Abstract
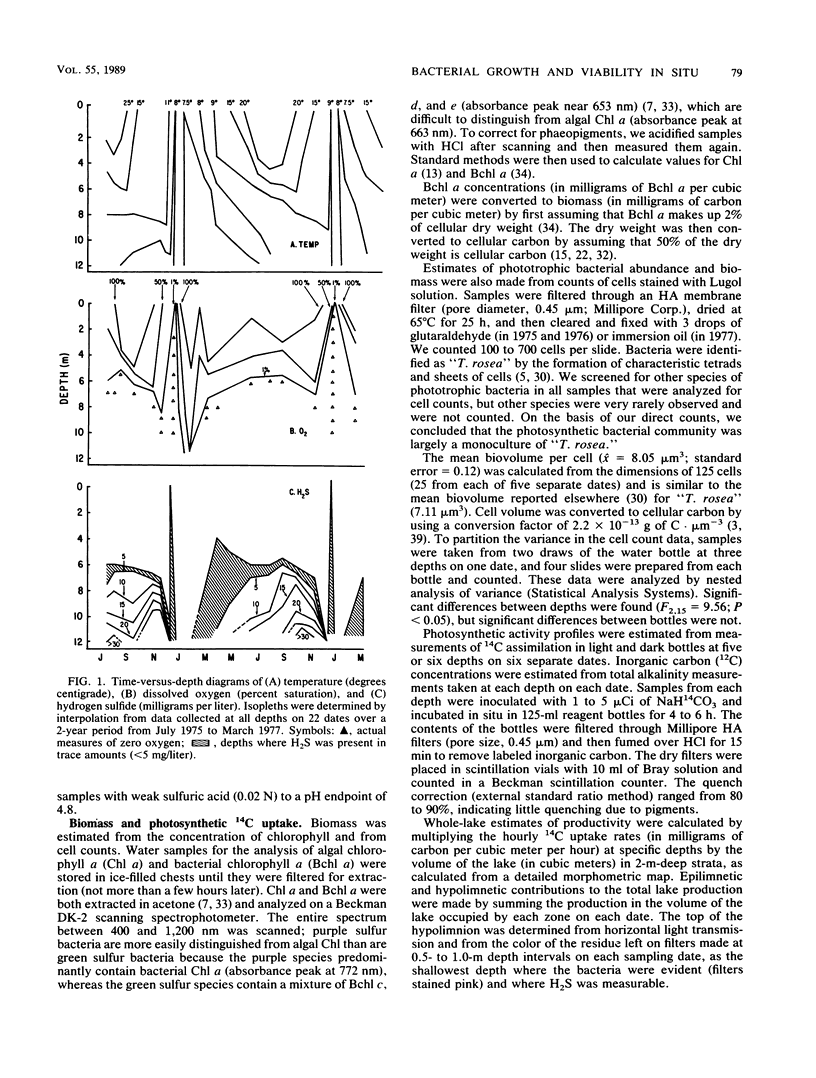
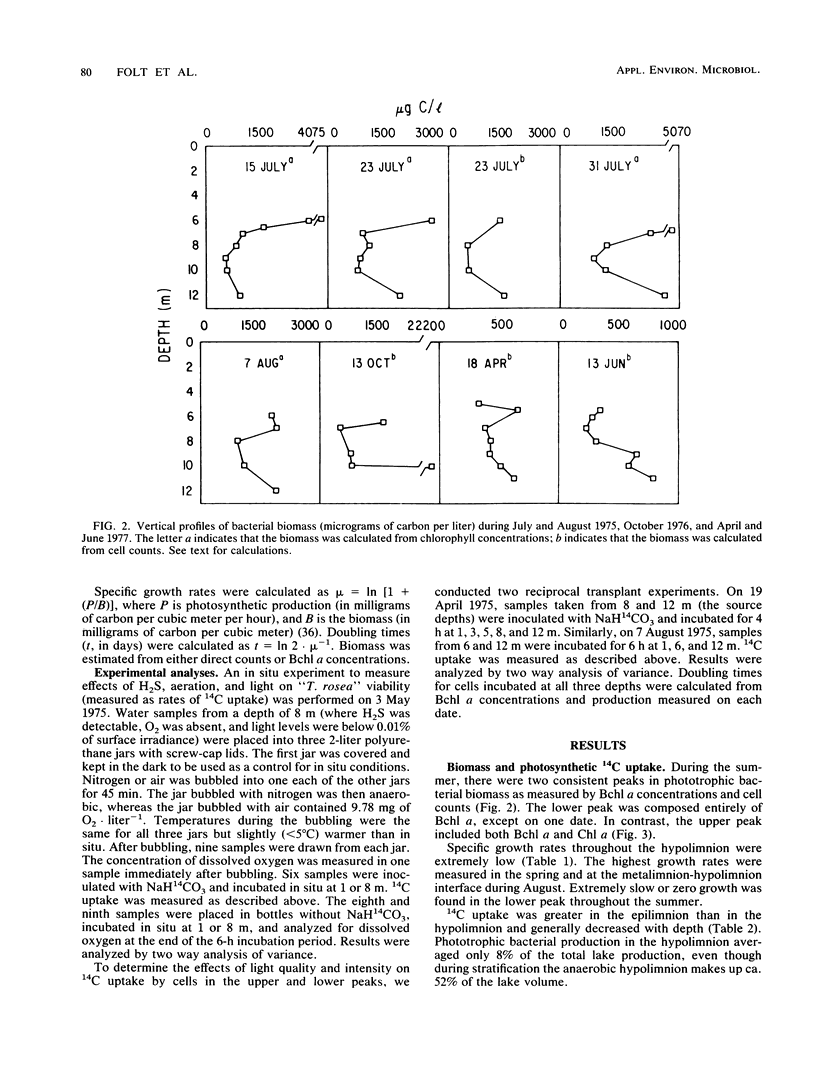
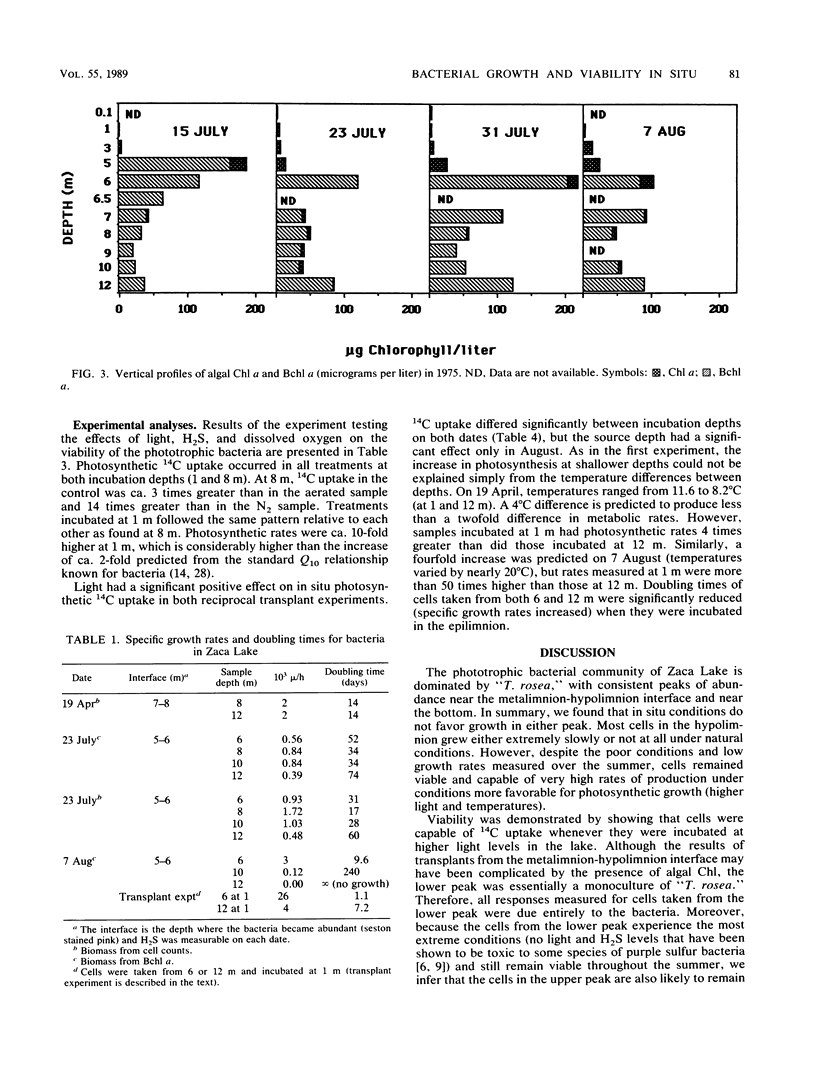
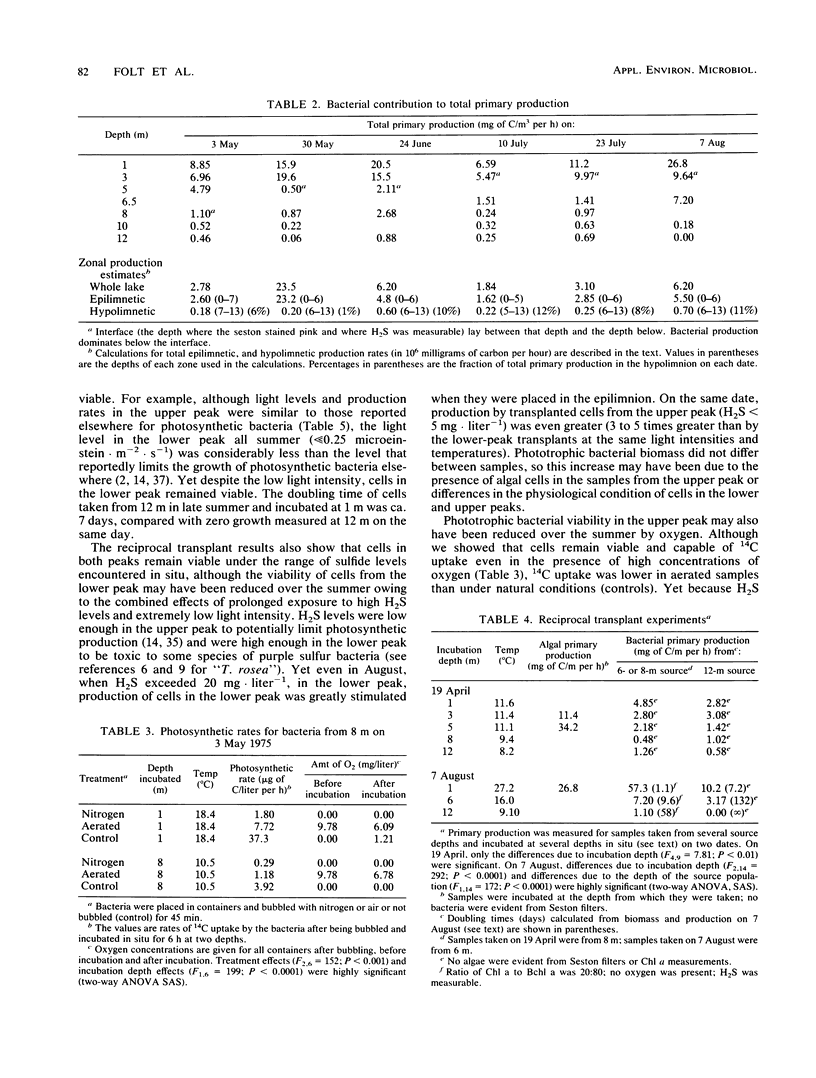
The growth and viability of an anoxygenic, phototrophic bacterial community in the hypolimnion of Zaca Lake, Calif., were compared throughout the summer. The community is dominated by a single species, “Thiopedia rosea,” that inhabits the entire hypolimnion (6 to 8 m) for approximately 11 months. Suboptimal conditions in the hypolimnion (extremely low light intensity, high or low H2S levels) result in zero or extremely low growth rates (doubling times > 1 month) for most of the population, most of the time, yet cells remain viable and capable of high specific growth rates (doubling times of 1 to 10 days) when placed under favorable conditions (higher light intensities and temperatures). We first conclude that phototrophic bacterial populations in situ may frequently exist in a viable yet nongrowing state. Second, the viability of cells is likely to be reduced with depth owing to higher concentrations of potentially toxic chemicals and to changes in the physiological state associated with the prolonged periods of darkness commonly found at the bottom of bacterial plates.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bergstein T., Henis Y., Cavari B. Z. Investigations on the photosynthetic sulfur bacterium Chlorobium phaeobacteroides causing seasonal blooms in Lake Kinneret. Can J Microbiol. 1979 Sep;25(9):999–1007. doi: 10.1139/m79-154. [DOI] [PubMed] [Google Scholar]
- Bratbak G., Dundas I. Bacterial dry matter content and biomass estimations. Appl Environ Microbiol. 1984 Oct;48(4):755–757. doi: 10.1128/aem.48.4.755-757.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caldwell D. E., Tiedje J. M. A morphological study of anaerobic bacteria from the hypolimnia of two Michigan lakes. Can J Microbiol. 1975 Mar;21(3):362–376. doi: 10.1139/m75-051. [DOI] [PubMed] [Google Scholar]
- Caldwell D. E., Tiedje J. M. The structure of anaerobic bacterial communities in the hypolimnia of several Michigan lakes. Can J Microbiol. 1975 Mar;21(3):377–385. doi: 10.1139/m75-052. [DOI] [PubMed] [Google Scholar]
- Harder W., Dijkhuizen L. Physiological responses to nutrient limitation. Annu Rev Microbiol. 1983;37:1–23. doi: 10.1146/annurev.mi.37.100183.000245. [DOI] [PubMed] [Google Scholar]
- Jannasch H. W. Estimations of bacterial growth rates in natural waters. J Bacteriol. 1969 Jul;99(1):156–160. doi: 10.1128/jb.99.1.156-160.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montesinos E. Change in Size of Chromatium minus Cells in Relation to Growth Rate, Sulfur Content, and Photosynthetic Activity: A Comparison of Pure Cultures and Field Populations. Appl Environ Microbiol. 1987 Apr;53(4):864–871. doi: 10.1128/aem.53.4.864-871.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Novitsky J. A., Morita R. Y. Survival of a psychrophilic marine Vibrio under long-term nutrient starvation. Appl Environ Microbiol. 1977 Mar;33(3):635–641. doi: 10.1128/aem.33.3.635-641.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roszak D. B., Colwell R. R. Survival strategies of bacteria in the natural environment. Microbiol Rev. 1987 Sep;51(3):365–379. doi: 10.1128/mr.51.3.365-379.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scherrer R., Shull V. E. Structure, partial elemental composition, and size of Thiopedia rosea cells and platelets. Can J Microbiol. 1986 Jul;32(7):607–610. doi: 10.1139/m86-113. [DOI] [PubMed] [Google Scholar]
- Wenke T. L., Vogt J. C. Temporal changes in a pink feedlot lagoon. Appl Environ Microbiol. 1981 Feb;41(2):381–385. doi: 10.1128/aem.41.2.381-385.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]


