Abstract
Background
Use of antimicrobials like sulfonamides in production of layers is a public health risk since it inevitably results in sulfonamide residues in eggs. The presence of the residues may be influenced by knowledge, attitudes and practices of farmers regarding use of sulfonamides (and other antimicrobials) in poultry.
Objective
The study aimed at assessing the possible contribution of the knowledge, attitudes and practices of poultry farmers to the presence/levels of sulfonamide residues in hen eggs.
Methods
A descriptive cross sectional study was done in the 5 political divisions (and surroundings) of Kampala district. Sixty farmers were systematically sampled from a list of poultry farmers in Kampala and a semi-structured questionnaire administered. Each farmer provided sixty eggs for analysis of sulfadiazine and sulfamethazine residues. Whole eggs were homogenized in acetonitrile and centrifuged twice, extracts evaporated and residues dissolved in mobile phase (32:68, methanol: potassium dihydrogen phosphate). Fats were removed using hexane while anhydrous sodium chloride was added to break emulsions. Extracts were analyzed by reverse phase high performance liquid chromatography with photodiode array detector.
Results
Ninety-five percent of the farmers never observed withdrawal periods although 80% of them knew the importance of withdrawal periods. However, farmers noted that they play a great role in ensuring a safe food supply. Most farmers attributed the non-observance of withdrawal periods to poverty and fear to lose their investments. Ninety-eight percent of the samples had detectable levels of the sulfonamides. Meanwhile, 98.3% of the samples that had detectable sulfonamide residues came from farmers who applied antimicrobials in feeds/ water.
Conclusion
Consumers of hen eggs in Kampala district are at high risk of sulfonamide residue exposure due to poor farming/regulatory practices.
Keywords: Farmers' knowledge, attitudes and practices, residues, sulfonamides, Kampala
Introduction
Most of the estimated 23–25 million poultry and their products in Uganda provide an important source of human nutrition to the country. Farmers use various antimicrobial agents with or without the guidance of veterinarians to prevent or treat diseases and enhance production. Approximately half of the antimicrobials produced today are used in human medicine and most of the remainder is used in animal production1. The increased use of antimicrobials may be associated with misuse and, therefore, increased risk of human exposure to harmful residues.
Farming practices in Uganda have not reached a level where primary health care, through extension services, can contain major epizootics. Therefore, control of animal diseases depends heavily on the use of drugs and vaccines, and about 10% of these drugs imported into Uganda are sulfonamides2. Sulfonamides are a diverse class of chemically related compounds and many like sulfamethazine (SMZ), sulfadiazine (SDZ), sulfaquinoxaline and sulfamethoxypyridazine have found widespread use in animal production3,4. Unfortunately, about 10–15% of individuals in a population, exhibit allergic reactions to antimicrobials especially penicillin and sulfonamides5. Furthermore, antimicrobial resistance may occur if antimicrobials are misused and the most common case of drug resistant bacteria that can infect humans is Salmonella, responsible for epidemics of Salmonellosis6,7.
Since administration of any chemically active pharmaceutical to food producing animals inevitably leads to presence of residues in foods from such animals, national and international legislation seeks to ensure that consumers of animal food products are not exposed to potentially harmful concentrations8. Expert bodies like Codex Alimentarius9, Joint FAO/WHO Expert Committee on Food Additives10, and the European Union have set a series of maximum residue limits (MRL) on edible tissues. For example, the MRL for all sulfonamides combined is 100µg/kg in muscle, liver, kidney and fat except eggs that lack sulfonamide MRL10. The purpose of the MRL is to limit the exposure of consumers to residues of medicines used in food animals, to concentrations that do not pose human health risk8.
If a licensed drug is used in accordance with its product license, and if the withdrawal periods are observed, harmful residues should not occur in human food. Therefore, good farming practices (which are associated with farmers' knowledge and attitudes) are paramount in ensuring food safety and public health. The purpose of this study was to assess the knowledge, attitudes and practices of poultry farmers in Kampala on sulfonamide use, and the possible contribution to residues (which were also analyzed) in animal foods. The assignment of a single MRL to all the sulfonamides (unlike other veterinary drugs) may pose serious health risks to consumers of animal products especially when sulfonamides are misused. The risk is even bigger if the sulfonamides are not recommended for use in certain food animals like laying hens.
Materials and Methods
A descriptive cross-sectional study was conducted in Kampala, district, Uganda from December 2002 to March 2003 to collect data from sixty poultry farmers, using a semi-structured questionnaire. Sample size determination and sample collection were according to Codex Alimentarius guidelines9 without regard of the Delphi technique11,12 Contrary to this study's objective, the Delphi technique principally emphasizes consensus-seeking using respondents with expert knowledge and it rarely involves face-to-face interaction12. The authors visited poultry and veterinary drug dealers in Kampala to establish contact with farmers in the district and its immediate surroundings. A list of layer farmers in Kampala district and the surroundings was drawn to include farms that regularly supplied eggs for consumption to Kampala district. The authors then used a systematic sampling technique to determine the sixty respondents who also provided 60 eggs each for residue analysis using high performance liquid chromatographic (HPLC).
Residue analysis
Preparation of mobile phase and drug standards
A thirty two percent mobile phase {320 mls methanol with potassium di-hydrogen phosphate (PDP) 0.1M solution added until the 1L mark} was prepared at least 1 hour before HPLC analysis or sonicated (using a 1510 Branson Sonicator) 15 minutes for immediate use. Stock drug standards (1mg/kg in methanol), mixed and separate intermediate standard solutions (10µg/ml in de-ionized water), fortification solutions (1µg/ml in water) and working standards (10, 50, 200, 300, 400 and 1000ng/ml in water) were also prepared for the HPLC analysis
Sample extraction and HPLC analysis
A liquid-liquid extraction method similar in principle to methods described by Shaikh et al13 and Howitz14 was used. Five milliliters of pre-homogenized eggs, 1ml PDP and 20ml of acetonitrile were homogenized (using an ultaturrax homogenizer) for 20 seconds after vigorous vortexing (Using a Hook and tucker rota mixture). The homogenate was centrifuged at 4100 relative centrifugal force at 4°C for 15 minutes. The supernatant was transferred into a 250ml flask, the pellet loosened and rehomogenized with 10ml acetonitrile and the mixture recentrifuged. The second supernatant was added to the first and the mixture evaporated just to dryness on a rotavapor at 35°C.
The residue was dissolved in 1ml mobile phase and 8mls of hexane added into the flask. The combination of the residue, mobile phase and hexane was re-centrifuged (in Scott Duran glass, Germany). Hexane layer was discarded. One gram of anhydrous NaCl was added to the lower aqueous layer, the mixture vortexed and re-centrifuged. The aqueous layer was filtered using Whatman 0.45µm nylon filter into small amber colored bottles for HPLC analysis.
The HPLC system (Gilson 1997) used consisted of Gilson pumps 305, 306, a manometric module model 806 and a dynamic mixture model 811C. The detector (spectral System detector, thermo-separation products, USA) had a lamp model UV6000LP. Detector run time was 10 minutes and scan wavelength was 250nm–298nm. The LC column was a Supelcosil, LC-18; length 2.5cm and diameter 4.6mm and particle size 12µm. The mobile phase flow rate was 1.3mls/min. System suitability was checked by injecting 10 samples spiked with sulfonamide standards (to determine the limit of detection) and concentrations of the sulfonamides as low as 1µg/kg were detected. Samples were injected in sequences sandwiched by groups of the sulfonamide standards.
Principle of the analytical procedure
Homogenized whole egg is extracted with PDP and acetonitrile. A portion of the extract is evaporated just to dryness using a rotavapour at 35°C, the residues dissolved in the isocratic mobile phase (methanol and PDP 32:68), and washed with hexane to remove the fats. The extract is analyzed without further cleanup by reverse phase HPLC. A photodiode array detector at wavelength 265nm is used to detect SDZ and SMZ.
Results
Knowledge, attitudes and practices
The minimum number of layers in the study was 80 and the maximum was 16,700. The mean was 2032. Fifty seven (95%) of the respondents experienced disease in the flocks, 3 (5%) did not experience diseases. While 17 (36%) respondents could not remember the frequency of disease occurrence in the flocks, 12 (20%) experienced diseases twice a year; 9 (15%) experienced 6 or 7 times a year, while 4 (6.7%) experienced the diseases 4 times a year.
Thirty two (53%) of the respondents said veterinarians treated the diseases in the flock, while 24 (40%) treated their flock for diseases. Only 1 farmer said s/he prevented diseases and never treated the birds because of improved management and regular vaccinations. Meanwhile, 30 (65%) of the respondents purchased and stocked sulfas among other drugs but 11 (23%) could not remember the antimicrobials used. Three sulfonamides namely sulfadiazine, sulfamethazine and sulfaquinoxaline were the commonest therapeutic and prophylactic preparations purchased by poultry farmers (according to preliminary interactions of the authors with selected drug sellers in Kampala). Thirty (50%) of the respondents administered sulfas in feed or water but 16 (27%) could not remember the antimicrobials they used.
Fifty seven (95%) of the respondents sold or ate their eggs immediately after use of veterinary drugs in the flock for treatment or prophylaxis. Fifty one (85%) of the respondents knew withdrawal periods for antimicrobial agents including (sulfas), while the rest of the respondents had no knowledge. Farmers were interviewed on the importance of withdrawal periods and the public health significance Half of the farmers thought drug residues in foods had economic implications. Respondents were also interviewed on the responsibilities of ensuring a safe/residue free food supply, and the responses are shown in table 1 below.
Table 1.
Farmer's response: People and/or bodies responsible for regulation of drug residues in animal food products
| Frequency | Percent | |
| All stakeholders | 9 | 15.0 |
| Farmerse | 16 | 26.7 |
| Veterinarians | 7 | 11.7 |
| Veterinarians and farmerse | 6 | 10.0 |
| Government (Uganda National Bureau of Statistics, National Drug Authority Local, Centre) and farmers | 3 | 5.0 |
| Drug dealers/manufacturers and other stakeholders | 4 | 6.7 |
| NDA or UNBS alone | 3 | 5.0 |
| Government (Ministries of Health and Agriculture) alone | 9 | 15.0 |
| Scientists | 1 | 1.7 |
| Don't know | 2 | 3.3 |
| Total | 60 | 100.0 |
Farmers were further interviewed on what they considered constraints in the regulation of veterinary drug residues in foods of animal origin and the responses are shown in table 2 below
Sulfonamide residues
Linear regression coefficients, using the least square regression were calculated for each sulfonamide to obtain the working equation Y = mX + b (Y = height of sample at the corresponding sulfonamide retention time; m = slope of the standard curve; and b = intercept; X = sulfonamide concentration of unknown sample). X was calculated by comparing the peak height of the sulfonamide standards injected before and after the study samples, at the corresponding retention times. The final concentration calculated was divided by a concentration factor. For example, if a 5ml sample was homogenized, the equivalent concentration of the drug residues would be determined by dividing the final concentration by 5. This is because the residue was dissolved in 1ml of the mobile phase so that the original sample present for extraction was concentrated by a factor of 5. Recovery test results done at 50µg/kg were 81% for SDZ and 81.5% for SMZ with standard deviations of 5.73 and 5.85 respectively. The relative standard deviations were 7.07 and 7.19 respectivel.
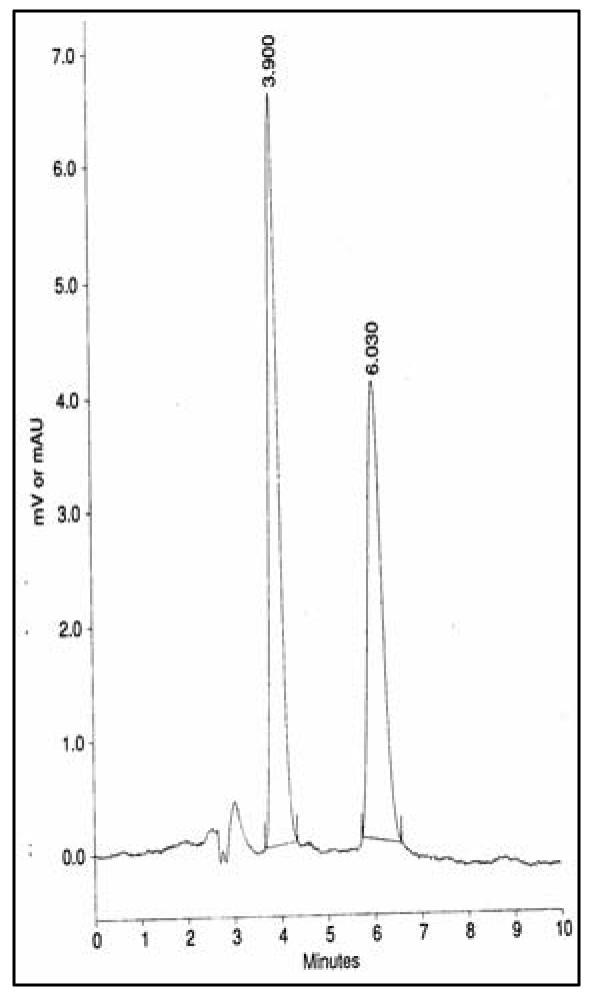
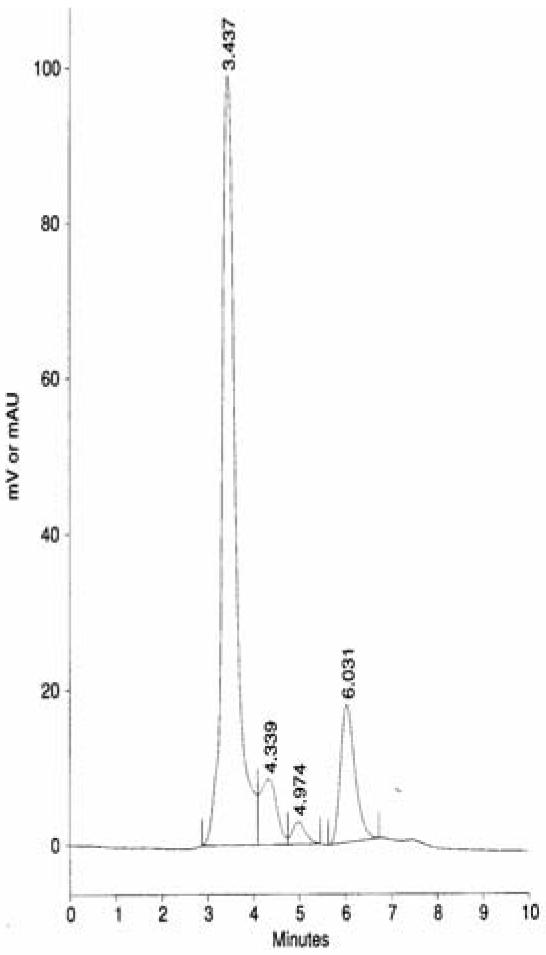
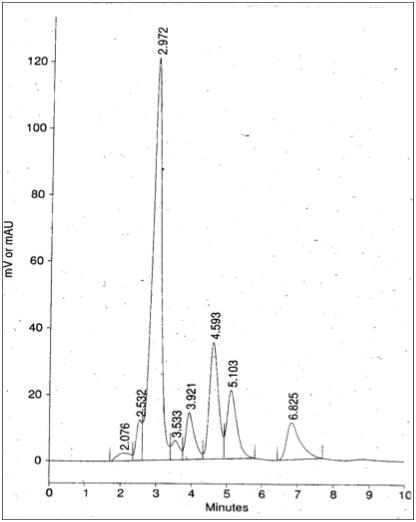
Selected chromatograms:
Fig 2: is chromatogram for drug standards SDZ (at retention time 3.9 minutes) and SMZ (retention time 6.0 minutes)
Fig 3 is chromatogram of sample with SMZ residue (peak detected at retention time 6.031 minutes)
Fig 4 is chromatogram of a sample with SDZ residue (peak detected at retention time 3.921 minutes)
Discussion
Since veterinary drugs can be administered by farmers in animal feeds/water for livestock production, a totally risk/residue-free food production system does not exist15,16. While this study could not verify use of antimicrobials for growth purposes, it was able to report therapeutic/prophylactic use of antimicrobials by most (58) farmers. Maximum recommended residue levels have been fixed for authorized drugs to ensure consumer safety and such MRLs would not be exceeded if good veterinary practices (GVPs) were used9. This observation is in agreement with the study's findings since the farmers confessed failure to adhere to GVPs and indeed most samples analyzed had detectable residues.
Although according to Speedy16 the assessment and containment of public health risks in livestock products is a matter of priority, such commitment was not evident in Kampala based on the farmers' attitude regarding the importance of GVPs. For example, most farmers where concerned with costs of production rather than public health risks and therefore could not think of discarding their eggs for purposes of observing withdrawal periods.
The intensification of livestock production and the increasing trade in livestock products demands a rigorous risk management program by all countries16. Unfortunately such a program is not evident in Kampala according to the farmers interviewed. While in developed countries like USA, governmental bodies like the United States Department of Agriculture (USDA) have a well developed chemical risk management program17, in Uganda the Ministry of Agriculture Animal Industry and Fisheries (MAAIF) and National Drug Authority (NDA) are yet to fully embrace a similar program18. Indeed farmers in this study said government had not addressed the issue of regulating drug residues in animal products. Treatment of an entire flock or herd with antimicrobials even in low concentrations and over long periods for prevention/treatment of disease or performance enhancement significantly increases antimicrobial resistance, which is a global problem19, 20. While this study could not show association of antimicrobial resistance (in humans and animals) and use of drugs, failure of the farmers to follow GVPs in the study predisposes to development of drug resistant organisms in Kampala. This is in view of the fact that most farmers reported extensive use of antimicrobials while disregarding withdrawal periods. Furthermore, failure by 95% of the farmers to observe withdrawal periods is clear indication of failure to observe GVPs. Most farmers interviewed complained of poverty, therefore, they could not afford to discard their eggs while observing withdrawal periods.
The emergence of a multi-drug resistant strain of Salmonella typhimurium DT 104 has caused a great deal of concern globally7. This organism is resistant to 5 antimicrobials including sulfonamides. Although misuse of antibiotics in human medicine is a big contributor to development of resistance, agriculture's contribution (through poor farming practices) is not dismissible15. Since GVPs are not strictly adhered to in Kampala, and since the study findings proved presence of sulfonamides (sulfadiazine and sulfamethazine) in the samples analyzed, the emergence or existence of Salmonella typhimurium DT 104 resistance in Uganda cannot be ruled out.
Residues can also lead to allergies20 a fact that farmers in this study knew. Unfortunately, with this knowledge, farmers in Kampala continued to ignore GVPs. Even in the case of products (like milk, meat) with sulfonamide MRLs10, failure to follow GVPs is a health risk. Absence of sulfonamide MRL for eggs implies that sulfas are not authorized for use in the layers and such drugs should not even be purchased for use in layers10. Therefore, use of sulfonamides in laying hens is a greater public health risk. Unfortunately, farmers in Kampala acknowledged purchase and use of sulfonamides for layer farming. Furthermore, residues of sulfamethazine and sulfadiaziane where detected in this study some of which exceeded 100µg/kg (a sulfonamide MRL in animal products other than eggs). This shows a very high public health risk.
Farmers in this study believed in multidisciplinary efforts to control use of antimicrobials so as to contain possible development of drug resistance and other sulfonamide-residue toxicities. However, the study indicates a poor definition of roles among residue regulators (like health inspectors and veterinarians) and that this poor definition has compromised the delivery of quality service through the multidisciplinary approach.
FAO21 defines the food chain approach as recognition that the responsibility for ensuring a safe, healthy and nutritious food supply is shared along the entire food chain (from farm to table) by all those involved in the production, processing, trade and consumption of food. Most respondents suggested that farmers were mainly responsible for a safe food supply, besides other stakeholders in the food chain. The study findings are thus in line with the FAO's food chain approach.
Implementation of this farm-to-table approach to food safety requires enabling policies and regulatory environment at national and international level with clear definition of roles. Although the study findings could not show participation/non-participation of relevant international bodies like FAO/WHO, farmers clearly emphasized the expected role of national bodies. According to farmers, government lacks vigilance and the desire needed to ensure food quality control yet this is necessary to ensure public health22.
Failure to attain international food safety standards (like low/no residue foods) usually results in significant financial losses for food exporting countries10,22. While Uganda intends to join the global market in the trade of food animal products (with Kampala benefiting), immediate realization of this wish requires a lot of national effort in food safety and veterinary drug residues. This is because government (MAAIF, NDA) is yet to enforce food quality standards, while farmers consider poverty and high egg production costs as the major constraints to ensuring food quality.
According to FAO21 governments are obliged to set, impose and control food safety standards in accordance with the Rome Declaration of world food security 1996, that “All people have the right to safe-food what ever the levels of their effective demand”. Respondents in the study emphasized the need for the Uganda government to comply with the Rome Declaration on safe-food supply. The farmers in this study proposed subsidized farming like government's will to compensate farmers (if foods have unacceptable residue levels and need discarding), as a way to ensuring safe-food supply. In this way, the issue of high production costs would be addressed and the global market easily accessed subsequently.
Conclusions
Consumers of animal products like eggs in Kampala district are at high risk of exposure to high and non-authorized drug residues in eggs in view of the knowledge, attitudes and practices of farmers in the district on use of antimicrobials and in view of the presence of sulfamethazine and sulfadiazine residues in the eggs. Such harmful effects as allergy and drug resistance are potential health risks to both local and international consumers. There is, therefore, need to massively educate farmers (and other stakeholders) about the public health significance of good farming practices.
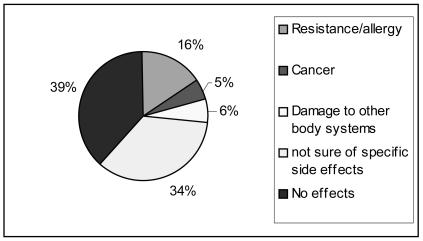
Fig 1.
Farmers' response on effect of drug residues in eggs (and other foods)
Table 2.
Farmer's reponse on: constraints in the regulation of drug residues in animal food products, identified by farmers
| Frequency | Percent | |
| Lack of cooperation | 14 | 23.3 |
| Lack of vigilance and poor facilitation | 11 | 18.3 |
| Ignorance of the importance of regulation | 4 | 6.7 |
| Poverty | 8 | 13.3 |
| Greed for money | 5 | 8.3 |
| Don't know, not sure | 6 | 10.0 |
| Ignorance and poverty | 8 | 13.3 |
| Lack of facilitation and high costs of production | 4 | 6.7 |
| Total | 60 | 100.0 |
Fifty nine (98.3%) of the 60 samples analyzed showed detectable levels of SDZ and SMZ (ranging from 6–992µg/kg). No SDZ and SMZ residues were detected in 1 (1.7%) sample. Onlythree (5%) of the 60 samples had SDZ and SMZ residues above 100µg/kg, the MRL for sulfas in foods other than eggs. Of all the detectable SDZ and SMZ residue levels, 41 (70%) were SDZ and 18 (30%) SMZ.
All samples from farmers who never observed withdrawal periods had detectable sulfonamide residues (Table 3). Three respondents who said they observed withdrawal period had samples with detectable sulfonamide residues. Meanwhile, sulfonamide residues where detected in samples provided by fifty eight (98.3%) of the farmers who administered antibiotics in water/feed.
References
- 1.WHO, author. The use of antimicrobials outside human medicine. 2001. [Google Scholar]
- 2.Ministry of Agriculture, Animal Industry and Fisheries/MAAIF, author. Annual Veterinary Report. Department of veterinary services epidemiological Unit; 1995. [Google Scholar]
- 3.Tarbin JA, Clarke P, Shearer G. Screening of Sulfonamides in eggs using gas chromatography-mass selective detection and liquid chromatography-mass spectrometry. J of Chromatogr. 1999;729:127–138. doi: 10.1016/s0378-4347(99)00142-5. [DOI] [PubMed] [Google Scholar]
- 4.Paige JC, Pell FBS. Drug residues in Food Producing Animals. FDA Veterinarian Newsletter. 1997 Jul–Aug;XII(IV) 1997. [Google Scholar]
- 5.Cochrane B, Doyle E M, Steinhart C E. Food Safety. New York, USA: 1995. p. 247. [Google Scholar]
- 6.Holmberg SD, Osterholm MT, Senger KA, Cohen ML. Drug resistant Salmonella from animals fed antimicrobials. New Eng J Med. 1984;31:617. doi: 10.1056/NEJM198409063111001. [DOI] [PubMed] [Google Scholar]
- 7.Threlfall EJ, Frost JA, Ward LR, Rowe B. In use of drugs in food animals, benefits and risks. Vol. 134. Washington D.C: National academic press; 1999. Epidemic in cattle and humans of Salmonella typhimurium DT 104 with chromosomally integrated multiple drug resistance veterinary record 1994; p. 577. [DOI] [PubMed] [Google Scholar]
- 8.Kennedy GD, Cannavan A, Mccracken RJ. Regulatory problems caused by contamination, a frequently overlooked cause of veterinary residue. J of Chromatogr A. 2000;882:37–52. doi: 10.1016/s0021-9673(00)00320-4. [DOI] [PubMed] [Google Scholar]
- 9.Residues of veterinary drugs in foods. 2nd edition. Rome: 1994. Codex Alimentarius. [Google Scholar]
- 10.Joint Fao/Who Expert Committee On Food Additives, author. A second meeting of the Joint FAO/WHO Expert Committee on Food Additives. 1992
- 11.Shaikh B, Rummel N, Donoghue D. Determination of sulfamethazine and its metabolites in egg albumin and yolk by HPLC. J liq Crom and Rel Technol. 1999;22(170):2651–2662. [Google Scholar]
- 12.Howitz W. Official Methods of analysis of AOAC international. 17th edition Vol. 1. 2000. [Google Scholar]
- 13.Fink A, Kosecoff J, Chassin M, Brook RH. Consensus methods: Characteristics and guidelines for use. American Journal of Public Health. 1984;74:979–983. doi: 10.2105/ajph.74.9.979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bowles N. The Delphi techniques. Nursing Standards. 1999;13(4):32–36. doi: 10.7748/ns1999.07.13.45.32.c2650. [DOI] [PubMed] [Google Scholar]
- 15.Mlot C. Antidotes for antibiotic use on the farm livestock; antibiotics in animal nutrition. drug resistance in microorganisms; agriculture. Bioscience. 2000;150:955. [Google Scholar]
- 16.Speedy WA. WHO Consultation on pre-harvest food safety. Berlin: 2001. pp. 26–28. [Google Scholar]
- 17.United States Department of Agriculture, author. Egg products Inspection Act title 21 food and drugs. 2002 Chapter 15.
- 18.The National Veterinary Drug Policy, MAAIF, UGANDA. 2002
- 19.Fedorka P J, Linda T, Reeves D. Manitoba Agricultural Food News. Canada: 2001. Antimicrobial dilemma in Animal Production. [Google Scholar]
- 20.COFFMAN RJ, BERAN GW. Committee on drug use in food animal. Panel on animal health, Food Safety and Public Health. Washington D.C: National academic press; 1999. Use of drugs in food animals, Benefits and Risks; pp. vii–87. [Google Scholar]
- 21.FAO/WHO/WTO/Codex, author. FAO Food law news. 2003 Mar 31;
- 22.WHO, author. Food Safety and the Globalization of trade, a challenge to the public health sector. 1998 WHO/FSF/FOS/97.8.rev.1.