Abstract
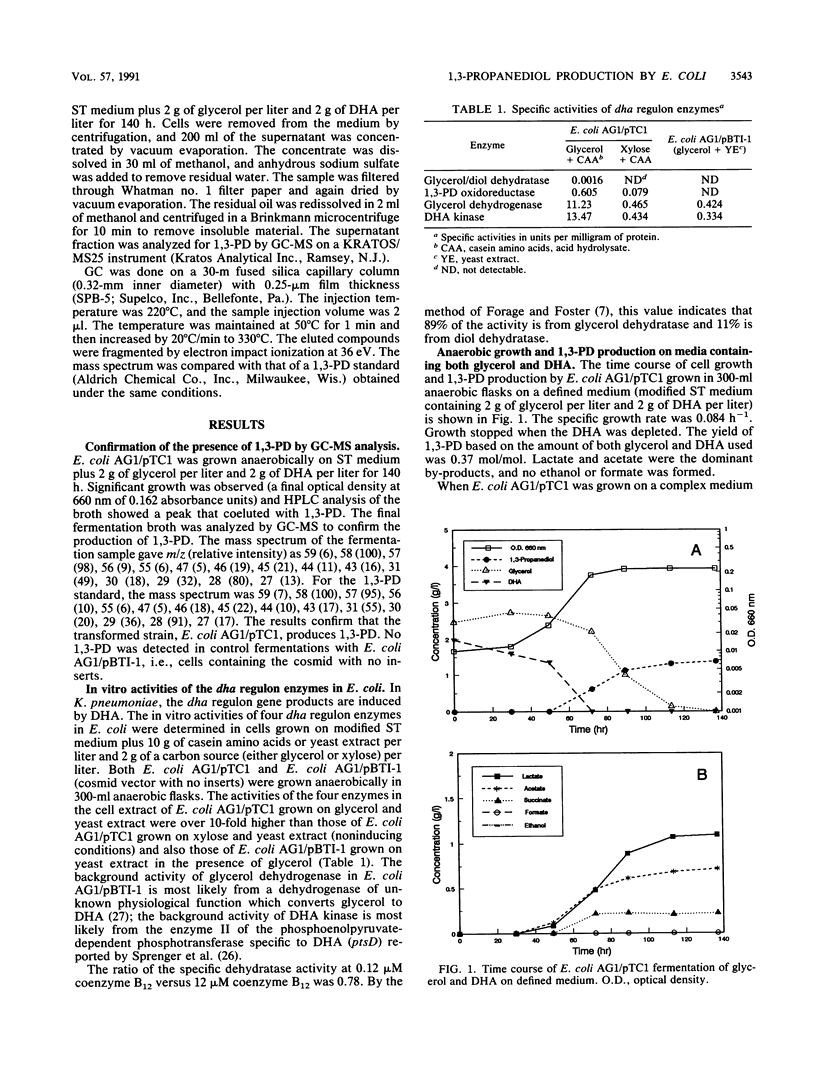
The dha regulon in Klebsiella pneumoniae enables the organism to grow anaerobically on glycerol and produce 1,3-propanediol (1,3-PD). Escherichia coli, which does not have a dha system, is unable to grow anaerobically on glycerol without an exogenous electron acceptor and does not produce 1,3-PD. A genomic library of K. pneumoniae ATCC 25955 constructed in E. coli AG1 was enriched for the ability to grow anaerobically on glycerol and dihydroxyacetone and was screened for the production of 1,3-PD. The cosmid pTC1 (42.5 kb total with an 18.2-kb major insert) was isolated from a 1,3-PD-producing strain of E. coli and found to possess enzymatic activities associated with four genes of the dha regulon: glycerol dehydratase (dhaB), 1,3-PD oxidoreductase (dhaT), glycerol dehydrogenase (dhaD), and dihydroxyacetone kinase (dhaK). All four activities were inducible by the presence of glycerol. When E. coli AG1/pTC1 was grown on complex medium plus glycerol, the yield of 1,3-PD from glycerol was 0.46 mol/mol. The major fermentation by-products were formate, acetate, and D-lactate. 1,3-PD is an intermediate in organic synthesis and polymer production. The 1,3-PD fermentation provides a useful model system for studying the interaction of a biochemical pathway in a foreign host and for developing strategies for metabolic pathway engineering.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bailey J. E. Toward a science of metabolic engineering. Science. 1991 Jun 21;252(5013):1668–1675. doi: 10.1126/science.2047876. [DOI] [PubMed] [Google Scholar]
- Cozzarelli N. R., Koch J. P., Hayashi S., Lin E. C. Growth stasis by accumulated L-alpha-glycerophosphate in Escherichia coli. J Bacteriol. 1965 Nov;90(5):1325–1329. doi: 10.1128/jb.90.5.1325-1329.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forage R. G., Foster M. A. Glycerol fermentation in Klebsiella pneumoniae: functions of the coenzyme B12-dependent glycerol and diol dehydratases. J Bacteriol. 1982 Feb;149(2):413–419. doi: 10.1128/jb.149.2.413-419.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forage R. G., Foster M. A. Resolution of the coenzyme B-12-dependent dehydratases of Klebsiella sp. and Citrobacter freundii. Biochim Biophys Acta. 1979 Aug 15;569(2):249–258. doi: 10.1016/0005-2744(79)90060-3. [DOI] [PubMed] [Google Scholar]
- Forage R. G., Lin E. C. DHA system mediating aerobic and anaerobic dissimilation of glycerol in Klebsiella pneumoniae NCIB 418. J Bacteriol. 1982 Aug;151(2):591–599. doi: 10.1128/jb.151.2.591-599.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Honda S., Toraya T., Fukui S. In situ reactivation of glycerol-inactivated coenzyme B12-dependent enzymes, glycerol dehydratase and diol dehydratase. J Bacteriol. 1980 Sep;143(3):1458–1465. doi: 10.1128/jb.143.3.1458-1465.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson E. A., Burke S. K., Forage R. G., Lin E. C. Purification and properties of dihydroxyacetone kinase from Klebsiella pneumoniae. J Bacteriol. 1984 Oct;160(1):55–60. doi: 10.1128/jb.160.1.55-60.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson E. A., Levine R. L., Lin E. C. Inactivation of glycerol dehydrogenase of Klebsiella pneumoniae and the role of divalent cations. J Bacteriol. 1985 Oct;164(1):479–483. doi: 10.1128/jb.164.1.479-483.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson E. A., Lin E. C. Klebsiella pneumoniae 1,3-propanediol:NAD+ oxidoreductase. J Bacteriol. 1987 May;169(5):2050–2054. doi: 10.1128/jb.169.5.2050-2054.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kohara Y., Akiyama K., Isono K. The physical map of the whole E. coli chromosome: application of a new strategy for rapid analysis and sorting of a large genomic library. Cell. 1987 Jul 31;50(3):495–508. doi: 10.1016/0092-8674(87)90503-4. [DOI] [PubMed] [Google Scholar]
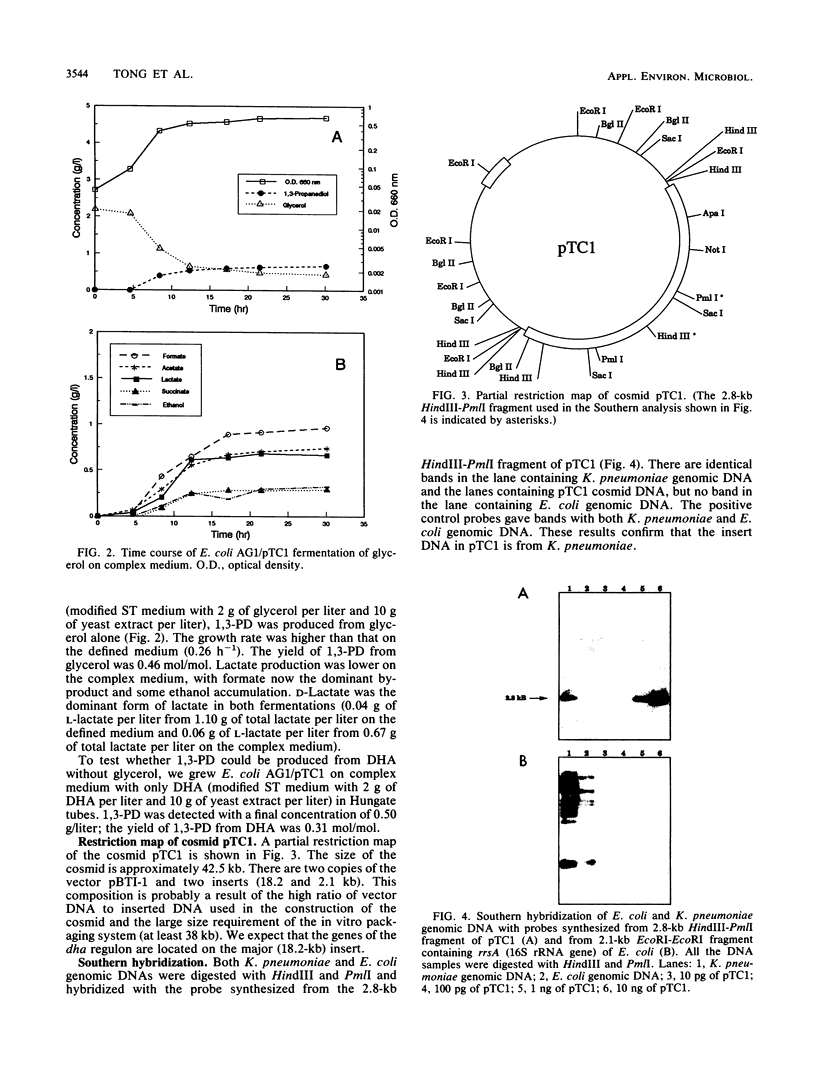
- LIN E. C., MAGASANIK B. The activation of glycerol dehydrogenase from Aerobacter aerogenes by monovalent cations. J Biol Chem. 1960 Jun;235:1820–1823. [PubMed] [Google Scholar]
- Lin E. C. Glycerol dissimilation and its regulation in bacteria. Annu Rev Microbiol. 1976;30:535–578. doi: 10.1146/annurev.mi.30.100176.002535. [DOI] [PubMed] [Google Scholar]
- Morris D. W., Noti J. D., Osborne F. A., Szalay A. A. Plasmid vectors capable of transferring large DNA fragments to yeast. DNA. 1981;1(1):27–36. doi: 10.1089/dna.1.1981.1.27. [DOI] [PubMed] [Google Scholar]
- Pawelkiewicz J., Zagalak B. Enzymic conversion of glycerol into beta-hydroxy-propionaldehyde in a cell-free extract from Aerobacter aerogenes. Acta Biochim Pol. 1965;12(3):207–218. [PubMed] [Google Scholar]
- Richey D. P., Lin E. C. Importance of facilitated diffusion for effective utilization of glycerol by Escherichia coli. J Bacteriol. 1972 Nov;112(2):784–790. doi: 10.1128/jb.112.2.784-790.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruch F. E., Lengeler J., Lin E. C. Regulation of glycerol catabolism in Klebsiella aerogenes. J Bacteriol. 1974 Jul;119(1):50–56. doi: 10.1128/jb.119.1.50-56.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneider Z., Pawelkiewicz J. The properties of glycerol dehydratase isolated from Aerobacter aerogenes, and the properties of the apoenzyme subunits. Acta Biochim Pol. 1966;13(4):311–328. [PubMed] [Google Scholar]
- Sprenger G. A., Hammer B. A., Johnson E. A., Lin E. C. Anaerobic growth of Escherichia coli on glycerol by importing genes of the dha regulon from Klebsiella pneumoniae. J Gen Microbiol. 1989 May;135(5):1255–1262. doi: 10.1099/00221287-135-5-1255. [DOI] [PubMed] [Google Scholar]
- Sridhara S., Wu T. T., Chused T. M., Lin E. C. Ferrous-activated nicotinamide adenine dinucleotide-linked dehydrogenase from a mutant of Escherichia coli capable of growth on 1, 2-propanediol. J Bacteriol. 1969 Apr;98(1):87–95. doi: 10.1128/jb.98.1.87-95.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stroinski A., Pawelkiewicz J., Johnson B. C. Allosteric interactions in glycerol dehydratase. Purification of enzyme and effects of positive and negative cooperativity for glycerol. Arch Biochem Biophys. 1974 Jun;162(2):321–330. doi: 10.1016/0003-9861(74)90189-1. [DOI] [PubMed] [Google Scholar]
- Toraya T., Ushio K., Fukui S., Hogenkamp P. C. Studies on the mechanism of the adenosylcobalamin-dependent diol dehydrase reaction by the use of analogs of the coenzyme. J Biol Chem. 1977 Feb 10;252(3):963–970. [PubMed] [Google Scholar]
- Zwaig N., Kistler W. S., Lin E. C. Glycerol kinase, the pacemaker for the dissimilation of glycerol in Escherichia coli. J Bacteriol. 1970 Jun;102(3):753–759. doi: 10.1128/jb.102.3.753-759.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]