Abstract
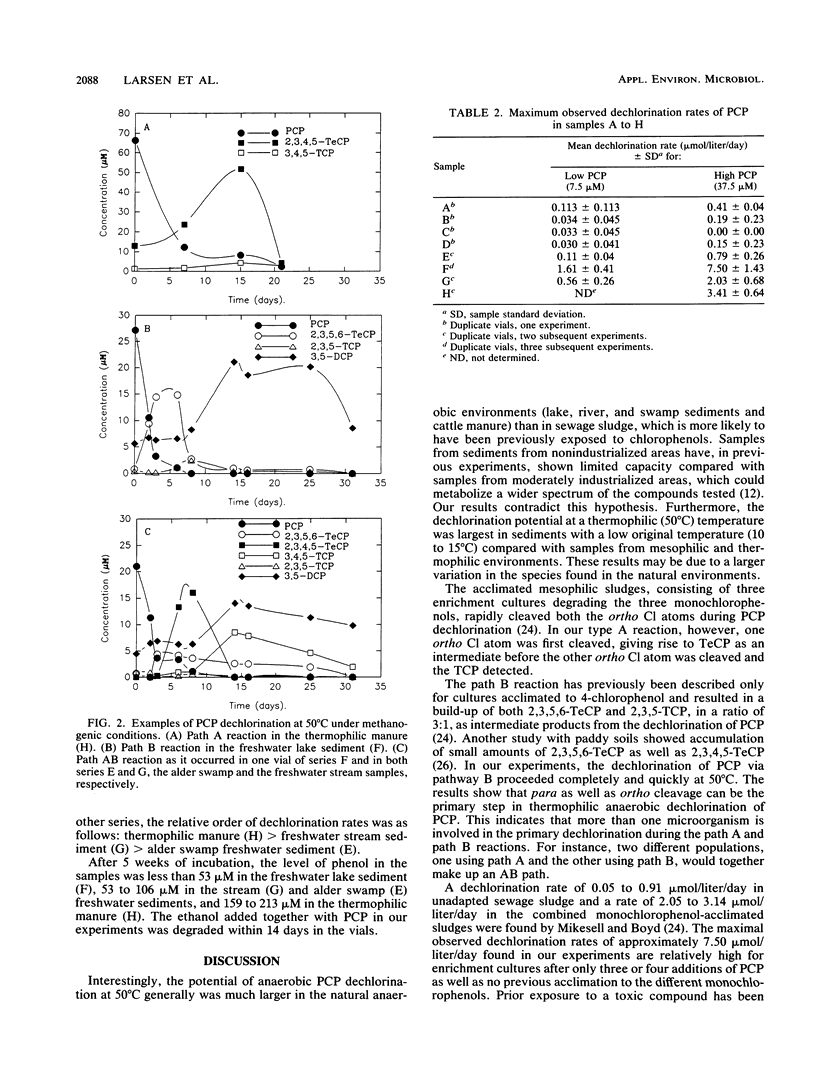
Thermophilic (50 degrees C) anaerobic biodegradation of pentachlorophenol (PCP) was investigated by using different inocula from natural ecosystems and anaerobic digesters. The inocula tested were three freshwater sediments, four anaerobic sewage sludge samples from digesters treating sludge from wastewater plants with various industrial inputs, and digested manure from an anaerobic reactor. Only one digested-sludge sample and the manure sample were from thermophilic environments. The initial PCP concentration was 7.5 or 37.5 microM. After 8 months, PCP had disappeared from the sediment samples and various, less chlorinated intermediates were present. Additions of extra PCP were degraded within 4 weeks, and a maximal observed dechlorination rate of 1.61 mumol/liter/day in the vials with addition of 7.5 microM PCP and 7.50 mumol/liter/day in the vials with addition of 37.5 microM PCP were measured for a freshwater sediment. In contrast, only 2.8 to 17.5% of the initial PCP added had disappeared from the sludge samples after 8 months of incubation. The complex pattern of intermediates formed indicated that the dechlorination of PCP proceeded via different pathways, involving at least two different populations in the dechlorination processes.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Angelidaki I., Petersen S. P., Ahring B. K. Effects of lipids on thermophilic anaerobic digestion and reduction of lipid inhibition upon addition of bentonite. Appl Microbiol Biotechnol. 1990 Jul;33(4):469–472. doi: 10.1007/BF00176668. [DOI] [PubMed] [Google Scholar]
- Attaway H. H., 3rd, Paynter M. J., Camper N. D. Degradation of selected phenylurea herbicides by anaerobic pond sediment. J Environ Sci Health B. 1982;17(6):683–699. doi: 10.1080/03601238209372350. [DOI] [PubMed] [Google Scholar]
- Boyd S. A., Shelton D. R. Anaerobic biodegradation of chlorophenols in fresh and acclimated sludge. Appl Environ Microbiol. 1984 Feb;47(2):272–277. doi: 10.1128/aem.47.2.272-277.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boyd S. A., Shelton D. R., Berry D., Tiedje J. M. Anaerobic biodegradation of phenolic compounds in digested sludge. Appl Environ Microbiol. 1983 Jul;46(1):50–54. doi: 10.1128/aem.46.1.50-54.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown M. P., Bush B., Rhee G. Y., Shane L. PCB dechlorination in Hudson River sediment. Science. 1988 Jun 17;240(4859):1674–1676. doi: 10.1126/science.3132740. [DOI] [PubMed] [Google Scholar]
- Dolfing J., Tiedje J. M. Growth yield increase linked to reductive dechlorination in a defined 3-chlorobenzoate degrading methanogenic coculture. Arch Microbiol. 1987;149(2):102–105. doi: 10.1007/BF00425073. [DOI] [PubMed] [Google Scholar]
- Fathepure B. Z., Tiedje J. M., Boyd S. A. Reductive dechlorination of hexachlorobenzene to tri- and dichlorobenzenes in anaerobic sewage sludge. Appl Environ Microbiol. 1988 Feb;54(2):327–330. doi: 10.1128/aem.54.2.327-330.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Genthner B. R., Price W. A., Pritchard P. H. Anaerobic Degradation of Chloroaromatic Compounds in Aquatic Sediments under a Variety of Enrichment Conditions. Appl Environ Microbiol. 1989 Jun;55(6):1466–1471. doi: 10.1128/aem.55.6.1466-1471.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Genthner B. R., Price W. A., Pritchard P. H. Characterization of anaerobic dechlorinating consortia derived from aquatic sediments. Appl Environ Microbiol. 1989 Jun;55(6):1472–1476. doi: 10.1128/aem.55.6.1472-1476.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibson S. A., Suflita J. M. Anaerobic biodegradation of 2,4,5-trichlorophenoxyacetic Acid in samples from a methanogenic aquifer: stimulation by short-chain organic acids and alcohols. Appl Environ Microbiol. 1990 Jun;56(6):1825–1832. doi: 10.1128/aem.56.6.1825-1832.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibson S. A., Suflita J. M. Extrapolation of biodegradation results to groundwater aquifers: reductive dehalogenation of aromatic compounds. Appl Environ Microbiol. 1986 Oct;52(4):681–688. doi: 10.1128/aem.52.4.681-688.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horowitz A., Suflita J. M., Tiedje J. M. Reductive dehalogenations of halobenzoates by anaerobic lake sediment microorganisms. Appl Environ Microbiol. 1983 May;45(5):1459–1465. doi: 10.1128/aem.45.5.1459-1465.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kohring G. W., Rogers J. E., Wiegel J. Anaerobic biodegradation of 2,4-dichlorophenol in freshwater lake sediments at different temperatures. Appl Environ Microbiol. 1989 Feb;55(2):348–353. doi: 10.1128/aem.55.2.348-353.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Linkfield T. G., Suflita J. M., Tiedje J. M. Characterization of the acclimation period before anaerobic dehalogenation of halobenzoates. Appl Environ Microbiol. 1989 Nov;55(11):2773–2778. doi: 10.1128/aem.55.11.2773-2778.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mikesell M. D., Boyd S. A. Complete reductive dechlorination and mineralization of pentachlorophenol by anaerobic microorganisms. Appl Environ Microbiol. 1986 Oct;52(4):861–865. doi: 10.1128/aem.52.4.861-865.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mohn W. W., Tiedje J. M. Strain DCB-1 conserves energy for growth from reductive dechlorination coupled to formate oxidation. Arch Microbiol. 1990;153(3):267–271. doi: 10.1007/BF00249080. [DOI] [PubMed] [Google Scholar]
- Quensen J. F., 3rd, Tiedje J. M., Boyd S. A. Reductive dechlorination of polychlorinated biphenyls by anaerobic microorganisms from sediments. Science. 1988 Nov 4;242(4879):752–754. doi: 10.1126/science.242.4879.752. [DOI] [PubMed] [Google Scholar]
- Shelton D. R., Tiedje J. M. Isolation and partial characterization of bacteria in an anaerobic consortium that mineralizes 3-chlorobenzoic Acid. Appl Environ Microbiol. 1984 Oct;48(4):840–848. doi: 10.1128/aem.48.4.840-848.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suflita J. M., Horowitz A., Shelton D. R., Tiedje J. M. Dehalogenation: a novel pathway for the anaerobic biodegradation of haloaromatic compounds. Science. 1982 Dec 10;218(4577):1115–1117. doi: 10.1126/science.218.4577.1115. [DOI] [PubMed] [Google Scholar]
- Suflita J. M., Robinson J. A., Tiedje J. M. Kinetics of microbial dehalogenation of haloaromatic substrates in methanogenic environments. Appl Environ Microbiol. 1983 May;45(5):1466–1473. doi: 10.1128/aem.45.5.1466-1473.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang X., Wiegel J. Sequential anaerobic degradation of 2,4-dichlorophenol in freshwater sediments. Appl Environ Microbiol. 1990 Apr;56(4):1119–1127. doi: 10.1128/aem.56.4.1119-1127.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]



