Abstract
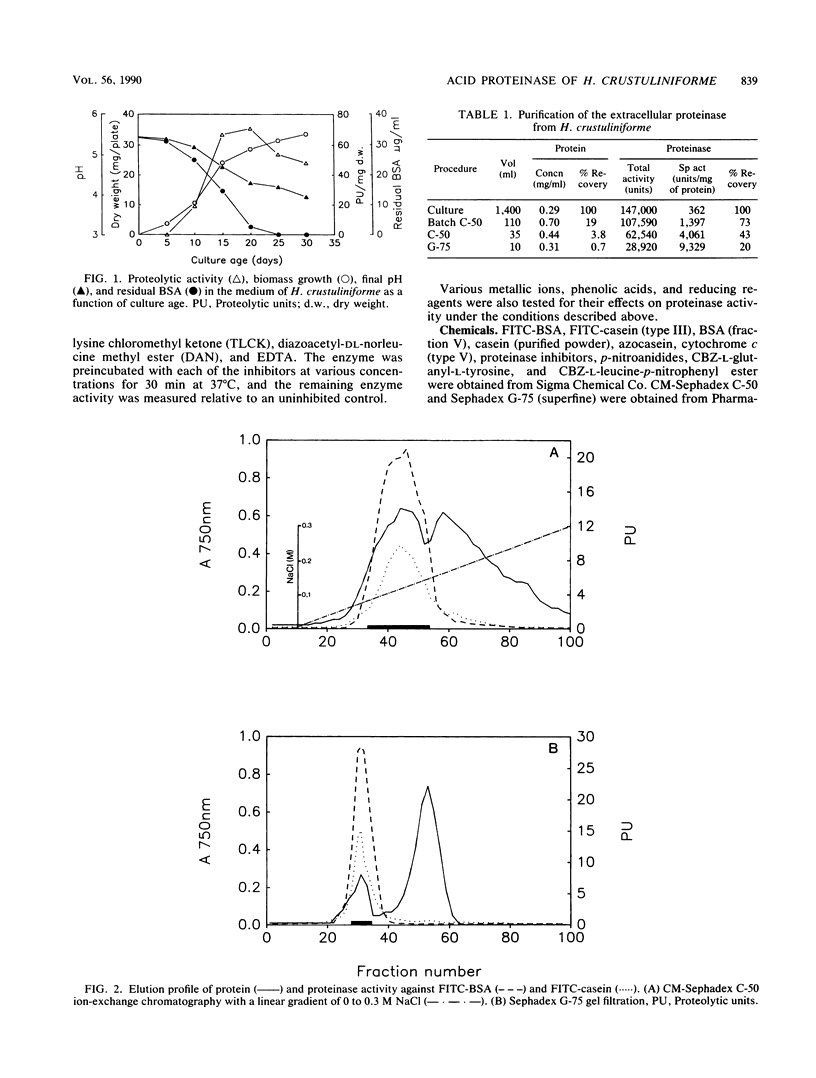
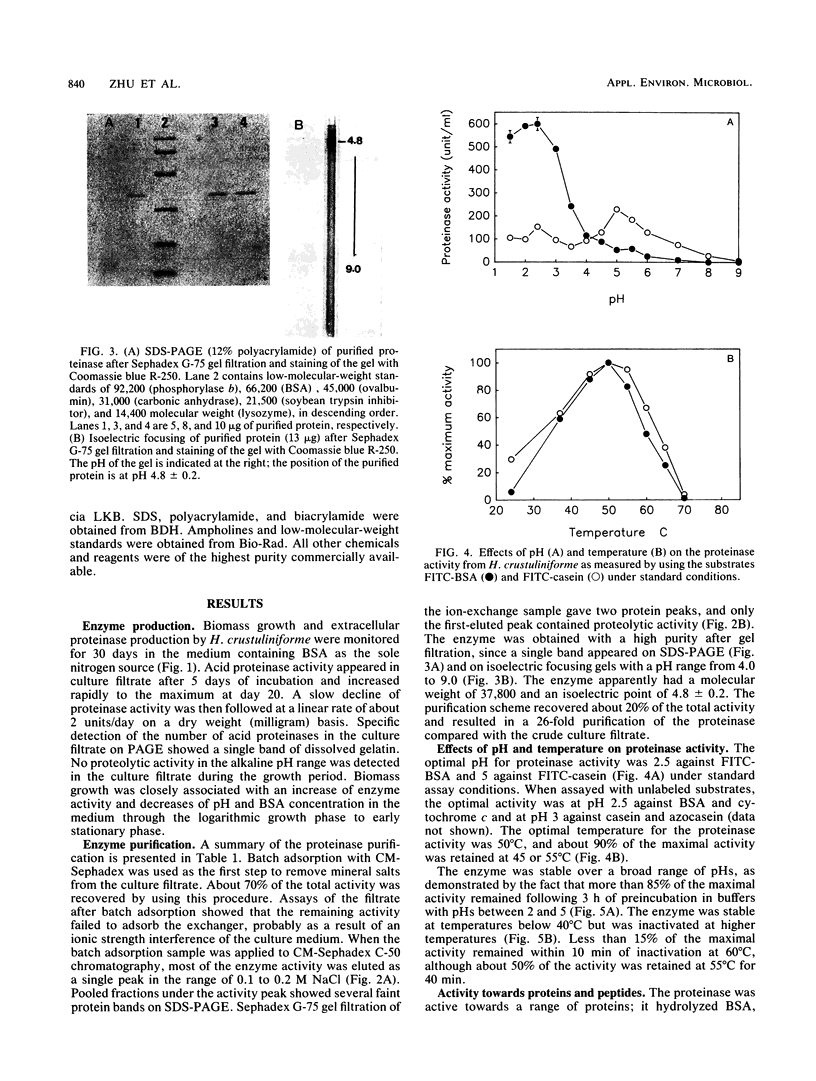
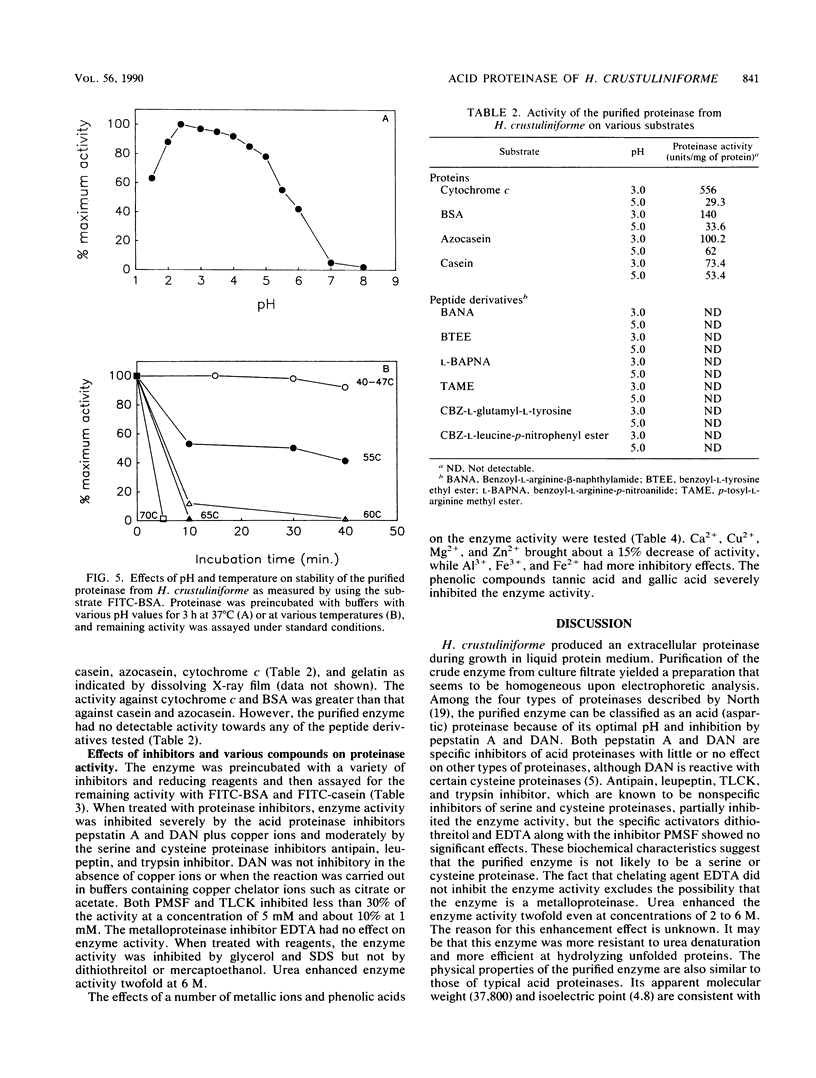
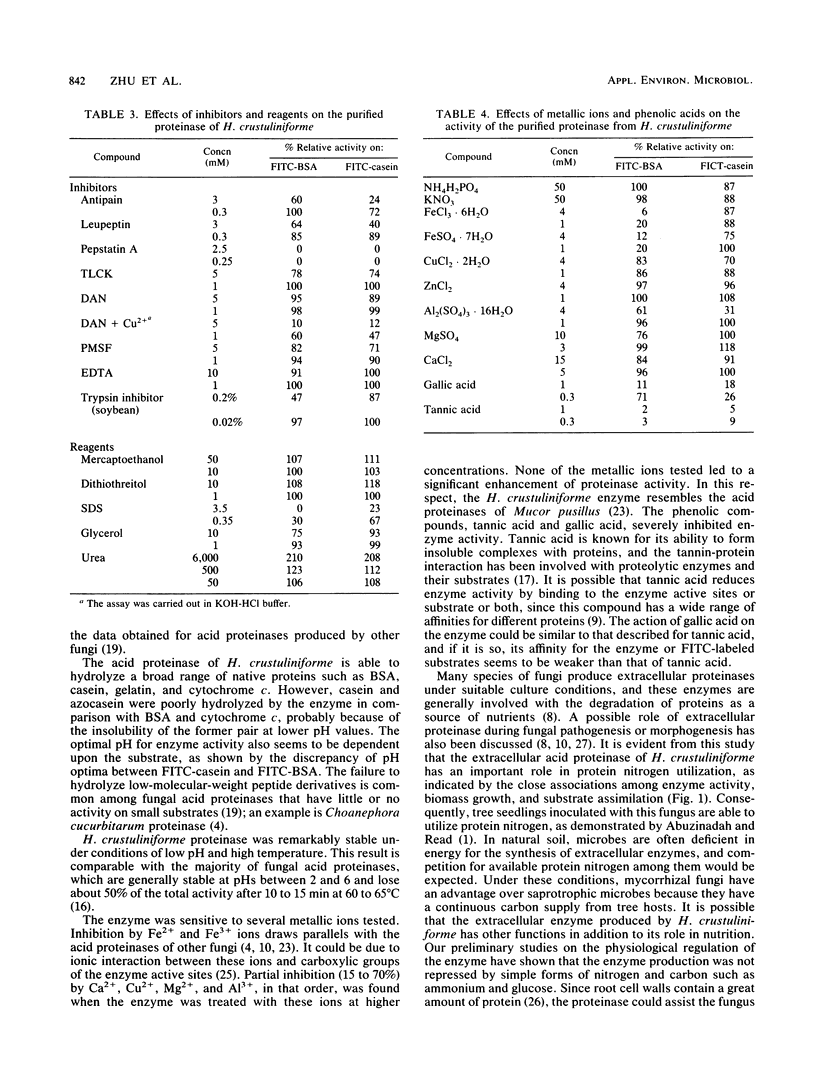
Hebeloma crustuliniforme produced an extracellular acid proteinase in a liquid medium containing bovine serum albumin as the sole nitrogen source. The proteinase was purified 26-fold with 20% activity recovery and was shown to have a molecular weight of 37,800 (as indicated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis) and an isoelectric point of 4.8 ± 0.2. The enzyme was most active at 50°C and pH 2.5 against bovine serum albumin and was stable in the absence of substrates at temperatures up to 45°C and pHs between 2.0 and 5.0. Pepstatin A, diazoacetyl-dl-norleucine methylester, metallic ions Fe2+ and Fe3+, and phenolic acids severely inhibited the enzyme activity, while antipain, leupeptin, N-α-p-tosyl-l-lysine chloromethyl ketone, and trypsin inhibitor inhibited the activity moderately. The proteinase hydrolyzed bovine serum albumin and cytochrome c rapidly compared with casein and azocasein but failed to hydrolyze any of the low-molecular-weight peptide derivatives tested.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Hagerman A. E., Butler L. G. The specificity of proanthocyanidin-protein interactions. J Biol Chem. 1981 May 10;256(9):4494–4497. [PubMed] [Google Scholar]
- Ichishima E. Purification and characterization of a new type of acid carboxypeptidase from Aspergillus. Biochim Biophys Acta. 1972 Jan 20;258(1):274–288. doi: 10.1016/0005-2744(72)90985-0. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- North M. J. Comparative biochemistry of the proteinases of eucaryotic microorganisms. Microbiol Rev. 1982 Sep;46(3):308–340. doi: 10.1128/mr.46.3.308-340.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Farrell P. H. High resolution two-dimensional electrophoresis of proteins. J Biol Chem. 1975 May 25;250(10):4007–4021. [PMC free article] [PubMed] [Google Scholar]
- REISFELD R. A., LEWIS U. J., WILLIAMS D. E. Disk electrophoresis of basic proteins and peptides on polyacrylamide gels. Nature. 1962 Jul 21;195:281–283. doi: 10.1038/195281a0. [DOI] [PubMed] [Google Scholar]
- Somkuti G. A., Babel F. J. Purification and properties of Mucor pusillus acid protease. J Bacteriol. 1968 Apr;95(4):1407–1414. doi: 10.1128/jb.95.4.1407-1414.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Twining S. S. Fluorescein isothiocyanate-labeled casein assay for proteolytic enzymes. Anal Biochem. 1984 Nov 15;143(1):30–34. doi: 10.1016/0003-2697(84)90553-0. [DOI] [PubMed] [Google Scholar]