. 1988. Analysis of the variable endpoints generated by one-ended transposition of Tn21. J. Bacteriol. 170:1350-1353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Avison, M. B., C. J. von Heldreich, C. S. Higgins, P. M. Bennett, and T. Walsh. 2000. TEM-2 β-lactamase encoded on an active Tn1-like transposon in the genome of a clinical isolate of Stenotrophomonas maltophilia. J. Antimicrob. Chemother. 46:879-884. [DOI] [PubMed] [Google Scholar]
- 5.Baraniak, A., J. Fiett, W. Hryniewicz, P. Nordmann, and M. Gniadkowski. 2002. Ceftazidime-hydrolysing CTX-M-15 extended-spectrum β-lactamase (ESBL) in Poland. J. Antimicrob. Chemother. 50:393-396. [DOI] [PubMed] [Google Scholar]
- 6.Bauernfeind, A., I. Scheider, R. Jungwirth, H Sahly, and U. Ullmann. 1999. A novel type of AmpC beta-lactamase, ACC-1, produced by a Klebsiella pneumoniae strain causing nosocomial pneumonia. Antimicrob. Agents Chemother. 43:1924-1931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bauernfeind, A., I. Stemplinger, R. Jungwirth, and H. Giamarellou. 1996. Characterization of the plasmidic β-lactamase CMY-2, which is responsible for cephamycin resistance. Antimicrob. Agents Chemother. 40:221-224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Blattner, F. R., G. Plunkett, C. A. Bloch, N. T. Perna, V. Burland, M. Ridley, J. Collado-Vides, J. D. Glasner, C. K. Rode, G. F. Mayhew, J. Gregor, N. W. Davis, H. A. Kirkpatrick, M. A. Goeden, D. J. Rose, B. Mau, and Y. Shao. 1997. The complete genome sequence of Escherichia coli K12. Science 277:1453-1474. [DOI] [PubMed] [Google Scholar]
- 9.Cao, V., T. Lambert, and P. Courvalin. 2002. ColE1-like plasmid pIP843 of Klebsiella pneumoniae encoding extended-spectrum β-lactamase CTX-M-17. Antimicrob. Agents Chemother. 46:1212-1217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chanawong, A., F. H. M'Zali, J. Heritage, J. H. Xiong, and P. M. Hawkey. 2002. Three cefotaximases, CTX-M-9, CTX-M-13, and CTX-M-14, among Enterobacteriaceae in the People's Republic of China. Antimicrob. Agents Chemother. 46:630-637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chang, A. C. Y., and S. N. Cohen. 1978. Construction and characterization of amplifiable multicopy DNA cloning vehicles derived from the P15A cryptic miniplasmid. J. Bacteriol. 134:1141-1156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Collis, C. M., and R. M. Hall. 1995. Expression of antibiotic resistance genes in the integrated cassettes of integrons. Antimicrob. Agents Chemother. 39:155-162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Craig, N. L. 1996. Transposition, p. 2339-2362. In F. C. Neidhardt, R. Curtiss III, J. L. Ingraham, E. C. C. Lin, K. B. Low, B. Magasanik, W. S. Reznikoff, M. Riley, M. Schaechter, and H. E. Umbarger (ed.), Escherichia coli and Salmonella: cellular and molecular biology, vol. 2. ASM Press, Washington, D.C.
- 14.Decousser, J. W., L. Poirel, and P. Nordmann. 2001. Characterization of a chromosomally encoded extended-spectrum class A β-lactamase from Kluyvera cryocrescens. Antimicrob. Agents Chemother. 45:3595-3598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Derbyshire, K. M., L. Hwang, and N. D. Grindley. 1987. Genetic analysis of the interaction of the insertion sequence IS903 transposase with its terminal inverted repeats. Proc. Natl. Acad. Sci. USA 84:8048-8053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Di Conza, J., J. A. Ayala, P. Power, M. Mollerach, and G. Gutkind. 2002. Novel class 1 integron (InS21) carrying blaCTX-M-2 in Salmonella enterica serovar Infantis. Antimicrob. Agents Chemother. 46:2257-2261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Dutour, C., R. Bonnet, H. Marchandin, M. Boyer, C. Chanal, D. Sirot, and J. Sirot. 2002. CTX-M-1, CTX-M-3, and CTX-M-14 β-lactamases from Enterobacteriaceae isolated in France. Antimicrob. Agents Chemother. 46:534-537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gazouli, M., E. Tzelepi, A. Markogiannakis, N. J. Legakis, and L. S. Tzouvelekis. 1998. Two novel plasmid-mediated cefotaxime-hydrolysing beta-lactamases (CTX-M-5 and CTX-M-6) from Salmonella typhimurium. FEMS Microbiol. Lett. 165:289-293. [DOI] [PubMed] [Google Scholar]
- 19.Goussard, S., W. Sougakoff, C. Mabilat, A. Bauernfeind, and P. Courvalin. 1991. An ISl-like element is responsible for high-level synthesis of extended-spectrum β-lactamase TEM-6 in Enterobacteriaceae. J. Gen. Microbiol. 137:2681-2687. [DOI] [PubMed] [Google Scholar]
- 20.Grindley, N. D. F., and C. M. Joyce. 1980. Genetic and DNA sequence analysis of the kanamycin resistance transposon Tn903. Proc. Natl. Acad. Sci. USA 77:7176-7180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Grindley, N. D. F., and R. R. Reed. 1985. Transpositional recombination in prokaryotes. Annu. Rev. Biochem. 54:863-896. [DOI] [PubMed] [Google Scholar]
- 22.Hall, R. M., D. E. Brookes, and H. W. Stokes. 1994. Integrons found in different locations have identical 5′ ends but variable 3′ ends. J. Bacteriol. 176:6286-6294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Heritage, J., P. M. Hawkey, N. Todd, and I. J. Lewis. 1992. Transposition of the gene encoding a TEM-12 extended-spectrum β-lactamase. Antimicrob. Agents Chemother. 36:1981-1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Humeniuk, C., G. Arlet, V. Gauthier, P. Grimont, R. Labia, and A. Philippon. 2002. β-Lactamases of Kluyvera ascorbata, probable progenitors of some plasmid-encoded CTX-M types. Antimicrob. Agents Chemother. 46:3045-3049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Karim, A., L. Poirel, S. Nagarajan, and P. Nordmann. 2001. Plasmid-mediated extended spectrum β-lactamase (CTX-M-3-like) from India and gene association with insertion sequence ISEcp1. FEMS Microbiol. Lett. 201:237-241. [DOI] [PubMed] [Google Scholar]
- 26.Lambert, T., M. C. Ploy, and P. Courvalin. 1994. A spontaneous point mutation in the aac(6′)-Ib′ gene results in altered substrate specificity of aminoglycoside 6′-N-acetyltransferase of a Pseudomonas fluorescens strain. FEMS Microbiol. Lett. 115:297-304. [DOI] [PubMed] [Google Scholar]
- 27.Lévesque, C., S. Brassard, J. Lapointe, and P. H. Roy. 1994. Diversity and relative strengh of tandem promoters for the antibiotic-resistance genes of several integrons. Gene 142:49-54. [DOI] [PubMed] [Google Scholar]
- 28.Mabilat, C., J. Lourencao-Vital, S. Goussard, and P. Courvalin. 1992. A new example of physical linkage between Tn1 and Tn21: the antibiotic multiple-resistance region of plasmid pCFF04 encoding extended-spectrum β-lactamase TEM-3. Mol. Gen. Genet. 235:113-121. [DOI] [PubMed] [Google Scholar]
- 29.Mahillon, J., and M. Chandler. 1998. Insertions sequences. Microbiol. Mol. Biol. Rev. 62:725-774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Maki, H., and K. Murakami. 1997. Formation of potent hybrid promoters of the mutant llm gene by IS256 transposition in methicillin-resistant Staphylococcus aureus. J. Bacteriol. 179:6944-6948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mollet, B., S. Iida, and W. Arber. 1985. An active variant of the prokaryotic transposable element IS903 carries an amber stop codon in the middle of an open reading frame. Mol. Gen. Genet. 199:534-536. [DOI] [PubMed] [Google Scholar]
- 32.Mötsch, S., R. Schmitt, P. Avila, F. De La Cruz, E. Ward, and J. Grinsted. 1985. Junction sequences generated by “one-ended transposition.” Nucleic Acids Res. 13:3335-3342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Naas, T., L. Philippon, L. Poirel, E. Ronco, and P. Nordmann. 1999. An SHV-derived extended-spectrum β-lactamase in Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 43:1281-1284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Nadjar, D., M. Rouveau, C. Verdet, L. Donay, J. L. Herrmann, P. H. Lagrange, A. Philippon, and G. Arlet. 2000. Outbreak of Klebsiella pneumoniae producing transferable AmpC-type beta-lactamase (ACC-1) originating from Hafnia alvei. FEMS Microbiol. Lett. 187:35-40. [DOI] [PubMed] [Google Scholar]
- 35.Podglajen, I., J. Breuil, and E. Collatz. 1994. Insertion of a novel DNA sequence, IS1186, upstream of the silent carbapenemase gene cfiA, promotes expression of carbapenem resistance in clinical isolates of Bacteroides fragilis. Mol. Microbiol 12:105-114. [DOI] [PubMed] [Google Scholar]
- 36.Poirel, L., P. Kämpfer and P. Nordmann. 2002. Chromosome-encoded Ambler class A β-lactamase of Kluyvera georgiana, a probable progenitor of a subgroup of CTX-M extended-spectrum β-lactamase. Antimicrob. Agents Chemother. 46:4038-4040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Poirel, L., I. Le Thomas, T. Naas, A. Karim, and P. Nordmann. 2000. Biochemical-sequence analyses of GES-1, a novel class A extended-spectrum β-lactamase, and the class 1 integron In 52 from Klebsiella pneumoniae. Antimicrob. Agents Chemother. 44:622-632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Poirel, L., T. Naas, M. Guibert, E. B. Chaibi, R. Labia, and P. Nordmann. 1999. Molecular and biochemical characterization of VEB-1, a novel class A extended-spectrum β-lactamase encoded by an Escherichia coli integron gene. Antimicrob. Agents Chemother. 43:573-581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Poirel, L., T. Naas, I. Le Thomas, A. Karim, E. Bingen, and P. Nordmann. 2001. CTX-M-type extended-spectrum β-lactamase that hydrolyzes ceftazidime through a single amino acid substitution in the omega loop. Antimicrob. Agents Chemother. 45:3355-3361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Sirot, D., C. De Champs, C. Chanal, R. Labia, A. Darfeuille-Michaud, R. Perroux, and J. Sirot. 1991. Translocation of antibiotic resistance determinants including an extended-spectrum β-lactamase between conjugative plasmids of Klebsiella pneumoniae and Escherichia coli. Antimicrob. Agents Chemother. 35:1576-1581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Stokes H. W., and R. M. Hall. 1991. Sequence analysis of the inducible chloramphenicol resistance determinant in Tn1696 integron suggests regulation by translation attenuation. Plasmid 26:10-19. [DOI] [PubMed] [Google Scholar]
- 42.Tzouvelekis, L. S., E. Tzelepi, P. T. Tassios, and N. J. Legakis. 2000. CTX-M-type β-lactamases: an emerging group of extended-spectrum enzymes. Int. J. Antimicrob. Agents 14:137-140. [DOI] [PubMed] [Google Scholar]
- 43.Van der Ploeg, J., M. Willemsem, G. Van Hall, and D. B. Janssen. 1995. Adaptation of Xanthobacter autotrophicus GJ10 to bromoacetate due to activation and mobilization of the halocetate dehalogenase gene by insertion element IS1247. J. Bacteriol. 177:1348-1356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wu, S. W., K. Dornbusch, G. Kronvall, and M. Norgren. 1999. Characterization and nucleotide sequence of a Klebsiella oxytoca cryptic plasmid encoding a CMY-type β-lactamase: confirmation that the plasmid-mediated cephamycinase originated from the Citrobacter freundii AmpC β-lactamase. Antimicrob. Agents Chemother. 43:1350-1357. [DOI] [PMC free article] [PubMed] [Google Scholar]