Abstract
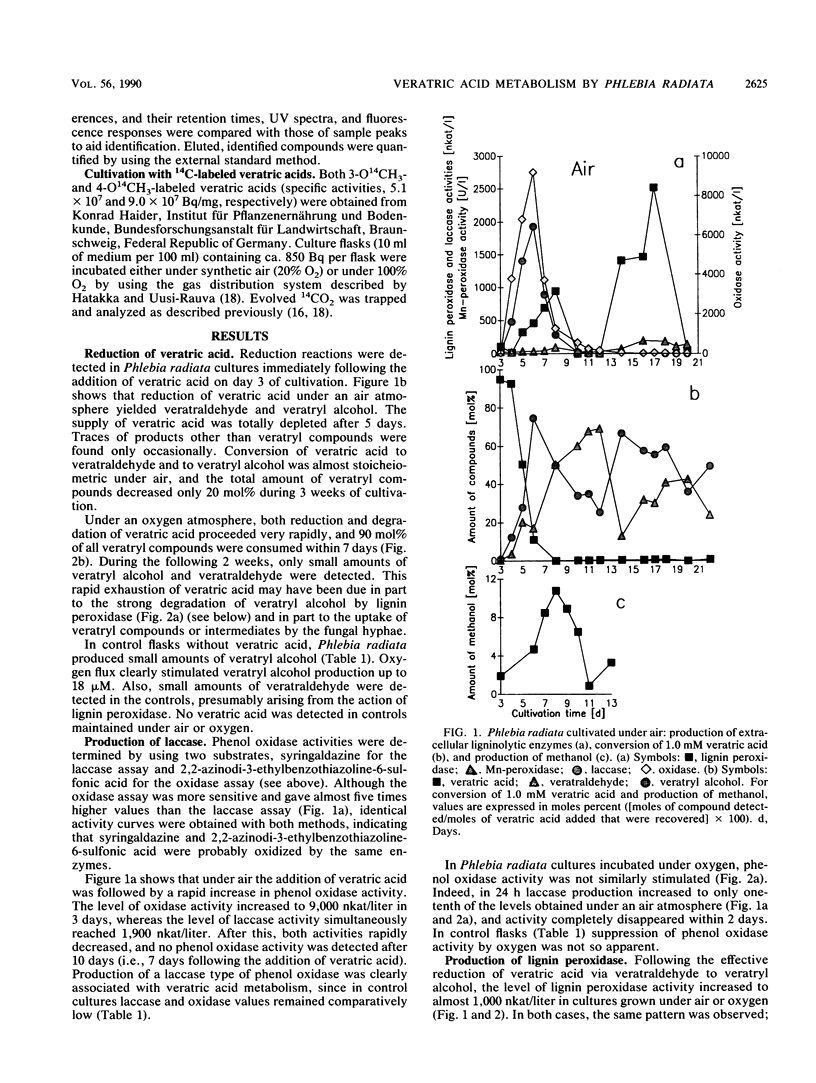
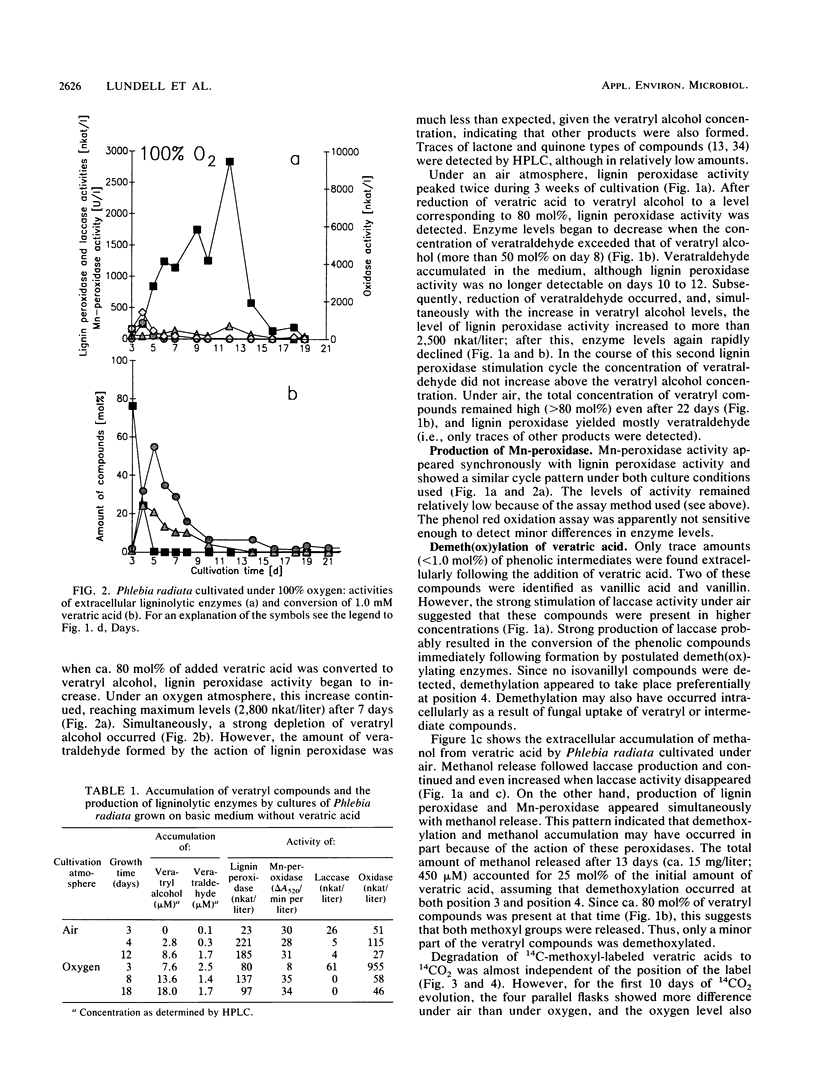
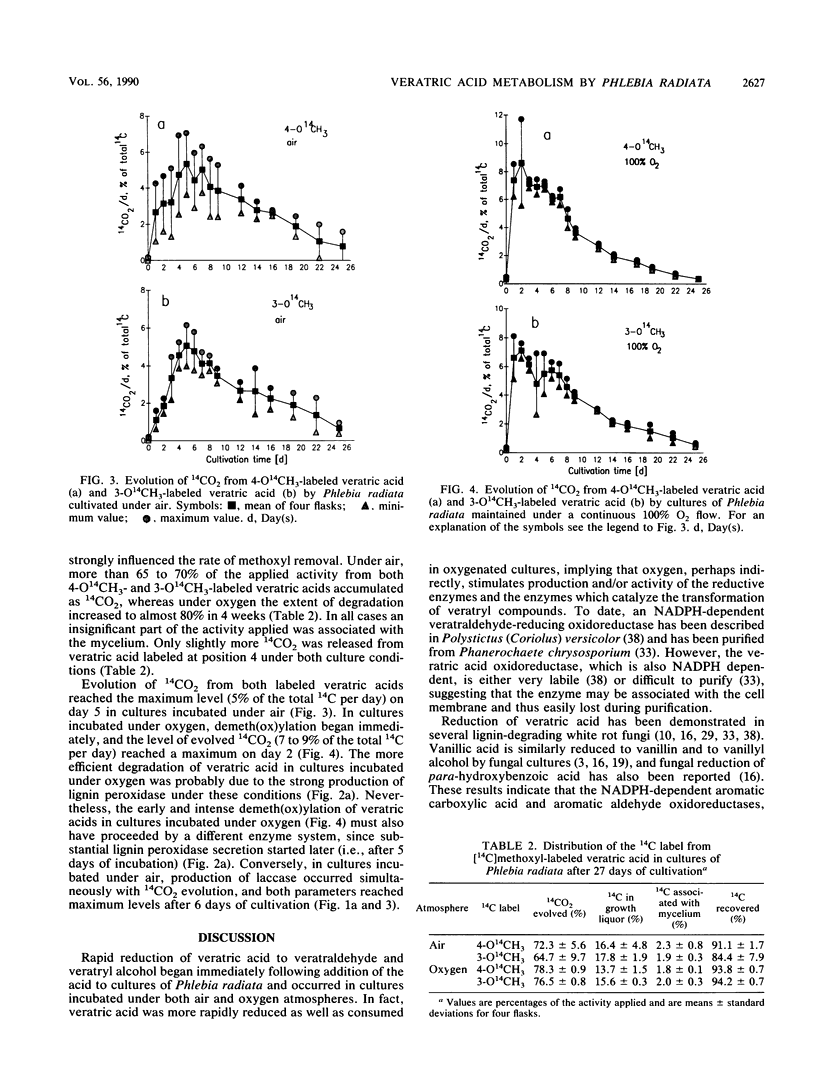
Transformation of veratric (3,4-dimethoxybenzoic) acid by the white rot fungus Phlebia radiata was studied to elucidate the role of ligninolytic, reductive, and demeth(ox)ylating enzymes. Under both air and a 100% O2 atmosphere, with nitrogen limitation and glucose as a carbon source, reducing activity resulted in the accumulation of veratryl alcohol in the medium. When the fungus was cultivated under air, veratric acid caused a rapid increase in laccase (benzenediol:oxygen oxidoreductase; EC 1.10.3.2) production, which indicated that veratric acid was first demethylated, thus providing phenolic compounds for laccase. After a rapid decline in laccase activity, elevated lignin peroxidase (ligninase) activity and manganese-dependent peroxidase production were detected simultaneously with extracellular release of methanol. This indicated apparent demethoxylation. When the fungus was cultivated under a continuous 100% O2 flow and in the presence of veratric acid, laccase production was markedly repressed, whereas production of lignin peroxidase and degradation of veratryl compounds were clearly enhanced. In all cultures, the increases in lignin peroxidase titers were directly related to veratryl alcohol accumulation. Evolution of 14CO2 from 3-O14CH3-and 4-O14CH3-labeled veratric acids showed that the position of the methoxyl substituent in the aromatic ring only slightly affected demeth(ox)ylation activity. In both cases, more than 60% of the total 14C was converted to 14CO2 under air in 4 weeks, and oxygen flux increased the degradation rate of the 14C-labeled veratric acids just as it did with unlabeled cultures.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Buswell J. A., Hamp S., Eriksson K. E. Intracellular quinone reduction in Sporotrichum pulverulentum by a NAD(P)H:quinone oxidoreductase: possible role in vanillic acid catabolism. FEBS Lett. 1979 Dec 1;108(1):229–232. doi: 10.1016/0014-5793(79)81216-8. [DOI] [PubMed] [Google Scholar]
- Faison B. D., Kirk T. K., Farrell R. L. Role of Veratryl Alcohol in Regulating Ligninase Activity in Phanerochaete chrysosporium. Appl Environ Microbiol. 1986 Aug;52(2):251–254. doi: 10.1128/aem.52.2.251-254.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glenn J. K., Gold M. H. Purification and characterization of an extracellular Mn(II)-dependent peroxidase from the lignin-degrading basidiomycete, Phanerochaete chrysosporium. Arch Biochem Biophys. 1985 Nov 1;242(2):329–341. doi: 10.1016/0003-9861(85)90217-6. [DOI] [PubMed] [Google Scholar]
- ISHIKAWA H., SCHUBERT W. J., NORD F. F. Investigations on lignins and lignification. 28. The degradation by Polyporus versicolor and Fomes fomentarius of aromatic compounds structurally related to softwood lignin. Arch Biochem Biophys. 1963 Jan;100:140–149. doi: 10.1016/0003-9861(63)90044-4. [DOI] [PubMed] [Google Scholar]
- Kirk T. K., Farrell R. L. Enzymatic "combustion": the microbial degradation of lignin. Annu Rev Microbiol. 1987;41:465–505. doi: 10.1146/annurev.mi.41.100187.002341. [DOI] [PubMed] [Google Scholar]
- Paszczyński A., Trojanowski J. An affinity-column procedure for the purification of veratrate O-demethylase from fungi. Microbios. 1977;18(72):111–121. [PubMed] [Google Scholar]
- Shimada M., Hattori T., Umezawa T., Higuchi T., Uzura K. Regiospecific oxygenations during ring cleavage of a secondary metabolite, 3,4-dimethoxybenzyl alcohol catalyzed by lignin peroxidase. FEBS Lett. 1987 Sep 14;221(2):327–331. doi: 10.1016/0014-5793(87)80950-x. [DOI] [PubMed] [Google Scholar]
- Tien M., Kirk T. K. Lignin-degrading enzyme from Phanerochaete chrysosporium: Purification, characterization, and catalytic properties of a unique H(2)O(2)-requiring oxygenase. Proc Natl Acad Sci U S A. 1984 Apr;81(8):2280–2284. doi: 10.1073/pnas.81.8.2280. [DOI] [PMC free article] [PubMed] [Google Scholar]


