Abstract
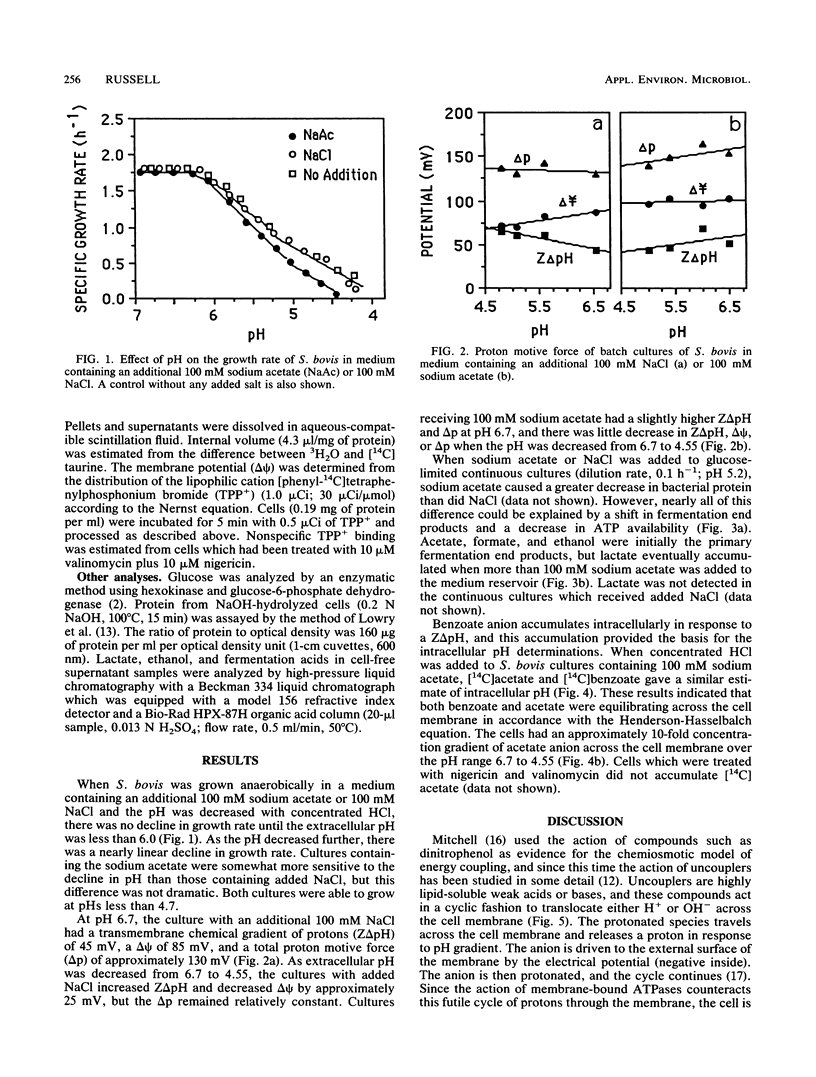
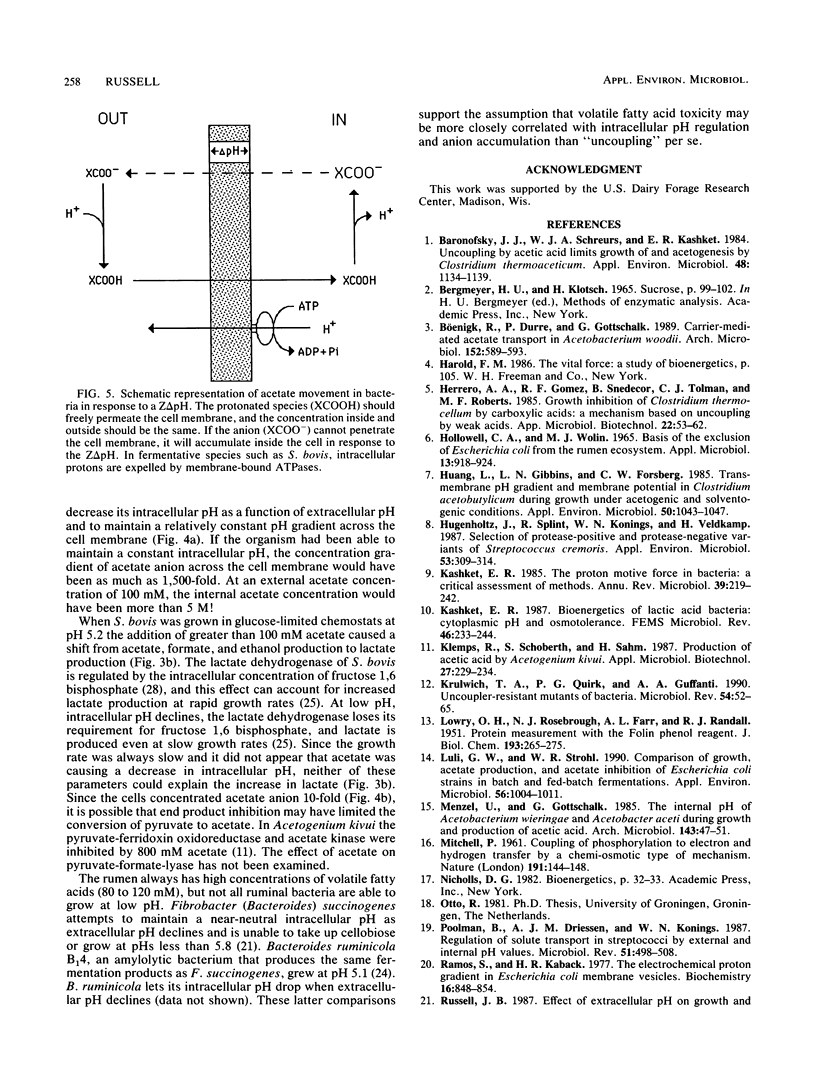
Streptococcus bovis JB1, an acid-tolerant ruminal bacterium, was able to grow at pHs from 6.7 to 4.5, and 100 mM acetate had little effect on growth rate or proton motive force across the cell membrane. When S. bovis was grown in glucose-limited chemostats at pH 5.2, the addition of sodium acetate (as much as 100 mM) had little effect on the production of bacterial protein. At higher concentrations of sodium acetate (100 to 360 mM), production of bacterial protein declined, but this decrease could largely be explained by a shift in fermentation products (acetate, formate, and ethanol production to lactate production) and a decline in ATP production (3 ATP per glucose versus 2 ATP per glucose). YATP (grams of cells per mole of ATP) was not decreased significantly even by high concentrations of acetate. Cultures supplemented with 100 mM sodium acetate took up [14C]acetate and [14C]benzoate in accordance with the Henderson-Hasselbalch equation and gave similar estimates of intracellular pH. As the extracellular pH declined, S. bovis allowed its intracellular pH to decrease and maintained a relatively constant pH gradient across the cell membrane (0.9 unit). The decrease in intracellular pH prevented S. bovis from accumulating large amounts of acetate anion. On the basis of these results it did not appear that acetate was acting as an uncoupler. The sensitivity of other bacteria to volatile fatty acids at low pH is explained most easily by a high transmembrane pH gradient and anion accumulation.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baronofsky J. J., Schreurs W. J., Kashket E. R. Uncoupling by Acetic Acid Limits Growth of and Acetogenesis by Clostridium thermoaceticum. Appl Environ Microbiol. 1984 Dec;48(6):1134–1139. doi: 10.1128/aem.48.6.1134-1139.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hollowell C. A., Wolin M. J. Basis for the exclusion of Escherichia coli from the rumen ecosystem. Appl Microbiol. 1965 Nov;13(6):918–924. doi: 10.1128/am.13.6.918-924.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang L., Gibbins L. N., Forsberg C. W. Transmembrane pH gradient and membrane potential in Clostridium acetobutylicum during growth under acetogenic and solventogenic conditions. Appl Environ Microbiol. 1985 Oct;50(4):1043–1047. doi: 10.1128/aem.50.4.1043-1047.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hugenholtz J., Splint R., Konings W. N., Veldkamp H. Selection of Protease-Positive and Protease-Negative Variants of Streptococcus cremoris. Appl Environ Microbiol. 1987 Feb;53(2):309–314. doi: 10.1128/aem.53.2.309-314.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kashket E. R. The proton motive force in bacteria: a critical assessment of methods. Annu Rev Microbiol. 1985;39:219–242. doi: 10.1146/annurev.mi.39.100185.001251. [DOI] [PubMed] [Google Scholar]
- Krulwich T. A., Quirk P. G., Guffanti A. A. Uncoupler-resistant mutants of bacteria. Microbiol Rev. 1990 Mar;54(1):52–65. doi: 10.1128/mr.54.1.52-65.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Luli G. W., Strohl W. R. Comparison of growth, acetate production, and acetate inhibition of Escherichia coli strains in batch and fed-batch fermentations. Appl Environ Microbiol. 1990 Apr;56(4):1004–1011. doi: 10.1128/aem.56.4.1004-1011.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MITCHELL P. Coupling of phosphorylation to electron and hydrogen transfer by a chemi-osmotic type of mechanism. Nature. 1961 Jul 8;191:144–148. doi: 10.1038/191144a0. [DOI] [PubMed] [Google Scholar]
- Poolman B., Driessen A. J., Konings W. N. Regulation of solute transport in streptococci by external and internal pH values. Microbiol Rev. 1987 Dec;51(4):498–508. doi: 10.1128/mr.51.4.498-508.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramos S., Kaback H. R. The electrochemical proton gradient in Escherichia coli membrane vesicles. Biochemistry. 1977 Mar 8;16(5):848–854. doi: 10.1021/bi00624a006. [DOI] [PubMed] [Google Scholar]
- Russell J. B., Baldwin R. L. Substrate preferences in rumen bacteria: evidence of catabolite regulatory mechanisms. Appl Environ Microbiol. 1978 Aug;36(2):319–329. doi: 10.1128/aem.36.2.319-329.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russell J. B., Dombrowski D. B. Effect of pH on the efficiency of growth by pure cultures of rumen bacteria in continuous culture. Appl Environ Microbiol. 1980 Mar;39(3):604–610. doi: 10.1128/aem.39.3.604-610.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russell J. B. Effect of extracellular pH on growth and proton motive force of Bacteroides succinogenes, a cellulolytic ruminal bacterium. Appl Environ Microbiol. 1987 Oct;53(10):2379–2383. doi: 10.1128/aem.53.10.2379-2383.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russell J. B. Low-affinity, high-capacity system of glucose transport in the ruminal bacterium Streptococcus bovis: evidence for a mechanism of facilitated diffusion. Appl Environ Microbiol. 1990 Nov;56(11):3304–3307. doi: 10.1128/aem.56.11.3304-3307.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russell J. B., Strobel H. J. ATPase-dependent energy spilling by the ruminal bacterium, Streptococcus bovis. Arch Microbiol. 1990;153(4):378–383. doi: 10.1007/BF00249009. [DOI] [PubMed] [Google Scholar]
- WOLIN M. J. FRUCTOSE-1,6-DIPHOSPHATE REQUIREMENT OF STREPTOCOCCAL LACTIC DEHYDROGENASES. Science. 1964 Nov 6;146(3645):775–777. doi: 10.1126/science.146.3645.775. [DOI] [PubMed] [Google Scholar]


