Abstract
Although there is substantial evidence indicating that platelets are released from megakaryocytes in the capillary bed of the lung, this concept has not been universally accepted because much of the evidence has been indirect. To more definitively substantiate that platelet production takes place in the lungs, megakaryocyte and platelet production was accelerated in mice by phlebotomy or by administration of thrombopoietin, and ultrastructural analysis was performed on lung specimens. Intact megakaryocytes, megakaryocyte fragments with numerous demarcated platelet fields, dissociating intact platelets, and denuded megakaryocyte nuclei were seen in the pulmonary capillaries of mice. In addition, some megakaryocyte nuclei exhibited the morphological counterpart of apoptosis. These observations provide evidence for platelet release in the capillary bed of the lungs during stimulated as well as reactive thrombocytosis without precluding observations that some “proplatelets” form in the sinusoids of the bone marrow before transmigration of intact megakaryocytes into the circulation.
The origin of platelets is no longer controversial. No one would question that platelets are derived from megakaryocytes, whether this occurs by fragmentation of long megakaryocyte processes forming on a solid substrate in vitro, 1 by fragmentation of long megakaryocyte pseudopods, so-called proplatelets, protruding into the sinusoidal lumen in vivo, 2,3 or by more global fragmentation of megakaryocyte cytoplasm into individual platelets as observed when the cells are kept in suspension. 4 Rather, the controversy concerns the site where the bulk of platelet release takes place in vivo. Evidence supporting the concept that megakaryocytes are migratory and able to exit intact via marrow sinusoids has been illustrated by numerous investigators. 5,6 It is also generally known that platelets are not motile and that few, if any, are found in normal bone marrow. The latter statement pertains equally to denuded or bare megakaryocyte nuclei. 7 Therefore, it has become increasingly clear that platelets are released from megakaryocytes into the bloodstream when long megakaryocyte processes protrude through the marrow sinusoids or that this process takes place in the lungs, the first capillary bed encountered by any cell leaving the bone marrow. Scattered reports of the latter observation have appeared in the literature for at least 30 years. 8-12 A more recent study by Levine et al 13 addressing this issue specifically has established that in humans, 10 times as many intact megakaryocytes are found in pulmonary artery blood than in blood obtained from the aorta. Moreover, it was observed that 98% of megakaryocytes leaving the lung are devoid of cytoplasm. These observations corroborated other studies quantitating megakaryocytes in the central and arterial circulations before, during, and after cardiopulmonary bypass in patients with normal platelet counts who underwent cardiac surgery. 14 Before cardiopulmonary bypass (CPB), the number of megakaryocytes in arterial blood was 0.25/ml, whereas it was 4.21/ml in venous blood. During CPB, the number of megakaryocytes in arterial blood rose to 19.49/ml. Whereas the number of megakaryocytes was always higher in central venous than in arterial blood before and after CPB, this difference was essentially lost during CPB. Last, on autopsy pathologists routinely find megakaryocytes in the lungs but not in any other organs. 15 Together, these reports have markedly strengthened the conclusion based on numerous smaller historical studies that platelets are released from megakaryocytes in the lung. Unfortunately, even the report by Levine et al was not able to lay the controversy to rest. A subsequent study carried out in mice, after perturbation of platelet production by several means, concluded that the fraction of thrombopoiesis occurring in murine lung, even during periods of greatly increased platelet production, is insignificant. 16 However, the agents used to increase thrombopoiesis in this study, such as platelet antiserum, 5-fluorouracil, and radioactive strontium, are known to affect megakaryocyte and platelet physiology in ways not commonly operative during physiological, reactive thrombopoiesis. For instance, platelet antiserum cross-reacts with megakaryocytes, which could affect their motility. The detrimental effect of 5-fluorouracil on megakaryocytes is well known to hematologists.
Thus, although the evidence that platelet release occurs in the lung is substantial, most of the data are indirect and are still questioned by some investigators. Therefore, we decided to reexamine the issue by increasing thrombopoiesis in mice with more physiological stimuli, namely by phlebotomy and by the administration of thrombopoietin, and subjecting lung specimens to ultrastructural analysis. The large number of intact megakaryocytes, megakaryocyte fragments, and denuded megakaryocyte nuclei seen in the pulmonary capillary bed of these animals was remarkable, leaving little room for further doubt that platelets are released at this site.
Materials and Methods
Six 8–12-week-old CB6Fl/J mice (Jackson Laboratories, Bar Harbor, ME) were given recombinant human megakaryocyte and development factor (polyethylene glycol conjugated), a truncated Mpl ligand containing the receptor-binding N-terminal domain of thrombopoietin (50 μg/kg/day) (a gift from Amgen, Thousand Oaks, CA) for 4 days subcutaneously as previously described. 17 Three mice were bled (200 μl) daily for 4 days via tail vein incision, and three mice were injected with saline for 4 days. Mice were sacrificed on day 5. After 5 days, platelet counts had risen from 1 × 106/μl to 1.23 × 106/μl in bled mice and 3.6 × 106/μl in thrombopoietin-treated animals. At this time, pegylated recombinant human megakaryocyte and development factor is active in stimulating megakaryocyte maturation and platelet production in mice. 18
Lung tissue was harvested for electron microscopy on day 5 from sacrificed mice and placed in 3% phosphate-buffered glutaraldehyde. Tissue was postfixed in osmium tetroxide, dehydrated, and embedded in PolyBed 812 (Polysciences, Warrentown, PA) as previously described. 19 Thin sections were stained with uranyl acetate and lead citrate, and sections from untreated mice, thromboietin-treated mice, and phlebotomized mice were examined with a Siemens Elmskop I electron microscope.
Results
Megakaryocyte fragments exhibiting many demarcated platelet fields were found within pulmonary capillaries (Figure 1) ▶ in phlebotomized mice, when the peripheral blood platelet count had risen from 1 × 106/ul to 1.23 × 106/μl. These findings were even more evident in thrombopoietin-treated mice, the peripheral blood platelet count of which had risen to 3.6 × l06/μl. Megakaryocyte nuclei and cytoplasmic fragments were also seen in the lungs of untreated mice, confirming reports by others. 11,14,15 However, finding megakaryocyte fragments required more extensive search and numerous ultrathin sections in specimens from control mice with normal platelet counts.
Figure 1.
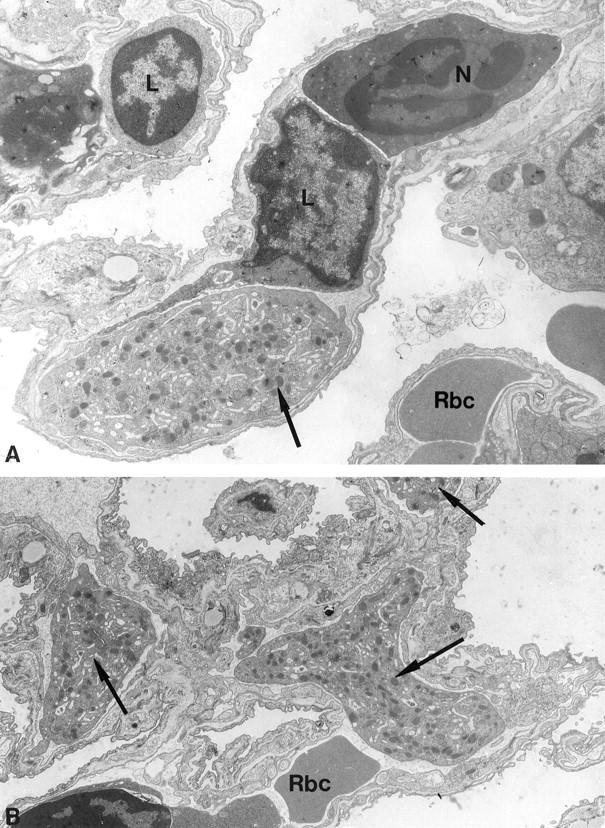
Representative sections of lung from bled mice, showing large fragments of megakaryocyte cytoplasm (arrows) within capillary lumen. The demarcated platelet fields are clearly seen. Also note that the megakaryocyte fragment in A is considerably larger than the lymphocyte (L) or neutrophil (N) within the same capillary lumen. In B an erythrocyte (RBC) is seen within the same lumen as the megakaryocyte fragment. Original magnifications: A, ×8000; B, ×7500.
Figure 2 ▶ illustrates an intact megakaryocyte, which fills the entire capillary lumen. Its peripheral zone appears to be uninterrupted. A large intraluminal megakaryocyte, which appears to have been fixed in the process of release of numerous platelets, is shown in Figure 3 ▶ . It should be noted that the platelets exhibit no evidence of activation, a conclusion that may be safely drawn from their uniformly lenticular shape and the even distribution of their granules. Fragmenting megakaryocytes were not seen in the bone marrow.
Figure 2.
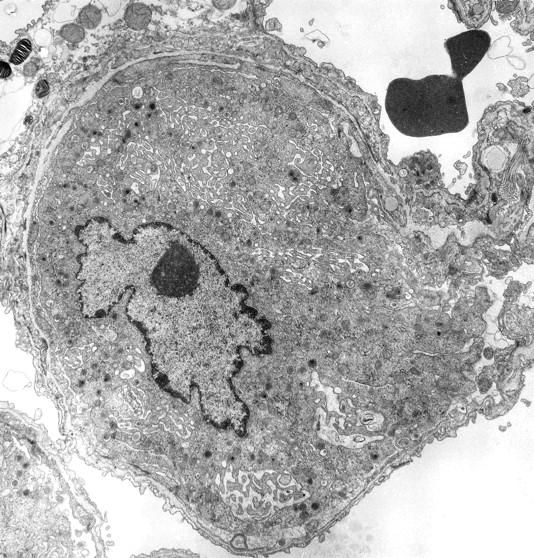
Intact megakaryocyte occupying the lumen of a pulmonary capillary. The specimen was obtained from a thrombopoietin-treated mouse. Original magnification, ×9000.
Figure 3.
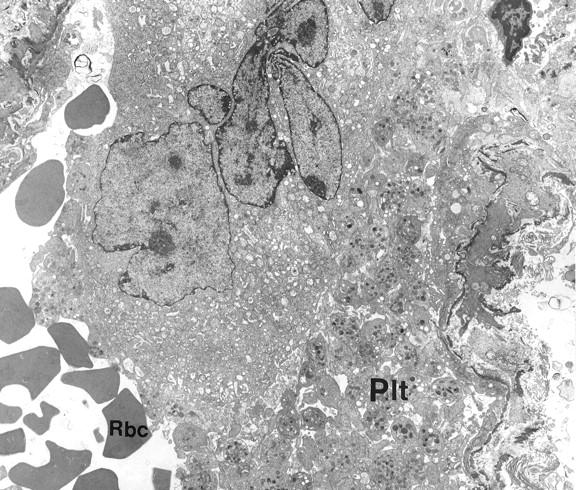
Megakaryocyte in the lumen of a pulmonary capillary adjacent to numerous platelets (Plt) likely to leave been released by the cell. RBC, erythrocyte. Original magnification, ×4000.
In addition, the pulmonary capillaries of animals with accelerated thrombopoiesis contained numerous megakaryocyte nuclei, which were almost completely denuded of cytoplasm (Figure 4) ▶ . Many of these nuclei also exhibited morphological evidence of apoptosis (see arrow in Figure 4 ▶ and inset). Although present, such nuclei were much more difficult to find in control animals with baseline platelet counts.
Figure 4.
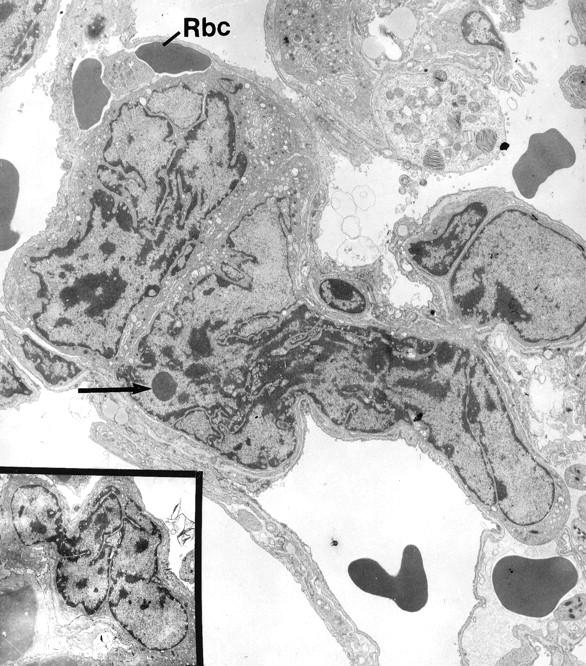
Denuded megakaryocyte nuclei seen in the capillaries of lungs obtained from thrombopoietin-treated mice. Note erythrocytes (RBC) within the same lumen. The chromatin distribution in the megakaryocyte nuclei is suggestive of apoptosis. The arrow indicates an “apoptic” body. Original magnification, ×4500; inset, ×3000.
Discussion
The illustrations presented in this report provide convincing evidence that intact megakaryocytes reach the pulmonary capillaries and that they release platelets by fragmentation of their cytoplasm at this site. The fragments of megakaryocyte cytoplasm seen in Figure 2 ▶ are likely to fragment further before reaching the systemic circulation, because they are several times larger than giant platelets or even leukocytes seen on routine blood smears. Although the uninitiated reader may question whether the large accumulation of platelets depicted in Figure 3 ▶ could represent a thrombus, this concern is obviated by the lack of adherence of one platelet to another, as well as the absence of shape changes and degranulation. Even centralization of granules, which occurs very early after activation and before degranulation takes place, is not evident.
These observations do not gainsay reports by several investigators showing that long megakaryocyte processes exhibiting demarcated platelet fields, referred to as proplatelets, can be seen to protrude into the lumen of marrow sinusoids (reviewed in ref 3 ). Presumably, the proplatelets subsequently fragment into individual platelets. In this case, the denuded megakaryocyte nucleus may not exit from the medullary cavity. However, although a few denuded megakaryocyte nuclei can be found in normal bone marrow, their number does not increase during reactive thrombocytosis ( ref 7 and unpublished observations).
Intravascular megakaryocytes in the lung were first described by Aschoff in 1893, 20 and the concept that platelet production occurs primarily in the lung was first published in 1937. 21 Since that time, numerous reports of the occurrence of pulmonary and circulating megakaryocytes in human subjects have been published. Interestingly, the largest number of these reports are found in the nonhematological literature. 15 For instance, the prevailing theory attempting to explain the etiopathology of hypertrophic pulmonary arthropathy associated with arteriovenous shunts, either in the heart or in the lungs, attributes the phenomenon to circulating megakaryocytes or their fragments. It is postulated that these reach the fingertips in the axial vascular stream, releasing growth factors there, such as platelet-derived growth factor, which hyperstimulates fibroblasts. 22,23
Together with convincing indirect evidence, provided by an unusually large number of reports, the illustrations presented here should leave no further doubt that in health, a substantial number of megakaryocytes arrest in the pulmonary capillary bed and release platelets at this site.
Footnotes
Address reprint requests to Dr. Dorothea Zucker-Franklin, New York University Medical Center, UH 445, 550 First Avenue, New York, NY 10016.
Supported in part by American Heart Association grant-in-aid 9707978A (NJ Affiliate), the Foundation of UMDNJ, and the Robert Wood Johnson Cardiovascular Institute.
References
- 1.Scurfield G, Radley JM: Aspects of platelet formation and release. Am J Hematol 1981, 10:285-296 [DOI] [PubMed] [Google Scholar]
- 2.Radley JM, Scurfield G: The mechanism of platelet release. Blood 1980, 56:996-999 [PubMed] [Google Scholar]
- 3.Choi E: Regulation of proplatelet and platelet formation in vitro. Kuter DJ Hunt P Sheridan W Zucker-Franklin D eds. Thrombopoiesis and Thrombopoietins, ch 17. 1997, :pp 271-284 Humana Press, Totowa NJ [Google Scholar]
- 4.Zucker-Franklin D, Petursson S: Thrombocytopoiesis analysis by membrane tracer and freeze-fracture studies on fresh human and mouse megakaryocytes. J Cell Biol 1984, 99:390-402 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lichtman MA, Chamberlain JK, Simon W, Santillo PA: Parasinusoidal location of megakaryocytes in marrow: a determinant of platelet release. Am J Hematol 1978, 4:303-312 [DOI] [PubMed] [Google Scholar]
- 6.Tavassoli M, Aoki M: Migration of entire megakaryocytes through the marrow-blood barrier. Br J Haematol 1981, 48:25-29 [DOI] [PubMed] [Google Scholar]
- 7.Zucker-Franklin D, Termin CS, Cooper MC: Structural changes in the megakaryocytes of patients infected with the human immune deficiency virus (HIV-1). Am J Pathol 1989, 134:1295-1304 [PMC free article] [PubMed] [Google Scholar]
- 8.Melamed MR, Clifften EE, Mercer C, Koss G: The megakaryocyte blood count. Am J Med Sci 1966, 252:301-309 [DOI] [PubMed] [Google Scholar]
- 9.Kaufman RM, Airo R, Pollack S, Crosby WH: Circulating megakaryocytes and platelets released in the lung. Blood 1965, 26:720-731 [PubMed] [Google Scholar]
- 10.Pedersen NT: Occurrence of megakaryocytes in various vessels and their retention in pulmonary capillaries in man. Scand J Haematol 1978, 21:369-375 [DOI] [PubMed] [Google Scholar]
- 11.Trowbridge EA, Martin JF, Slater DN: Evidence for a theory of physical fragmentation of megakaryocytes implying that all platelets are produced in the pulmonary circulation. Thromb Res 1982, 28:461-472 [DOI] [PubMed] [Google Scholar]
- 12.Slater DN, Trowbridge EA, Martin JF: The megakaryocyte in thrombocytopenia: a microscopic study which supports the theory that platelets are produced in the pulmonary circulation. Thromb Res 1963, 31:163-176 [DOI] [PubMed] [Google Scholar]
- 13.Levine RF, Eldor A, Shoff PK, Kirwin S, Tenza D, Cramer EM: Circulating megakaryocytes. Delivery of large numbers of intact, mature megakaryocytes to the lungs. Eur J Haematol 1993, 51:233-246 [DOI] [PubMed] [Google Scholar]
- 14.Wilde NT, Burgess R, Keenan DJM, Lucas GS: The effect of cardiopulmonary bypass on circulating megakaryocytes. Br J Haematol 1997, 98:322-327 [DOI] [PubMed] [Google Scholar]
- 15.Aabo K, Hansen B: Megakaryocytes in pulmonary blood vessels. Acta Pathol Microbial Scand A 1978, 86:285-291 [PubMed] [Google Scholar]
- 16.Davis RE, Stenberg PE, Levin J, Beckstead JA: Localization of megakaryocytes in normal mice and following administration of platelet antiserum, 5-fluorouracil, or radiostrontium: evidence for the site of platelet production. Exp Hematol 1997, 25:638-648 [PubMed] [Google Scholar]
- 17.Philipp CS, Remmler J, Zucker-Franklin D: The effects of MPL-ligand, interleukin-6 and interleukin-11 on megakaryocyte and platelet alpha-granule proteins. Thromb Haemost 1998, 80:968-975 [PubMed] [Google Scholar]
- 18.Ulich TR, del Castillo J, Senaldi G, Kinstler O, Yin S, Kaufman S, Tarpley J, Choi E, Kirley T, Hunt P, Sheridan WP: Systemic hematologic effects of PEG-rHuMGDF-induced megakaryocyte hyperplasia in mice. Blood 1996, 87:5006-5015 [PubMed] [Google Scholar]
- 19.Stahl CP, Zucker-Franklin D, Evatt BL, Winton EF: Effects of human interleukin-6 on megakaryocyte development and thrombocytopoiesis in primates. Blood 1991, 78:1467-1475 [PubMed] [Google Scholar]
- 20.Aschoff L: Über capillare Embolie von riesenkemhaltigen Zellen. Arch Pathol Anat Phys 1893, 134:11-14 [Google Scholar]
- 21.Howell WH, Donohue DD: The production of blood platelets in the lungs. J Exp Med 1937, 65:171-203 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Vasquez-Abad D, Martinez-Lavin M: Macrothrombocytes in the peripheral circulation of patients with cardiogenic hypertrophic osteoarthropathy. Clin Exp Rheumatol 1991, 9:59-62 [PubMed] [Google Scholar]
- 23.Dickinson CJ: The aetiology of clubbing and hypertrophic osteoarthropathy. Eur J Clin Invest 1993, 23:330-338 [DOI] [PubMed] [Google Scholar]


