Abstract
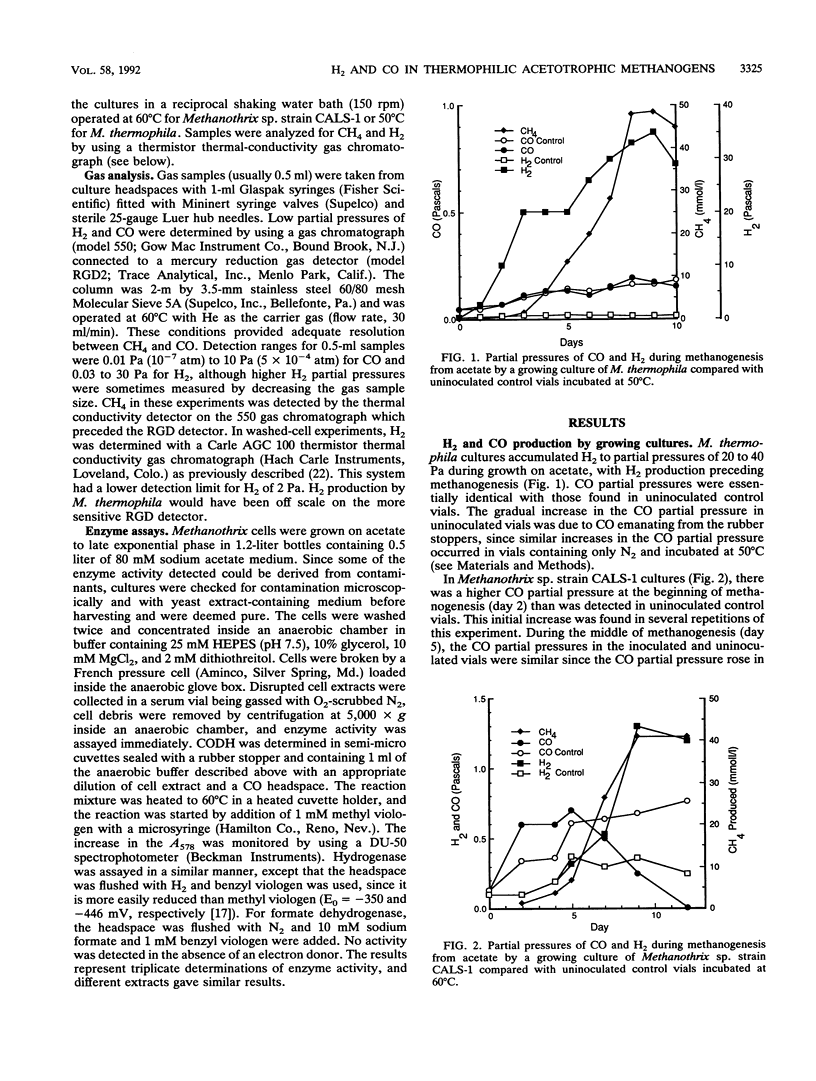
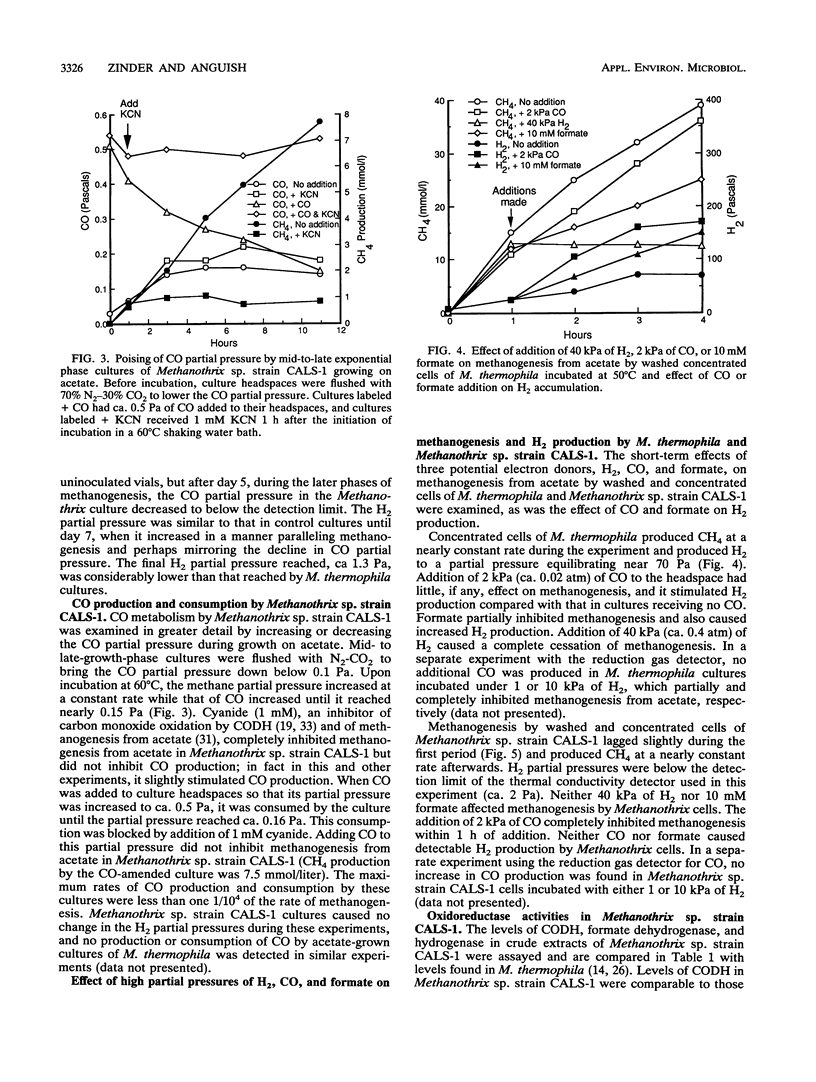
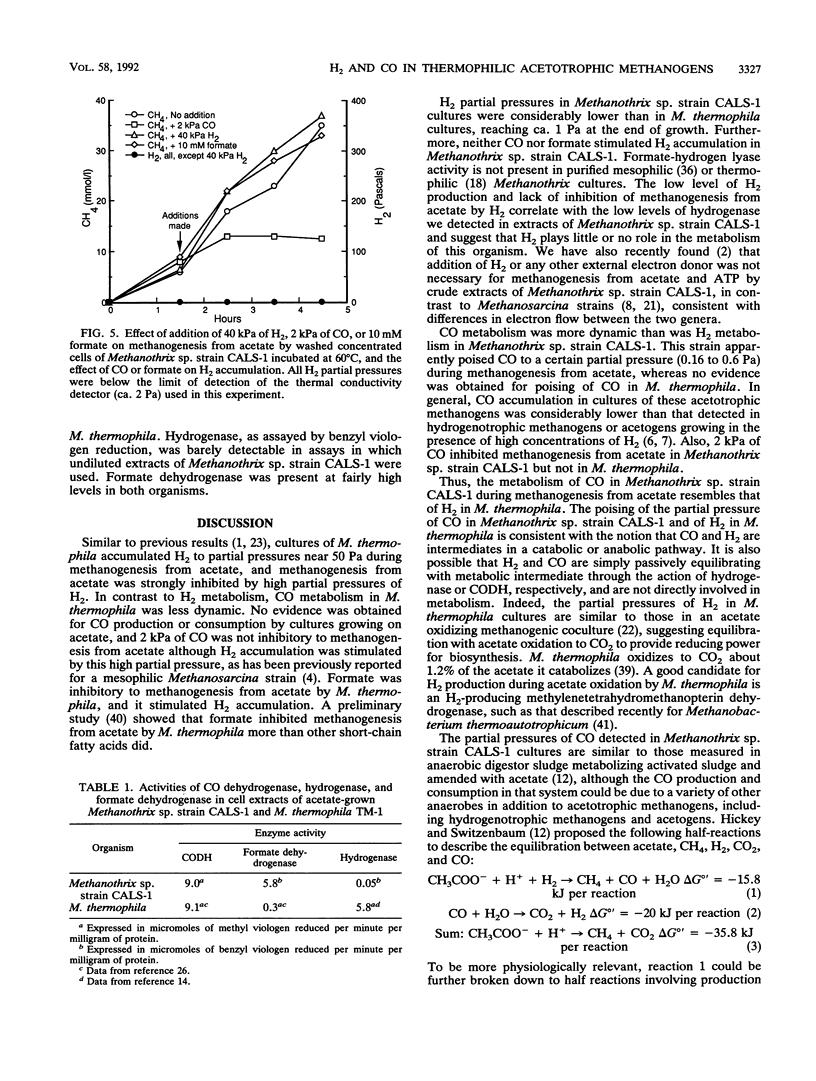
CO and H2 have been implicated in methanogenesis from acetate, but it is unclear whether they are directly involved in methanogenesis or electron transfer in acetotrophic methanogens. We compared metabolism of H2, CO, and formate by cultures of the thermophilic acetotrophic methanogens Methanosarcina thermophila TM-1 and Methanothrix sp. strain CALS-1. M. thermophila accumulated H2 to partial pressures of 40 to 70 Pa (1 Pa = 0.987 × 10-5 atm), as has been previously reported for this and other Methanosarcina cultures. In contrast, Methanothrix sp. strain CALS-1 accumulated H2 to maximum partial pressures near 1 Pa. Growing cultures of Methanothrix sp. strain CALS-1 initially accumulated CO, which reached partial pressures near 0.6 Pa (some CO came from the rubber stopper) during the middle of methanogenesis; this was followed by a decrease in CO partial pressures to less than 0.01 Pa by the end of methanogenesis. Accumulation or consumption of CO by cultures of M. thermophila growing on acetate was not detected. Late-exponential-phase cultures of Methanothrix sp. strain CALS-1, in which the CO partial pressure was decreased by flushing with N2-CO2, accumulated CO to 0.16 Pa, whereas cultures to which ca. 0.5 Pa of CO was added consumed CO until it reached this partial pressure. Cyanide (1 mM) blocked CO consumption but not production. High partial pressures of H2 (40 kPa) inhibited methanogenesis from acetate by M. thermophila but not by Methanothrix sp. strain CALS-1, and 2 kPa of CO was not inhibitory to M. thermophila but was inhibitory to Methanothrix sp. strain CALS-1. Levels of CO dehydrogenase, hydrogenase, and formate dehydrogenase in Methanothrix sp. strain CALS-1 were 9.1, 0.045, and 5.8 μmol of viologen reduced min-1 mg of protein-1. These results suggest that CO plays a role in Methanothrix sp. strain CALS-1 similar to that of H2 in M. thermophila and are consistent with the conclusion that CO is an intermediate in a catabolic or anabolic pathway in Methanothrix sp. strain CALS-1; however, they could also be explained by passive equilibration of CO with a metabolic intermediate.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Boone D. R., Menaia J. A., Boone J. E., Mah R. A. Effects of Hydrogen Pressure during Growth and Effects of Pregrowth with Hydrogen on Acetate Degradation by Methanosarcina Species. Appl Environ Microbiol. 1987 Jan;53(1):83–87. doi: 10.1128/aem.53.1.83-87.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bott M., Eikmanns B., Thauer R. K. Coupling of carbon monoxide oxidation to CO2 and H2 with the phosphorylation of ADP in acetate-grown Methanosarcina barkeri. Eur J Biochem. 1986 Sep 1;159(2):393–398. doi: 10.1111/j.1432-1033.1986.tb09881.x. [DOI] [PubMed] [Google Scholar]
- Bott M., Thauer R. K. Proton-motive-force-driven formation of CO from CO2 and H2 in methanogenic bacteria. Eur J Biochem. 1987 Oct 15;168(2):407–412. doi: 10.1111/j.1432-1033.1987.tb13434.x. [DOI] [PubMed] [Google Scholar]
- Fukuzaki S., Nishio N., Nagai S. Kinetics of the methanogenic fermentation of acetate. Appl Environ Microbiol. 1990 Oct;56(10):3158–3163. doi: 10.1128/aem.56.10.3158-3163.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grahame D. A. Catalysis of acetyl-CoA cleavage and tetrahydrosarcinapterin methylation by a carbon monoxide dehydrogenase-corrinoid enzyme complex. J Biol Chem. 1991 Nov 25;266(33):22227–22233. [PubMed] [Google Scholar]
- Jablonski P. E., DiMarco A. A., Bobik T. A., Cabell M. C., Ferry J. G. Protein content and enzyme activities in methanol- and acetate-grown Methanosarcina thermophila. J Bacteriol. 1990 Mar;172(3):1271–1275. doi: 10.1128/jb.172.3.1271-1275.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jetten M. S., Stams A. J., Zehnder A. J. Purification and characterization of an oxygen-stable carbon monoxide dehydrogenase of Methanothrix soehngenii. Eur J Biochem. 1989 May 1;181(2):437–441. doi: 10.1111/j.1432-1033.1989.tb14744.x. [DOI] [PubMed] [Google Scholar]
- Jones R. W., Garland P. B. Sites and specificity of the reaction of bipyridylium compounds with anaerobic respiratory enzymes of Escherichia coli. Effects of permeability barriers imposed by the cytoplasmic membrane. Biochem J. 1977 Apr 15;164(1):199–211. doi: 10.1042/bj1640199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krzycki J. A., Lehman L. J., Zeikus J. G. Acetate catabolism by Methanosarcina barkeri: evidence for involvement of carbon monoxide dehydrogenase, methyl coenzyme M, and methylreductase. J Bacteriol. 1985 Sep;163(3):1000–1006. doi: 10.1128/jb.163.3.1000-1006.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee M. J., Zinder S. H. Hydrogen partial pressures in a thermophilic acetate-oxidizing methanogenic coculture. Appl Environ Microbiol. 1988 Jun;54(6):1457–1461. doi: 10.1128/aem.54.6.1457-1461.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lovley D. R., Ferry J. G. Production and Consumption of H(2) during Growth of Methanosarcina spp. on Acetate. Appl Environ Microbiol. 1985 Jan;49(1):247–249. doi: 10.1128/aem.49.1.247-249.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Min H., Zinder S. H. Kinetics of Acetate Utilization by Two Thermophilic Acetotrophic Methanogens: Methanosarcina sp. Strain CALS-1 and Methanothrix sp. Strain CALS-1. Appl Environ Microbiol. 1989 Feb;55(2):488–491. doi: 10.1128/aem.55.2.488-491.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson M. J., Ferry J. G. Carbon monoxide-dependent methyl coenzyme M methylreductase in acetotrophic Methosarcina spp. J Bacteriol. 1984 Nov;160(2):526–532. doi: 10.1128/jb.160.2.526-532.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohtsubo S., Demizu K., Kohno S., Miura I., Ogawa T., Fukuda H. Comparison of acetate utilization among strains of an aceticlastic methanogen, Methanothrix soehngenii. Appl Environ Microbiol. 1992 Feb;58(2):703–705. doi: 10.1128/aem.58.2.703-705.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith M. R., Lequerica J. L., Hart M. R. Inhibition of methanogenesis and carbon metabolism in Methanosarcina sp. by cyanide. J Bacteriol. 1985 Apr;162(1):67–71. doi: 10.1128/jb.162.1.67-71.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanner R. S., McInerney M. J., Nagle D. P., Jr Formate auxotroph of Methanobacterium thermoautotrophicum Marburg. J Bacteriol. 1989 Dec;171(12):6534–6538. doi: 10.1128/jb.171.12.6534-6538.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Terlesky K. C., Nelson M. J., Ferry J. G. Isolation of an enzyme complex with carbon monoxide dehydrogenase activity containing corrinoid and nickel from acetate-grown Methanosarcina thermophila. J Bacteriol. 1986 Dec;168(3):1053–1058. doi: 10.1128/jb.168.3.1053-1058.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thauer R. K., Jungermann K., Decker K. Energy conservation in chemotrophic anaerobic bacteria. Bacteriol Rev. 1977 Mar;41(1):100–180. doi: 10.1128/br.41.1.100-180.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thauer R. K., Möller-Zinkhan D., Spormann A. M. Biochemistry of acetate catabolism in anaerobic chemotrophic bacteria. Annu Rev Microbiol. 1989;43:43–67. doi: 10.1146/annurev.mi.43.100189.000355. [DOI] [PubMed] [Google Scholar]
- Westermann P., Ahring B. K., Mah R. A. Threshold acetate concentrations for acetate catabolism by aceticlastic methanogenic bacteria. Appl Environ Microbiol. 1989 Feb;55(2):514–515. doi: 10.1128/aem.55.2.514-515.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zinder S. H., Mah R. A. Isolation and Characterization of a Thermophilic Strain of Methanosarcina Unable to Use H(2)-CO(2) for Methanogenesis. Appl Environ Microbiol. 1979 Nov;38(5):996–1008. doi: 10.1128/aem.38.5.996-1008.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]



