Abstract
We report a novel in vitro method which, through application of appropriate nutrient limitations, enables growth rate control of adherent bacterial populations. Exponentially growing cells are collected by pressure filtration onto cellulose acetate membranes. Following inversion into the bases of modified fermentors, membranes and bacteria are perfused with fresh medium. Newly formed and loosely attached cells are eluted with spent medium. Steady-state conditions (dependent upon the medium flow rate) at which the adherent bacterial biomass is constant and proportional to the limiting nutrient concentrations are rapidly achieved, and within limits, the growth rate is proportional to the medium flow rate. Scanning electron microscopic studies showed that such populations consist of individual cells embedded within an extracellular polymer matrix.
Full text
PDF
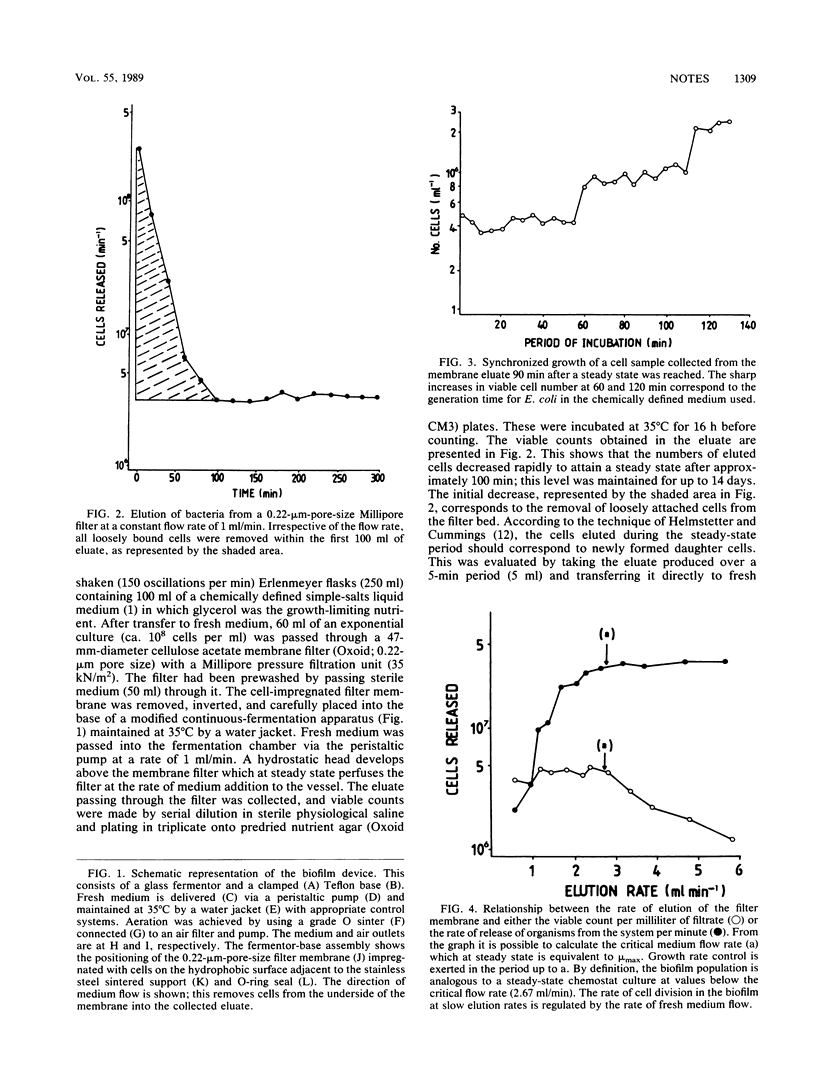
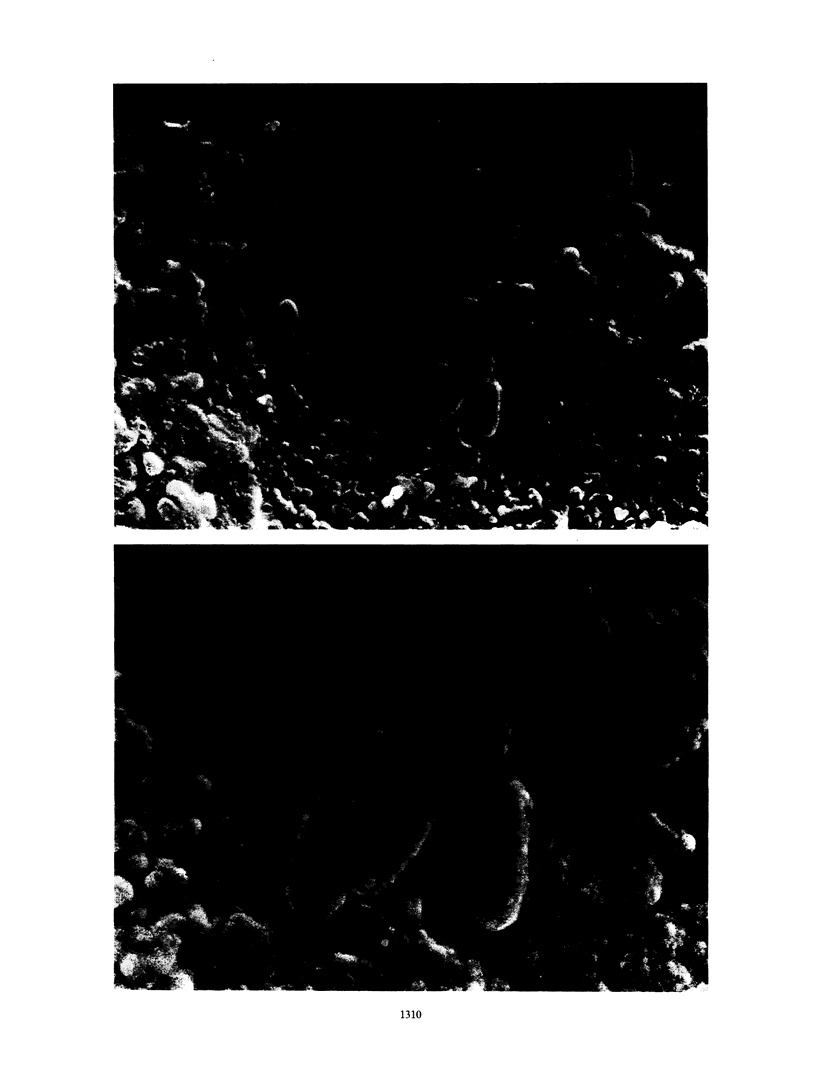

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Al-Hiti M. M., Gilbert P. A note on inoculum reproducibility: a comparison between solid and liquid culture. J Appl Bacteriol. 1983 Aug;55(1):173–175. doi: 10.1111/j.1365-2672.1983.tb02662.x. [DOI] [PubMed] [Google Scholar]
- Brown M. R., Allison D. G., Gilbert P. Resistance of bacterial biofilms to antibiotics: a growth-rate related effect? J Antimicrob Chemother. 1988 Dec;22(6):777–780. doi: 10.1093/jac/22.6.777. [DOI] [PubMed] [Google Scholar]
- Brown M. R., Williams P. The influence of environment on envelope properties affecting survival of bacteria in infections. Annu Rev Microbiol. 1985;39:527–556. doi: 10.1146/annurev.mi.39.100185.002523. [DOI] [PubMed] [Google Scholar]
- Costerton J. W., Cheng K. J., Geesey G. G., Ladd T. I., Nickel J. C., Dasgupta M., Marrie T. J. Bacterial biofilms in nature and disease. Annu Rev Microbiol. 1987;41:435–464. doi: 10.1146/annurev.mi.41.100187.002251. [DOI] [PubMed] [Google Scholar]
- Costerton J. W. The etiology and persistence of cryptic bacterial infections: a hypothesis. Rev Infect Dis. 1984 Sep-Oct;6 (Suppl 3):S608–S616. doi: 10.1093/clinids/6.supplement_3.s608. [DOI] [PubMed] [Google Scholar]
- Evans R. C., Holmes C. J. Effect of vancomycin hydrochloride on Staphylococcus epidermidis biofilm associated with silicone elastomer. Antimicrob Agents Chemother. 1987 Jun;31(6):889–894. doi: 10.1128/aac.31.6.889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilbert P., Brown M. R., Costerton J. W. Inocula for antimicrobial sensitivity testing: a critical review. J Antimicrob Chemother. 1987 Aug;20(2):147–154. doi: 10.1093/jac/20.2.147. [DOI] [PubMed] [Google Scholar]
- Gristina A. G., Hobgood C. D., Webb L. X., Myrvik Q. N. Adhesive colonization of biomaterials and antibiotic resistance. Biomaterials. 1987 Nov;8(6):423–426. doi: 10.1016/0142-9612(87)90077-9. [DOI] [PubMed] [Google Scholar]
- Gristina A. G., Oga M., Webb L. X., Hobgood C. D. Adherent bacterial colonization in the pathogenesis of osteomyelitis. Science. 1985 May 24;228(4702):990–993. doi: 10.1126/science.4001933. [DOI] [PubMed] [Google Scholar]
- HELMSTETTER C. E., CUMMINGS D. J. AN IMPROVED METHOD FOR THE SELECTION OF BACTERIAL CELLS AT DIVISION. Biochim Biophys Acta. 1964 Mar 16;82:608–610. doi: 10.1016/0304-4165(64)90453-2. [DOI] [PubMed] [Google Scholar]
- Marrie T. J., Costerton J. W. Mode of growth of bacterial pathogens in chronic polymicrobial human osteomyelitis. J Clin Microbiol. 1985 Dec;22(6):924–933. doi: 10.1128/jcm.22.6.924-933.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCoy W. F., Bryers J. D., Robbins J., Costerton J. W. Observations of fouling biofilm formation. Can J Microbiol. 1981 Sep;27(9):910–917. doi: 10.1139/m81-143. [DOI] [PubMed] [Google Scholar]
- Nickel J. C., Ruseska I., Wright J. B., Costerton J. W. Tobramycin resistance of Pseudomonas aeruginosa cells growing as a biofilm on urinary catheter material. Antimicrob Agents Chemother. 1985 Apr;27(4):619–624. doi: 10.1128/aac.27.4.619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pringle J. H., Fletcher M. Influence of substratum wettability on attachment of freshwater bacteria to solid surfaces. Appl Environ Microbiol. 1983 Mar;45(3):811–817. doi: 10.1128/aem.45.3.811-817.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shaw J. C., Bramhill B., Wardlaw N. C., Costerton J. W. Bacterial fouling in a model core system. Appl Environ Microbiol. 1985 Mar;49(3):693–701. doi: 10.1128/aem.49.3.693-701.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]




