Abstract
Background
Oligodeoxynucleotides containing unmethylated CpG motifs (CpG ODN) are known to exert a strong adjuvant effect on Th1 immune responses. Although several genes have been reported, no comprehensive study of the gene expression profiles in human cells after stimulation with CpG ODN has been reported.
Results
This study was designed to identify a CpG-inducible gene cluster that potentially predicts for the molecular mechanisms of clinical efficacy of CpG ODN, by determining mRNA expression in human PBMC after stimulation with CpG ODN. PBMCs were obtained from the peripheral blood of healthy volunteers and cultured in the presence or absence of CpG ODN 2006 for up to 24 hours. The mRNA expression profile was evaluated using a high-density oligonucleotide probe array, GeneChip®. Using hierarchical clustering-analysis, out of a total of 10,000 genes we identified a cluster containing 77 genes as having been up-regulated by CpG ODN. This cluster was further divided into two sub-clusters by means of time-kinetics. (1) Inflammatory cytokines such as IL-6 and GM-CSF were up-regulated predominantly 3 to 6 hours after stimulation with CpG ODN, presumably through activation of a transcription factor, NF-κB. (2) Interferon (IFN)-inducible anti-viral proteins, including IFIT1, OAS1 and Mx1, and Th1 chemoattractant IP-10, were up-regulated predominantly 6 to 24 hours after stimulation. Blocking with mAb against IFN-α/β receptor strongly inhibited the induction of these IFN-inducible genes by CpG ODN.
Conclusion
This study provides new information regarding the possible immunomodulatory effects of CpG ODN in vivo via an IFN-α/β receptor-mediated paracrine pathway.
Background
Several natural and synthetic compounds are known to act as adjuvants that enhance immune responses when administered with antigens, both in vitro and in vivo [1,2]. Some of these adjuvants elicit predominantly Th1 type immune responses and are used clinically to treat patients with viral infections, malignant neoplasms, and recently also those with allergic diseases [3,4]. BCG, a vaccine used to protect against tuberculosis infection, can also trigger an anti-tumor response when administered in vivo via increased immunoglobulin synthesis and altered NK cell activity [4]. The anti-tumor effect of BCG is attributed in part to a DNA fraction named MY-1 found in BCG extracts [5]. MY-1, which contains unmethylated CpG motifs, enhances NK cell activity through induction of both type I and type II interferons (IFNs) [4,5]. Synthetic oligodeoxynucleotides containing unmethylated CpG motifs (CpG ODN) have also been shown to mimic the effect of bacterial DNA in activating immune systems [6].
Recently, innate immunity has received attention not only because it participates in the prevention of microbial infection but also because it is capable of shifting host immunity away from allergic immune responses (Th2) toward non-allergic immune responses (Th1) [7,8]. Molecules that are highly conserved among species are involved in the innate immune system as specific receptors [7]. Human molecules responsible for innate immunity are called Toll-like receptors (TLRs) [9], homologues of Toll in Drosophila [10], which recognize pathogen-associated molecular patterns (PAMPs) of microbial organisms [7]. To date, 10 human TLRs have been identified [11]. Among them, TLR9 recognizes the CpG motif from bacterial DNA [12].
The fact that TLR9 is localized to the endocytic vesicles suggests that CpG ODN enhances the immune response of the host by acting as an adjuvant to ingested antigen in antigen-presenting cells [13]. TLRs and IL-1R share similar intracytoplasmic molecular associations [11]. TLRs initiate signaling via sequential recruitment of myeloid differentiation factor 88 (MyD88), IL-1R associated kinase (IRAK) and tumor necrosis factor receptor-associated factor 6 (TRAF), which in turn activate downstream mediators such as mitogen-activated protein kinase (MAPK) and transcription factors such as NF-κB, AP-1 and ATF-2 [11]. According to this signal transduction pathway, CpG ODN would be expected to act as other PAMPs do [11,12]. However, administration of CpG ODN results in multiple immunostimulatory effects in vivo without inducing serious endotoxin shock. This suggests that CpG ODN can be used as a therapeutic adjuvant in several human diseases.
Recently, at least two phenotypes of CpG ODN have been found [14-16]. A/D-type CpG ODNs promote production of large amounts of type I IFN by plasmacytoid dendritic cells (PDC). In contrast, B/K-type CpG ODNs such as CpG ODN 2006 strongly activate human B cells, promote their survival and up-regulate the expression of costimulatory molecules on their cell surface. However, it is known that administration of high doses of type I IFN may directly induce various side effects, such as fever, chills and depression [17]. Therefore, to investigate clinical use of CpG ODN, we selected CpG ODN 2006, which is B/K-type CpG ODN and so less likely to cause side effects [6,15].
In this study, out of 10,000 genes we identified a gene cluster containing 77 genes that were up-regulated by CpG ODN. One sub-cluster included genes for inflammatory cytokines and was up-regulated predominantly 3 to 6 hours after stimulation with CpG ODN. The second sub-cluster included several genes for IFN-inducible anti-viral and anti-tumor proteins and was up-regulated predominantly 6 to 24 hours after stimulation. These data suggest a possible association between these molecules and the immunomodulatory effects of CpG ODN in vivo.
Results
Identification of CpG ODN-inducible genes
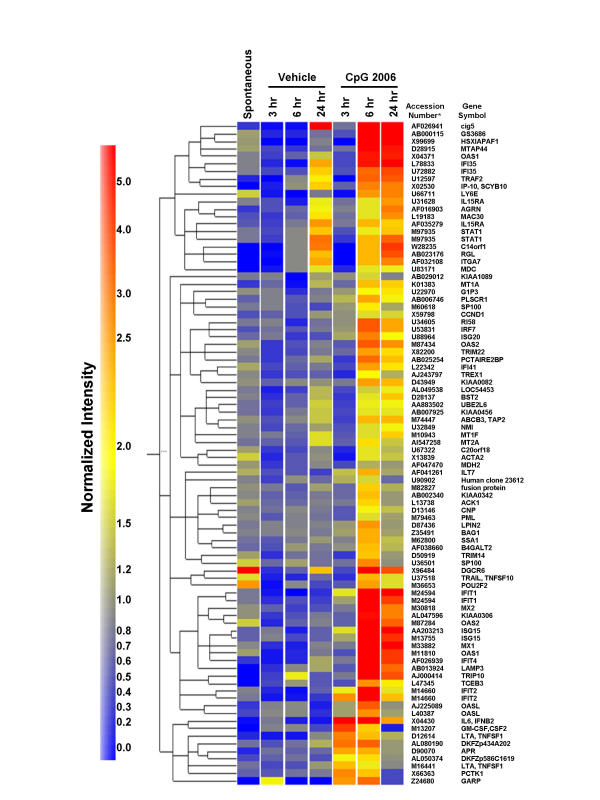
Using hierarchical clustering-analysis of the gene expression profiles of approximately 10,000 genes, we identified a cluster containing 77 genes which were up-regulated by CpG ODN (Fig. 1). This cluster was further sub-divided into two clusters on the basis of time-kinetics. Early-response genes, including IL-6, lymphotoxin α (LTA) and granulocyte-macrophage colony-stimulating factor (GM-CSF, CSF2), were up-regulated predominantly 3 to 6 hours after stimulation with CpG ODN. In contrast, late-response genes, including IFN-induced protein with tetratricopeptide repeats 1 (IFIT1), IFIT2, 2',5'-oligoadenylate synthetase 1 (OAS1), OAS2, myxovirus (influenza virus) resistance 1 (Mx1), Mx2 and IFN-γ inducible protein 10 (IP-10), were up-regulated predominantly 6 to 24 hours after stimulation with CpG ODN. Some CpG ODN-inducible genes, especially such late-response genes as IFIT1, OAS1 and IP-10, have been shown to be regulated by type I IFN and/or type II IFNs [18]. However, mRNAs for all type I IFNs were judged to be 'Absent' by Microarray Suite software, which means that the mRNAs are below the detection limit of the GeneChip® system (data not shown).
Figure 1.
CpG ODN mediated up-regulation of a gene cluster in PBMC. A gene cluster containing 77 genes was up-regulated more than two-fold in PBMC after stimulation with CpG ODN. Data were analyzed by applying a hierarchical-tree algorithm to the normalized intensity. The color code for the signal strength in the classification scheme is shown in the box on the left. Induced genes are indicated by shades of red; repressed genes are indicated by shades of blue. Exactly the same experiments were performed with PBMCs from three individual donors to confirm the reproducibility of the present experiments. All of these genes were consistently included in a CpG-inducible cluster in the same fashion even though cRNA elongation was not completed in a single experiment. * GenBank accession number
Quantitative analysis of mRNA for CpG ODN-inducible genes
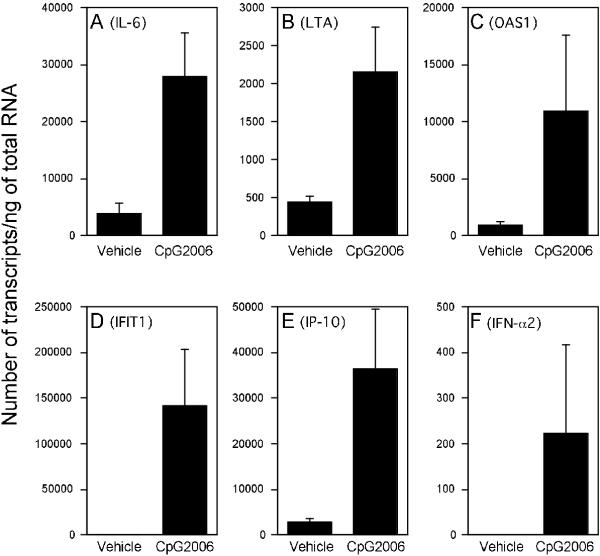
Up-regulation of the mRNA levels of five genes, IL-6, LTA, OAS1, IFIT1 and IP-10, was further confirmed by real-time PCR (Fig. 2). The magnitude of enhancement of the mRNA for IL-6, LTA, OAS1, IFIT1 and IP-10 by CpG ODN was 7-fold, 5-fold, 14-fold, 289-fold and 18-fold, respectively. However, induction of inflammatory genes such as IL-6 and GM-CSF by CpG ODN is very weak compared with LPS (Fig. 3). Up-regulation of the mRNA levels of CpG ODN-inducible genes, such as IP-10 and IL-6, was scarcely induced by GpC control ODN, being less than 1:10 when compared to the enhancement by CpG ODN 2006 (data not shown). In addition, induction of a low level of IFN-α was detectable by real-time PCR (Fig. 2).
Figure 2.
CpG ODN mediated up-regulation of mRNA for IL-6, LTA, OAS1, IFIT1, IP-10 and IFN-α2. PBMC were incubated in the presence or absence of 2 μg/ml CpG ODN 2006 for 6 hours. The mRNA levels of IL-6 (A), LTA (B), OAS1 (C), IFIT1 (D), IP-10 (E) and IFN-α2 (F) were determined by SYBR® green-based real-time quantitative RT-PCR. The results are shown as the mean ± SEM from four independent experiments. The copy number is expressed as the number of transcripts/ng of total RNA.
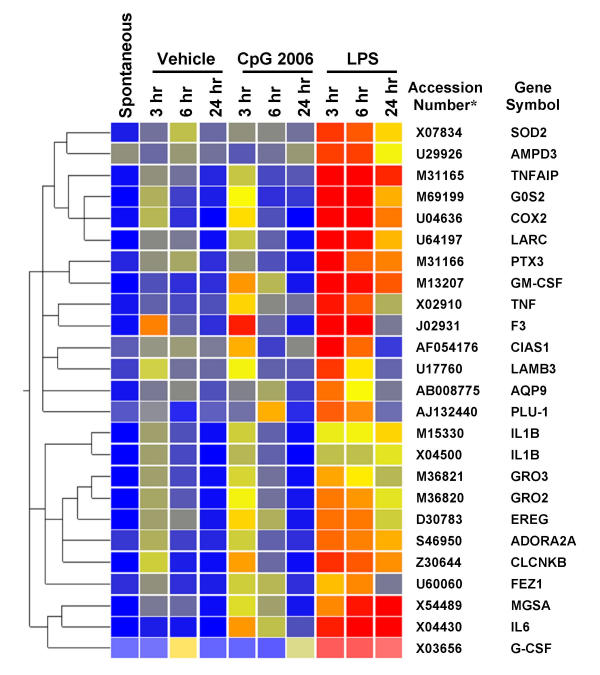
Figure 3.
LPS mediated up-regulation of a gene cluster in PBMC, compared with CpG ODN. A gene cluster containing proinflammatory genes was up-regulated in PBMC after stimulation with LPS (100 ng/ml). For information regarding data analysis and color code, see Figure 1. * GenBank accession number
Role of type I IFNs in the up-regulation of IFN-inducible genes by CpG ODN
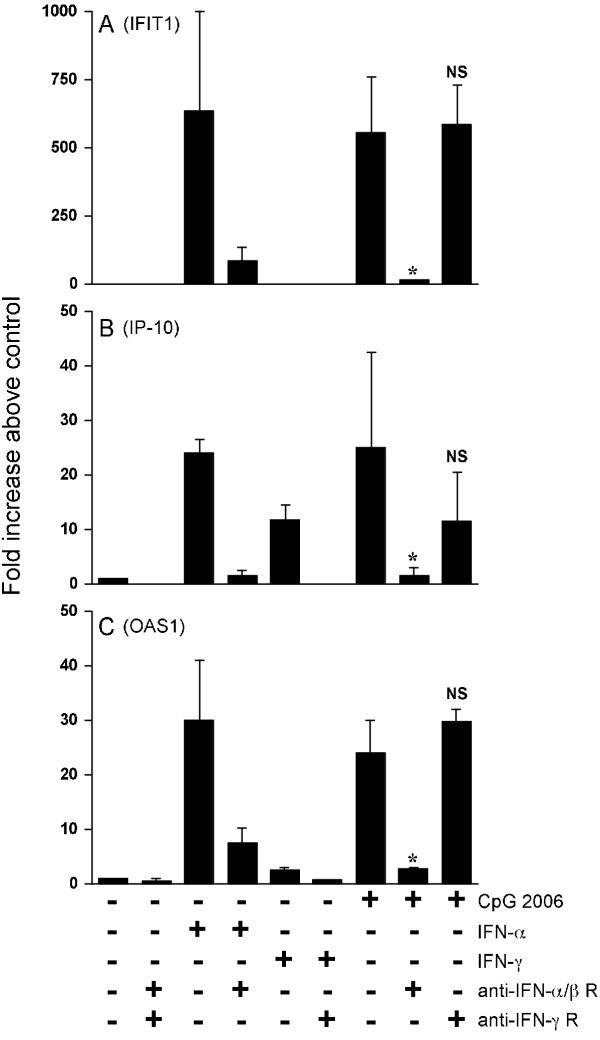
To elucidate whether IFN-inducible genes such as IFIT1, OAS1 and IP-10 were up-regulated directly by CpG ODN or as a result of autocrine/paracrine pathways of IFNs, blocking experiments were performed using anti-IFN receptor mAbs. Up-regulation of mRNA for IFIT1 by IFN-α2 and CpG ODN was significantly inhibited by the addition of anti-IFN-α/β receptor mAb (Fig. 4). With respect to IP-10, mRNA expression was up-regulated by IFN-α2, IFN-γ and CpG ODN. Induction of IP-10 by IFN-α2 or IFN-γ was significantly inhibited by the addition of mAbs specific for each IFN receptor (p < 0.05). The induction by CpG ODN was significantly inhibited by anti-IFN-α/β receptor mAb, whereas it was partially inhibited, though not statistically significant, by anti-IFN-γ receptor mAb (Fig. 4).
Figure 4.
Effects of blocking mAbs against IFN receptors on up-regulation of mRNA expression by CpG ODN. PBMC were stimulated with IFN-α2 (200 IU/ml), IFN-γ (100 IU/ml) or CpG ODN 2006 (2 μg/ml) in the presence or absence of blocking mAbs against IFN-α/β receptor or IFN-γ receptor for 6 hours. The mRNA levels of IFIT1 (A), IP-10 (B) and OAS1 (C) were determined by SYBR® green-based real-time quantitative RT-PCR. The results are shown as the mean ± SEM of three independent experiments and expressed as -fold increase above the mRNA level in the control cells. In this experiment, up-regulation of IFIT1 and IP-10 by CpG ODN 2006 was not blocked by the addition of the isotype-matched control mAbs (data not shown). * p < 0.05 vs CpG ODN 2006 stimulated. NS: not significant.
Moreover, we confirmed that up-regulation of mRNA for OAS1, Mx1 and IFN regulatory factor 7 (IRF-7) by CpG ODN was also strongly inhibited by the addition of anti-IFN-α/β receptor mAb (Fig. 4C for OAS1; other data not shown), whereas up-regulation of mRNA for IL-6 and LTA by CpG ODN was not inhibited by the addition of these anti-IFN receptor mAbs (data not shown). Moreover, induction of each of IFIT1 and IP-10 by CpG ODN 2006 was not inhibited by the addition of these isotype control mAbs (data not shown).
Discussion
Using the GeneChip® system, we identified a gene cluster consisting of 88 probe sets in PBMC after stimulation with CpG ODN. These genes are thought to be responsible for the immunomodulatory effects of CpG ODN in vivo. Among the 88 probe sets, 11 pairs of probe sets were designed to recognize different parts of single genes. These genes, including OAS1, IFN-induced protein 35 (IFI35), IL-15Rα chain, STAT1, OAS2, IFIT1, IFN-stimulated gene 15 (ISG15), IFIT2, OASL, nuclear antigen Sp100 (SP100) and LTA, were expressed at high levels. The fact that most of these pair sets are located next- or very close- to each other in this cluster after hierarchical clustering (Fig. 1) indicated that the results of our experiment are highly reproducible and reliable.
A recent study observed that human monocytes, NK cells and T cells are not sensitive to CpG ODN because of their poor expression of TLR9 [19]. However, they are activated by CpG ODN-induced B cell- and PDC-derived cytokines [19,20]. Since the focus of this study was to predict the molecular mechanisms of clinically administered CpG ODN, we selected a mixed population of cells (PBMC) to allow for the effects of multiple cell interactions.
The gene sub-cluster that was predominantly up-regulated at 3 to 6 hours after stimulation with CpG ODN contains genes that encode inflammatory cytokines or chemokines such as IL-6 and GM-CSF. Because the promotor regions of these genes contain the NF-κB binding site, the up-regulation of these gene transcripts may be mediated by activation of NF-κB [21]. In addition, we report for the first time that CpG ODN enhanced expression of the mRNA for LTA in this sub-cluster. LTA is released from Th1 cells and exerts proinflammatory effects [22]. However, induction of other inflammatory cytokines such as IL-6 by CpG ODN seems to be very weak when compared to induction by LPS (Fig. 3): the magnitude of enhancement of the transcript for IL-6 was 242-fold and 7-fold by LPS and CpG ODN 2006, respectively (data not shown). Moreover, induction of further inflammatory genes such as IL-1β, TNFα and cyclooxygenase-2 (COX2), and neutrophil chemoattractants such as growth-related oncogene 2 (GRO2) and GRO3 by LPS is of high magnitude and is maintained for a long time (Fig. 3).
The majority of genes in the second sub-cluster (up-regulated at 6 to 24 hours after stimulation with CpG ODN) are known to be regulated by type I and/or type II IFNs. These include: IFIT1, OAS1, Mx1, cig5, ISG15, IFI35, lymphocyte antigen 6 complex, locus E (LY6E), phospholipid scramblase 1 (PLSCR1), SP100, metallothionein, TNF-related apoptosis-inducing ligand (TRAIL, TNFSF10) and IP-10 [18,23-28]. Interestingly, both the mRNA levels of type I IFN measured by the GeneChip system and protein levels of IFNs measured by ELISA were undetectably low (data not shown). However, real-time quantitative RT-PCR, the most sensitive way to measure mRNA levels, did detect low levels of IFN-α2. To clarify whether or not the enhancement of such mRNA expression is a direct effect of CpG ODN or mediated by an IFN-autocrine/paracrine pathway, we performed blocking experiments. As a result, anti-IFN-α/β receptor mAb strongly inhibited the up-regulation of type I IFN-inducible genes such as IFIT1 and OAS1, whereas anti-IFN-γ receptor mAb or isotype-matched control mAbs did not elicit this response. Induction of IP-10, a type I/II IFN-inducible gene, by CpG ODN 2006 was also significantly inhibited by anti-IFN-α/β receptor mAb. Induction of IP-10 was partially inhibited by anti-IFN-γ receptor mAb, although this effect did not reach statistical significance. Some publications suggest that type I IFNs enhance production of IFN-γ by NK cells and T cells [29,30]. Thus, some part of IP-10 induction by CpG ODN may be regulated by type I IFN-induced IFN-γ.
In addition, our GeneChip® data indicate that CpG ODN up-regulated the expression of IRF-7 (Fig. 1). With respect to type I IFN synthesis, constitutive expression of IRF-3 along with inducible IRF-7 is known to play a key role [31,32]. Furthermore, PDC – natural type I IFN producing cells – are a very rare cell population (less than 0.1% of PBMC) [33,34]. CpG ODN 2006 selectively stimulates the early phase type I IFN induction in PDC but is unable to initiate the IFN-α/β receptor-mediated autocrine feedback loop [35]. Taken together, these results strongly suggest that the effect of CpG ODN is at least in part mediated through an IFN-α/β receptor-dependent paracrine pathway. In contrast, NF-κB regulated genes, including IL-6, were not affected by the IFN receptor mAbs. The significant induction of IFN-inducible genes in correlation with a low level of type I IFN suggests involvement of other synergistic molecular mechanisms which have yet to be elucidated.
Several groups have investigated the immunomodulatory effects of CpG ODN in the clinical context including chronic viral infection, malignant disease and allergy, and initial results have been encouraging [36]. These results, together with those of our study suggest a number of genes may have possible roles in the clinical efficacy of CpG ODN in the treatment of these diseases.
With respect to the treatment of chronic viral infections, our study demonstrated that CpG ODN induces production of mRNA for anti-viral proteins, including IFIT1, OAS1, Mx1, ISG15 and TRAIL. IFIT1, a type I IFN-inducible gene, was recently described to interact with eukaryotic translation factor 3 (eIF-3), thus inhibiting translation [37]. OAS1, a type I IFN-inducible gene, regulates the RNase L dependent anti-viral defense pathway [38]. Mx1, a type I IFN-inducible GTPase, has been shown to inhibit replication of certain RNA viruses [39]. ISG15, also known as an ubiquitin cross-reactive protein, is known to be induced by type I IFNs and to form conjugates with intracellular proteins in a process analogous to that for ubiquitin [40]. TRAIL is thought to be involved in the selective killing of virus-infected cells and tumor cells [27,28]. CpG ODN also induces mRNA for IP-10, which recruits NK cells, and enhances the production of virus-specific antibodies when administered as an adjuvant with vaccine [36]. Up-regulation of these genes by CpG ODN suggests potential prophylactic and therapeutic use of CpG ODN in the treatment of viral infections.
In addition, CpG ODN is currently under trial in the treatment of patients with allergic diseases through down-regulation of pre-existing Th2 type immune responses and up-regulation of Th1 type immune responses [41-44]. Recently, it has become clear that type I IFNs are capable of inducing Th1 type immune responses in humans but not in mice [45]. The up-regulation of mRNA for IP-10 by CpG ODN in our study supports the findings of previous studies [20,46]. IP-10 is a natural ligand for CXCR3 expressed on Th1 cells and acts as an important regulator in recruiting Th1 cells [47,48]. In addition, IP-10 acts as an antagonist to CCR3 expressed on activated Th2 cells and eosinophils [49]. These results suggest that type I IFNs and IP-10 may have possible roles in the clinical efficacy of CpG ODN in allergic diseases.
Conclusion
We applied the GeneChip® system to PBMC and identified, out of 10,000 genes, a gene cluster containing 77 genes which are up-regulated by CpG ODN 2006. This cluster was further divided into two sub-clusters by means of time-kinetics. One sub-cluster contains genes for inflammatory cytokines such as IL-6 and LTA, which were up-regulated predominantly 3 to 6 hours after stimulation with CpG ODN. However induction of these inflammatory cytokines by CpG ODN is very weak when compared with LPS, giving CpG ODN a favorable side effect profile. The second cluster contains several genes for IFN-inducible anti-viral proteins, including IFIT1, OAS1 and Mx1, and Th1 chemoattractant IP-10, which were up-regulated predominantly 6 to 24 hours after stimulation with CpG ODN. Our experiments strongly suggest that induction of these genes by CpG ODN was regulated through an IFN-α/β receptor-dependent paracrine pathway involving a sub-threshold level of type I IFN. This study provides new information regarding the possible immunomodulatory effects of CpG ODN in vivo via an IFN-α/β receptor-mediated paracrine pathway.
Methods
Oligodeoxynucleotides
Phosphorothioate ODNs were synthesized at Operon Technologies (Alameda, CA). The sequence of the CpG ODN 2006 used in the present study was 5'-TCG TCG TTT TGT CGT TTT GTC GTT-3' [6]. The sequence of the GpC control ODN was 5'-TGC TGC TTT TGT GCT TTT GTG CTT-3', which was modified by substitutions of GpC for the CpG in CpG ODN 2006. ODNs were dissolved in pyrogen-free double distilled water.
Preparation and cell culture
All human subjects in this study provided written informed consent, which was approved by the Ethical Review Board at National Children's Hospital in Tokyo, Japan. Human peripheral blood mononuclear cells (PBMC) were isolated by centrifugation on a Ficoll-Isopaque density gradient (d = 1.077 g/ml, Lymphoprep, Nycomed, Oslo, Norway). After washing with PBS (Gibco BRL, Grand Island, NY), the cells were suspended in Iscove modified Dulbecco medium (IMDM; Gibco BRL) supplemented with 100 U/ml penicillin and 100 μg/ml streptomycin (Gibco BRL) at a cell density of 1 × 106 cells/ml. PBMC were incubated in a 25 cm2 tissue culture flask in the presence or absence of 2 μg/ml CpG ODN 2006 or 100 ng/ml LPS (Salmonella typhimurium; Sigma, St. Louis, MO) for up to 24 hours. For real-time PCR analysis, PBMC from four healthy volunteers were cultured in 6-well plates in the presence or absence of 2 μg/ml CpG ODN 2006 for 6 hours. As a control for a neutralization study, PBMC were also incubated in the presence of 200 IU/ml IFN-α2 (Takeda Chemical Industries, Osaka, Japan) or 100 IU/ml IFN-γ (Shionogi & Co, Ltd, Osaka, Japan) without CpG ODN 2006 for 6 hours. In some experiments, 10 μg/ml mouse anti-human IFN-α/β receptor chain 2 (CD118) mAb (clone: MMHAR-2, IgG2a, PBL Biomedical Laboratories, New Brunswick, NJ), 10 μg/ml mouse anti-human IFN-γ receptor α-chain (CD119) mAb (clone: GIR-208, IgG1, Pharmingen, San Diego, CA) or isotype-matched control mAbs (IgG1: SouthernBiotech, Birmingham, AL, IgG2a: eBioscience, San Diego, CA) were added simultaneously with CpG ODN or IFNs throughout the culture period.
GeneChip® expression analysis
Total RNA was extracted by RNeasy® (Qiagen, Hilden, Germany) according to the manufacturer's instructions. Total RNAs were pooled from three individual experiments for each time point. Five micrograms of total RNA from each sample (0, 3, 6 or 24 hours) were used to prepare cRNA. Gene expression was analyzed with the GeneChip® Human Genome U95Av2 probe array (Affymetrix, Santa Clara, CA), which contains the oligonucleotide probe sets for approximately 10,000 genes, according to the manufacturer's protocol (Expression Analysis Technical Manual). Data analysis was performed with a Microarray Suite version 4.0 software (Affymetrix) and a GeneSpring software version 4.1 (Silicon Genetics, San Carlos, CA). To normalize the staining intensity variations among chips, the average difference values for all genes on a given chip were divided by the median of all measurements on that chip. Hierarchical-clustering analysis was performed using a minimum distance value of 0.001, a separation ratio of 0.5 and the standard definition of the correlation distance.
Real-time quantitative RT-PCR
Primer sets for 7 genes, IL-6 (sense, 5'-CCA CAC AGA CAG CCA CTC AC-3'; antisense, 5'-AGG TTG TTT TCT GCC AGT GC-3'), LTA (sense, 5'-GGA GACC AGC TAT CCA CCC AC-3'; antisense, 5'-TTC CTT CTT TGA AAG CTC CGG-3'), IFIT1 (sense, 5'-GCC ATT TTC TTT GCT TCC CCT A-3'; antisense, 5'-TGC CCT TTT GTA GCC TCC TTG-3'), OAS1 (sense, 5'-CAT CCG CCT AGT CAA GCA CTG-3'; antisense, 5'-CCA CCA CCC AAG TTT CCT GTA G-3'), IP-10 (sense, 5'-CGC TGT ACC TGC ATC AGC ATT-3'; antisense, 5'-GCT CCC CTC TGG TTT TAA GGA G-3'), IFN-α2 (sense, 5'-GGA TGA GAC CCT CCT AGA CAA AT-3'; antisense, 5'-ATG ATT TCT GCT CTG ACA ACC TC-3') and GAPDH (sense, 5'-GAA GGT GAA GGT CGG AGT C-3'; antisense, 5'-GAA GAT GGT GAT GGG ATT TC-3') were synthesized at Sawady Technology (Tokyo, Japan). These molecules were chosen by means of an NF-κB-responsible gene, a type I IFN-inducible gene, a type II IFN-inducible gene, an IFN-α gene itself and a housekeeping gene, respectively. Total RNA was analyzed after DNase I (Qiagen) digestion. Single-strand cDNA was synthesized with SuperScript II reverse transcriptase (Gibco BRL). Real-time quantitative RT-PCR was performed with a double-stranded DNA-binding dye, SYBR® Green I, using an Applied Biosystems 7700 Sequence Detection System (Applied Biosystems, Foster City, CA) in 50 μL reactions (25 μL 2 × SYBR® Green Master Mix (Applied Biosystems), 50 nM of each primer plus cDNA). The expression levels of mRNA were normalized by the average expression of a housekeeping gene (GAPDH). The copy number is expressed as the number of transcripts/ng total RNA.
Statistical analysis
All data are presented as the mean ± SEM unless otherwise noted. Differences between groups were analyzed using the Mann-Whitney U-test and considered to be significant if p < 0.05.
Authors' contributions
AK and TH participated in the design of the experiments and contributed to the GeneChip analysis. AK performed the blocking experiment and wrote the manuscript. JB carried out revision of the manuscript. NH contributed to the GeneChip analysis. SI and HW participated in its design and coordination. HS and KM participated in experimental design, interpretation of the results, and revision of the manuscript. All authors read and approved the final version of the manuscript.
Acknowledgments
Acknowledgements
This work was supported in part by a grant from the Organization for Pharmaceutical Safety and Research and the Ministry of Health, Labour and Welfare (the Millennium Genome Project, MPJ-5) and by a grant from RIKEN Research Center for Allergy & Immunology.
Contributor Information
Atsushi Kato, Email: atkato@nch.go.jp.
Toshiki Homma, Email: thomma@nch.go.jp.
Jonathan Batchelor, Email: jmb@doctors.org.uk.
Noriko Hashimoto, Email: nhashimoto@nch.go.jp.
Shosuke Imai, Email: shoimai@med.kochi-ms.ac.jp.
Hiroshi Wakiguchi, Email: wakiguti@med.kochi-ms.ac.jp.
Hirohisa Saito, Email: hsaito@nch.go.jp.
Kenji Matsumoto, Email: kmatsumoto@nch.go.jp.
References
- Whittle HC, Lamb WH, Ryder RW. Trials of intradermal hepatitis B vaccines in Gambian children. Ann Trop Paediatr. 1987;7:6–9. doi: 10.1080/02724936.1987.11748464. [DOI] [PubMed] [Google Scholar]
- Uede T, Huff TF, Ishizaka K. Formation of IgE-binding factors by rat T lymphocytes. V. Effect of adjuvant for the priming immunization on the nature of IgE binding factors formed by antigenic stimulation. J Immunol. 1982;129:1384–90. [PubMed] [Google Scholar]
- Koh YI, Choi IS, Kim WY. BCG infection in allergen-presensitized rats suppresses Th2 immune response and prevents the development of allergic asthmatic reaction. J Clin Immunol. 2001;21:51–9. doi: 10.1023/A:1006745116360. [DOI] [PubMed] [Google Scholar]
- Tokunaga T, Yamamoto T, Yamamoto S. How BCG led to the discovery of immunostimulatory DNA. Jpn J Infect Dis. 1999;52:1–11. [PubMed] [Google Scholar]
- Tokunaga T, Yamamoto H, Shimada S, Abe H, Fukuda T, Fujisawa Y, Furutani Y, Yano O, Kataoka T, Sudo T, et al. Antitumor activity of deoxyribonucleic acid fraction from Mycobacterium bovis BCG. I. Isolation, physicochemical characterization, and antitumor activity. J Natl Cancer Inst. 1984;72:955–62. [PubMed] [Google Scholar]
- Hartmann G, Weeratna RD, Ballas ZK, Payette P, Blackwell S, Suparto I, Rasmussen WL, Waldschmidt M, Sajuthi D, Purcell RH, et al. Delineation of a CpG phosphorothioate oligodeoxynucleotide for activating primate immune responses in vitro and in vivo. J Immunol. 2000;164:1617–24. doi: 10.4049/jimmunol.164.3.1617. [DOI] [PubMed] [Google Scholar]
- Medzhitov R, Janeway CA., Jr Innate immunity: the virtues of a nonclonal system of recognition. Cell. 1997;91:295–8. doi: 10.1016/s0092-8674(00)80412-2. [DOI] [PubMed] [Google Scholar]
- Krieg AM. The role of CpG motifs in innate immunity. Curr Opin Immunol. 2000;12:35–43. doi: 10.1016/S0952-7915(99)00048-5. [DOI] [PubMed] [Google Scholar]
- Rock FL, Hardiman G, Timans JC, Kastelein RA, Bazan JF. A family of human receptors structurally related to Drosophila Toll. Proc Natl Acad Sci U S A. 1998;95:588–93. doi: 10.1073/pnas.95.2.588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lemaitre B, Nicolas E, Michaut L, Reichhart JM, Hoffmann JA. The dorsoventral regulatory gene cassette spatzle/Toll/cactus controls the potent antifungal response in Drosophila adults. Cell. 1996;86:973–83. doi: 10.1016/s0092-8674(00)80172-5. [DOI] [PubMed] [Google Scholar]
- Akira S, Takeda K, Kaisho T. Toll-like receptors: critical proteins linking innate and acquired immunity. Nat Immunol. 2001;2:675–80. doi: 10.1038/90609. [DOI] [PubMed] [Google Scholar]
- Hemmi H, Takeuchi O, Kawai T, Kaisho T, Sato S, Sanjo H, Matsumoto M, Hoshino K, Wagner H, Takeda K, et al. A Toll-like receptor recognizes bacterial DNA. Nature. 2000;408:740–5. doi: 10.1038/35047123. [DOI] [PubMed] [Google Scholar]
- Takeshita F, Leifer CA, Gursel I, Ishii KJ, Takeshita S, Gursel M, Klinman DM. Role of Toll-like receptor 9 in CpG DNA-induced activation of human cells. J Immunol. 2001;167:3555–8. doi: 10.4049/jimmunol.167.7.3555. [DOI] [PubMed] [Google Scholar]
- Krieg AM. CpG motifs in bacterial DNA and their immune effects. Annu Rev Immunol. 2002;20:709–60. doi: 10.1146/annurev.immunol.20.100301.064842. [DOI] [PubMed] [Google Scholar]
- Krug A, Rothenfusser S, Hornung V, Jahrsdorfer B, Blackwell S, Ballas ZK, Endres S, Krieg AM, Hartmann G. Identification of CpG oligonucleotide sequences with high induction of IFN-alpha/beta in plasmacytoid dendritic cells. Eur J Immunol. 2001;31:2154–63. doi: 10.1002/1521-4141(200107)31:7<2154::AID-IMMU2154>3.0.CO;2-U. [DOI] [PubMed] [Google Scholar]
- Gursel M, Verthelyi D, Gursel I, Ishii KJ, Klinman DM. Differential and competitive activation of human immune cells by distinct classes of CpG oligodeoxynucleotide. J Leukoc Biol. 2002;71:813–20. [PubMed] [Google Scholar]
- Zdilar D, Franco-Bronson K, Buchler N, Locala JA, Younossi ZM. Hepatitis C, interferon alfa, and depression. Hepatology. 2000;31:1207–11. doi: 10.1053/jhep.2000.7880. [DOI] [PubMed] [Google Scholar]
- Der SD, Zhou A, Williams BR, Silverman RH. Identification of genes differentially regulated by interferon alpha, beta, or gamma using oligonucleotide arrays. Proc Natl Acad Sci U S A. 1998;95:15623–8. doi: 10.1073/pnas.95.26.15623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hornung V, Rothenfusser S, Britsch S, Krug A, Jahrsdorfer B, Giese T, Endres S, Hartmann G. Quantitative expression of toll-like receptor 1–10 mRNA in cellular subsets of human peripheral blood mononuclear cells and sensitivity to CpG oligodeoxynucleotides. J Immunol. 2002;168:4531–7. doi: 10.4049/jimmunol.168.9.4531. [DOI] [PubMed] [Google Scholar]
- Blackwell SE, Krieg AM. CpG-A-Induced Monocyte IFN-gamma-Inducible Protein-10 Production Is Regulated by Plasmacytoid Dendritic Cell-Derived IFN-alpha. J Immunol. 2003;170:4061–8. doi: 10.4049/jimmunol.170.8.4061. [DOI] [PubMed] [Google Scholar]
- Munoz C, Pascual-Salcedo D, Castellanos MC, Alfranca A, Aragones J, Vara A, Redondo MJ, de Landazuri MO. Pyrrolidine dithiocarbamate inhibits the production of interleukin-6, interleukin-8, and granulocyte-macrophage colony-stimulating factor by human endothelial cells in response to inflammatory mediators: modulation of NF-kappa B and AP-1 transcription factors activity. Blood. 1996;88:3482–90. [PubMed] [Google Scholar]
- Ohshima Y, Yang LP, Avice MN, Kurimoto M, Nakajima T, Sergerie M, Demeure CE, Sarfati M, Delespesse G. Naive human CD4+ T cells are a major source of lymphotoxin alpha. J Immunol. 1999;162:3790–4. [PubMed] [Google Scholar]
- Shan X, Bourdeau A, Rhoton A, Wells DE, Cohen EH, Landgraf BE, Palfree RG. Characterization and mapping to human chromosome 8q24.3 of Ly-6-related gene 9804 encoding an apparent homologue of mouse TSA-1. J Immunol. 1998;160:197–208. [PubMed] [Google Scholar]
- Grotzinger T, Jensen K, Will H. The interferon (IFN)-stimulated gene Sp100 promoter contains an IFN-gamma activation site and an imperfect IFN-stimulated response element which mediate type I IFN inducibility. J Biol Chem. 1996;271:25253–60. doi: 10.1074/jbc.271.41.25253. [DOI] [PubMed] [Google Scholar]
- Sciavolino PJ, Vilcek J. Regulation of metallothionein gene expression by TNF-alpha and IFN-beta in human fibroblasts. Cytokine. 1995;7:242–50. doi: 10.1006/cyto.1995.0028. [DOI] [PubMed] [Google Scholar]
- Silverman RH, Halloum A, Zhou A, Dong B, Al-Zoghaibi F, Kushner D, Zhou Q, Zhao J, Wiedmer T, Sims PJ. Suppression of ovarian carcinoma cell growth in vivo by the interferon-inducible plasma membrane protein, phospholipid scramblase 1. Cancer Res. 2002;62:397–402. [PubMed] [Google Scholar]
- Sedger LM, Shows DM, Blanton RA, Peschon JJ, Goodwin RG, Cosman D, Wiley SR. IFN-gamma mediates a novel antiviral activity through dynamic modulation of TRAIL and TRAIL receptor expression. J Immunol. 1999;163:920–6. [PubMed] [Google Scholar]
- Kayagaki N, Yamaguchi N, Nakayama M, Eto H, Okumura K, Yagita H. Type I interferons (IFNs) regulate tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) expression on human T cells: A novel mechanism for the antitumor effects of type I IFNs. J Exp Med. 1999;189:1451–60. doi: 10.1084/jem.189.9.1451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brinkmann V, Geiger T, Alkan S, Heusser CH. Interferon alpha increases the frequency of interferon gamma-producing human CD4+ T cells. J Exp Med. 1993;178:1655–63. doi: 10.1084/jem.178.5.1655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunter CA, Gabriel KE, Radzanowski T, Neyer LE, Remington JS. Type I interferons enhance production of IFN-gamma by NK cells. Immunol Lett. 1997;59:1–5. doi: 10.1016/S0165-2478(97)00091-6. [DOI] [PubMed] [Google Scholar]
- Sato M, Suemori H, Hata N, Asagiri M, Ogasawara K, Nakao K, Nakaya T, Katsuki M, Noguchi S, Tanaka N, et al. Distinct and essential roles of transcription factors IRF-3 and IRF-7 in response to viruses for IFN-alpha/beta gene induction. Immunity. 2000;13:539–48. doi: 10.1016/s1074-7613(00)00053-4. [DOI] [PubMed] [Google Scholar]
- Le Bon A, Tough DF. Links between innate and adaptive immunity via type I interferon. Curr Opin Immunol. 2002;14:432–6. doi: 10.1016/S0952-7915(02)00354-0. [DOI] [PubMed] [Google Scholar]
- Siegal FP, Kadowaki N, Shodell M, Fitzgerald-Bocarsly PA, Shah K, Ho S, Antonenko S, Liu YJ. The nature of the principal type 1 interferon-producing cells in human blood. Science. 1999;284:1835–7. doi: 10.1126/science.284.5421.1835. [DOI] [PubMed] [Google Scholar]
- Cella M, Jarrossay D, Facchetti F, Alebardi O, Nakajima H, Lanzavecchia A, Colonna M. Plasmacytoid monocytes migrate to inflamed lymph nodes and produce large amounts of type I interferon. Nat Med. 1999;5:919–23. doi: 10.1038/11360. [DOI] [PubMed] [Google Scholar]
- Kerkmann M, Rothenfusser S, Hornung V, Towarowski A, Wagner M, Sarris A, Giese T, Endres S, Hartmann G. Activation with CpG-A and CpG-B Oligonucleotides Reveals Two Distinct Regulatory Pathways of Type I IFN Synthesis in Human Plasmacytoid Dendritic Cells. J Immunol. 2003;170:4465–74. doi: 10.4049/jimmunol.170.9.4465. [DOI] [PubMed] [Google Scholar]
- Krieg AM. From A to Z on CpG. Trends Immunol. 2002;23:64–5. doi: 10.1016/S1471-4906(01)02150-0. [DOI] [PubMed] [Google Scholar]
- Guo J, Hui DJ, Merrick WC, Sen GC. A new pathway of translational regulation mediated by eukaryotic initiation factor 3. Embo J. 2000;19:6891–9. doi: 10.1093/emboj/19.24.6891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diaz-Guerra M, Rivas C, Esteban M. Inducible expression of the 2-5A synthetase/RNase L system results in inhibition of vaccinia virus replication. Virology. 1997;227:220–8. doi: 10.1006/viro.1996.8294. [DOI] [PubMed] [Google Scholar]
- Ponten A, Sick C, Weeber M, Haller O, Kochs G. Dominant-negative mutants of human MxA protein: domains in the carboxy-terminal moiety are important for oligomerization and antiviral activity. J Virol. 1997;71:2591–9. doi: 10.1128/jvi.71.4.2591-2599.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malakhov MP, Malakhova OA, Kim KI, Ritchie KJ, Zhang DE. UBP43 (USP18) specifically removes ISG15 from conjugated proteins. J Biol Chem. 2002;277:9976–81. doi: 10.1074/jbc.M109078200. [DOI] [PubMed] [Google Scholar]
- Park Y, Chang YS, Lee SW, Cho SY, Kim YK, Min KU, Kim YY, Cho SH, Sung YC. The enhanced effect of a hexameric deoxyriboguanosine run conjugation to CpG oligodeoxynucleotides on protection against allergic asthma. J Allergy Clin Immunol. 2001;108:570–6. doi: 10.1067/mai.2001.118517. [DOI] [PubMed] [Google Scholar]
- Marshall JD, Abtahi S, Eiden JJ, Tuck S, Milley R, Haycock F, Reid MJ, Kagey-Sobotka A, Creticos PS, Lichtenstein LM, et al. Immunostimulatory sequence DNA linked to the Amb a 1 allergen promotes T(H)1 cytokine expression while downregulating T(H)2 cytokine expression in PBMCs from human patients with ragweed allergy. J Allergy Clin Immunol. 2001;108:191–7. doi: 10.1067/mai.2001.116984. [DOI] [PubMed] [Google Scholar]
- Shirota H, Sano K, Hirasawa N, Terui T, Ohuchi K, Hattori T, Tamura G. B cells capturing antigen conjugated with CpG oligodeoxynucleotides induce Th1 cells by elaborating IL-12. J Immunol. 2002;169:787–94. doi: 10.4049/jimmunol.169.2.787. [DOI] [PubMed] [Google Scholar]
- Santeliz JV, Van Nest G, Traquina P, Larsen E, Wills-Karp M. Amb a 1-linked CpG oligodeoxynucleotides reverse established airway hyperresponsiveness in a murine model of asthma. J Allergy Clin Immunol. 2002;109:455–62. doi: 10.1067/mai.2002.122156. [DOI] [PubMed] [Google Scholar]
- Farrar JD, Murphy KM. Type I interferons and T helper development. Immunol Today. 2000;21:484–9. doi: 10.1016/S0167-5699(00)01710-2. [DOI] [PubMed] [Google Scholar]
- Krug A, Towarowski A, Britsch S, Rothenfusser S, Hornung V, Bals R, Giese T, Engelmann H, Endres S, Krieg AM, et al. Toll-like receptor expression reveals CpG DNA as a unique microbial stimulus for plasmacytoid dendritic cells which synergizes with CD40 ligand to induce high amounts of IL-12. Eur J Immunol. 2001;31:3026–37. doi: 10.1002/1521-4141(2001010)31:10<3026::AID-IMMU3026>3.0.CO;2-H. [DOI] [PubMed] [Google Scholar]
- Loetscher M, Loetscher P, Brass N, Meese E, Moser B. Lymphocyte-specific chemokine receptor CXCR3: regulation, chemokine binding and gene localization. Eur J Immunol. 1998;28:3696–705. doi: 10.1002/(SICI)1521-4141(199811)28:11<3696::AID-IMMU3696>3.3.CO;2-N. [DOI] [PubMed] [Google Scholar]
- Weng Y, Siciliano SJ, Waldburger KE, Sirotina-Meisher A, Staruch MJ, Daugherty BL, Gould SL, Springer MS, DeMartino JA. Binding and functional properties of recombinant and endogenous CXCR3 chemokine receptors. J Biol Chem. 1998;273:18288–91. doi: 10.1074/jbc.273.29.18288. [DOI] [PubMed] [Google Scholar]
- Loetscher P, Pellegrino A, Gong JH, Mattioli I, Loetscher M, Bardi G, Baggiolini M, Clark-Lewis I. The ligands of CXC chemokine receptor 3, I-TAC, Mig, and IP10, are natural antagonists for CCR3. J Biol Chem. 2001;276:2986–91. doi: 10.1074/jbc.M005652200. [DOI] [PubMed] [Google Scholar]