Abstract
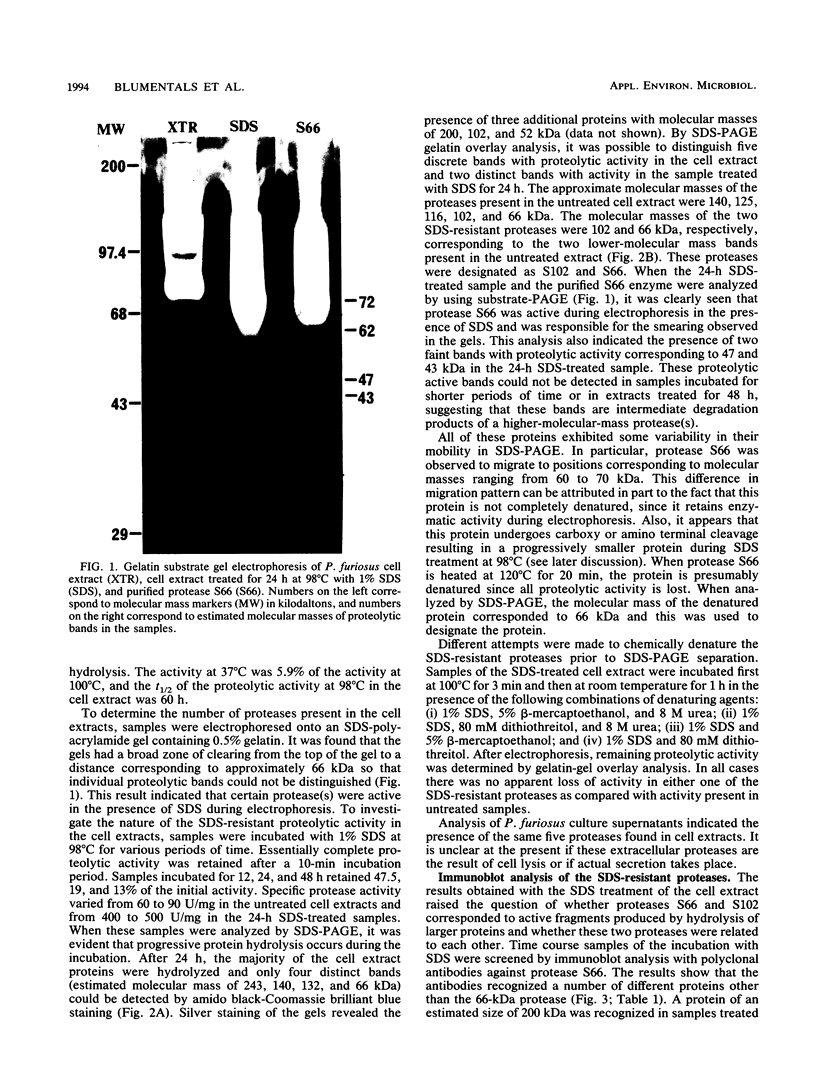
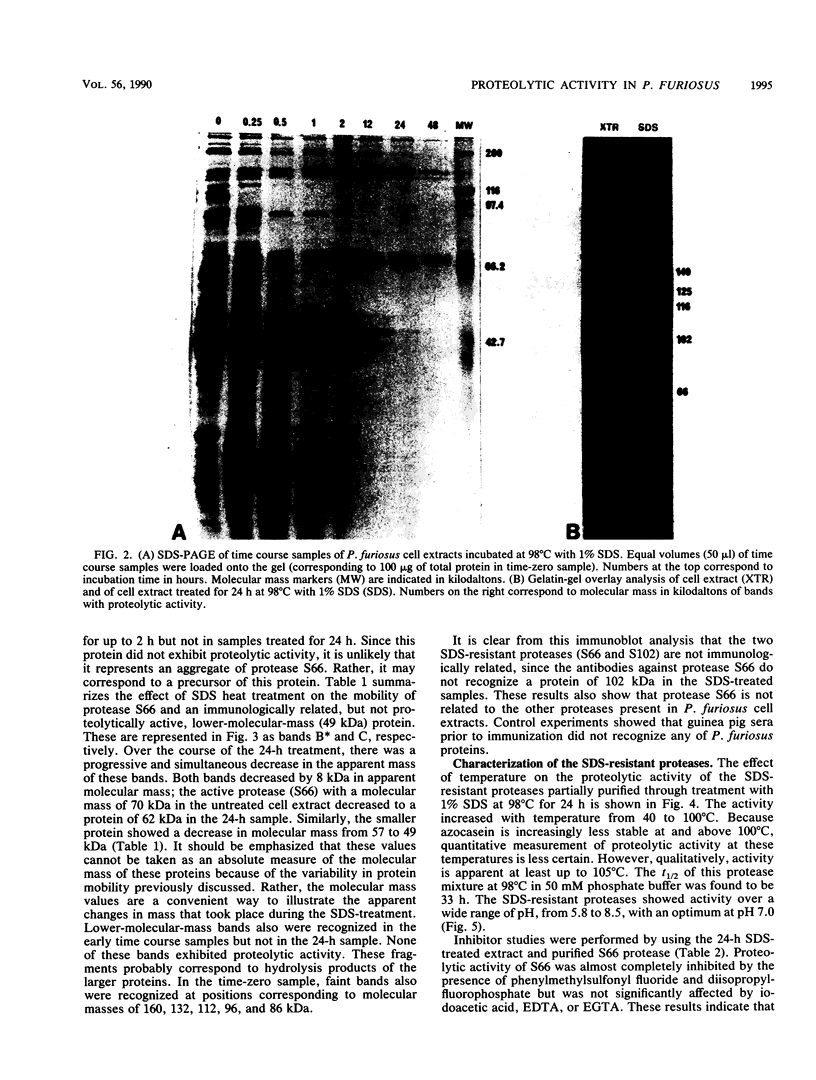
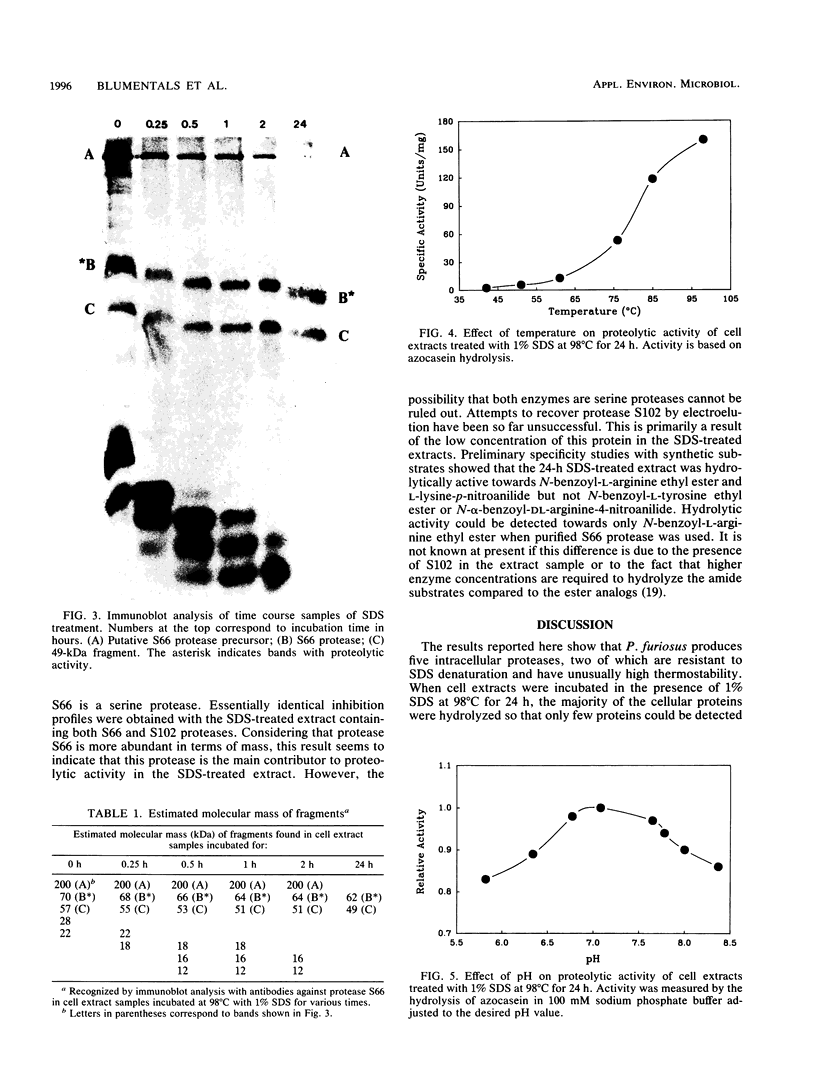
Cell extracts from Pyrococcus furiosus were found to contain five proteases, two of which (S66 and S102) are resistant to sodium dodecyl sulfate (SDS) denaturation. Cell extracts incubated at 98 degrees C in the presence of 1% SDS for 24 h exhibited substantial cellular proteolysis such that only four proteins could be visualized by amido black-Coomassie brilliant blue staining of SDS-polyacrylamide gels. The SDS-treated extract retained 19% of the initial proteolytic activity as represented by two proteases, S66 (66 kilodaltons [kDa]) and S102 (102 kDa). Immunoblot analysis with guinea pig sera containing antibodies against protease S66 indicated that S66 is related neither to S102 nor to the other proteases. The results of this analysis also suggest that S66 might be the hydrolysis product of a 200-kDa precursor which does not have proteolytic activity. The 24-h SDS-treated extract showed unusually thermostable proteolytic activity; the measured half-life at 98 degrees C was found to be 33 h. Proteases S66 and S102 were also resistant to denaturation by 8 M urea, 80 mM dithiothreitol, and 5% beta-mercaptoethanol. Purified protease S66 was inhibited by phenylmethylsulfonyl fluoride and diisopropyl fluorophosphate but not by EDTA, ethylene glycol-bis(beta-aminoethyl ether)-N,N,N',N'-tetraacetic acid, or iodoacetic acid. These results indicate that S66 is a serine protease. Amino acid ester hydrolysis studies showed that protease S66 was hydrolytically active towards N-benzoyl-L-arginine ethyl ester.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aono S., Bryant F. O., Adams M. W. A novel and remarkably thermostable ferredoxin from the hyperthermophilic archaebacterium Pyrococcus furiosus. J Bacteriol. 1989 Jun;171(6):3433–3439. doi: 10.1128/jb.171.6.3433-3439.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Brown S. H., Costantino H. R., Kelly R. M. Characterization of Amylolytic Enzyme Activities Associated with the Hyperthermophilic Archaebacterium Pyrococcus furiosus. Appl Environ Microbiol. 1990 Jul;56(7):1985–1991. doi: 10.1128/aem.56.7.1985-1991.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bryant F. O., Adams M. W. Characterization of hydrogenase from the hyperthermophilic archaebacterium, Pyrococcus furiosus. J Biol Chem. 1989 Mar 25;264(9):5070–5079. [PubMed] [Google Scholar]
- Cowan D. A., Daniel R. M. Purification and some properties of an extracellular protease (caldolysin) from an extreme thermophile. Biochim Biophys Acta. 1982 Aug 10;705(3):293–305. doi: 10.1016/0167-4838(82)90251-5. [DOI] [PubMed] [Google Scholar]
- Cowan D. A., Smolenski K. A., Daniel R. M., Morgan H. W. An extremely thermostable extracellular proteinase from a strain of the archaebacterium Desulfurococcus growing at 88 degrees C. Biochem J. 1987 Oct 1;247(1):121–133. doi: 10.1042/bj2470121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heussen C., Dowdle E. B. Electrophoretic analysis of plasminogen activators in polyacrylamide gels containing sodium dodecyl sulfate and copolymerized substrates. Anal Biochem. 1980 Feb;102(1):196–202. doi: 10.1016/0003-2697(80)90338-3. [DOI] [PubMed] [Google Scholar]
- Khoo T. C., Cowan D. A., Daniel R. M., Morgan H. W. Interactions of calcium and other metal ions with caldolysin, the thermostable proteinase from Thermus aquaticus strain T351. Biochem J. 1984 Jul 15;221(2):407–413. doi: 10.1042/bj2210407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwon S. T., Matsuzawa H., Ohta T. Determination of the positions of the disulfide bonds in aqualysin I (a thermophilic alkaline serine protease) of Thermus aquaticus YT-1. J Biochem. 1988 Oct;104(4):557–559. doi: 10.1093/oxfordjournals.jbchem.a122509. [DOI] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]






