Abstract
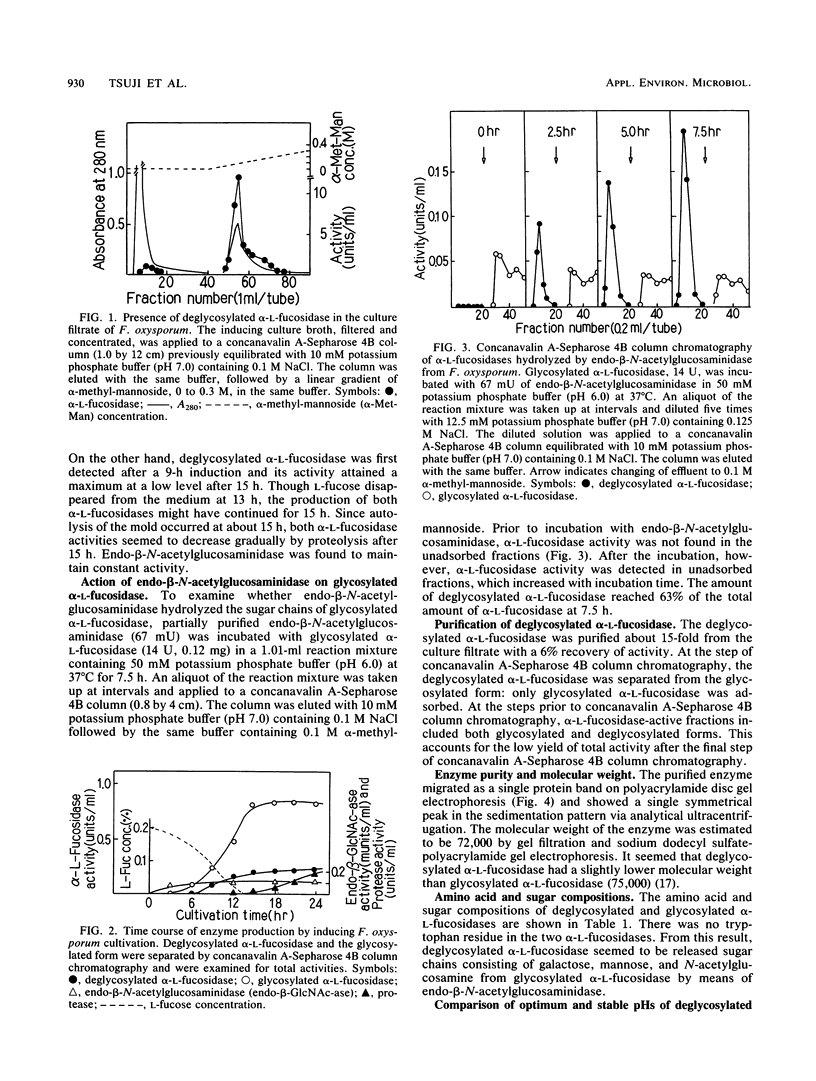
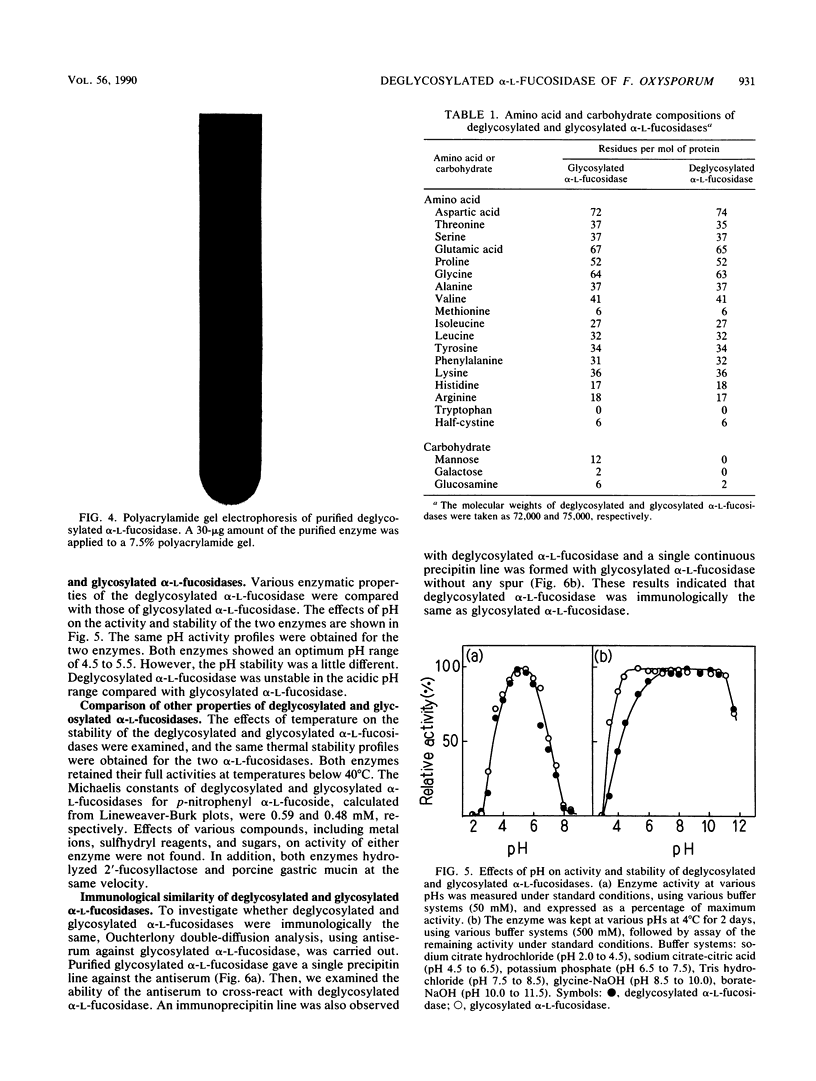
Two forms of alpha-L-fucosidase, deglycosylated and glycosylated, were found in the fucose-inducing culture broth of Fusarium oxysporum. Endo-beta-N-acetylglucosaminidase was also found in the same culture broth. The deglycosylated alpha-L-fucosidase was purified from the culture broth to homogeneity on polyacrylamide disc gel electrophoresis and analytical ultracentrifugation. Purified deglycosylated alpha-L-fucosidase was compared in chemical composition and immunological homology with glycosylated alpha-L-fucosidase which had been reported previously (K. Yamamoto, Y. Tsuji, H. Kumagai, and T. Tochikura, Agric. Biol. Chem. 50: 1689, 1986). Both enzymes had nearly the same amino acid compositions and were immunologically identical. Glycosylated alpha-L-fucosidase had mannose, galactose, and N-acetylglucosamine residues. In contrast, the deglycosylated enzyme had only N-acetylglucosamine residues. These results suggest that the deglycosylated alpha-L-fucosidase is formed by the release of sugar chains from the glycosylated form by Fusarium endo-beta-N-acetylglucosaminidase. Furthermore, various enzymatic properties were compared: the two alpha-L-fucosidases were found to exhibit similar catalytic activities and thermal stability profiles. The deglycosylated enzyme, however, was slightly unstable in the acidic pH range compared with the glycosylated enzyme.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andrews P. The gel-filtration behaviour of proteins related to their molecular weights over a wide range. Biochem J. 1965 Sep;96(3):595–606. doi: 10.1042/bj0960595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DAVIS B. J. DISC ELECTROPHORESIS. II. METHOD AND APPLICATION TO HUMAN SERUM PROTEINS. Ann N Y Acad Sci. 1964 Dec 28;121:404–427. doi: 10.1111/j.1749-6632.1964.tb14213.x. [DOI] [PubMed] [Google Scholar]
- Furbish F. S., Krett N. L., Barranger J. A., Brady R. O. Fucose plays a role in the clearance and uptake of glucocerebrosidase by rat liver cells. Biochem Biophys Res Commun. 1980 Aug 29;95(4):1768–1774. doi: 10.1016/s0006-291x(80)80103-3. [DOI] [PubMed] [Google Scholar]
- Goodwin T. W., Morton R. A. The spectrophotometric determination of tyrosine and tryptophan in proteins. Biochem J. 1946;40(5-6):628–632. doi: 10.1042/bj0400628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hitomi J., Murakami Y., Saitoh F., Shigemitsu N., Yamaguchi H. Purification and characterization of endo-beta-N-acetylglucosaminidase of Aspergillus oryzae. J Biochem. 1985 Aug;98(2):527–533. doi: 10.1093/oxfordjournals.jbchem.a135307. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Prieels J. P., Pizzo S. V., Glasgow L. R., Paulson J. C., Hill R. L. Hepatic receptor that specifically binds oligosaccharides containing fucosyl alpha1 leads to 3 N-acetylglucosamine linkages. Proc Natl Acad Sci U S A. 1978 May;75(5):2215–2219. doi: 10.1073/pnas.75.5.2215. [DOI] [PMC free article] [PubMed] [Google Scholar]




