Abstract
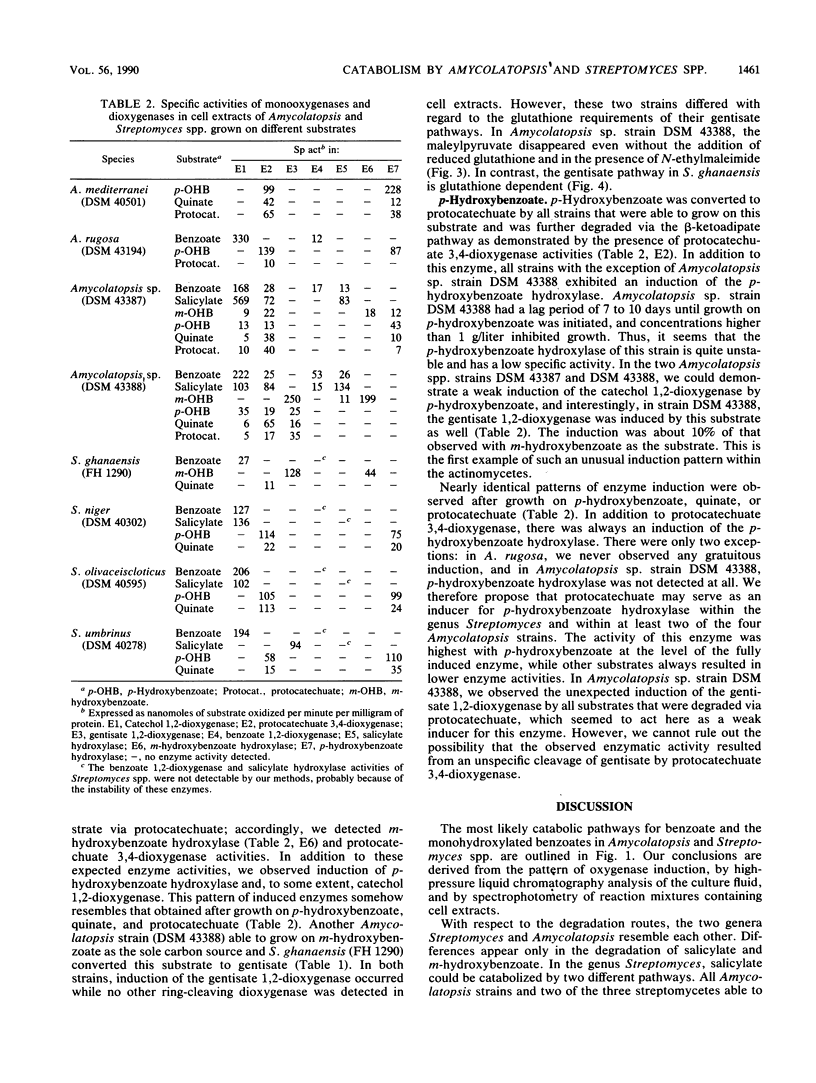
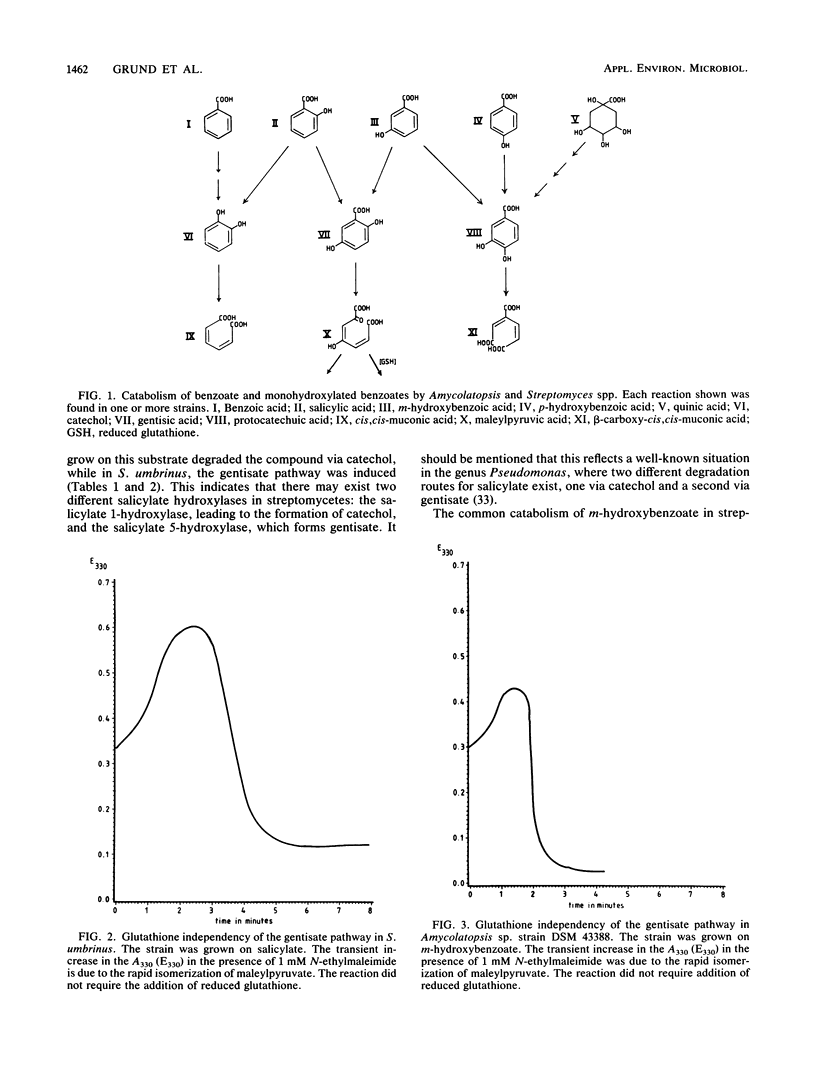
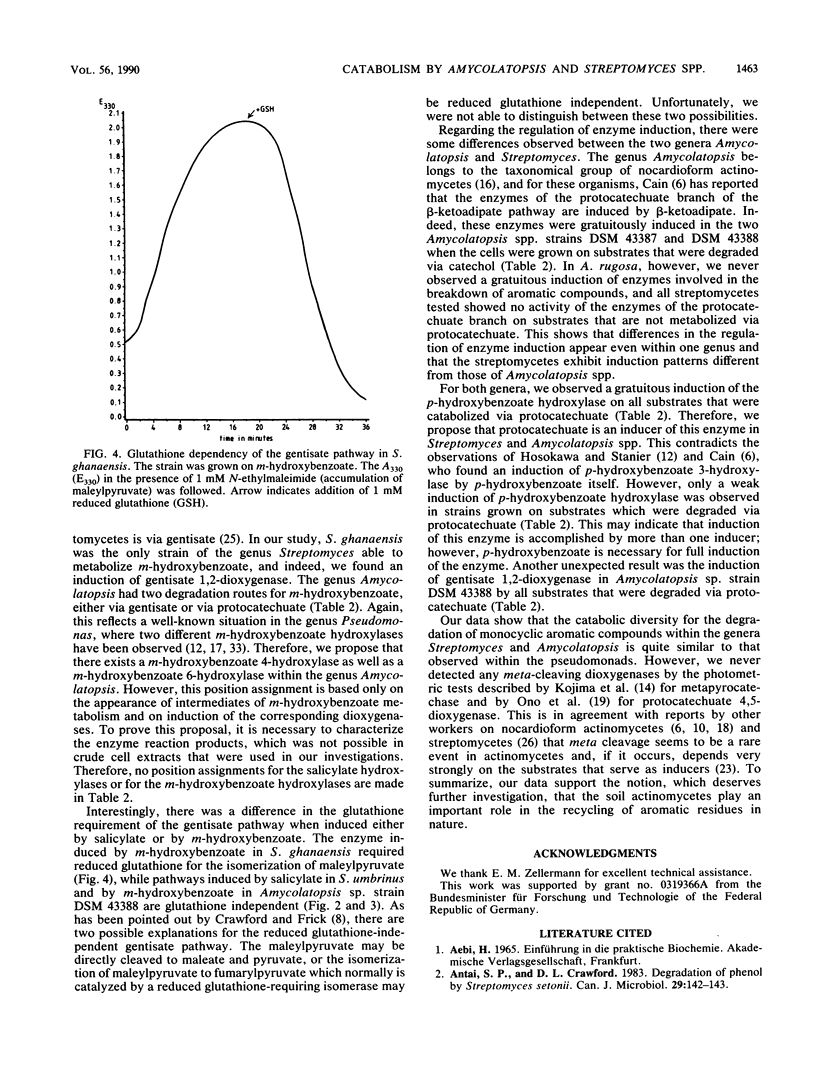
Eight actinomycetes of the genera Amycolatopsis and Streptomyces were tested for the degradation of aromatic compounds by growth in a liquid medium containing benzoate, monohydroxylated benzoates, or quinate as the principal carbon source. Benzoate was converted to catechol. The key intermediate in the degradation of salicylate was either catechol or gentisate, while m-hydroxybenzoate was metabolized via gentisate or protocatechuate. p-Hydroxybenzoate and quinate were converted to protocatechuate. Catechol, gentisate, and protocatechuate were cleaved by catechol 1,2-dioxygenase, gentisate 1,2-dioxygenase, and protocatechuate 3,4-dioxygenase, respectively. The requirement for glutathione in the gentisate pathway was dependent on the substrate and the particular strain. The conversion of p-hydroxybenzoate to protocatechuate by p-hydroxybenzoate hydroxylase was gratuitously induced by all substrates that were metabolized via protocatechuate as an intermediate, while protocatechuate 3,4-dioxygenase was gratuitously induced by benzoate and salicylate in two Amycolatopsis strains.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Apajalahti J. H., Salkinoja-Salonen M. S. Dechlorination and para-hydroxylation of polychlorinated phenols by Rhodococcus chlorophenolicus. J Bacteriol. 1987 Feb;169(2):675–681. doi: 10.1128/jb.169.2.675-681.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CAIN R. B. The microbial metabolism of nitro-aromatic compounds. J Gen Microbiol. 1958 Aug;19(1):1–14. doi: 10.1099/00221287-19-1-1. [DOI] [PubMed] [Google Scholar]
- Cain R. B. Utilization of anthranilic and nitrobenzoic acids by Nocardia opaca and a flavobacterium. J Gen Microbiol. 1966 Feb;42(2):219–235. doi: 10.1099/00221287-42-2-219. [DOI] [PubMed] [Google Scholar]
- Crawford R. L., Frick T. D. Rapid spectrophotometric differentiation between glutathione-dependent and glutathione-independent gentisate and homogentisate pathways. Appl Environ Microbiol. 1977 Aug;34(2):170–174. doi: 10.1128/aem.34.2.170-174.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crawford R. L., Hutton S. W., Chapman P. J. Purification and properties of gentisate 1,2-dioxygenase from Moraxella osloensis. J Bacteriol. 1975 Mar;121(3):794–799. doi: 10.1128/jb.121.3.794-799.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Groseclose E. E., Ribbons D. W., Hughes H. 3-Hydroxybenzoate 6-hydroxylase from Pseudomonas aeruginosa. Biochem Biophys Res Commun. 1973 Dec 10;55(3):897–903. doi: 10.1016/0006-291x(73)91228-x. [DOI] [PubMed] [Google Scholar]
- Hosokawa K., Stanier R. Y. Crystallization and properties of p-hydroxybenzoate hydroxylase from Pseudomonas putida. J Biol Chem. 1966 May 25;241(10):2453–2460. [PubMed] [Google Scholar]
- KOJIMA Y., ITADA N., HAYAISHI O. Metapyrocatachase: a new catechol-cleaving enzyme. J Biol Chem. 1961 Aug;236:2223–2228. [PubMed] [Google Scholar]
- LACK L. The enzymic oxidation of gentisic acid. Biochim Biophys Acta. 1959 Jul;34:117–123. doi: 10.1016/0006-3002(59)90239-2. [DOI] [PubMed] [Google Scholar]
- Michalover J. L., Ribbons D. W., Hughes H. 3-Hydroxybenzoate 4-hydroxylase from Pseudomonas testosteroni. Biochem Biophys Res Commun. 1973 Dec 10;55(3):888–896. doi: 10.1016/0006-291x(73)91227-8. [DOI] [PubMed] [Google Scholar]
- Ono K., Nozaki M., Hayaishi O. Purification and some properties of protocatechuate 4,5-dioxygenase. Biochim Biophys Acta. 1970 Nov 11;220(2):224–238. doi: 10.1016/0005-2744(70)90008-2. [DOI] [PubMed] [Google Scholar]
- Ornston L. N., Stanier R. Y. The conversion of catechol and protocatechuate to beta-ketoadipate by Pseudomonas putida. J Biol Chem. 1966 Aug 25;241(16):3776–3786. [PubMed] [Google Scholar]
- Pometto A. L., 3rd, Crawford D. L. L-Phenylalanine and L-tyrosine catabolism by selected Streptomyces species. Appl Environ Microbiol. 1985 Mar;49(3):727–729. doi: 10.1128/aem.49.3.727-729.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pometto A. L., 3rd, Sutherland J. B., Crawford D. L. Streptomyces setonii: catabolism of vanillic acid via guaiacol and catechol. Can J Microbiol. 1981 Jun;27(6):636–638. doi: 10.1139/m81-097. [DOI] [PubMed] [Google Scholar]
- Seiler H. Identification key for coryneform bacteria derived by numerical taxonomic studies. J Gen Microbiol. 1983 May;129(5):1433–1471. doi: 10.1099/00221287-129-5-1433. [DOI] [PubMed] [Google Scholar]
- Sutherland J. B., Crawford D. L., Pometto A. L., 3rd Metabolism of cinnamic, p-coumaric, and ferulic acids by Streptomyces setonii. Can J Microbiol. 1983 Oct;29(10):1253–1257. doi: 10.1139/m83-195. [DOI] [PubMed] [Google Scholar]
- Sutherland J. B., Crawford D. L., Pometto A. L. Catabolism of substituted benzoic acids by streptomyces species. Appl Environ Microbiol. 1981 Feb;41(2):442–448. doi: 10.1128/aem.41.2.442-448.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watson G. K., Cain R. B. Microbial metabolism of the pyridine ring. Metabolic pathways of pyridine biodegradation by soil bacteria. Biochem J. 1975 Jan;146(1):157–172. doi: 10.1042/bj1460157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wheelis M. L., Palleroni N. J., Stanier R. Y. The metabolism of aromatic acids by Pseudomonas testosteroni and P. acidovorans. Arch Mikrobiol. 1967;59(1):302–314. doi: 10.1007/BF00406344. [DOI] [PubMed] [Google Scholar]
- YAMAMOTO S., KATAGIRI M., MAENO H., HAYAISHI O. SALICYLATE HYDROXYLASE, A MONOOXYGENASE REQUIRING FLAVIN ADENINE DINUCLEOTIDE. I. PURIFICATION AND GENERAL PROPERTIES. J Biol Chem. 1965 Aug;240:3408–3413. [PubMed] [Google Scholar]
- Yamaguchi M., Fujisawa H. Purification and characterization of an oxygenase component in benzoate 1,2-dioxygenase system from Pseudomonas arvilla C-1. J Biol Chem. 1980 Jun 10;255(11):5058–5063. [PubMed] [Google Scholar]



