Abstract
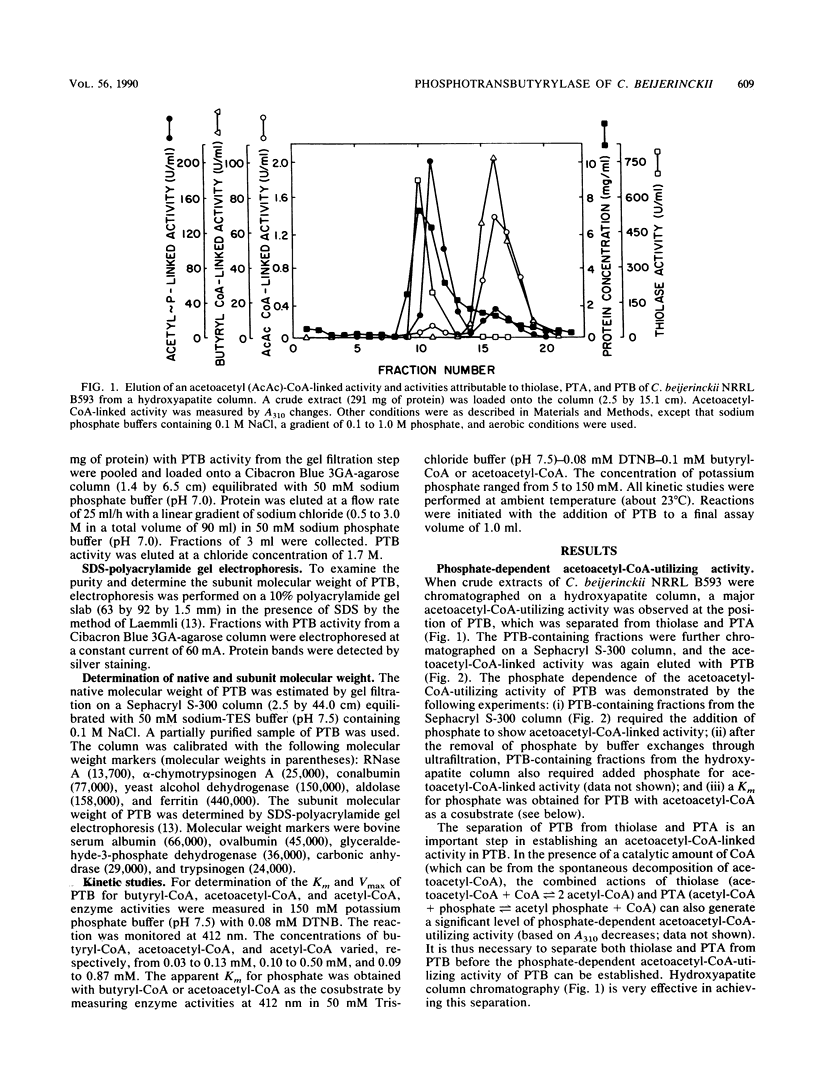
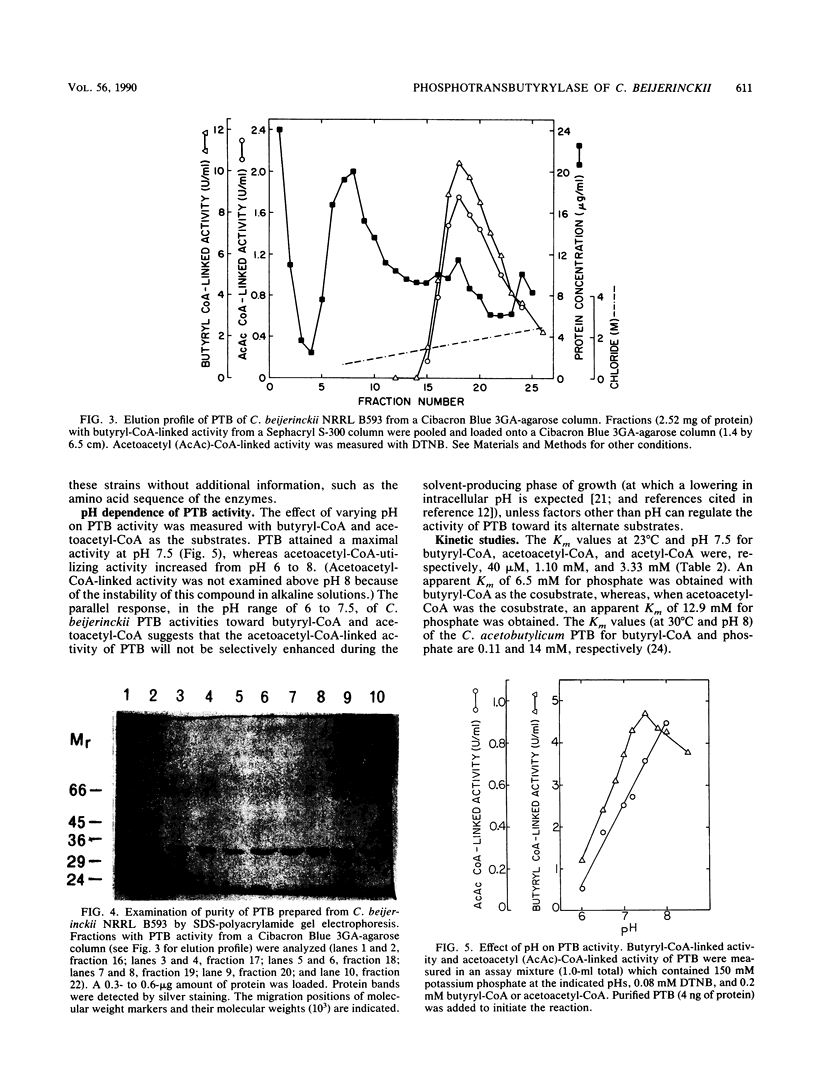
During the study of acetoacetyl coenzyme A (CoA)-reacting enzymes of Clostridium beijerinckii NRRL B593, a phosphate-dependent acetoacetyl-CoA-utilizing activity was detected in protein fractions devoid of thiolase and phosphotransacetylase. Further purification of this acetoacetyl-CoA-utilizing activity yielded an enzyme which may be designated as phosphotransbutyrylase (PTB; phosphate butyryltransferase [EC 2.3.1.19]). PTB from C. beijerinckii NRRL B593 was purified 160-fold with a yield of 14% and, with the best fractions, purified 190-fold to near homogeneity. It showed a native Mr of 205,000 and a subunit Mr of 33,000. PTB activity was sensitive to pH changes within the physiological range of 6 to 8. PTB exhibited a broad substrate specificity. The Km values at pH 7.5 for butyryl-CoA, acetoacetyl-CoA, and acetyl-CoA were 0.04, 1.10, and 3.33 mM, respectively. The Vmax values with butyryl-CoA and acetoacetyl-CoA were comparable, but the Vmax/Km was higher for butyryl-CoA than for acetoacetyl-CoA. An apparent Km of 6.5 mM for phosphate was obtained with butyryl-CoA as the cosubstrate, whereas it was 12.9 mM with acetoacetyl-CoA as the cosubstrate. It remains to be established whether the putative compound acetoacetyl phosphate is produced in the PTB-catalyzed reaction with acetoacetyl-CoA.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BERGMEYER H. U., HOLZ G., KLOTZSCH H., LANG G. PHOSPHOTRANSACETYLASE AUS CLOSTRIDIUM KLUYVERI. ZUECHTUNG DES BACTERIUMS, ISOLIERUNG, KRISTALLISATION UND EIGENSCHAFTEN DES ENZYMS. Biochem Z. 1963;338:114–121. [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Cary J. W., Petersen D. J., Papoutsakis E. T., Bennett G. N. Cloning and expression of Clostridium acetobutylicum phosphotransbutyrylase and butyrate kinase genes in Escherichia coli. J Bacteriol. 1988 Oct;170(10):4613–4618. doi: 10.1128/jb.170.10.4613-4618.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ELLMAN G. L. Tissue sulfhydryl groups. Arch Biochem Biophys. 1959 May;82(1):70–77. doi: 10.1016/0003-9861(59)90090-6. [DOI] [PubMed] [Google Scholar]
- GAVARD R., HAUTECOEUR B., DESCOURTIEUX H. Phosphotransbutyrylase de Clostridium acetobutylicum. C R Hebd Seances Acad Sci. 1957 Apr 29;244(18):2323–2326. [PubMed] [Google Scholar]
- George H. A., Chen J. S. Acidic Conditions Are Not Obligatory for Onset of Butanol Formation by Clostridium beijerinckii (Synonym, C. butylicum). Appl Environ Microbiol. 1983 Aug;46(2):321–327. doi: 10.1128/aem.46.2.321-327.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hartmanis M. G. Butyrate kinase from Clostridium acetobutylicum. J Biol Chem. 1987 Jan 15;262(2):617–621. [PubMed] [Google Scholar]
- Hartmanis M. G., Gatenbeck S. Intermediary Metabolism in Clostridium acetobutylicum: Levels of Enzymes Involved in the Formation of Acetate and Butyrate. Appl Environ Microbiol. 1984 Jun;47(6):1277–1283. doi: 10.1128/aem.47.6.1277-1283.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones D. T., Woods D. R. Acetone-butanol fermentation revisited. Microbiol Rev. 1986 Dec;50(4):484–524. doi: 10.1128/mr.50.4.484-524.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Robinson J. R., Sagers R. D. Phosphotransacetylase from Clostridium acidiurici. J Bacteriol. 1972 Oct;112(1):465–473. doi: 10.1128/jb.112.1.465-473.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- STERN J. R. Optical properties of aceto-acetyl-S-coenzyme A and its metal chelates. J Biol Chem. 1956 Jul;221(1):33–44. [PubMed] [Google Scholar]
- Sargent M. G. Fiftyfold amplification of the Lowry protein assay. Anal Biochem. 1987 Jun;163(2):476–481. doi: 10.1016/0003-2697(87)90251-x. [DOI] [PubMed] [Google Scholar]
- Senior P. J., Dawes E. A. The regulation of poly-beta-hydroxybutyrate metabolism in Azotobacter beijerinckii. Biochem J. 1973 May;134(1):225–238. doi: 10.1042/bj1340225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Srere P. A. Complexes of sequential metabolic enzymes. Annu Rev Biochem. 1987;56:89–124. doi: 10.1146/annurev.bi.56.070187.000513. [DOI] [PubMed] [Google Scholar]
- Terracciano J. S., Kashket E. R. Intracellular Conditions Required for Initiation of Solvent Production by Clostridium acetobutylicum. Appl Environ Microbiol. 1986 Jul;52(1):86–91. doi: 10.1128/aem.52.1.86-91.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thauer R. K., Jungermann K., Decker K. Energy conservation in chemotrophic anaerobic bacteria. Bacteriol Rev. 1977 Mar;41(1):100–180. doi: 10.1128/br.41.1.100-180.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- VALENTINE R. C., WOLFE R. S. Purification and role of phosphotransbutyrylase. J Biol Chem. 1960 Jul;235:1948–1952. [PubMed] [Google Scholar]
- Wiesenborn D. P., Rudolph F. B., Papoutsakis E. T. Phosphotransbutyrylase from Clostridium acetobutylicum ATCC 824 and its role in acidogenesis. Appl Environ Microbiol. 1989 Feb;55(2):317–322. doi: 10.1128/aem.55.2.317-322.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan Run-Tao, Zhu Chang-Xi, Golemboski Christine, Chen Jiann-Shin. Expression of Solvent-Forming Enzymes and Onset of Solvent Production in Batch Cultures of Clostridium beijerinckii ("Clostridium butylicum"). Appl Environ Microbiol. 1988 Mar;54(3):642–648. doi: 10.1128/aem.54.3.642-648.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]




