Abstract
Stable mixed donor/host chimerism has been reliably established in dogs given a sublethal dose (2 Gy) of total body irradiation (TBI) before and immunosuppression with mycophenolate mofetil (MMF) or rapamycin combined with cyclosporine (CSP) after marrow transplantation from dog leukocyte antigen (DLA)-identical littermates (hematopoietic cell transplantation [HCT]). When TBI was reduced to 1 Gy, only transient engraftment was observed. Here we investigated whether stable engraftment after 1-Gy TBI could be accomplished by reducing host-versus-donor immune responsiveness through preceding CD154 blockade and infusion of donor peripheral blood mononuclear cells (PBMCs). We found that the anti-human CD154 antibody, 5c8, cross-reacted with canine lymphocytes and blocked alloimmune responses in vitro. Based on pharmacokinetic studies, 6 dogs received a single intravenous injection of 5 mg/kg anti-CD154 antibody (on day −5), followed 1 day later by donor PBMCs. On day 0, the dogs were given 1 Gy of TBI and underwent DLA-identical marrow grafts. Postgraft immunosuppression consisted of MMF and CSP. All 6 dogs demonstrated initial engraftment; 3 dogs sustained the engraftment for >26 weeks, whereas 3 dogs rejected their grafts, after 9, 22, and 24 weeks, and survived with autologous recovery. Graft survival was significantly improved over that in 11 historical controls conditioned with 1-Gy TBI and given either MMF or rapamycin with CSP after HCT, all of which rejected their grafts between 3 and 12 weeks (P = .03). Preceding donor PBMC infusion and CD154 blockade improved survival of DLA-identical marrow grafts after 1-Gy TBI.
Key words: Anti-CD154 antibody, Costimulatory blockade, Marrow transplantation, Dogs, Nonmyeloablative conditioning
Introduction
Sustained engraftment of dog leukocyte antigen (DLA)-identical littermate marrow was the rule in dogs conditioned with a nonmyeloablative dose of 2 Gy of total body irradiation (TBI) and given short courses of postgraft immunosuppression with either mycophenolate mofetil (MMF) or rapamycin along with cyclosporine (CSP) [1, 2]. However, uniform graft rejections were seen when TBI conditioning was decreased to 1 Gy. In contrast, most dogs showed sustained engraftment when 1-Gy TBI was preceded by intravenous injections of both peripheral blood mononuclear cells (PBMCs) from the marrow donor and T-cell costimulatory blocker CTLA4-Ig [3]. Here we used 1-Gy TBI conditioning to evaluate whether blockade of another costimulatory pathway—the interaction between CD40 and CD154—was equally effective in enhancing marrow engraftment. Blockade of this pathway has been successful in murine models of hematopoietic stem cell transplantation and different animal models of solid organ transplantation [4, 5, 6].
Materials and methods
Dogs
Litters of beagle, mini-mongrel, basenji, and golden retriever crossbreeds, either raised at the Fred Hutchinson Cancer Research Center (FHCRC) or purchased from commercial kennels, were assessed for disease and enrolled in a veterinary preventive medicine program against worms, distemper, parvovirus, adenovirus type 2, parainfluenza virus, corona virus, rabies, and canine papillomavirus. The dogs were age 6-24 months and weighed 8.1-15.1 kg (median, 9.6 kg).
The study was approved by the FHCRC’s Institutional Animal Care and Use Committee, which is accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. DLA-identical littermates were selected as donor–recipient pairs on the basis of matching for highly polymorphic microsatellite markers within DLA class I and class II regions [7], which was confirmed by DLA-DRB1 sequencing [8].
Monoclonal Antibody 5c8
The hybridoma cell line obtained from American Type Culture Collection (Manassas, VA) produced the mouse anti-human CD154 monoclonal antibody (mAb) 5c8 [9]. Hybridoma cells were cultivated according to the guidelines of the provider. The mAb was purified from supernatant and checked for endotoxin by the FHCRC biologic production facility. Endotoxin-free antibody was diluted in phosphate-buffered saline (PBS) without calcium and magnesium at a concentration of 1.4-1.5 mg/mL and immediately frozen at −70°C until use.
Cross-Reactivity with Canine CD154
Cross-reactivity of the mAb 5c8 was determined by flow cytometry using canine lymphocytes stimulated with 4α-phorbol 12-myristate 13-acetate (PMA) and ionomycin. For these studies, canine PBMCs were separated from 20-30 mL of blood using Ficoll gradient centrifugation (specific density, 1.074 g/dL). The cells were washed, counted, and resuspended in RPMI medium supplemented with 10% heat-inactivated canine serum, 2 mmol of L-glutamine, 100 U/mL of penicillin, 100 μg/mL of streptomycin, and 50 μmol of β-mercaptoethanol at a concentration of 4 × 106 cells per mL. Then 1-mL cell suspension samples were plated on a 24-well plate (1 sample per plate). Ionomycin and PMA were added to the lymphocyte culture at final concentrations of 5 μM and 100 μg/mL, respectively. Medium and supplements were obtained from Gibco Invitrogen (Carlsbad, CA) and Sigma-Aldrich (St Louis, MO). The PBMCs were cultivated for 4.5 hours, then harvested, washed, and resuspended in PBS with 2% goat serum and 0.2% sodium azide. Cells were counted, the concentration was adjusted to 1 × 106/mL, and the cells were centrifuged at 800 rpm for 10 minutes. After removal of the supernatant, the cells were resuspended in 100 μL of mAb solution (concentration of 10 μg/mL), incubated on ice for 30 minutes, and then washed with 1 mL of PBS + 2% goat serum and 0.2% sodium azide. After centrifugation, the supernatant was removed, and the cells resuspended in fluorescein isothiocyanate (FITC)-labeled goat anti-mouse secondary antibody solution (dilution 1:100; Jackson ImmunoResearch Laboratories, West Grove, PA), incubated in the dark at 4°C for 30 minutes, washed, and resuspended in 1 mL of PBS with 2% goat serum and 0.2% sodium azide. The cells were analyzed using a FACS-Calibur machine and Cell Quest Pro software (Becton Dickinson, Franklin Lakes, NJ). Corresponding isotype controls were obtained from Dako North America (Carpinteria, CA).
Mixed Leukocyte Reactions
Mixed leukocyte reactions (MLRs) were performed as described previously [10]. Purified anti-CD154 mAb 5c8 was added to the medium in a concentration of 10 μg/mL for initial testing and in doses increasing from 1 μg/mL to 50 μg/mL to determine dose effects. CTLA4-Ig (courtesy of Dr. Richard Boismenu, Repligen Corp, Waltham, MA) was used as a positive control, and the irrelevant mouse antibody 31A was used as a negative control.
Pharmacokinetic Studies
Three dogs received single intravenous (IV) doses of anti-CD154 mAb 5c8 of 1, 5, and 10 mg/kg. These dogs had not been previously exposed to mouse immunoglobulin. After injection, serial blood samples were obtained at 10 and 30 minutes; at 1, 2, 4, 6 and 9 hours; and at 1, 2, 3, 4, 5, 6, 7, 8, 10, and 13 days. The blood was centrifuged and the serum immediately stored at −70°C.
Antibody Concentrations and Immune Responses
Serum concentrations of mAb 5c8 were measured by enzyme-linked immunosorbent assay (ELISA). Briefly, anti-mouse IgG–precoated 96-well plates (R&D Biosystems, Minneapolis, MN) were blocked with 5% chicken serum and 0.5% Tween 20 containing PBS and incubated with canine serum from treated dogs (dilution: sample-dependent, 1:25000-1:100). Bound 5c8 was detected by horseradish peroxidase–conjugated goat anti-mouse IgG (H+L; Pierce, Rockford, IL) as the secondary antibody. The color reagent was 2,2′azino-bis (3-ethylbenzthiazoline-6-sulfonic acid) (Sigma). Plates were read with a Vmax microtiter plate reader (Molecular Devices, Menlo Park, CA) at 405 nm. Standard curves were established with known concentrations of 5c8; sera obtained from dogs before mAb infusion served as controls. A similar procedure was used to detect canine anti-5c8 antibodies, except the plates were coated with 5c8 solution, and bound antibodies were detected by horseradish peroxidase–conjugated goat anti-dog IgM (μ-chain) and goat anti-dog IgG (H+L chain) (Immunology Consultants Laboratory, Newberg, OR) to distinguish between the 2 isotypes. Active pooled normal dog serum served as a negative control. The canine serum samples were diluted by 1:100.
Hematopoietic Cell Transplantation
On days −6 and −5 before marrow transplantation, recipients were given 0.5 mg/kg ketorolac IV as prophylaxis against thrombosis, as described for anti-CD154 5c8 treatment [11, 12]. On day −5, the animals received 5 mg/kg of anti-CD154 5c8 IV. On day −4, 1 × 107 donor PBMCs/kg were administered; half of the cells were infused IV and the other half were given subcutaneously for optimal antigen presentation.
On day 0, recipients were given 1-Gy TBI and donor marrow as described previously [2, 13]. Marrow grafts contained 2.26-8.08 × 108 nucleated cells (NCs)/kg (median, 4.22 × 108 NCs/kg). This is comparable to the cell doses used in previous studies, with a median of 4.0 × 108 total NCs/kg in the study with CSP and MMF [1] and 3.8 × 108 NCs/kg in the study with CSP and sirolimus [2]. The dogs received standard posttransplantation care. Immunosuppression consisted of CSP 15 mg/kg twice a day orally from day −1 to day 35 and MMF 10 mg/kg twice a day subcutaneously from day 0 to day 27. CSP levels were measured once weekly, and results were used for CSP dose adjustments. MMF doses were adjusted according to clinical toxicity, which was mainly gastrointestinal.
Assessment of Chimerism
Hematopoietic engraftment was assessed by documentation of donor dinucleotide and tetranucleotide variable-number tandem repeat (VNTR) polymorphisms in granulocyte and mononuclear cells from peripheral blood using a polymerase chain reaction (PCR)-based assay as described previously [14]. Samples were drawn weekly for the first 12 weeks and then every 2 weeks thereafter until 6 months posttransplantation. PCR products were analyzed by gel electrophoresis, and percentages of donor chimerism in the recipients were determined using Image-quant software (AMPL Software Pty Ltd, Turramurra, NSW, Australia) after autoradiography.
Antibody Concentrations and Immune Responses
On days −5, −4, and 0, and then weekly for the first 7-8 weeks posttransplantation, serum samples were obtained to determine mAb 5c8 serum concentrations and the dogs’ immune responses against the antibody.
Statistical Analysis
Responses between anti-CD154–treated MLR cultures and controls were compared using the paired Student t test. The durations of engraftment among current dogs were compared with those in historical controls (Table 1) [2] using the log-rank test. Associations among transplanted marrow cell doses, donor chimerism levels at week 6, and duration of donor chimerism were evaluated using Spearman’s rank correlation coefficient. All reported P values were 2-sided, and those <.05 were considered significant.
Table 1.
Results in Dogs Given DLA-Identical Littermate Marrow Grafts after Conditioning⁎
| TBI Dose (Gy) | Pretransplantation Immunomodulation | Posttransplantation Immunosuppression | Sustained Engraftment (%) | Median Time of Rejection (Weeks) | P Value | References |
|---|---|---|---|---|---|---|
| 2 | None | MMF + CSP | 11/12 (92) | 12 | .0373 | [1] |
| 2 | None | SRL + CSP | 6/7 (86) | 11 | [2] | |
| 1 | None | MMF + CSP | 0/6 (0) | 10 | .0014 | [1] |
| 1 | None | SRL + CSP | 0/5 (0) | 9 | [2] | |
| 1 | ATG | MMF + CSP | 1/5 (20) | 9.5 | .145 | [13] |
| 1 | MTX + donor PBMCs | MMF + CSP | 2/6 (33) | 8 | .315 | [25] |
| 1 | CTLA4-Ig + donor PBMCs | MMF + CSP | 4/6 (66) | 14 | .721 | [3] |
Conditioning consisted of either 1 or 2 Gy of TBI and posttransplantation immunosuppression with CSP combined with MMF or sirolimus (SRL). The P values refer to comparisons of current results with those in previous studies using the log-rank test.
Results
Flow Cytometry, MLR, Pharmacokinetic Studies, and Dog Anti-mAb 5c8 Responses
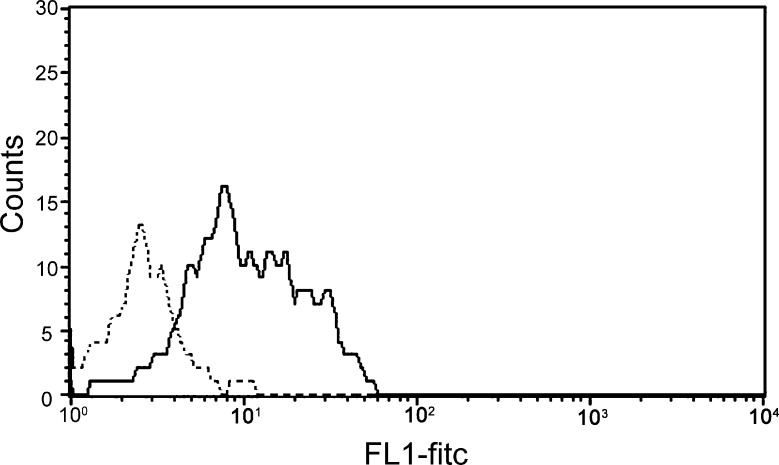
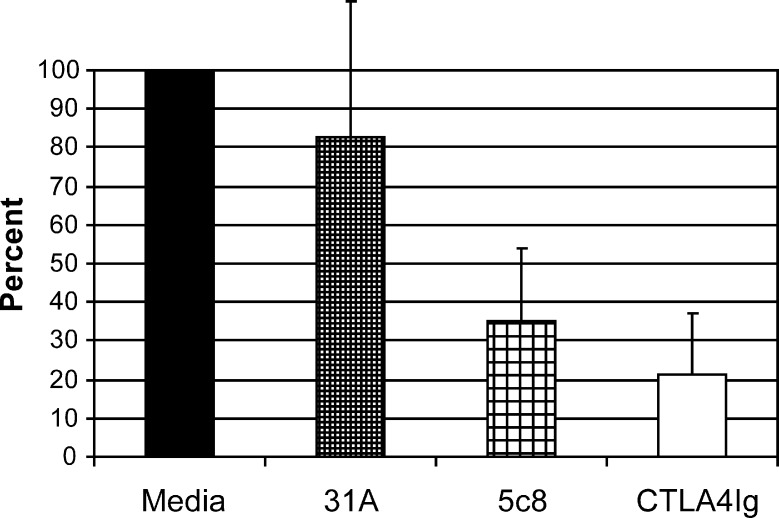
Figure 1 illustrates cross-reactivity of the anti-CD154 mAb 5c8 with activated canine lymphocytes compared with a murine isotype control antibody. Flow cytometry results indicate nearly a log increase in the intensity of binding of mAb 5c8 over the negative control. The MLR studies used cells from DLA-mismatched dogs after addition of irrelevant antibody 31A, anti-CD154 mAb 5c8, or CTLA4-Ig at concentrations of 10 μg/mL medium each (Figure 2). Anti-CD154 mAb 5c8 reduced the 3H-thymidine uptake to 35% compared with medium alone (P < .0001). CTLA4-Ig reduced the MLR response to 20% (P < .0001). The difference in MLR reactivities between mAb 5c8 and CTLA4-Ig was significant (P = .04). Increasing the mAb 5c8 concentration beyond 10 μg/mL did not further decrease 3H-thymidine uptake, whereas 5 μg/mL mAb 5c8 also led to significant MLR suppression (P = .002; data not shown). A concentration of 1 μg/mL did not significantly decrease 3H-thymidine uptake.
Figure 1.
Distribution of PMA and ionomycin- activated canine lymphocytes stained with FITC-labeled anti-CD154 antibody 5c8 (solid line) and a mouse isotype control (dotted line). Events are gated for living lymphocytes (vital stain: propidium iodide).
Figure 2.
MLR results. Shown are averages of 6 experiments, with each individual experiment done in triplicate. Anti-CD154 mAb 5c8, irrelevant mAb 31A (negative control), and CTLA4-Ig (positive control) were added at concentrations of 10 μg/mL each. The maximum 3H-thymidine uptake of the medium control in each experiment was set at 100%. Error bars represent 1 standard deviation. P values were calculated using the paired Student t test. Medium versus irrelevant mAb 31A, P = .14; medium versus 5c8, P < .0001; medium versus CTLA4Ig, P < .0001; mAb 5c8 versus CTLA4Ig, P = .04.
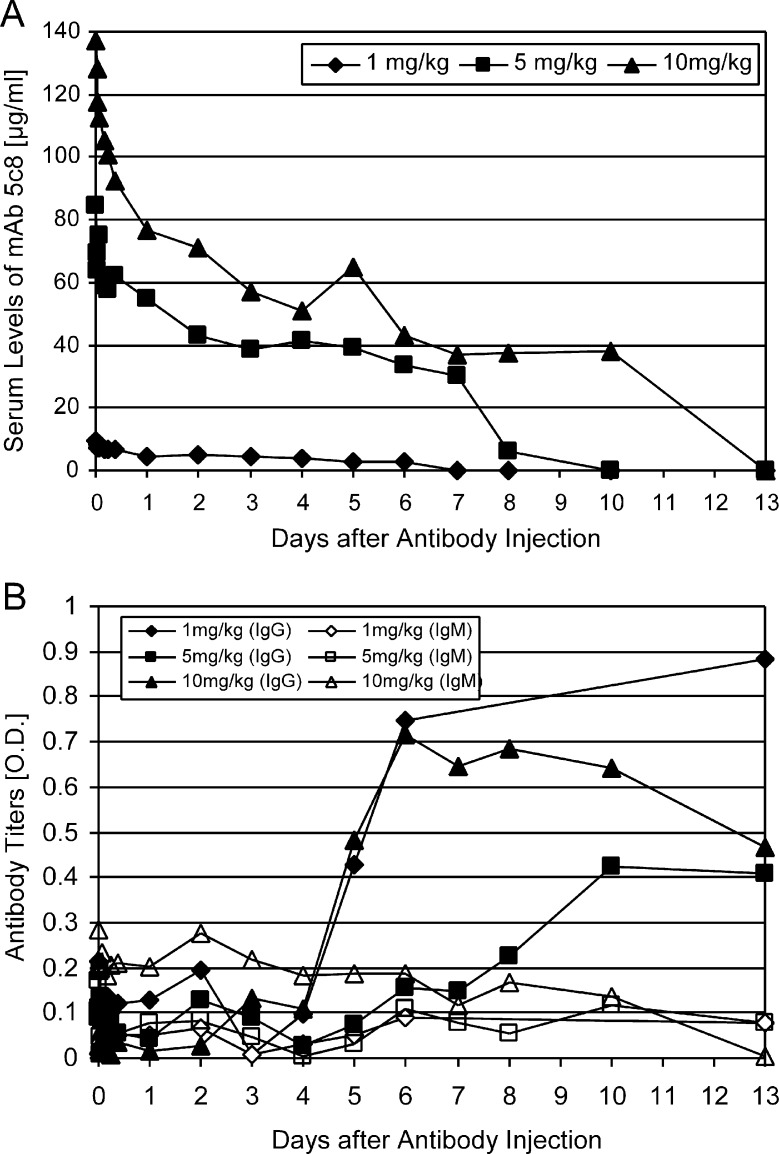
Ten minutes after each injection of mAb 5c8, serum concentrations reached maximum levels of 9.7 μg/mL (dog G397, 1 mg/kg dose), 84.6 μg/mL (dog G229, 5 mg/kg dose), and 137.4 μg/mL (dog G389, 10 mg/kg dose) (Figure 3A). These serum levels declined rapidly within 24 hours, to 4.7 μg/mL, 54.9 μg/mL, and 76.8 μg/mL, respectively, likely reflecting distribution of the antibody. Serum levels continued to decline gradually over the ensuing days, followed by rapid clearing of the mAb between days 8 and 10. No side effects of the mAb injections were seen.
Figure 3.
(A) Serum concentrations of anti-CD154 mAb 5c8 in 3 dogs given 1, 5, and 10 mg mAb/kg. Concentrations were calculated against a standard of purified mAb 5c8. (B) Antibody responses to anti-CD154 mAb 5c8 in 3 dogs given 1, 5, and 10 mg mAb/kg. IgG and IgM responses were assessed by isotype-specific secondary antibodies.
Anti-mAb 5c8 antibodies began to appear at days 5-8 after injection, and their titers increased as mAb 5c8 was cleared from the circulation (Figure 3B). The immune responses were predominantly of the IgG isotype, with lesser contributions of IgM antibodies. Parallel ELISA testing on mAb 5c8– and mouse IgG2a–coated plates suggested that 2 animals had either pure (dog G397) or predominantly (dog G389) anti-idiotype IgG-responses, whereas 1 animal had an anti-mouse isotype response (dog G229).
Hematopoietic Cell Transplantation
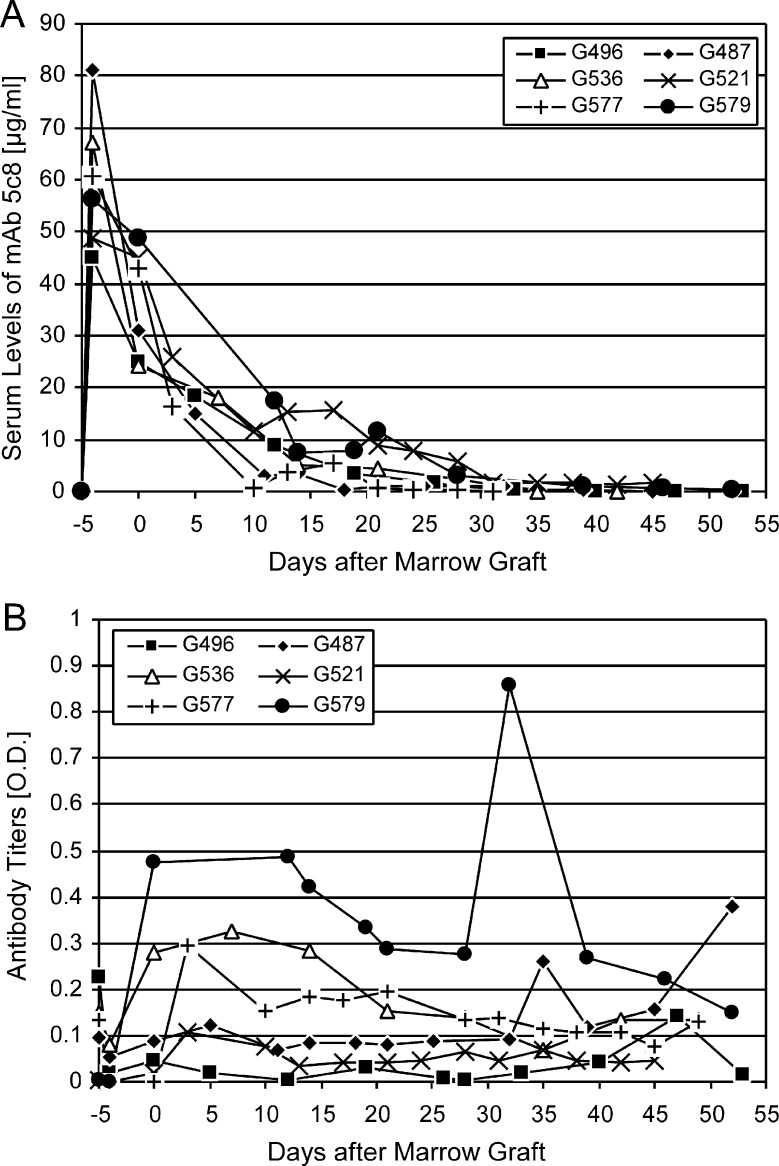
The 6 recipient dogs’ characteristics and outcomes of HCT are summarized in Table 2. Figure 4 summarizes the dogs’ serum concentrations of mAb 5c8 and canine antibody responses against mAb 5c8. The former ranged from 45 to 81 μg/mL (median, 59 μg/mL) on day −4 and from 24 to 48 μg/mL (median, 37 μg/mL) on day 0. The antibody clearances were considerably slower than those seen in the pharmacokinetic studies (days 35-52). Four dogs developed IgG anti-5c8 antibodies. In 3 of these 4 dogs, antibodies were detectable on the day of transplantation, with a second peak occurring after cessation of the postgrafting immunosuppression. In 1 dog, anti-mAb 5c8 antibodies appeared beyond day 35. No correlations between antibody response and duration of hematopoietic engraftment were seen.
Table 2.
HCT Outcomes in 6 Dogs Given 5 mg/kg Anti-CD154 mAb 5c8 and Donor PBMCs before 1-Gy TBI Conditioning and DLA-Identical Marrow Grafts⁎
| Maximum Donor Chimerism (%) |
|||||||
|---|---|---|---|---|---|---|---|
| Dog ID No. | Nucleated Marrow Cells (× 108/kg) | Toxicities | Mononuclear Cells | Granulocytes | Cumulative CSP/MMF Dosing (% of Intended Dose) | Rejection (Week) | Outcome |
| G487 | 3.32 | GI† | 9.4 | 10.8 | 100/79 | Yes (22) | Alive, autologous recovery |
| G496 | 7.84 | GI, infection allergic reaction to mAb 5c8 | 71.0 | 82.8 | 100/64 | No | Alive, mixed chimerism |
| G521 | 8.08 | ∅ | 51.2 | 73.0 | 100/100 | No | Alive, mixed chimerism |
| G536 | 4.22 | GI | 24.5 | 33.9 | 100/75 | Yes (24) | Alive, autologous recovery |
| G577 | 2.26 | GI, intussusception | 10.2 | 28.9 | 89/79 | Yes (9) | Alive, autologous recovery |
| G579 | 3.59 | GI, allergic reaction to mAb 5c8 | 31.9 | 66.6 | 100/74 | No | Alive, mixed chimerism |
GI indicates gastrointestinal.
MMF + CSP were administered after grafting.
GI toxicities usually manifested as diarrhea, nausea, or anorexia.
Figure 4.
(A) Serum concentrations of mAb 5c8 in the 6 dogs that underwent transplantation. Concentrations were calculated against a standard of purified mAb 5c8. (B) IgG antibody responses against mAb 5c8, assessed by isotype-specific secondary antibodies.
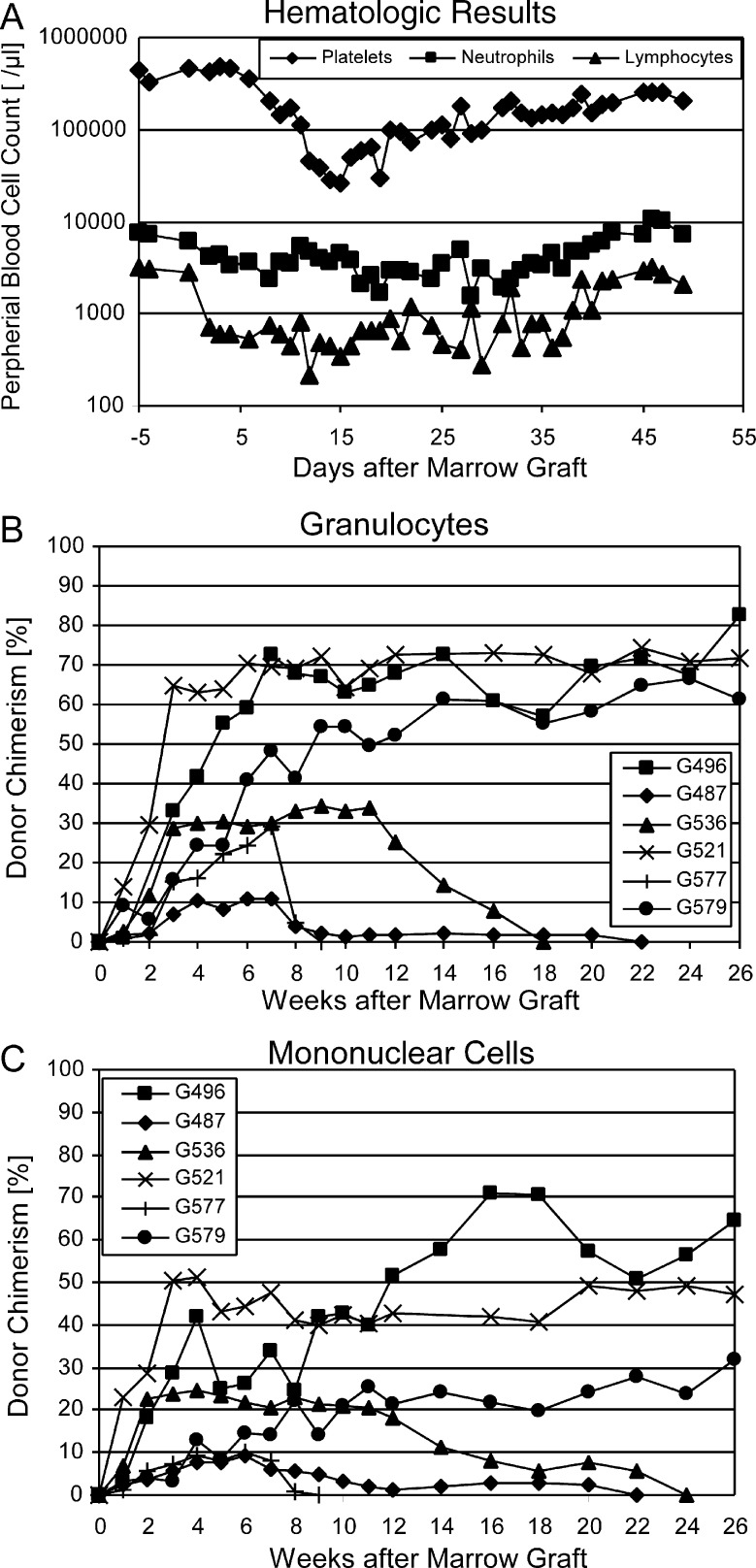
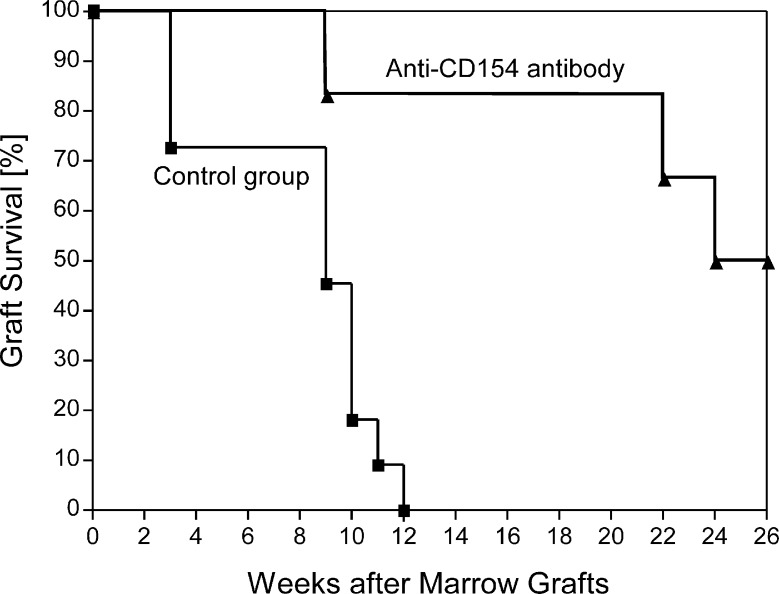
All 6 dogs exhibited prompt initial marrow engraftment. Both neutrophil and platelet counts showed nadirs at around day 20 after HCT before eventual blood count recovery occurred (Figure 5A). Lymphocyte counts remained <1000/μL between days 3 and 40 and then recovered. Peak donor chimerism levels were 10.8%-82.8% (median, 50.5%) for granulocytes (weeks 7-26; Figure 5B and Table 2) and 9.4%-71.0% (median, 28%) for mononuclear cells (weeks 4-26; Figure 5C and Table 2). Three of the 6 dogs demonstrated graft rejection, at weeks 9, 22, and 24 respectively. The remaining 3 were stable respectively mixed donor/host chimeras for >26 weeks of follow-up. The median durations of engraftment in the 6 dogs were significantly greater than those in 11 historical controls [1, 2] not given pretransplantation treatment (P = .0014; log-rank test; Table 1 and Figure 6) but were shorter than those achieved with 2-Gy TBI. There were no statistically significant differences between the current results and those of 3 regimens that used treatment before TBI with antithymocyte globulin, donor PBMCs, and methotrexate, or donor PBMCs and CTLA4-Ig (Table 1). The 50% cumulative rate of engraftment was significantly higher than the 0% rate in historical controls that received no pretransplantation treatment (P = .03; Fisher’s exact test; Table 1). High granulocyte chimerism levels at week 6 correlated significantly with sustained engraftment (Spearman’s rank correlation coefficient = 0.8783; P = .033).
Figure 5.
Hematologic changes in 6 dogs receiving DLA-identical littermate marrow grafts. (A) Median neutrophil, lymphocyte, and platelet counts. (B) Donor granulocyte chimerism levels. (C) Donor mononuclear cell chimerism levels.
Figure 6.
Kaplan-Meyer estimates of graft survival in the 6 dogs pretreated with anti-CD154 mAb 5c8 and donor PBMCs and 11 control dogs that did not receive pretransplantation treatment. All dogs were conditioned with 1-Gy TBI, received marrow grafts from DLA-identical littermates, and were given MMF or rapamycin for 28 days and CSP for 35 days posttransplantation.
The hematologic and nonhematologic toxicities were mild and comparable to those seen in previous studies using 1-Gy TBI and postgrafting MMF and CSP [1, 3, 13]. The first dog showed allergic reactions to the mAb infusion, which resolved with diphenylhydramine and hydration. Therefore, subsequent dogs received diphenylhydramine 2.5 mg/kg IV 5 minutes before mAb infusion as prophylaxis against allergic reactions. One dog (G577) developed an intussusception that was likely related to CSP and required surgical correction and suspension of postgrafting immunosuppression for 24 hours. GVHD was not observed in any of the dogs.
Discussion
The current study demonstrated that pretreatment of recipients with the anti-CD154 mAb 5c8 and donor PBMCs led to prolonged engraftment of DLA-identical marrow in dogs conditioned with 1-Gy TBI and treated with short courses of MMF and CSP after transplantation. However, sustained engraftment was seen only in half of the recipients. The results are similar to those seen in studies of mice [4, 5], in which uniform engraftment depended on both the strain combinations used and total marrow cell doses injected. A dose of 1 × 108 marrow cells (∼4 × 109 cells/kg) resulted in 100% engraftment even in the most resistant mouse strain combination, whereas injection of 0.5 × 108 marrow cells led to chimerism in only 68% of the mice [4]. Comparable marrow cell doses could not be achieved in dogs. Of note, the 2 dogs with the highest marrow cell doses (7.84 and 8.08 × 108 cells/kg) were among the 3 dogs that exhibited stable engraftment for >26 weeks.
Previous studies of marrow transplantation in mice [4, 5] and solid organ transplantation in nonhuman primates [15, 16] used higher total doses of anti-CD154 mAb than those administered in the current study. Only limited pharmacokinetic data for anti-CD154 mAb have been reported in the literature [17, 18]. Two studies examined the pharmacokinetics of humanized anti-CD154 mAbs (5c8 [17] and IDEC-131 [17, 18]) in monkeys. No antibody responses against these mAbs were detected in the primates. In both studies, the suppression of T-cell–dependent B-cell responses against soluble antigens (tetanus toxoid [17] or ovalbumin [17, 18]) were used to determine the in vivo efficacy of the antibodies. In both studies, single 5-mg/kg doses of anti-CD154 mAb effectively suppressed the antibody responses against the foreign antigens, although the humanized mAb 5c8 did not result in complete suppression, even at a mAb dose of 20 mg/kg. In the present study, complete MLR suppression was not observed even with high doses of mAb 5c8. We chose the smallest antibody dose for in vivo studies that achieved serum antibody levels >10 μg/mL for at least 5 days. This was realizable with a single mAb dose of 5 mg/kg IV, because the serum concentration of mAb 5c8 was 39 μg/mL even 5 days after injection.
All dogs in the pharmacokinetic studies and 4 of the 6 dogs in the transplantation study developed IgG antibody responses against mAb 5c8 beginning as early as 5 days after injection. The emerging canine antibodies likely determined the tempo of clearance of mAb 5c8 from the circulation. The development of IgG responses without preceding IgM responses was surprising, because the dogs had no known previous exposure to mouse immunoglobulin. In 2 of the 3 dogs analyzed, anti-idiotype or idiotype-like antibodies were found. Unlike the mice, the dogs were not maintained in pathogen-free conditions, which might have resulted in more educated immune systems [19, 20].
The results achieved with CD154 blockade in dogs were not significantly different than those previously seen with B7-CD28 blockade using CTLA4-Ig [3]; both studies failed to accomplish uniform sustained engraftment. Given the increasing number of newly discovered T-cell regulatory and costimulatory molecules [21], it seems likely that the blockade of only one system might not have been sufficient to overcome host resistance after a low-intensity or very-low-intensity conditioning regimen. Perhaps combining anti-CD154 mAb and CTLA4-Ig might be synergistic or additive and lead to more uniform sustained engraftment, as has been described in murine models of marrow and islet transplantation [22, 23, 24].
In conclusion, blocking the CD40–CD154 costimulatory signal was feasible and partially successful in establishing sustained allogenic canine marrow grafts after conditioning with only 1-Gy TBI. Canine-specific molecules and combinations of different costimulatory blockers may be required for more uniform success.
Acknowledgments
This work was supported in part by National Institutes of Health grants CA78902, CA15704, and AI067770. Additional support was provided by the Laura Landro Salomon Endowment Fund (R.S.), the Jose Carreras International Leukemia Foundation (R.S.), the Lupin Foundation (R.S.), and Deutsche Akademie der Naturforscher Leopoldina (grant BMBF-LPD 9901/8-63 to C.J.). We thank Drs. Roland Buelow and Elizabeth Squires (SangStat Medical Corporation, Fremont, CA) for the gift of oral cyclosporine, and Dr. Sabine Hadulco (Roche Biosciences, Nutley, NJ) for the gift of mycophenolate mofetil. We also thank Michele Spector, DVM, Alix Joslyn, and the technicians in the canine facilities of the Fred Hutchinson Cancer Research Center; Drs. Baron, Bethge, Burroughs, Diaconescu, Georges, Kiem, Nash, Mielcarek, and Sorror, who participated in the weekend treatments; Serina Gisburne, Sam Shin, and Patrice Stroup for performing DLA typing, Erlinda Santos for assisting with the MLR; and the technicians of the hematology and pathology laboratories of Seattle Cancer Care Alliance.
References
- 1.Storb R., Yu C., Wagner J.L. Stable mixed hematopoietic chimerism in DLA-identical littermate dogs given sublethal total body irradiation before and pharmacological immunosuppression after marrow transplantation. Blood. 1997;89:3048–3054. [PubMed] [Google Scholar]
- 2.Hogan W.J., Little M.-T., Zellmer E. Postgrafting immunosuppression with sirolimus and cyclosporine facilitates stable mixed hematopoietic chimerism in dogs given sublethal total body irradiation before marrow transplantation from DLA-identical littermates. Biol Blood Marrow Transplant. 2003;9:489–495. doi: 10.1016/s1083-8791(03)00148-4. [DOI] [PubMed] [Google Scholar]
- 3.Storb R., Yu C., Zaucha J.M. Stable mixed hematopoietic chimerism in dogs given donor antigen, CTLA4Ig, and 100-cGy total body irradiation before and pharmacologic immunosuppression after marrow transplant. Blood. 1999;94:2523–2529. [PubMed] [Google Scholar]
- 4.Seung E., Mordes J.P., Rossini A.A. Hematopoietic chimerism and central tolerance created by peripheral-tolerance induction without myeloablative conditioning. J Clin Invest. 2003;112:795–808. doi: 10.1172/JCI18599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Pan Y., Luo B., Sozen H. Blockade of the CD40/CD154 pathway enhances T-cell–depleted allogeneic bone marrow engraftment under nonmyeloablative and irradiation-free conditioning therapy. Transplantation. 2003;76:216–224. doi: 10.1097/01.TP.0000069602.30162.A1. [DOI] [PubMed] [Google Scholar]
- 6.Elster E.A., Hale D.A., Mannon R.B. The road to tolerance: renal transplant tolerance induction in nonhuman primate studies and clinical trials. Transplant Immunol. 2004;13:87–99. doi: 10.1016/j.trim.2004.05.010. [DOI] [PubMed] [Google Scholar]
- 7.Wagner J.L., Burnett R.C., DeRose S.A. Histocompatibility testing of dog families with highly polymorphic microsatellite markers. Transplantation. 1996;62:876–877. doi: 10.1097/00007890-199609270-00032. [DOI] [PubMed] [Google Scholar]
- 8.Wagner J.L., Works J.D., Storb R. DLA-DRB1 and DLA-DQB1 histocompatibility typing by PCR-SSCP and sequencing. Tissue Antigens. 1998;52:397–401. doi: 10.1111/j.1399-0039.1998.tb03063.x. [DOI] [PubMed] [Google Scholar]
- 9.Lederman S., Yellin M.J., Krichevsky A. Identification of a novel surface protein on activated CD4+ T cells that induces contact-dependent B-cell differentiation (help) J Exp Med. 1992;175:1091–1101. doi: 10.1084/jem.175.4.1091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Raff R.F., Deeg H.J., Farewell V.T. The canine major histocompatibility complex: Population study of DLA-D alleles using a panel of homozygous typing cells. Tissue Antigens. 1983;21:360–373. [PubMed] [Google Scholar]
- 11.Koyama I., Kawai T., Andrews D. Thrombophilia associated with anti-CD154 monoclonal antibody treatment and its prophylaxis in nonhuman primates. Transplantation. 2004;77:460–462. doi: 10.1097/01.TP.0000110291.29370.C0. [DOI] [PubMed] [Google Scholar]
- 12.Buhler L., Alwayn I.P., Appel J.Z., III Anti-CD154 monoclonal antibody and thromboembolism. Transplantation. 2001;71:491. doi: 10.1097/00007890-200102150-00028. [DOI] [PubMed] [Google Scholar]
- 13.Diaconescu R., Little M.-T., Leisenring W. What role is there for antithymocyte globulin in allogeneic nonmyeloablative canine hematopoietic cell transplantation? Biol Blood Marrow Transplant. 2005;11:335–344. doi: 10.1016/j.bbmt.2005.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yu C., Ostrander E., Bryant E. Use of (CA)n polymorphisms to determine the origin of blood cells after allogeneic canine marrow grafting. Transplantation. 1994;58:701–706. [PubMed] [Google Scholar]
- 15.Elster E.A., Xu H., Tadaki D.K. Treatment with the humanized CD154-specific monoclonal antibody, hu5C8, prevents acute rejection of primary skin allografts in nonhuman primates. Transplantation. 2001;72:1473–1478. doi: 10.1097/00007890-200111150-00001. [DOI] [PubMed] [Google Scholar]
- 16.Kirk A.D., Harlan D.M., Armstrong N.N. CTLA4-Ig and anti-CD40 ligand prevent renal allograft rejection in primates. Proc Natl Acad Sci U S A. 1997;94:8789–8794. doi: 10.1073/pnas.94.16.8789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gobburu J.V., Tenhoor C., Rogge M.C. Pharmacokinetics/dynamics of 5c8, a monoclonal antibody to CD154 (CD40 ligand) suppression of an immune response in monkeys. J Pharmacol Exp Ther. 1998;286:925–930. [PubMed] [Google Scholar]
- 18.Brams P., Black A., Padlan E.A. A humanized anti-human CD154 monoclonal antibody blocks CD154–CD40-mediated human B-cell activation. Int Immunopharmacol. 2001;1:277–294. doi: 10.1016/s1567-5769(00)00020-5. [DOI] [PubMed] [Google Scholar]
- 19.Adams A.B., Pearson T.C., Larsen C.P. Heterologous immunity: an overlooked barrier to tolerance. Immunol Rev. 2003;196:147–160. doi: 10.1046/j.1600-065x.2003.00082.x. [DOI] [PubMed] [Google Scholar]
- 20.Adams A.B., Williams M.A., Jones T.R. Heterologous immunity provides a potent barrier to transplantation tolerance. J Clin Invest. 2003;111:1887–1895. doi: 10.1172/JCI17477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Clarkson M.R., Sayegh M.H. T-cell costimulatory pathways in allograft rejection and tolerance. Transplantation. 2005;80:555–563. doi: 10.1097/01.tp.0000168432.60022.99. [DOI] [PubMed] [Google Scholar]
- 22.Wekerle T., Kurtz J., Ito H. Allogeneic bone marrow transplantation with co-stimulatory blockade induces macrochimerism and tolerance without cytoreductive host treatment. Nat Med. 2000;6:464–469. doi: 10.1038/74731. [DOI] [PubMed] [Google Scholar]
- 23.Wekerle T., Sayegh M.H., Ito H. Anti-CD154 or CTLA4Ig obviates the need for thymic irradiation in a non-myeloablative conditioning regimen for the induction of mixed hematopoietic chimerism and tolerance. Transplantation. 1999;68:1348–1355. doi: 10.1097/00007890-199911150-00022. [DOI] [PubMed] [Google Scholar]
- 24.Jin Y.Z., Xie S.S. Bicistronic adenovirus-mediated gene transfer of the CTLA4Ig and CD40Ig genes results in indefinite survival of islet xenograft. Transplant Proc. 2003;35:3165–3166. doi: 10.1016/j.transproceed.2003.10.064. [DOI] [PubMed] [Google Scholar]
- 25.Jochum C., Beste M., Zellmer E. Donor-specific mononuclear cell transfusion and methotrexate as pretransplant treatment in dogs given DLA-identical marrow grafts after nonmyeloablative conditioning. Biol Blood Marrow Transplant. 2006;12:885–886. doi: 10.1016/j.bbmt.2006.04.002. [letter] [DOI] [PubMed] [Google Scholar]