Abstract
Fibronectin (FN), expressed primarily by macrophages, endothelial cells, and smooth muscle cells, represents an integral feature of the rejection response in transplant recipients. Here we demonstrate a unique pattern of cellular FN expression in rat recipients of cardiac allografts rendered tolerant in an infectious manner with either nondepleting CD4 mAb or regulatory spleen cells. Unlike in rejecting controls, cellular FN in tolerant hosts was restricted to the graft vessels and no vascular cell adhesion molecule-1 or intercellular adhesion molecule-1 expression could be found, supporting the role of FN in leukocyte sequestration at the graft site. The lack of myocardial FN in tolerant rats, despite dense macrophage infiltration, correlated with profound depression of Th1 (interleukin-2 and interferon-γ) cytokines. Treatment with CD4-depleting mAb prevented tolerance induction and restored myocardial expression of FN in parallel with marked increase in the expression of interleukin-2 and interferon-γ mRNA/protein. Furthermore, connective segment-1 peptide-facilitated adjunctive blockade of FN-α4β1 interactions in recipients conditioned with CD4 depleting mAb, significantly depressed intragraft expression of interleukin-2 and interferon-γ mRNA/protein. Hence, the lack of FN associated with infiltrating leukocytes plays an important role in the maintenance of tolerance in transplant recipients by depressing local expression of Th1 cytokines that otherwise facilitate acute graft rejection.
Lymphocytes recirculate between blood and lymph until the trigger of signals that stimulate their adhesion to vascular endothelium or extracellular matrix (ECM) proteins. 1,2 To emigrate through vascular endothelium, lymphocytes must acquire strong adhesion interactions to the vessel wall while resisting continuous sheer forces. 3 The emerging relevance of lymphocyte-ECM interactions to cell adhesion, migration, and positioning within specific tissue microenvironments emphasizes the role ECM proteins may play in the functioning of the immune system in both physiological and pathological conditions. 1 Fibronectin (FN) is a key ECM protein involved in these events. Indeed, the importance of FN in cell migration and differentiation is underscored by the observation that mice made null for this ECM protein die during embryonic development. 4,5 The role of FN in lymphocyte adhesion, migration, and activation has been extensively studied. 6-10 Circulating leukocytes, such as T cells, use β1 integrins for adhesion and migration through endothelial cells as well as through distinct tissue microenvironments. 11,12 Thus, adhesion of T cells to FN occurs primarily through α4β1 and α5β1 integrins, and it is regulated by the engagement of T cell receptor (TCR)/CD3, CD2, CD7, and CD28. 13-15 The ability of the C-C chemokines, such as RANTES, monocyte chemoattractant protein-1 (MCP-1), and macrophage inflammatory protein-1 (MIP-1) to induce integrin-dependent biding of T lymphocytes to FN has also been demonstrated. 16-18 The fact that TCR engagement up-regulates adhesion of T lymphocytes to FN highlights the importance of this protein in host immune reactivity.
Major histocompatibility complex (MHC)-incompatible organ allograft represents a useful in vivo experimental system to examine the role of FN in the immune response in which the effector phase is dependent on the migration of alloreactive cells into the foreign tissue. 19,20 In our previous studies, we have shown markedly increased deposition of FN in rat cardiac allografts in the early post-transplant period. 21,22 Interestingly, leukocytes infiltrating rejecting cardiac allografts were preferentially detected in close association with FN deposits. The prime source of cellular FN in rejecting cardiac allografts were macrophages in the myocardium and smooth muscle and endothelial cells in the vessels. 22,23 It has been demonstrated that IL-1β and tumor necrosis factor (TNF)-α up-regulate FN expression in vitro by both endothelial and smooth muscle cells. 24,25 Indeed, in recipients treated with anti-TNF-α antibody, we have shown that the diminution in the number of infiltrating cells was correlated with decreased FN accumulation. 23 More recently, we have documented that the blockade of in vivo interactions between the connective segment-1 (CS1) splicing domain of FN and its α4β1 receptors on circulating cells, by using CS1 peptides, abrogated acute rejection and prolonged cardiac allograft survival in rat recipients. 26 Furthermore, the use of CS1 peptides effectively prevented progressive allograft failure associated with chronic rejection. 27
The evidence for FN as an integral part of the host allograft rejection cascade has prompted us to test our hypothesis that acquisition and maintenance of tolerance, despite leukocyte infiltration observed in so-called tolerant grafts, depend on the inhibition of cellular FN expression. We have developed a model in which administration of RIB-5/2, a nondepleting anti-rat CD4 mAb, abrogates otherwise accelerated (<36 hours) rejection of cardiac allografts in presensitized rats and induces donor-specific transplantation tolerance. 28 RIB-5/2 is a novel mouse mAb that exerts its effects by modulating and blocking the CD4 antigen (Ag) on rat target cells. 29 RIB-5/2 mAb therapy has also been highly efficient in acute skin 29 and renal 30 allograft models in normal rats not otherwise sensitized. The present study is the first to document that, unlike in untreated accelerated or progressive late chronic cardiac allograft rejection, the maintenance of tolerance in CD4 mAb-conditioned recipients results from the failure of graft-infiltrating macrophages to express FN. The lack of FN expression in the myocardium of tolerant hosts correlated with down-regulation of intragraft interleukin (IL)-2 and interferon (IFN)-γ expression. Furthermore, this study demonstrates an important role of FN-α4β1 integrin interactions in the regulation of Th1-type cytokine expression at the graft site. Although FN expression was depressed in the myocardium of long-term and well-functioning tolerant heart transplants, cellular FN was up-regulated in the vessels in the absence of intercellular adhesion molecule-1 (ICAM-1) and vascular cell adhesion molecule-1 (VCAM-1), supporting the role of this ECM protein in the recruitment of mononuclear cells (MNC) into the graft itself.
Materials and Methods
Animals and Grafting Techniques
Male inbred adult rats (Harlan Sprague-Dawley, Indianapolis, IN) weighing 200 to 220 g were used. Lewis (LEW; RTl) and Fisher (F344; RT1lvl) were recipients of cardiac allografts from Lewis × Brown Norway F1 hybrids (LBNF1; RT1l/n) and LEW donors, respectively. Brown Norway (BN; RT1n) served as a source of sensitizing skin grafts. LEW isografts served as controls. Orthotopic skin grafts (3 × 3 cm) were placed bilaterally to appropriate defects on the dorsal thorax of prospective recipients. Donor hearts were transplanted to the abdominal great vessels of the recipients by using standard microvascular techniques, and their function was monitored by palpitation. Rejection was defined as occurring on the day of cessation of heartbeat and confirmed by histology.
Accelerated Rejection Model
LEW rats were sensitized with BN skin grafts (day −7), followed 1 week later by transplantation of LBNF1 hearts (day 0). These cardiac allografts are rejected in an accelerated manner in <36 hours, compared with 7- to 8-day acute rejection in untreated hosts. 31 In this rat model, the sequence of cellular and humoral immune events 31,32 is relevant to the clinical cases of graft rejection, in which patients are sensitized to MHC antigens before transplantation. 33
Tolerance Model
LEW rats were treated with RIB-5/2, a nondepleting anti-rat CD4 mAb (5 mg/kg/day i.v.), at days −7 (the day of skin graft), −4, −1, 0 (the day of LBNF1 heart transplant), and days 1, 4, 7, 14, and 21 thereafter. This protocol abrogates rejection at <36 hours and results in permanent (>200 days) acceptance of cardiac allografts in sensitized rat recipients. 28 RIB-5/2 mAb treatment induces a true donor-specific tolerance, as evidenced by the acceptance of secondary donor type (but not third party) test allografts, 28 depression of anti-donor antibody responses, 28 and lack of chronic rejection. 34 The long-term beneficial effects of RIB-5/2 mAb therapy contrast with marginal prolongation of cardiac allograft survival (about 11 days) in sensitized rat recipients treated with BWH-4, a CD4-depleting mAb. 32 RIB-5/2 mAb precipitates a 53-kd polypeptide expressed on rat thymocytes and splenocytes and defines an epitope on rat CD4+ T cells that is distinct from those recognized by W3/25 or MRC OX-35 mAbs. 29
Chronic Rejection Model
LEW hearts were transplanted to the abdominal great vessels of F344 rats. A subtherapeutic dose of cyclosporin A (1.5 mg/kg/day, i.m.) was administered for 10 days post-transplant. This therapy prevents early acute rejection and increases the incidence of long-term (>100 days) allograft survival without abrogating cardinal features of chronic rejection. 35-37
Cell Transfer Studies and Host Immunomodulation
Spleens were harvested from long-term (>100 days) RIB-5/2 mAb-pretreated tolerant LEW recipients of LBNF1 cardiac allografts. Single erythrocyte-free cell suspensions were prepared in RPMI-1640 medium, filtered through cotton wool, treated with 0.83% hypotonic ammonium chloride (pH 7.21), and extensively washed. In the first group, splenocyte suspensions (100 × 10 6 cells/ml) were administrated i.v. into lightly total body γ-irradiated (450 R) syngeneic secondary LEW rats. These test animals were challenged 24 hours later with a donor-specific (LBNF1) cardiac allograft. This adoptive transfer (AT) protocol induces indefinite (>200 days) survival of cardiac allografts with features of infectious tolerance in test recipients. 28 In a second group, adoptively transferred test recipients were challenged with a depleting anti-CD4 antibody (OX-36) at days 1 and 2 post-transplant (2 mg/rat i.v.). This protocol prevents the development of infectious tolerance and recreates rejection in adoptively transferred test animals. 28,38 In the third group, in an attempt to block in vivo interactions between FN and its α4β1 integrin receptor in rejecting cardiac allografts, adoptively transferred secondary hosts were given a course of synthetic CS1 peptides (1 mg/rat/day i.v. × 6 days) in concert with CD4-depleting OX36 mAb (2 mg/rat i.v. × 2 days). The CS1 peptides corresponding to the full length of CS1 alternative splicing domain of FN (25-mer, DELPQLVTLPHPNLHGPEILDVPST) were synthesized on a Beckman System 990 peptide synthesizer (Beckman Instruments, Inc., Fullerton, CA) as described. 39 These peptides specifically block the interactions between FN and α4β1 integrins on MNC. 40-42 Finally, a fourth control group received, instead of bioactive CS1 peptides, a course of the scrambled version of the FN peptides in which the last 10 residues were modified (VIPDLTESPG).
Histology and Immunohistology
Hematoxylin/eosin and immunoperoxidase stainings were used for assessment of myocardial and vascular preservation, as well as detection of inflammatory cell infiltration/activation, cellular FN, and adhesion molecules. 21-23 The presence of cytokine gene products was demonstrated by immunostaining using the method of Anderson et al. 43 Briefly, cardiac tissues were fixed in buffered formalin or embedded in Tissue Tec OCT compound (Miles, Elkhart, IN), snap-frozen in liquid nitrogen, and stored at −70°C. Serial cryostat sections (5 μm) were fixed in acetone or 2% paraformaldehyde for cell or cytokine detection, respectively. Endogenous peroxidase activity was blocked by incubating the slides with 1% H2O2 in phosphate-buffered saline. Normal heat-inactivated donkey or rabbit serum (10%) were used for blocking. Primary mouse antibodies against rat FN (IST-9, does not cross with plasma FN; Accurate Chemical, Westbury, NY), VCAM-1 (courtesy of Dr. R. Lobb, Biogen, Inc., Cambridge, MA), ICAM-1 (CD54, Harlan Bioproducts for Science, Indianapolis, IN), T cells (TCR-α/β chains, R73, Harlan), monocytes/macrophages (ED1, Harlan), and CD25 (IL-2 receptor (IL-2R); ART-18; courtesy of Dr. T. Diamantstein, Berlin, Germany) were added at optimal dilutions. Antibodies against rat IL-2 (R&D Systems, Minneapolis, MN) and IFN-γ (DB-1, Harlan) were added at optimal concentration (0.5–5.0 μg/ml) in phosphate-buffered saline/saponin and incubated overnight. Bound primary antibody was detected using biotinylated donkey anti-mouse IgG and streptavidin peroxidase-conjugated complexes obtained from DAKO (Carpinteria, CA) or using biotinylated rabbit anti-goat IgG followed by application of avidin-biotin complex (Vector Laboratories). The control sections were performed by replacing the primary mAb with either dilution buffer or isotype-matched control immunoglobulin. The peroxidase reaction was developed with 3,3-diaminobenzidine tetrahydrochloride (Sigma, St. Louis, MO). The sections were evaluated blindly by counting the labeled cells in triplicates by within 10 high-power fields per section. Some antigens were analyzed in a semiquantitative fashion where the relative abundance of each one was judged as negative (−), minimal (±), little (+), moderately abundant (++), or very abundant (+++).
Competitive Template Reverse Transcriptase-Polymerase Chain Reaction (CT RT-PCR)
For evaluation of cytokine gene expression, cardiac grafts were harvested and the cellular RNA was prepared by the guanidinium thiocyanate-cesium chloride method. CT RT-PCR from total RNA was performed as described. 44 Briefly, RNA was reverse-transcribed into cDNA in 40 μl of reaction mixture containing 200 ng of total RNA. Quantification of cytokine cDNA derived from this RNA was carried out using multispecific control fragment as an internal standard for competitive PCR. First, the cDNA samples to be compared were equilibrated according to their β-actin cDNA content. Then, the relative concentration of cytokine cDNA in each sample was estimated from the concentration of control fragment DNA that achieved equilibrium between its own amplification and that of the target cDNA. The level of cytokine gene expression was measured in arbitrary units (AU) per μl of cDNA. One AU was defined as the highest dilution of the control fragment that yielded a detectable amplification product under the conditions used. Control RT-PCR without template DNA was performed in all experiments to exclude contamination. As an additional control, reverse transcriptase was omitted from the reaction mixture during cDNA synthesis to prove the absence of genomic DNA. The sense and antisense primer sequences derived from rat CD3, CD25, IL-2, IFN-γ, IL-4, IL-10, and β-actin cDNA were used as described. 30
Results
Intramyocardial Leukocyte Infiltration in Tolerant Hosts
CD4-targeted therapy with nondepleting RIB-5/2 mAb (10× between day −7 and 21) abrogated accelerated rejection at <36 hours and induced indefinite (>200 days) survival of LBNF1 cardiac allografts in presensitized LEW rat recipients (n = 8), consistent with our previous findings. 38 We analyzed infiltrating MNC in cardiac transplants harvested during the induction (day 40) and maintenance (day 120) phase of CD4 mAb-induced tolerance by histology and immunohistology (Table 1) ▶ . Cardiac allografts undergoing accelerated rejection (ACCR) harvested at 24 hours from untreated sensitized recipients, as well as cardiac allografts undergoing chronic rejection harvested at 120 days, cardiac isografts, and native hearts served as controls. Cardiac specimens from RIB-5/2 mAb-treated recipients at 24 hours after transplantation morphologically resembled native hearts with minimal MNC infiltration. 45 First, we confirmed our previous findings 38 by demonstrating that unlike rejecting transplants, those in tolerant hosts (day 40 or 120) showed good preservation of histological detail, with no infarcts, myocardial necrosis, or signs of vascular disease. However, despite myocardial preservation, long-term cardiac allografts in tolerant hosts were characterized by persistent MNC infiltration. Hence, the number of intramyocardial infiltrating MNC cells at day 40 in the treated group (T cells, 39 ± 23; monocytes/macrophages, 108 ± 13) was comparable to that in untreated rejecting controls (ACCR 24 hours: T cells, 76 ± 23; monocytes/macrophages, 156 ± 60; CR 120 days: T cells, 80 ± 17; monocytes/macrophages, 170 ± 41). In long-term (120 days) cardiac allografts of CD4 mAb-treated recipients, both infiltrating T cells (22 ± 10) and monocytes/macrophages (47 ± 9) were somewhat decreased, as compared with rejecting controls (P < 0.04 and P < 0.06, respectively). However, despite such decreased cell numbers, tolerant grafts remained densely infiltrated as compared with respective long-term (120 day) isograft controls (T cells, 4 ± 4, monocytes/macrophages, 15 ± 7; P < 0.02 and P < 0.04, respectively). CD4-targeted therapy profoundly depressed MNC activation, as evidenced by decreased frequency of infiltrating IL2R+ cells in tolerant hosts at day 40 (3 ± 1) or day 120 (1 ± 1), and contrasted with untreated rejecting ACCR (49 ± 9) and CR (43 ± 8) controls (P < 0.009 and P < 0.002 respectively).
Table 1.
Immunohistochemical Analysis of Intramyocardial Infiltrating MNC in Rat Cardiac Allografts
| Feature | Naïve | ACCR 24 h | CR 120 d | RIB-Rx. 40 d | RIB-Rx. 120 d | ISO 120 d |
|---|---|---|---|---|---|---|
| T cells | 5 ± 1 | 76 ± 23 | 80 ± 17 | 39 ± 23 | 22 ± 10 | 4 ± 4 |
| Mono/Mac | 15 ± 2 | 156 ± 60 | 170 ± 41 | 108 ± 13 | 47 ± 9 | 15 ± 7 |
| IL-2R+ cells | — | 49 ± 9 | 43 ± 8 | 3 ± 1 | 1 ± 1 | — |
Immunoperoxidase staining analysis of cardiac grafts from sensitized recipients undergoing acute rejection (ACCR 24 h) and chronic rejection (CR 120 days), RIB-5/2 mAb-treated hosts (RIB-Rx; day 40 and day 120) and isografts (ISO; day 120). The results are expressed as mean ± SD of stained cells in 10 HPF/section/rat. n = 3–4 rats/group.
FN Expression and Associated Adhesion Molecules in Tolerant Hosts
Our earlier immunohistochemical and laser scanning microscopy studies have shown that infiltrating MNC localize selectively in FN-rich interstitial and perivascular areas of rejecting cardiac allografts. 21,22 Further, results of our in situ hybridization and immunocytochemistry studies have shown that endothelial cells, smooth muscle cells, and macrophages represent the prime FN producers during the rejection response. 22,23 In the present study, the expression of FN and associated adhesion molecules VCAM-1 and ICAM-1 in well-functioning cardiac allografts of tolerant hosts was contrasted with those in rejecting transplants. Unlike in rejecting (24 hours) controls, in which vascular expression of FN, VCAM-1 and ICAM-1 was high (++/+++; Table 2 ▶ and Figure 1, A and B ▶ ), cardiac allografts in tolerant hosts showed moderate vascular FN deposition (+/++; Table 2 ▶ and Figure 1, E and I ▶ ), whereas VCAM-1 and ICAM-1 were both negative (Table 2 ▶ and Figure 1F ▶ ). Cardiac isografts showed very weak deposition of FN in the vessels (±), comparable to naïve hearts, 22 and ICAM-1 and VCAM-1 expression were both negative (Table 2) ▶ . In contrast with the observed up-regulation of the vascular FN expression, graft-infiltrating macrophages in CD4 mAb-treated tolerant hosts failed to express FN (Figure 1, H and J) ▶ . As can be seen in Figure 1, C and G ▶ , cardiac allografts in both untreated rejecting and RIB-5/2-treated tolerant recipients were considerably infiltrated, primarily by monocytes/macrophages. However, in contrast with high expression of FN by macrophages in rejecting grafts (+++), macrophages infiltrating functioning cardiac allografts in tolerant rats failed to express FN (Figure 1, D, H, and J) ▶ .
Table 2.
Immunohistochemical Analysis of Vascular Expression of Cellular FN, VCAM-1, and ICAM-1 in Rat Cardiac Allografts
| Feature | ACCR 24 h | CR 120 d | RIB-Rx. 40 d | RIB-Rx. 120 d | ISO 120 d |
|---|---|---|---|---|---|
| FN | ++/+++ | +++ | ++ | +/++ | ± |
| VCAM-1 | ++/+++ | ++ | − | − | − |
| ICAM-1 | ++/+++ | +++ | − | − | − |
Immunoperoxidase staining analysis of cardiac grafts from sensitized recipients undergoing rejection (ACCR 24 h and CR 120 days), RIB-5/2 mAb-treated hosts (RIB-Rx; day 40 and 120) and isografts (ISO; day 120). Staining intensity was evaluated semiquantitatively on a scale from − (negative) to +++ (strong); n = 3–4 rats/group.
Figure 1.
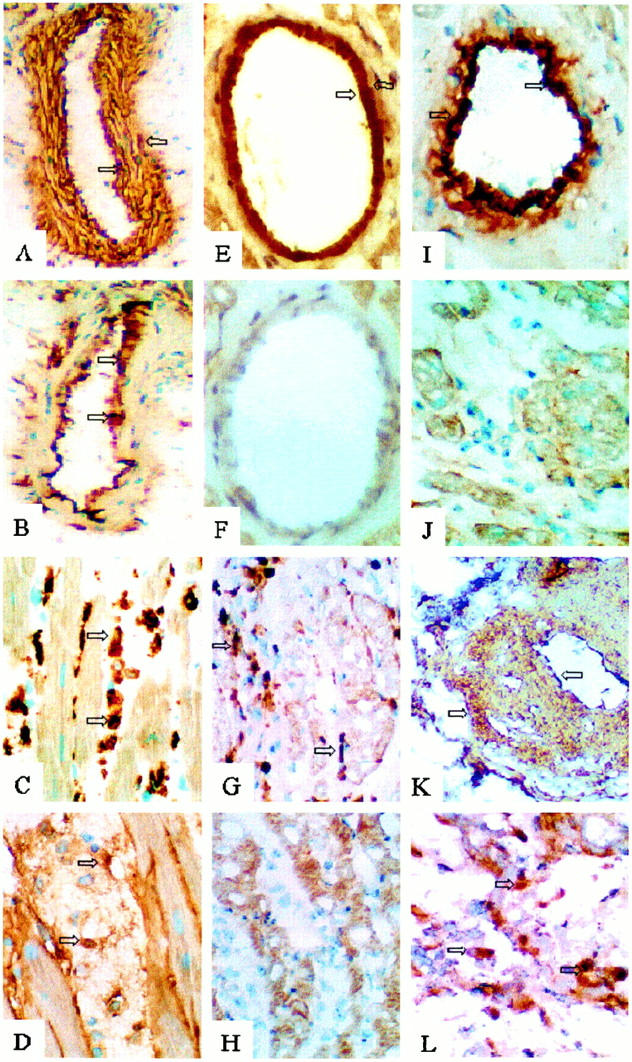
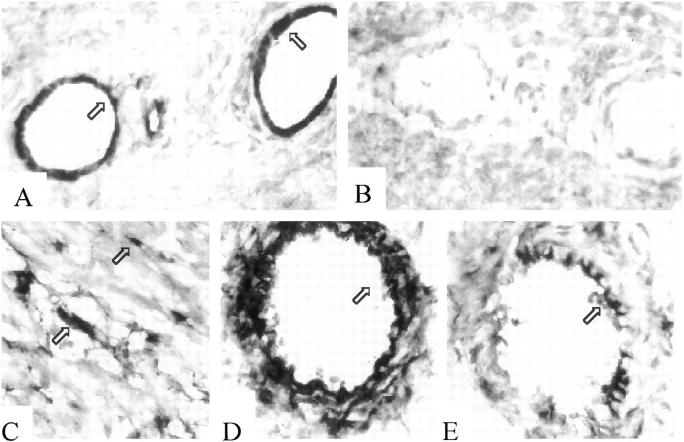
Immunohistochemical analysis of FN and VCAM-1 expression in rat cardiac allografts. LBNF1 cardiac allografts in LEW rats undergoing accelerated rejection (ACCR) at 24 hours (A−D) and surviving long-term at day 40 (E−H) and day 120 (I and J) in tolerant rats after treatment with nondepleting CD4 (RIB 5/2) mAb. LEW cardiac allografts in F344 rats served as long-term controls undergoing chronic rejection (CR) at day 120 post-transplant (K and L). Analysis of serial cryostat sections showed moderate to high deposition of cellular FN in the vessels of rejecting (A and K) and tolerant (E and I) cardiac allografts. Allografts of tolerant recipients lacked endothelial VCAM-1 expression (F), in contrast with elevated VCAM-1 expression during rejection (B). Both rejecting and CD4 mAb-treated groups were characterized by extensive MNC infiltration, consisting primarily of macrophages (C and G). In cardiac allografts undergoing ACCR (D) or CR (L), infiltrating MNC were labeled and/or were associated with cellular FN. In contrast, macrophages in cardiac allografts of tolerant hosts failed to express FN (H and J). Arrows indicate positive labeling. Original magnifications, ×100 (A, B, and L), ×230 (C, D, G, H, J, and L), and ×300 (E, F and I).
To demonstrate that FN expression is an ongoing myocardial feature of nontolerant long-term grafts, we evaluated the expression of this ECM protein in control recipients undergoing chronic rejection. We used an established model in which LEW cardiac allografts undergo progressive chronic rejection in F344 rat recipients pretreated with a low, subtherapeutic dose of cyclosporin A. Indeed, unlike long-term cardiac allografts in CD4 mAb-pretreated tolerant recipients, those undergoing chronic rejection and harvested at 120 days showed markedly enhanced expression of cellular FN in both vascular and myocardial compartments (+++; Table 2 ▶ and Figure 1, K and L ▶ ). Up-regulation of FN deposition in the chronic rejection group correlated with enhanced endothelial VCAM-1 and ICAM-1 expression.
FN Expression by Macrophages Is Required for Allograft Rejection
To document the functional significance of the lack of FN expression by macrophages in cardiac allografts of tolerant recipients, we evaluated FN expression after recreation of rejection in this transplant model. Since we were unable to abolish tolerance in original RIB-5/2 mAb-treated hosts, 28 we have used a model of infectious adoptively transferred tolerance. We have recently documented that spleen cells from long-term RIB-5/2 mAb-pretreated engrafted hosts adoptively transfer donor-specific unresponsiveness in an infectious manner into new sets of secondary test recipients, and that such a tolerant state can be broken by challenging adoptively transferred recipients with OX-36, a depleting CD4 antibody. 28,38 In fact, animals treated with OX-36 mAb (2 mg/rat i.v. on days 1 and 2 post-transplant) reject their transplants within 7 days. First, we analyzed the pattern of MNC infiltration, FN deposition, and VCAM-1/ICAM-1 expression in long-term (day 80) LBNF1 test cardiac allografts that have been rendered tolerant in an infectious manner by regulatory cells from original CD4 mAb-pretreated recipients. Indeed, both tolerant CD4 mAb-pretreated hosts and adoptively transferred hosts showed comparable results, with moderate to high intramyocardial MNC infiltration and minimal cellular activation (AT: T cells, 25 ± 2; monocytes/macrophages, 68 ± 4; IL-2R+ cells, 1 ± 1) (Table 3) ▶ , moderate vascular expression of FN (AT; ++) and negative expression of VCAM-1 (AT; −) (Figure 2, A and B) ▶ and ICAM-1 (AT; −) (not shown). The tolerant state in AT hosts was also found to be associated with the failure of graft-infiltrating macrophages to express FN. Abrogation of tolerance and recreation of graft rejection following infusion of CD4-depleting (OX-36) mAb, as shown in Table 3 ▶ , was accompanied by a significant increase in intramyocardial MNC infiltration and cellular activation (AT+OX-36; T cells: 44 ± 2; monocytes/macrophages: 138 ± 57; IL-2R+ cells 44 ± 10). Unlike in untreated AT recipients, interstitial macrophages in those receiving OX-36 mAb readily expressed FN (+/++; Figure 2C ▶ ). Moreover, recreation of rejection was associated with a further up-regulation of FN deposition (++/+++) and VCAM-1 (++/+++) and ICAM-1 (++/+++) expression in the vessels (Figure 2, D and E) ▶ .
Table 3.
Immunohistochemical Analysis of Intramyocardial Infiltrating MNC in Cardiac Allografts of Adoptively Transferred Secondary Tolerant versus Rejecting Hosts
| Feature | AT | AT + OX36 |
|---|---|---|
| T cells | 25 ± 2 | 44 ± 2* |
| Mono/Mac | 68 ± 4 | 138 ± 57* |
| IL-2R+ cells | 1 ± 1 | 44 ± 10** |
Cardiac allografts were harvested from test rat recipients following transfer of spleen cells from RIB-5/2 mAb-treated tolerant hosts (AT) or from rat recipients undergoing cardiac allograft rejection after adjunctive infusion of CD4 depleting mAb (AT + OX36). The results are expressed as mean ± SD of stained cells in 10 HPF/section/rat. n = 2–3 rats/group.
*P < 0.05
**P < 0.001
Figure 2.
Immunohistochemical analysis of FN and VCAM-1 expression after abrogation of the infectious tolerance pathway. Representative immunohistochemical analysis of LBNF1 cardiac allografts in LEW recipients after adoptive transfer of spleen cells from tolerant RIB-5/2 mAb-pretreated hosts (A and B), and after adjunctive treatment with a depleting CD4 (OX36) mAb that abrogates the ability of spleen cells to confer tolerance (C−E). Analysis of cryostat serial sections showed moderate and negative staining for FN in the vessels and myocardium, respectively (A). VCAM-1 expression was negative in cardiac allografts of tolerant recipients (B). Abrogation of tolerance was accompanied by myocardial expression of cellular FN (C) and up-regulation of FN (D) and VCAM-1 (E) expression in the vessels. Arrows indicate positive labeling. Original magnifications, ×200 (A−C), and ×160 (D and E).
Increased Deposition of FN Is Accompanied by Up-Regulation of Th1-Type Cytokines at the Graft Site
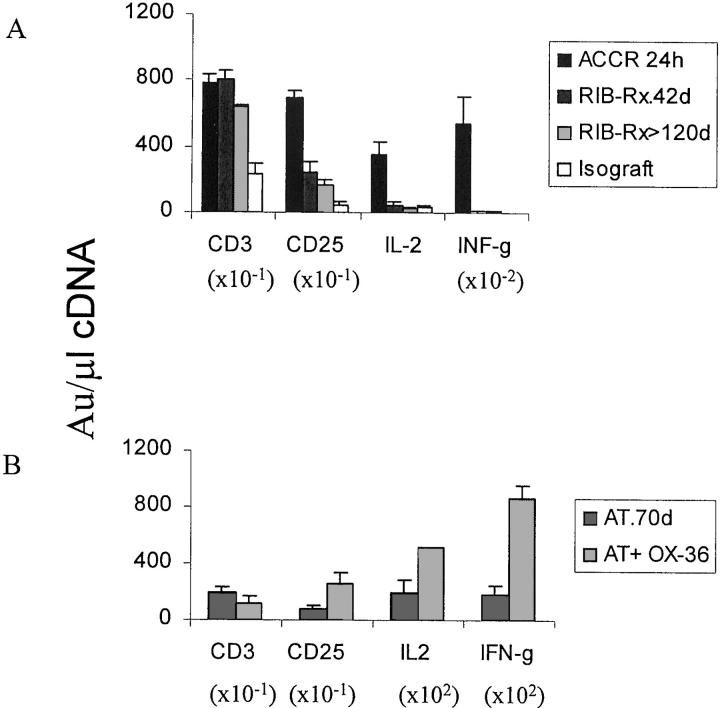
To address a possible link between intragraft FN expression and cytokine release, we have used CT RT-PCR and immunohistology techniques to analyze the expression of cytokines at mRNA and protein levels in different cardiac transplant groups. We have previously shown that intragraft IL-2 and IFN-γ levels are very low in RIB-5/2 mAb-treated hosts at 24 hours and 7 days after transplantation. 38,45 In our present study, the levels of mRNA coding for both IL-2 and IFN-γ were profoundly depressed in long-term tolerant recipients (P < 0.005) as compared with grafts rejected in an accelerated manner (Figure 3A) ▶ or with those undergoing chronic rejection (not shown). The immunohistochemical analysis confirmed the translation of mRNAs into relevant proteins (Figure 5, A, B, E, and F) ▶ . This marked decrease in Th1-type cytokine expression in tolerant hosts was accompanied by a lack of FN expression by infiltrating macrophages. In addition, in agreement with our immunohistological findings, CD3 mRNA levels were elevated in rejecting controls as well as in well-functioning CD4 mAb treated hosts at both 40 and 120 days post-transplant (Figure 3A) ▶ . The 24-hour cardiac allografts in RIB-5/2 mAb-treated rats show only modest level of CD3 mRNA expression, 45 consistent with our present histological observations.
Figure 3.
CT RT-PCR-assisted analysis of intragraft mRNA coding for CD3, CD25, IL-2 and IFN-γ. A shows mRNA levels in sensitized ACCR recipients, in RIB-5/2 mAb-treated hosts (RIB-Rx; at day 40 and 120), and in control cardiac isografts (day 120). B shows mRNA levels in recipients after transfer of infectious tolerance by spleen cells from RIB 5/2 mAb-treated hosts (AT) and in recipients undergoing rejection following adjunctive infusion of CD4-depleting mAb (AT + OX36). OX36 mAb-induced abrogation of the tolerant state prompted 2.5-fold and fivefold increase in the level of IL-2 and IFN-γ mRNA expression, respectively. Each value represents the mean ± SD of 2 to 3 analyses of 2 to 3 tissue samples/group.
Figure 5.
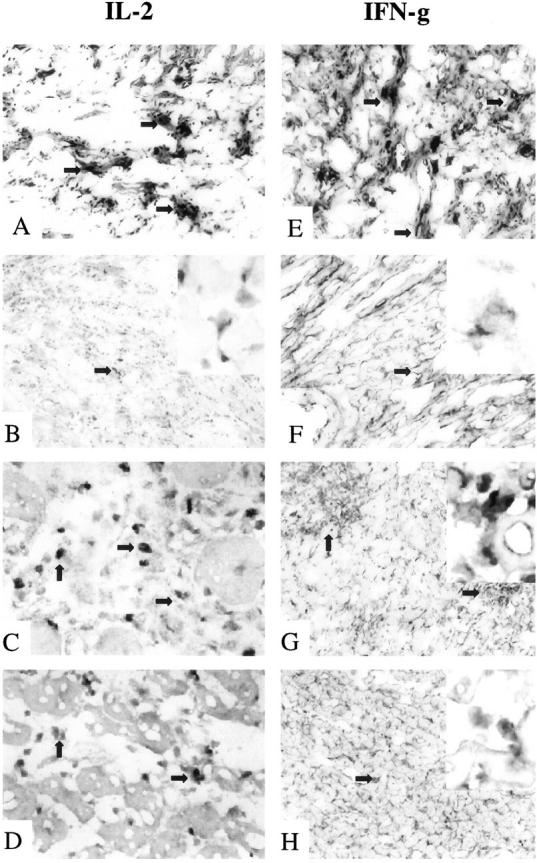
Immunohistochemical analysis of IL-2 and IFN-γ expression in rat cardiac allografts. Examination of IL-2 (A−D) and IFN-γ (E−H) shows that the tolerant state in RIB-5/2 mAb-treated hosts at 120 days was accompanied by basal staining for Th1 cytokines (B and F). In contrast, recipients undergoing chronic rejection showed dense expression of both IL-2 and IFN-γ (A and E). Adoptively transferred recipients undergoing rejection of test cardiac allografts after infusion of CD4-depleting mAb (OX36) showed moderate levels of Th1 cytokines (C and G). This contrasted with only mild IL-2 and IFN-γ deposition (D and H) after adjunctive course of CS1 peptides (OX36 + CS1). Original magnifications, ×100 (A, B, E−H) and ×270 (C and D) (n = 2). Arrows indicate positive labeling, and enlargements of those areas are included in the upper right insets of B, F, G, and H (original magnifications, ×420).
Then, to address a possible functional correlation between macrophage-derived FN and cytokine release at the graft site, we analyzed IL-2 and IFN-γ mRNA levels after recreation of graft rejection in adoptively transferred test recipients (Figure 3B) ▶ . Rats infused with spleen cells from tolerant hosts show basal intragraft Th-1 type cytokine levels at day 7. 38 However, OX-36 mAb-induced abrogation of the tolerant state prompted a 2.5-fold and fivefold increase in the level of mRNA coding for IL-2 and IFN-γ, respectively, in parallel with enhanced FN expression. Interestingly, we did not find a correlation between FN expression and Th2-type cytokine program. We confirmed our previous findings, 38 by demonstrating that intragraft expression of IL-4 mRNA was virtually abolished, and that of IL-10 mRNA (P < 0.0005) was depressed after RIB-5/2 mAb therapy as compared with the respective rejecting controls. However, in marked contrast with the latter group, the levels of mRNA coding for IL-4 and IL-10 were greatly increased (P < 0.0001) in recipients that were adoptively made tolerant by spleen cells, as compared with the respective rejecting controls. All together, these observations led us to postulate that FN expressed by macrophages might have a regulatory role in Th-1 type IL-2 and IFN-γ expression at the graft site.
Blockade of FN-α4β1 Integrin Interactions Depresses Intragraft Th1-Type Cytokine Program
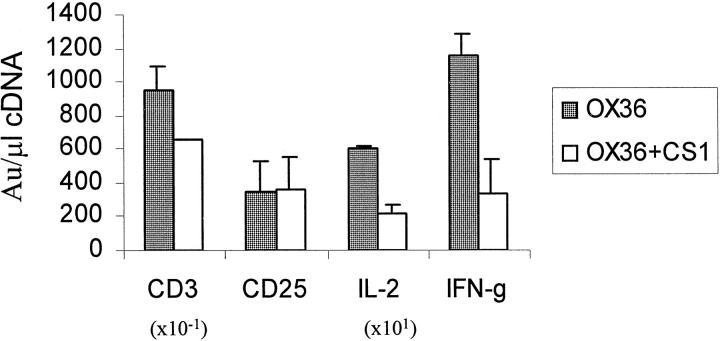
Finally, we examined the role of interactions between FN and its α4β1 integrin receptor on cellular events and Th1-type cytokine programs at the graft site. In an attempt to block FN-α4β1 integrin interactions in rejecting cardiac allografts, adoptively transferred recipients of test cardiac allografts were conditioned with a course of synthetic CS1 peptides (1 mg/rat/day i.v. × 6 days) in concert with CD4-depleting mAb (OX36; 2 mg/rat i.v. × 2 days). Control recipients received adjunctive scrambled peptides instead of bioactive CS1 peptides. The CS1−α4β1 integrin blockage decreased the number of intragraft T cells by 1/3 (76 ± 15 vs. 120 ± 27, P < 0.04), without significantly affecting infiltrating macrophages (>200 in both groups), and IL-2R+ cells (45 ± 4 vs. 73 ± 22) (not shown). We then used CT RT-PCR to test for the expression of mRNA coding for T cells, IL-2R+ cells, IL-2, and IFN-γ (Figure 4) ▶ . Adjunctive infusion of CS1 peptides reduced the level of transcripts for CD3 (P < 0.05) without affecting IL-2R mRNA expression, confirming our immunohistochemical findings. However, the blockade of FN-α4β1 integrin interactions markedly depressed intragraft expression of Th1-type cytokines. As shown in Figure 4 ▶ , IL-2 and IFN-γ mRNA levels in CS1 peptide-treated recipients decreased by factors of 2.8 (P < 0.05) and 3.4 (P < 0.02), respectively. The immunohistology analysis confirmed the translation of intragraft mRNAs into relevant proteins (Figure 5, C, D, G, and H) ▶ .
Figure 4.
CT RT-PCR-assisted analysis of intragraft mRNA coding for CD3, CD25, IL-2, and IFN-γ (day 6 post-transplant). Adoptively transferred secondary recipients undergoing rejection of cardiac allografts after infusion of CD4-depleting mAb (OX36) are contrasted with those which received adjunctive course of synthetic CS1 peptides (OX36 + CS1). The levels of mRNA coding for IL-2 and IFN-γ after infusion of CS1 decreased by a factor of 2.8 and 3.4, respectively. Each value represents the mean ± SD of 2 to 3 analyses of 2 to 3 tissue samples/group.
Discussion
We report here the results of our comprehensive studies on the role of FN during the acquisition of tolerance in a well-defined rat cardiac allograft model, and demonstrate a unique pattern of FN expression that may be pivotal to long-term transplant acceptance. The principal findings of this work are as follows. First, myocardial macrophages failed to express FN despite dense graft infiltration in tolerant hosts. Second, moderate deposits of FN were readily detectable in the allograft vessels in the absence of VCAM-1 and ICAM-1 expression. Third, failure of FN expression by infiltrating macrophages was associated with depressed levels of intragraft IL-2 and IFN-γ. Fourth, recreation of acute rejection after depletion of putative CD4 regulatory T cells that maintain infectious tolerance in this model, was correlated with up-regulated expression of FN by macrophages and Th1-type cytokines by graft infiltrating MNC. Finally, blockade of FN-α4β1 integrin interactions after adjunctive treatment with synthetic CS1 peptides significantly reduced intragraft IL-2 and IFN-γ mRNA/protein expression in animals given CD4-depleting mAb.
The role of interactions between β1 integrins and ECM proteins in regulating lymphocyte adhesion, migration, and activation is well established. 13,46 Although most of the attention has been focused on integrins activity rather than their ligands, the variations in ECM composition may also play an important regulatory role. Indeed, there is good evidence that ECM protein binding can modulate integrin expression. 47,48 We have previously reported that up-regulation of FN expression is a very early feature triggered by transplantation of MHC-incompatible organ, and that infiltrating MNC preferentially accumulate in interstitial and perivascular areas of rejecting cardiac transplants. 21 Moreover, there is a molecular heterogeneity in the FN found in cardiac allografts, where incorporation of the splicing domains EIIIA, EIIIB, and V have distinct temporal and spatial distribution patterns during rejection. 22 The V domain that includes the CS1 region, a sequence recognized by α4β1 integrin, is known to mediate lymphocyte adhesion. 11,12 We also demonstrated that FN is expressed primarily by macrophages in the myocardium and by endothelial and smooth muscle cells in the arteries of cardiac allografts. 22,23 Indeed, enhanced expression of FN has been reported in a number of pathological states, including cutaneous wounds, 49,50 vascular intimal proliferation, myocardial infarctions, hypertensive arteries, 51,52 glomerulonephritis, 53 and liver fibrosis. 54 The present study corroborates and extends our earlier findings by documenting that local synthesis of cellular FN occurs in the myocardium and in the vessels of cardiac allografts undergoing accelerated or chronic rejection. The observation that macrophages failed to express FN despite high number of these cells in long-term grafts of tolerant hosts represents direct evidence for an active role of FN in the rejection cascade. The demonstration of increased FN expression and the absence of ICAM-1 and VCAM-1 in the vessels during the acquisition of transplantation tolerance supports the role this ECM protein may play in the recruitment of MNC at the graft site. 21,23,55,56 Interestingly, the pattern of FN expression in tolerant grafts was comparable to that in cardiac isografts. In the latter model, there is an initial up-regulation of FN expression in the vessels in the absence of VCAM-1, which correlates with intragraft appearance of scattered MNC, and negative staining for cellular FN in the myocardium. 22 These observations suggest a role for VCAM-1-independent collateral adhesion system(s) to mediate leukocyte sequestration at the graft site. 57 The ischemia/reperfusion injury is mostly resolved by 24 hours in cardiac isografts transplanted into skin-sensitized rat recipients. 21 Taken together, there is evidence that mechanisms regulating FN expression are likely to vary between individual cell types, and FN expressed by different sources may have different functional roles. Indeed, our present observation that MNC accumulate in the myocardium in the absence of cellular FN in tolerant hosts indicates a role for this ECM protein that is unrelated to cell migration or tissue positioning. The demonstration that i) treatment with a nondepleting CD4 mAb induced tolerance in our transplant model; ii) tolerant state could be then transferred to new cohorts of test recipients in an infectious manner by CD4+ regulatory T cells, and iii) depletion of these regulatory T cells abrogated infectious tolerance pathway, points to alloreactive regulatory CD4 cells as potential regulators of FN expression. Furthermore, our observation that enhanced FN expression up-regulates production of IL-2 and IFN-γ indicates that FN by itself may play an active role in cellular activation. Our results are consistent with the idea that FN exerts synergistic effects on T cell activation by acting as a co-stimulator for both CD4+ and CD8+ T cells through TCR 9,58 and cytokine release. For example, the density of immobilized CD3 or TCR mAb required to induce degranulation and tyrosine phosphorylation of cellular proteins by CD8+ T cells is about 10-fold lower in the presence of FN. 9 Several studies have also shown that adhesion to FN activates tyrosine phosphorylation of several T cell proteins. 9,59,60 The production of IL-2, IFN-γ, and tumor necrosis factor-α are also stimulated in vitro by interactions between CD4+ cells and FN. 61-64 Indeed, it has been reported that binding of CD4+ cells to ECM proteins, most likely through conformational changes resulting in better presentation to their receptor, may in turn enhance cytokine activity. 65-67
To address the role of FN-α4β1 integrin interactions in our transplant model, we administered CS1 peptides into recipients in which tolerance had been abrogated and cardiac allograft rejection recreated after treatment with a CD4-depleting mAb. Adjunctive infusion of bioactive CS1 peptides not only reduced the number of infiltrating MNC, but also diminished intragraft IL-2 and IFN-γ expression, consistent with the regulatory role of FN in Th1-type cytokine network. 26 Our results corroborate previous reports showing that CS1 peptides partially inhibited T cell migration to skin inflammatory areas in a murine contact hypersensivity model, 68 and interrupted multiorgan cellular infiltration in transforming growth factor-β1-deficient mice. 69 Moreover, CS1 splicing variant of FN has also been implicated in the pathology of rheumatoid arthritis. 70 As CS1 motif was selectively expressed in the endothelium of rheumatoid arthritis synovium, treatment with CS1 peptides abrogated the binding of T lymphoblastoid cells to RA synovial sections. 71 Moreover, CS1 peptides inhibited leukocyte recruitment and resulted in partial attenuation of acute inflammatory response, with almost complete blockade of chronic inflammation in an erosive rat model of polyarthritis. 42 Treatment with CS1 peptides prevented the development of accelerated coronary arteriopathy and reduced T cell infiltration in heart transplants of cholesterol-fed rabbits. 55 Finally, we have recently showed that treatment of rats with CS1 peptides not only abrogated acute rejection, 26 but also prevented the development of chronic rejection 27,72 in rat cardiac allograft recipients.
In addition to the CS1 binding domain, FN has other active sites, such as KLDAPT, a recently described ligand for activated α4 integrins, 73 or the RGD domain, the ligand for α5β1. 74 In α5-null cells plated on FN, αV integrins can compensate functionally the loss of α5β1 in focal contacts. 75 It is also becoming appreciated that integrins are promiscuous, ie, an individual integrin can bind different ligands. For example, adhesion of B lymphocytes to FN is mediated exclusively by the interaction between α4β1 and the CS1 domain. 76 However, antigen activation can induce α4β1 integrin expression on B cells to recognize the RGD domain. 77 These data show the flexibility of α4β1 integrin in recognizing other domains in the FN molecule, and support the important role this ECM protein may play in an array of lymphocyte biological functions.
It remains to be determined how different cell types regulate FN expression in vivo. Although transforming growth factor-β1 is well known to increase FN expression by fibroblasts, its role in modulating FN expression in other cell types remains unclear. 78,79 Our present results suggest a participation of the CD4 cell in the regulation of FN expression by macrophages; however, the signals involved in the communication T lymphocytes and macrophages toward FN expression remain unknown. Future studies on regulation of FN expression by different cell types, in particular macrophages, should provide much needed functional criteria for the design of novel therapeutic strategies.
Acknowledgments
We thank Drs. Liz Messersmith and M. Lehmann for providing us with fibronectin CS1 peptides and RIB-5/2 mAb, respectively. We also thank Dr. Terry Strom for his invaluable comments on the manuscript and acknowledge the technical assistance of Mr. L. Barwari.
Footnotes
Address reprint requests to Dr. Ana J. Coito, The Dumont-UCLA Transplant Center Room 77–120 CHS, 10833 Le Conte Avenue, Los Angeles, CA 90095. E-mail: acoito@mednet.ucla.edu.
Supported by National Institutes of Health grants RO1 AI23847 and RO1 AI42223 (to J.W.K.-W.) and American Heart Association grant 003006N (to A.J.C.) and by The Dumont Research Foundation. A. J. C. is a recipient of the Faculty Development Award, American Society of Transplantation (1999).
References
- 1.de Sousa M, Tilney NL, Kupiec-Weglinski JW: Recognition of self within self: specific lymphocyte positioning and the extracellular matrix. Immunol Today 1991, 12:262-266 [DOI] [PubMed] [Google Scholar]
- 2.Springer TA: Traffic signals for lymphocyte recirculation and leukocyte emigration: the multistep paradigm. Cell 1994, 76:301-314 [DOI] [PubMed] [Google Scholar]
- 3.Diamond MS, Springer TA: The dynamic regulation of integrin adhesiveness. Curr Biol 1994, 4:506-517 [DOI] [PubMed] [Google Scholar]
- 4.George EL, Georges-Labouesse EN, Patel-King RS, Rayburn H, Hynes RO: Defects in mesoderm, neural tube and vascular development in mouse embryos lacking fibronectin. Development 1993, 119:1079-1091 [DOI] [PubMed] [Google Scholar]
- 5.Georges-Labouesse EN, George EL, Rayburn H, Hynes RO: Mesodermal development in mouse embryos mutant for fibronectin. Dev Dyn 1996, 207:145-156 [DOI] [PubMed] [Google Scholar]
- 6.Shimizu Y, van Seventer GA, Horgan KJ, Shaw S: Costimulation of proliferative responses of resting CD4+ T cells by the interaction of VLA-4 and VLA-5 with fibronectin or VLA-6 with laminin. J Immunol 1990, 145:59-67 [PubMed] [Google Scholar]
- 7.Hauzenberger D, Klominek J, Sundqvist KG: Functional specialization of fibronectin-binding beta 1-integrins in T lymphocyte migration. J Immunol 1994, 153:960-971 [PubMed] [Google Scholar]
- 8.Ybarrondo B, O’Rourke AM, Brian AA, Mescher MF: Contribution of lymphocyte function-associated-1/intercellular adhesion molecule-1 binding to the adhesion/signaling cascade of cytotoxic T lymphocyte activation. J Exp Med 1994, 179:359-363 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ostergaard HL, Ma EA: Fibronectin induces phosphorylation of a 120-kDa protein and synergizes with the T cell receptor to activate cytotoxic T cell clones. Eur J Immunol 1995, 25:252-256 [DOI] [PubMed] [Google Scholar]
- 10.Hunter AJ, Shimizu Y: Alpha 4 beta 1 integrin-mediated tyrosine phosphorylation in human T cells: characterization of Crk- and Fyn-associated substrates (pp105, pp115, and human enhancer of filamentation-1) and integrin-dependent activation of p59fyn1. J Immunol 1997, 159:4806-4814 [PubMed] [Google Scholar]
- 11.Wayner EA, Garcia-Pardo A, Humphries MJ, McDonald JA, Carter WG: Identification and characterization of the T lymphocyte adhesion receptor for an alternative cell attachment domain (CS-1) in plasma fibronectin. J Cell Biol 1989, 109:1321-1330 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Guan JL, Hynes RO: Lymphoid cells recognize an alternatively spliced segment of fibronectin via the integrin receptor alpha 4 beta 1. Cell 1990, 60:53-61 [DOI] [PubMed] [Google Scholar]
- 13.Shimizu Y, van Seventer GA, Ennis E, Newman W, Horgan KJ, Shaw S: Crosslinking of the T cell-specific accessory molecules CD7 and CD28 modulates T cell adhesion. J Exp Med 1992, 175:577-582 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Shimizu Y, Mobley JL, Finkelstein LD, Chan AS: A role for phosphatidylinositol 3-kinase in the regulation of beta 1 integrin activity by the CD2 antigen. J Cell Biol 1995, 131:1867-1880 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chan AS, Mobley JL, Fields GB, Shimizu Y: CD7-mediated regulation of integrin adhesiveness on human T cells involves tyrosine phosphorylation-dependent activation of phosphatidylinositol 3-kinase. J Immunol 1997, 159:934-942 [PubMed] [Google Scholar]
- 16.Weber C, Alon R, Moser B, Springer TA: Sequential regulation of alpha 4 beta 1 and alpha 5 beta 1 integrin avidity by CC chemokines in monocytes: implications for transendothelial chemotaxis. J Cell Biol 1996, 134:1063-1073 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Carr MW, Alon R, Springer TA: The C-C chemokine MCP-1 differentially modulates the avidity of beta 1 and beta 2 integrins on T lymphocytes. Immunity 1996, 4:179-187 [DOI] [PubMed] [Google Scholar]
- 18.Carr MW, Roth SJ, Luther E, Rose SS, Springer TA: Monocyte chemoattractant protein 1 acts as a T-lymphocyte chemoattractant. Proc Natl Acad Sci USA 1994, 91:3652-3656 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kupiec-Weglinski JW, Heemann UW, Coito AJ, Tullius SG, Tilney NL, de Sousa M: Adhesion molecule interaction with extracellular matrix. Exp Nephrol 1993, 1:78-82 [PubMed] [Google Scholar]
- 20.Coito AJ, de Sousa M, Kupiec-Weglinski JW: The role of cellular and extracellular matrix adhesion proteins in organ transplantation. Cell Adhes Commun 1994, 2:249-255 [DOI] [PubMed] [Google Scholar]
- 21.Coito AJ, Binder J, de Sousa M, Kupiec-Weglinski JW: The expression of extracellular matrix proteins during accelerated rejection of cardiac allografts in sensitized rats. Transplantation 1994, 57:599-605 [PubMed] [Google Scholar]
- 22.Coito AJ, Brown LF, Peters JH, Kupiec-Weglinski JW, Van de Water L: Expression of fibronectin splicing variants in organ transplantation: a differential pattern between rat cardiac allografts and isografts. Am J Pathol 1997, 150:1757-1772 [PMC free article] [PubMed] [Google Scholar]
- 23.Coito AJ, Binder J, Brown LF, de Sousa M, Van de Water L, Kupiec-Weglinski JW: Anti-TNF-alpha treatment down-regulates the expression of fibronectin and decreases cellular infiltration of cardiac allografts in rats. J Immunol 1995, 154:2949-2958 [PubMed] [Google Scholar]
- 24.Clausell N, Molossi S, Rabinovitch M: Increased interleukin-1 beta and fibronectin expression are early features of the development of the postcardiac transplant coronary arteriopathy in piglets. Am J Pathol 1993, 142:1772-1786 [PMC free article] [PubMed] [Google Scholar]
- 25.Clausell N, Molossi S, Sett S, Rabinovitch M: In vivo blockade of tumor necrosis factor-alpha in cholesterol-fed rabbits after cardiac transplant inhibits acute coronary artery neointimal formation. Circulation 1994, 89:2768-2779 [DOI] [PubMed] [Google Scholar]
- 26.Coito AJ, Korom S, Graser E, Volk HD, Van de Water L, Kupiec-Weglinski JW: Blockade of very late antigen-4 integrin binding to fibronectin in allograft recipients: I. Treatment with connecting segment-1 peptides prevents acute rejection by suppressing intragraft mononuclear cell accumulation, endothelial activation, and cytokine expression. Transplantation 1998, 65:699-706 [DOI] [PubMed] [Google Scholar]
- 27.Korom S, Hancock WW, Coito AJ, Kupiec-Weglinski JW: Blockade of very late antigen-4 integrin binding to fibronectin in allograft recipients. II. Treatment with connecting segment-1 peptides prevents chronic rejection by attenuating arteriosclerotic development and suppressing intragraft T cell and macrophage activation. Transplantation 1998, 65:854-859 [DOI] [PubMed] [Google Scholar]
- 28.Onodera K, Lehmann M, Akalin E, Volk HD, Sayegh MH, Kupiec-Weglinski JW: Induction of “infectious” tolerance to MHC-incompatible cardiac allografts in CD4 monoclonal antibody-treated sensitized rat recipients. J Immunol 1996, 157:1944-1950 [PubMed] [Google Scholar]
- 29.Lehmann M, Sternkopf F, Metz F, et al: Induction of long-term survival of rat skin allografts by a novel, highly efficient anti-CD4 monoclonal antibody. Transplantation 1992, 54:959-962 [DOI] [PubMed] [Google Scholar]
- 30.Siegling A, Lehmann M, Platzer C, Emmrich F, Volk HD: A novel multispecific competitor fragment for quantitative PCR analysis of cytokine gene expression in rats. J Immunol Methods 1994, 177:23-28 [DOI] [PubMed] [Google Scholar]
- 31.Kupiec-Weglinski JW, Sablinski T, Hancock WW, DiStefano R, Mariani G, Mix CT, Tilney NL: Modulation of accelerated rejection of cardiac allografts in sensitized rats by anti-interleukin 2 receptor monoclonal antibody and cyclosporine therapy. Transplantation 1991, 51:300-305 [DOI] [PubMed] [Google Scholar]
- 32.Sablinski T, Sayegh MH, Hancock WW, Kut JP, Milford EL, Tilney NL, Kupiec-Weglinski JW: Differential role of CD4+ cells in the sensitization and effector phases of accelerated graft rejection. Transplantation 1991, 51:226-231 [DOI] [PubMed] [Google Scholar]
- 33.Sanfilippo FP, Vaughn WK, Peters TG, Shield CF, Adams PL, Lorber MI, Williams GM: Factors affecting the waiting time of cadaveric kidney transplant candidates in the United States. JAMA 1992, 267:247-252 [PubMed] [Google Scholar]
- 34.Onodera K, Chandraker A, Volk HD, Ritter T, Lehmann M, Kato H, Sayegh MH, Kupiec-Weglinski JW: Distinct tolerance pathways in sensitized allograft recipients after selective blockade of activation signal 1 or signal 2. Transplantation 1999, 68:288-293 [DOI] [PubMed] [Google Scholar]
- 35.Adams DH, Russell ME, Hancock WW, Sayegh MH, Wyner LR, Karnovsky MJ: Chronic rejection in experimental cardiac transplantation: studies in the Lewis-F344 model. Immunol Rev 1993, 134:5-19 [DOI] [PubMed] [Google Scholar]
- 36.Tilney NL, Whitley WD, Diamond JR, Kupiec-Weglinski JW, Adams DH: Chronic rejection: an undefined conundrum. Transplantation 1991, 52:389-398 [DOI] [PubMed] [Google Scholar]
- 37.Cramer DV, Wu GD, Chapman FA, Cajulis E, Wang HK, Makowka L: Lymphocytic subsets and histopathologic changes associated with the development of heart transplant arteriosclerosis. J Heart Lung Transplant 1992, 11:458-466 [PubMed] [Google Scholar]
- 38.Onodera K, Hancock WW, Graser E, Lehmann M, Sayegh MH, Strom TB, Volk HD, Kupiec-Weglinski JW: Type 2 helper T cell-type cytokines and the development of: “infectious” tolerance in rat cardiac allograft recipients. J Immunol 1997, 158:1572-1581 [PubMed] [Google Scholar]
- 39.McCarthy JB, Chelberg MK, Mickelson DJ, Furcht LT: Localization and chemical synthesis of fibronectin peptides with melanoma adhesion and heparin binding activities. Biochemistry 1988, 27:1380-1388 [DOI] [PubMed] [Google Scholar]
- 40.Elices MJ, Osborn L, Takada Y, Crouse C, Luhowskyj S, Hemler ME, Loob RR: VCAM-1 on activated endothelium interacts with the leukocyte integrin VLA-4 at a site distinct from the VLA-4/fibronectin binding site. Cell 1990, 60:577-584 [DOI] [PubMed] [Google Scholar]
- 41.Elices MJ, Tsai V, Strahl D, Goel AS, Tollefson V, Arrhenius T, Wayner EA, Gaeta FC, Fikes JD, Firestein GS: Expression and functional significance of alternatively spliced CS1 fibronectin in rheumatoid arthritis microvasculature. J Clin Invest 1994, 93:405-416 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wahl SM, Allen JB, Hines KL, Imamichi T, Wahl AM, Furcht LT, McCarthy JB: Synthetic fibronectin peptides suppress arthritis in rats by interrupting leukocyte adhesion and recruitment. J Clin Invest 1994, 94:655-662 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lore K, Sonnerborg A, Spetz AL, Andersson U, Andersson J: Immunocytochemical detection of cytokines and chemokines in Langerhans cells and in vitro derived dendritic cells (corrected and republished in J Immunol Methods 1998, 218: 173–187). J Immunol Methods 1998, 214:97-111 [DOI] [PubMed] [Google Scholar]
- 44.Chirgwin JM, Przybyla AE, MacDonald RJ, Rutter WJ: Isolation of biologically active ribonucleic acid from sources enriched in ribonuclease. Biochemistry 1979, 18:5294-5299 [DOI] [PubMed] [Google Scholar]
- 45.Binder J, Lehmann M, Graser E, Hancock WW, Watschinger B, Onodera K, Sayegh MH, Volk HD, Kupiec-Weglinski JW: The effects of nondepleting CD4 targeted therapy in presensitized rat recipients of cardiac allografts. Transplantation 1996, 61:804-811 [DOI] [PubMed] [Google Scholar]
- 46.Finkelstein LD, Reynolds PJ, Hunt SW, Shimizu Y: Structural requirements for beta1 integrin-mediated tyrosine phosphorylation in human T cells. J Immunol 1997, 159:5355-5363 [PubMed] [Google Scholar]
- 47.LaFlamme SE, Akiyama SK, Yamada KM: Regulation of fibronectin receptor distribution (published erratum appears in J Cell Biol 1992, 118: 491). J Cell Biol 1992, 117:437-447 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Delcommenne M, Streuli CH: Control of integrin expression by extracellular matrix. J Biol Chem 1995, 270:26794-26801 [DOI] [PubMed] [Google Scholar]
- 49.Brown LF, Dubin D, Lavigne L, Logan B, Dvorak HF, Van de Water L: Macrophages and fibroblasts express embryonic fibronectins during cutaneous wound healing. Am J Pathol 1993, 142:793-801 [PMC free article] [PubMed] [Google Scholar]
- 50.Ffrench-Constant C, Van de Water L, Dvorak HF, Hynes RO: Reappearance of an embryonic pattern of fibronectin splicing during wound healing in the adult rat. J Cell Biol 1989, 109:903-914 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Glukhova MA, Frid MG, Shekhonin BV, Vasilevskaya TD, Grunwald J, Saginati M, Koteliansky VE: Expression of extra domain A fibronectin sequence in vascular smooth muscle cells is phenotype dependent. J Cell Biol 1989, 109:357-366 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Knowlton AA, Connelly CM, Romo GM, Mamuya W, Apstein CS, Brecher P: Rapid expression of fibronectin in the rabbit heart after myocardial infarction with and without reperfusion. J Clin Invest 1992, 89:1060-1068 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Nickeleit V, Zagachin L, Nishikawa K, Peters JH, Hynes RO, Colvin RB: Embryonic fibronectin isoforms are synthesized in crescents in experimental autoimmune glomerulonephritis. Am J Pathol 1995, 147:965-978 [PMC free article] [PubMed] [Google Scholar]
- 54.Jarnagin WR, Rockey DC, Koteliansky VE, Wang SS, Bissell DM: Expression of variant fibronectins in wound healing: cellular source and biological activity of the EIIIA segment in rat hepatic fibrogenesis. J Cell Biol 1994, 127:2037-2048 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Molossi S, Elices M, Arrhenius T, Diaz R, Coulber C, Rabinovitch M: Blockade of very late antigen-4 integrin binding to fibronectin with connecting segment-1 peptide reduces accelerated coronary arteriopathy in rabbit cardiac allografts (see comments). J Clin Invest 1995, 95:2601-2610 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Cowan B, Baron O, Crack J, Coulber C, Wilson GJ, Rabinovitch M: Elafin, a serine elastase inhibitor, attenuates post-cardiac transplant coronary arteriopathy and reduces myocardial necrosis in rabbits afer heterotopic cardiac transplantation. J Clin Invest 1996, 97:2452-2468 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Orosz CG, Ohye RG, Pelletier RP, Van Buskirk AM, Huang G, Morgan C, Kincade PW, Ferguson RM: Treatment with anti-vascular cell adhesion molecule 1 monoclonal antibody induces long-term murine cardiac allograft acceptance. Transplantation 1993, 56:453-460 [DOI] [PubMed] [Google Scholar]
- 58.Matsuyama T, Yamada A, Kay J, Yamada KM, Akiyama SK, Schlossman SF, Morimoto C: Activation of CD4 cells by fibronectin and anti-CD3 antibody: A synergistic effect mediated by the VLA-5 fibronectin receptor complex. J Exp Med 1989, 170:1133-1148 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Nojima Y, Rothstein DM, Sugita K, Schlossman SF, Morimoto C: Ligation of VLA-4 on T cells stimulates tyrosine phosphorylation of a 105-kD protein. J Exp Med 1992, 175:1045-1053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Ticchioni M, Deckert M, Bernard G, et al: Comitogenic effects of very late activation antigens on CD3-stimulated human thymocytes: involvement of various tyrosine kinase pathways. J Immunol 1995, 154:1207-1215 [PubMed] [Google Scholar]
- 61.Yamada A, Nikaido T, Nojima Y, Schlossman SF, Morimoto C: Activation of human CD4 T lymphocytes: interaction of fibronectin with VLA-5 receptor on CD4 cells induces the AP-1 transcription factor. J Immunol 1991, 146:53-56 [PubMed] [Google Scholar]
- 62.McCarthy JB, Vachhani BV, Wahl SM, Finbloom DS, Feldman GM: Human monocyte binding to fibronectin enhances IFN-gamma-induced early signaling events. J Immunol 1997, 159:2424-2430 [PubMed] [Google Scholar]
- 63.Brunmark A, O’Rourke AM: Augmentation of mature CD4+ T cell responses to isolated antigenic class II proteins by fibronectin and intercellular adhesion molecule-1. J Immunol 1997, 159:1676-1685 [PubMed] [Google Scholar]
- 64.Hershkoviz R, Gilat D, Miron S, Mekori YA, Aderka D, Wallach D, Vlodavsky I, Cohen IR, Lider O: Extracellular matrix induces tumour necrosis factor-alpha secretion by an interaction between resting rat CD4+ T cells and macrophages. Immunology 1993, 78:50-57 [PMC free article] [PubMed] [Google Scholar]
- 65.Crawford SE, Stellmach V, Murphy-Ullrich JE, Ribeiro SM, Lawler J, Hynes RO, Boivin GP, Bouck N: Thrombospondin-1 is a major activator of TGF-beta1 in vivo. Cell 1998, 93:1159-1170 [DOI] [PubMed] [Google Scholar]
- 66.Lortat-Jacob H, Grimaud JA: Interferon-gamma C-terminal function: new working hypothesis. Heparan sulfate and heparin, new targets for IFN-gamma, protect, relax the cytokine and regulate its activity. Cell Mol Biol 1991, 37:253-260 [PubMed] [Google Scholar]
- 67.Lortat-Jacob H, Kleinman HK, Grimaud JA: High-affinity binding of interferon-gamma to a basement membrane complex (matrigel). J Clin Invest 1991, 87:878-883 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Ferguson TA, Mizutani H, Kupper TS: Two integrin-binding peptides abrogate T cell-mediated immune responses in vivo. Proc Natl Acad Sci USA 1991, 88:8072-8076 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Hines KL, Kulkarni AB, McCarthy JB, Tian H, Ward JW, Christ M, McCartney-Francis NL, Furcht LT, Karlsson S, Wahl SM: Synthetic fibronectin peptides interrupt inflammatory cell infiltration in transforming growth factor beta 1 knockout mice. Proc Natl Acad Sci USA 1994, 91:5187-5191 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Laffon A, Garcia-Vicuna R, Humbria A, Postigo AA, Corbi AL, de Landazuri MO, Sanchez-Madrid F: Upregulated expression and function of VLA-4 fibronectin receptors on human activated T cells in rheumatoid arthritis. J Clin Invest 1991, 88:546-552 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Elices MJ, Tsai V, Strahl D, Goel AS, Tollefson V, Arrhenius T, Wayner EA, Gaeta FC, Fikes JD, Firestein GS: Expression and functional significance of alternatively spliced CS1 fibronectin in rheumatoid arthritis microvasculature. J Clin Invest 1994, 93:405-416 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Coito AJ, Korom S, Hancock WW, Kupiec-Weglinski JW: Blockade of alpha 4 beta 1-integrin-fibronectin adhesive interactions prevents chronic allograft rejection in sensitized recipients. Transplant Proc 1998, 30:939-940 [DOI] [PubMed] [Google Scholar]
- 73.Moyano JV, Carnemolla B, Dominguez-Jimenez C, Garcia-Gila M, Albar JP, Sanchez-Aparicio P, Leprini A, Querze G, Zardi L, Garcia-Pardo A: Fibronectin type III5 repeat contains a novel cell adhesion sequence, KLDAPT, which binds activated alpha4beta1 and alpha4beta7 integrins. J Biol Chem 1997, 272:24832-24836 [DOI] [PubMed] [Google Scholar]
- 74.Hynes RO: Integrins: a family of cell surface receptors. Cell 1987, 48:549-554 [DOI] [PubMed] [Google Scholar]
- 75.Yang JT, Hynes RO: Fibronectin receptor functions in embryonic cells deficient in alpha 5 beta 1 integrin can be replaced by alpha V integrins. Mol Biol Cell 1996, 7:1737-1748 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Garcia-Pardo A, Wayner EA, Carter WG, Ferreira OCJ: Human B lymphocytes define an alternative mechanism of adhesion to fibronectin. The interaction of the alpha 4 beta 1 integrin with the LHGPEILDVPST sequence of the type III connecting segment is sufficient to promote cell attachment. J Immunol 1990, 144:3361-3366 [PubMed] [Google Scholar]
- 77.Sanchez-Aparicio P, Dominguez-Jimenez C, Garcia-Pardo A: Activation of the alpha 4 beta 1 integrin through the beta 1 subunit induces recognition of the RGDS sequence in fibronectin. J Cell Biol 1994, 126:271-279 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Roberts CJ, Birkenmeier TM, McQuillan JJ, Akiyana SK, Yamada SS, Chen WT, Yamada KM, McDonald JA: Transforming growth factor beta stimulates the expression of fibronectin and of both subunits of the human fibronectin receptor by cultured human lung fibroblasts. J Biol Chem 1988, 263:4586-4592 [PubMed] [Google Scholar]
- 79.Ignotz RA, Endo T, Massague J: Regulation of fibronectin and type I collagen mRNA levels by transforming growth factor-beta. J Biol Chem 1987, 262:6443-6446 [PubMed] [Google Scholar]