Abstract
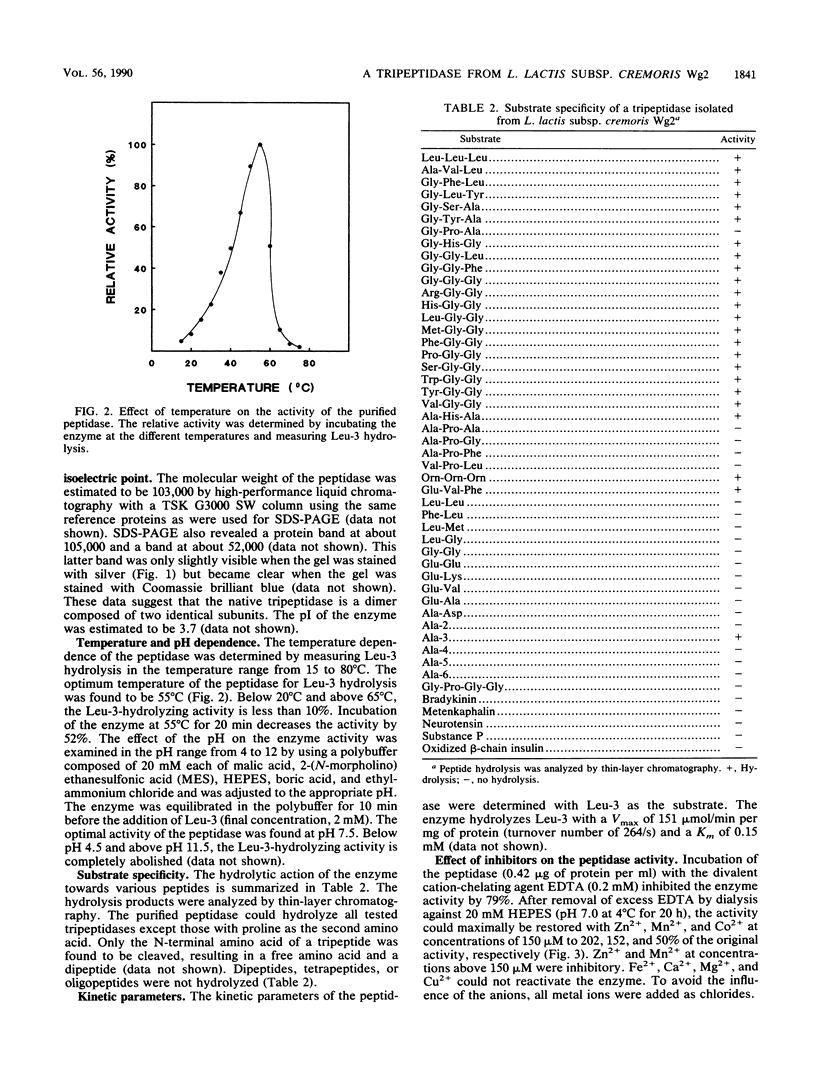
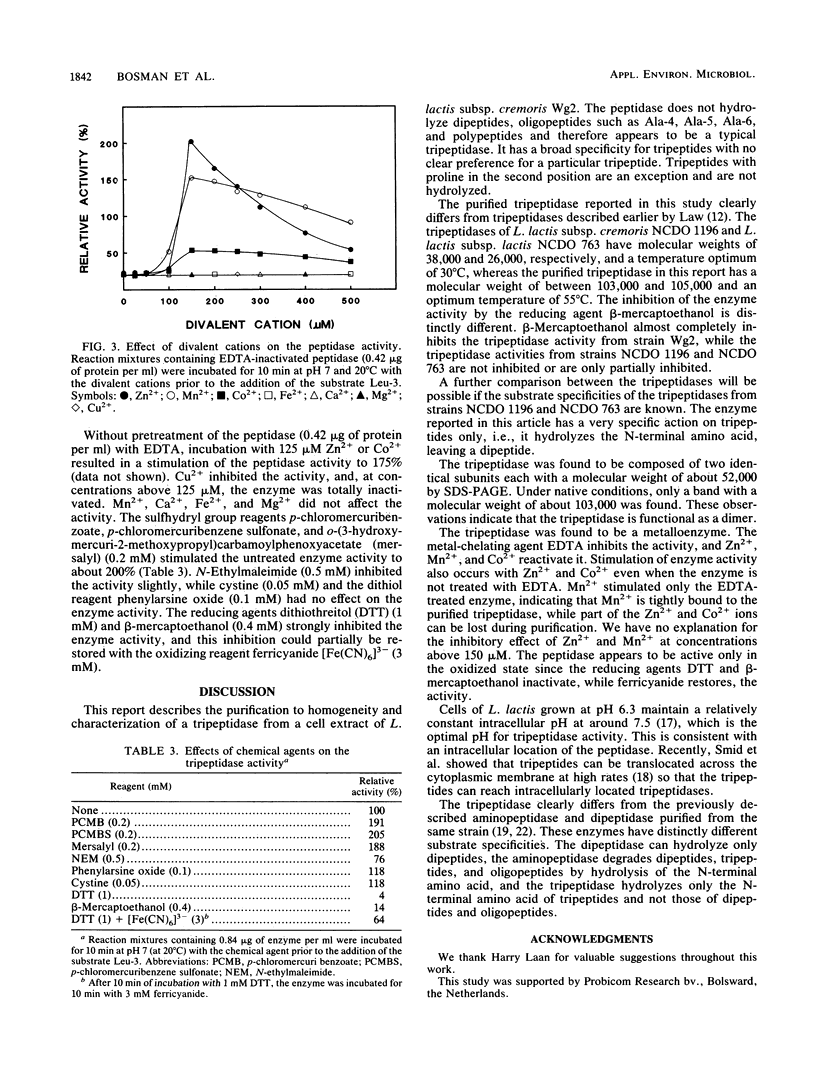
A tripeptidase from a cell extract of Lactococcus lactis subsp. cremoris Wg2 has been purified to homogeneity by DEAE-Sephacel and phenyl-Sepharose chromatography followed by gel filtration over a Sephadex G-100 SF column and a high-performance liquid chromatography TSK G3000 SW column. The enzyme appears to be a dimer with a molecular weight of between 103,000 and 105,000 and is composed of two identical subunits each with a molecular weight of about 52,000. The tripeptidase is capable of hydrolyzing only tripeptides. The enzyme activity is optimal at pH 7.5 and at 55°C. EDTA inhibits the activity, and this can be reactivated with Zn2+, Mn2+, and partially with Co2+. The reducing agents dithiothreitol and β-mercaptoethanol and the divalent cation Cu2+ inhibit tripeptidase activity. Kinetic studies indicate that the peptidase hydrolyzes leucyl-leucyl-leucine with a Km of 0.15 mM and a Vmax of 151 μmol/min per mg of protein.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Doi E., Shibata D., Matoba T. Modified colorimetric ninhydrin methods for peptidase assay. Anal Biochem. 1981 Nov 15;118(1):173–184. doi: 10.1016/0003-2697(81)90175-5. [DOI] [PubMed] [Google Scholar]
- Exterkate F. A. Location of Peptidases Outside and Inside the Membrane of Streptococcus cremoris. Appl Environ Microbiol. 1984 Jan;47(1):177–183. doi: 10.1128/aem.47.1.177-183.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Exterkate F. A., de Veer G. J. Purification and Some Properties of a Membrane-Bound Aminopeptidase A from Streptococcus cremoris. Appl Environ Microbiol. 1987 Mar;53(3):577–583. doi: 10.1128/aem.53.3.577-583.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hugenholtz J., van Sinderen D., Kok J., Konings W. N. Cell Wall-Associated Proteases of Streptococcus cremoris Wg2. Appl Environ Microbiol. 1987 Apr;53(4):853–859. doi: 10.1128/aem.53.4.853-859.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K., Favre M. Maturation of the head of bacteriophage T4. I. DNA packaging events. J Mol Biol. 1973 Nov 15;80(4):575–599. doi: 10.1016/0022-2836(73)90198-8. [DOI] [PubMed] [Google Scholar]
- Law B. A., Kolstad J. Proteolytic systems in lactic acid bacteria. Antonie Van Leeuwenhoek. 1983 Sep;49(3):225–245. doi: 10.1007/BF00399500. [DOI] [PubMed] [Google Scholar]
- Monnet V., Le Bars D., Gripon J. C. Purification and characterization of a cell wall proteinase from Streptococcus lactis NCDO 763. J Dairy Res. 1987 May;54(2):247–255. doi: 10.1017/s0022029900025383. [DOI] [PubMed] [Google Scholar]
- Neviani E., Boquien C. Y., Monnet V., Thanh L. P., Gripon J. C. Purification and Characterization of an Aminopeptidase from Lactococcus lactis subsp. cremoris AM2. Appl Environ Microbiol. 1989 Sep;55(9):2308–2314. doi: 10.1128/aem.55.9.2308-2314.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poolman B., Driessen A. J., Konings W. N. Regulation of solute transport in streptococci by external and internal pH values. Microbiol Rev. 1987 Dec;51(4):498–508. doi: 10.1128/mr.51.4.498-508.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smid E. J., Plapp R., Konings W. N. Peptide uptake is essential for growth of Lactococcus lactis on the milk protein casein. J Bacteriol. 1989 Nov;171(11):6135–6140. doi: 10.1128/jb.171.11.6135-6140.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan P. S., Konings W. N. Purification and Characterization of an Aminopeptidase from Lactococcus lactis subsp. cremoris Wg2. Appl Environ Microbiol. 1990 Feb;56(2):526–532. doi: 10.1128/aem.56.2.526-532.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wray W., Boulikas T., Wray V. P., Hancock R. Silver staining of proteins in polyacrylamide gels. Anal Biochem. 1981 Nov 15;118(1):197–203. doi: 10.1016/0003-2697(81)90179-2. [DOI] [PubMed] [Google Scholar]
- Yan T. R., Azuma N., Kaminogawa S., Yamauchi K. Purification and Characterization of a Substrate-Size-Recognizing Metalloendopeptidase from Streptococcus cremoris H61. Appl Environ Microbiol. 1987 Oct;53(10):2296–2302. doi: 10.1128/aem.53.10.2296-2302.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan T. R., Azuma N., Kaminogawa S., Yamauchi K. Purification and characterization of a novel metalloendopeptidase from Streptococcus cremoris H61. A metalloendopeptidase that recognizes the size of its substrate. Eur J Biochem. 1987 Mar 2;163(2):259–265. doi: 10.1111/j.1432-1033.1987.tb10796.x. [DOI] [PubMed] [Google Scholar]
- van Boven A., Tan P. S. T., Konings W. N. Purification and Characterization of a Dipeptidase from Streptococcus cremoris Wg2. Appl Environ Microbiol. 1988 Jan;54(1):43–49. doi: 10.1128/aem.54.1.43-49.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]



