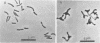Abstract
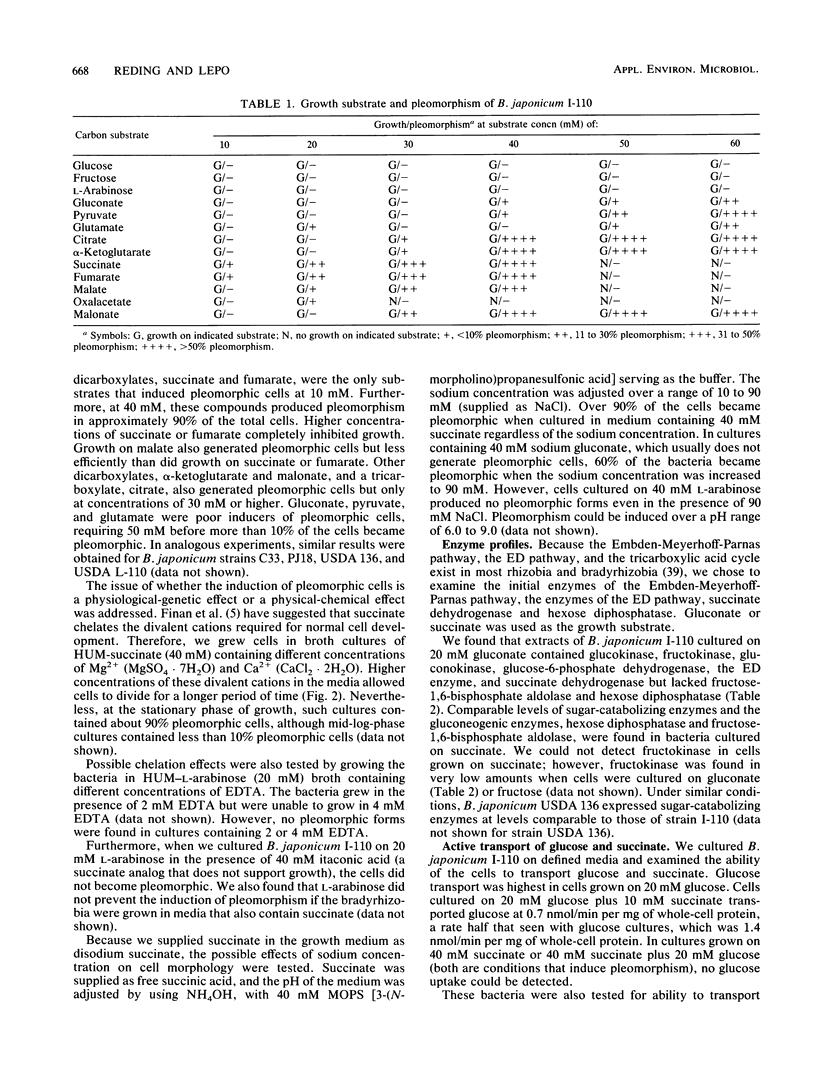
When Bradyrhizobium japonicum I-110 was transferred into medium containing 40 mM succinate or 40 mM fumarate, over 90% of the bacteria acquired a swollen, pleomorphic form similar to that of bacteroids. The induction of pleomorphism was dependent on the carbon substrate and concentration but was independent of the hydrogen ion and sodium ion concentration. Cell extracts of rod-shaped and pleomorphic cells contained enzymes required for sugar catabolism and gluconeogenesis. Variations in these enzyme profiles were correlated with the carbon source used and not with the conversion to the bacteroid-like morphology. Rod-shaped cells cultured on glucose or 10 mM succinate transported glucose and succinate; however, the pleomorphic cells behaved similarly to symbiotic bacteroids in that they lacked the ability to transport glucose and transported succinate at lower rates than did rod-shaped cells.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bach M. K., Magee W. E., Burris R. H. Translocation of Photosynthetic Products to Soybean Nodules and Their Role in Nitrogen Fixation. Plant Physiol. 1958 Mar;33(2):118–124. doi: 10.1104/pp.33.2.118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birkenhead K., Manian S. S., O'Gara F. Dicarboxylic acid transport in Bradyrhizobium japonicum: use of Rhizobium meliloti dct gene(s) to enhance nitrogen fixation. J Bacteriol. 1988 Jan;170(1):184–189. doi: 10.1128/jb.170.1.184-189.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Dazzo F. B., Napoli C. A., Hubbell D. H. Adsorption of bacteria to roots as related to host specificity in the Rhizobium-clover symbiosis. Appl Environ Microbiol. 1976 Jul;32(1):166–171. doi: 10.1128/aem.32.1.166-171.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finan T. M., Wood J. M., Jordan D. C. Succinate transport in Rhizobium leguminosarum. J Bacteriol. 1981 Oct;148(1):193–202. doi: 10.1128/jb.148.1.193-202.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finan T. M., Wood J. M., Jordan D. C. Symbiotic properties of C4-dicarboxylic acid transport mutants of Rhizobium leguminosarum. J Bacteriol. 1983 Jun;154(3):1403–1413. doi: 10.1128/jb.154.3.1403-1413.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gardiol A. E., Truchet G. L., Dazzo F. B. Requirement of succinate dehydrogenase activity for symbiotic bacteroid differentiation of Rhizobium meliloti in alfalfa nodules. Appl Environ Microbiol. 1987 Aug;53(8):1947–1950. doi: 10.1128/aem.53.8.1947-1950.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gardiol A., Arias A., Cerveñansky C., Martínez-Drets G. Succinate dehydrogenase mutant of Rhizobium meliloti. J Bacteriol. 1982 Sep;151(3):1621–1623. doi: 10.1128/jb.151.3.1621-1623.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hederstedt L., Holmgren E., Rutberg L. Characterization of a succinate dehydrogenase complex solubilized from the cytoplasmic membrane of Bacillus subtilis with the nonionic detergent Triton X-100. J Bacteriol. 1979 May;138(2):370–376. doi: 10.1128/jb.138.2.370-376.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- JORDAN D. C. The bacteroids of the genus Rhizobium. Bacteriol Rev. 1962 Jun;26:119–141. [PMC free article] [PubMed] [Google Scholar]
- Jordan D. C., Coulter W. H. On the cytology and synthetic capacities of natural and artificially produced bacteroids of Rhizobium leguminosarum. Can J Microbiol. 1965 Aug;11(4):709–720. doi: 10.1139/m65-094. [DOI] [PubMed] [Google Scholar]
- Keele B. B., Jr, Hamilton P. B., Elkan G. H. Gluconate catabolism in Rhizobium japonicum. J Bacteriol. 1970 Mar;101(3):698–704. doi: 10.1128/jb.101.3.698-704.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keele B. B., Jr, Hamilton P. B., Elkan G. H. Glucose catabolism in Rhizobium japonicum. J Bacteriol. 1969 Mar;97(3):1184–1191. doi: 10.1128/jb.97.3.1184-1191.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lynch W. H., MacLeod J., Franklin M. Effect of temperature on the activity and synthesis of glucose-catabolizing enzymes in Pseudomonas fluorescens. Can J Microbiol. 1975 Oct;21(10):1560–1572. doi: 10.1139/m75-229. [DOI] [PubMed] [Google Scholar]
- Maier R. J., Campbell N. E., Hanus F. J., Simpson F. B., Russell S. A., Evans H. J. Expression of hydrogenase activity in free-living Rhizobium japonicum. Proc Natl Acad Sci U S A. 1978 Jul;75(7):3258–3262. doi: 10.1073/pnas.75.7.3258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martínez-De Drets G., Arias A. Enzymatic basis for differentiation of Rhizobium into fast- and slow-growing groups. J Bacteriol. 1972 Jan;109(1):467–470. doi: 10.1128/jb.109.1.467-470.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McAllister C. F., Lepo J. E. Succinate transport by free-living forms of Rhizobium japonicum. J Bacteriol. 1983 Mar;153(3):1155–1162. doi: 10.1128/jb.153.3.1155-1162.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mulongoy K., Elkan G. H. Glucose catabolism in two derivatives of a Rhizobium japonicum strain differing in nitrogen-fixing efficiency. J Bacteriol. 1977 Jul;131(1):179–187. doi: 10.1128/jb.131.1.179-187.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peterson J. B., Larue T. A. Utilization of aldehydes and alcohols by soybean bacteroids. Plant Physiol. 1981 Aug;68(2):489–493. doi: 10.1104/pp.68.2.489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reibach P. H., Streeter J. G. Evaluation of active versus passive uptake of metabolites by Rhizobium japonicum bacteroids. J Bacteriol. 1984 Jul;159(1):47–52. doi: 10.1128/jb.159.1.47-52.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reibach P. H., Streeter J. G. Metabolism of C-labeled photosynthate and distribution of enzymes of glucose metabolism in soybean nodules. Plant Physiol. 1983 Jul;72(3):634–640. doi: 10.1104/pp.72.3.634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ronson C. W., Lyttleton P., Robertson J. G. C(4)-dicarboxylate transport mutants of Rhizobium trifolii form ineffective nodules on Trifolium repens. Proc Natl Acad Sci U S A. 1981 Jul;78(7):4284–4288. doi: 10.1073/pnas.78.7.4284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- STICKLAND L. H. The determination of small quantities of bacteria by means of the biuret reaction. J Gen Microbiol. 1951 Oct;5(4):698–703. doi: 10.1099/00221287-5-4-698. [DOI] [PubMed] [Google Scholar]
- Salminen S. O., Streeter J. G. Uptake and Metabolism of Carbohydrates by Bradyrhizobium japonicum Bacteroids. Plant Physiol. 1987 Mar;83(3):535–540. doi: 10.1104/pp.83.3.535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stowers M. D. Carbon metabolism in Rhizobium species. Annu Rev Microbiol. 1985;39:89–108. doi: 10.1146/annurev.mi.39.100185.000513. [DOI] [PubMed] [Google Scholar]
- Strijdom B. W., Allen O. N. Medium supplementation with L- and D-amino acids relative to growth and efficiency of Rhizobium meliloti. Can J Microbiol. 1966 Apr;12(2):275–283. doi: 10.1139/m66-038. [DOI] [PubMed] [Google Scholar]
- Urban J. E., Dazzo F. B. Succinate-Induced Morphology of Rhizobium trifolii 0403 Resembles That of Bacteroids in Clover Nodules. Appl Environ Microbiol. 1982 Jul;44(1):219–226. doi: 10.1128/aem.44.1.219-226.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]