Abstract
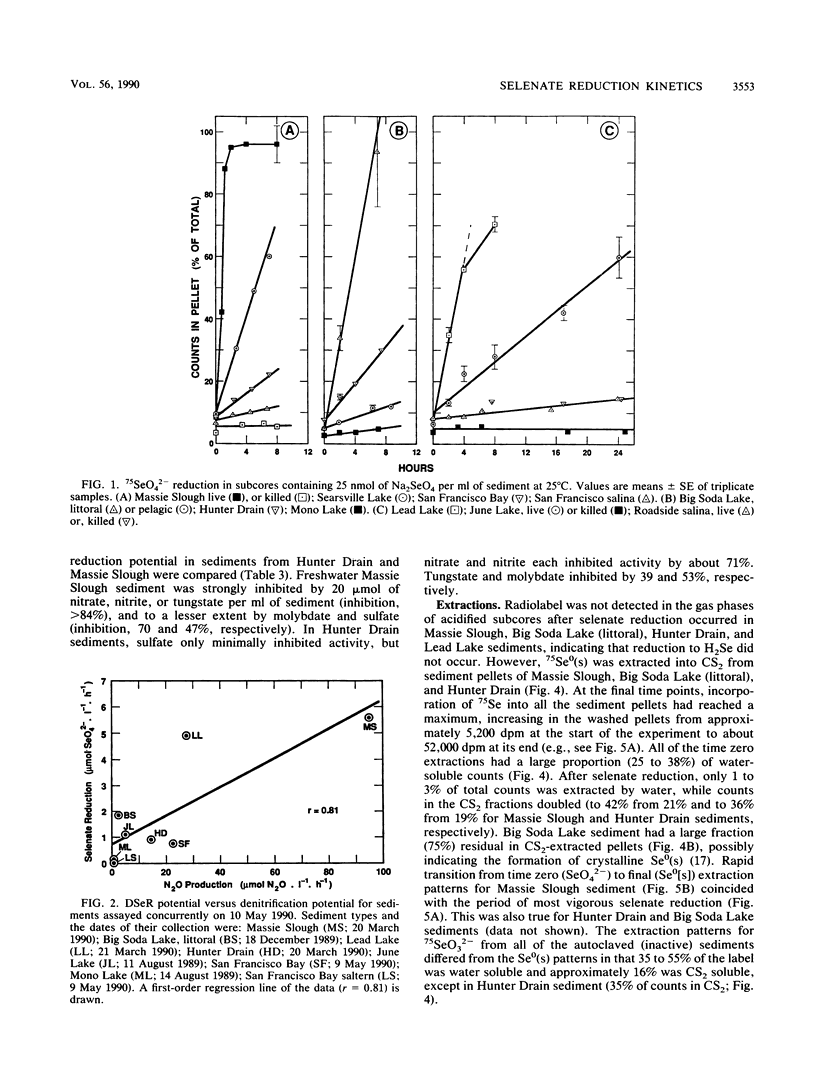
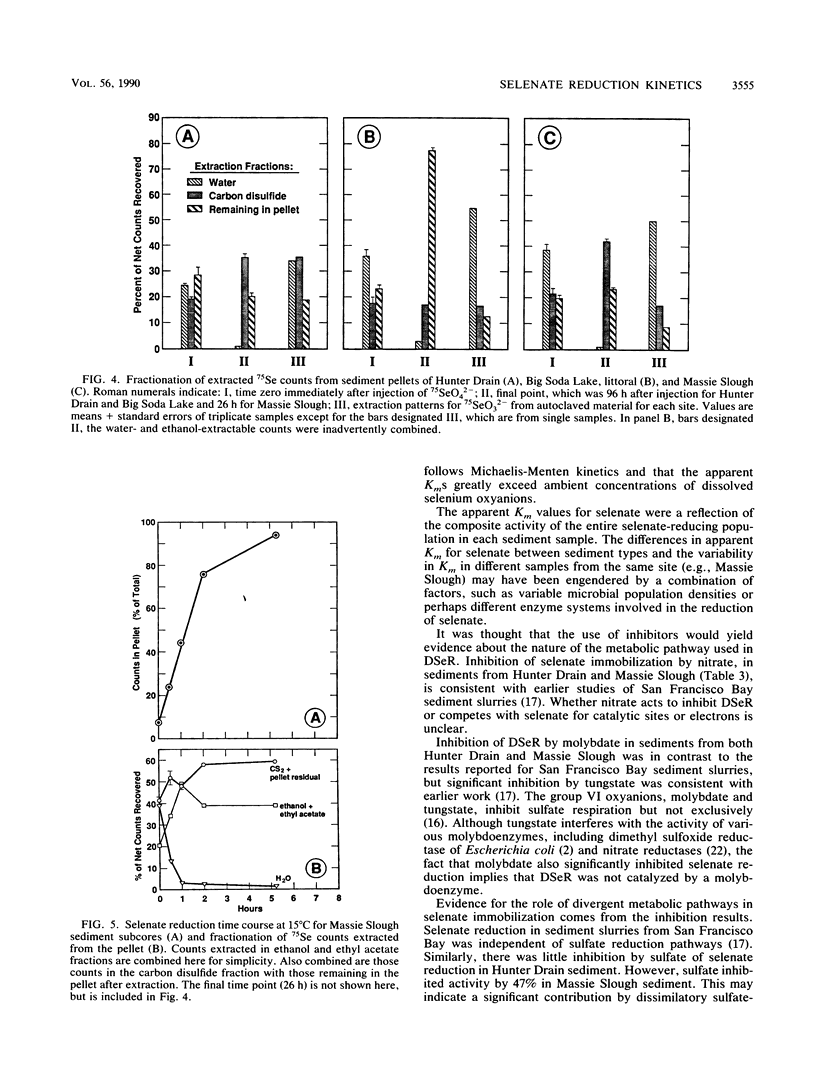
We measured potential rates of bacterial dissimilatory reduction of 75SeO42− to 75Se0 in a diversity of sediment types, with salinities ranging from freshwater (salinity = 1 g/liter) to hypersaline (salinity = 320 g/liter and with pH values ranging from 7.1 to 9.8. Significant biological selenate reduction occurred in all samples with salinities from 1 to 250 g/liter but not in samples with a salinity of 320 g/liter. Potential selenate reduction rates (25 nmol of SeO42− per ml of sediment added with isotope) ranged from 0.07 to 22 μmol of SeO42− reduced liter−1 h−1. Activity followed Michaelis-Menten kinetics in relation to SeO42− concentration (Km of selenate = 7.9 to 720 μM). There was no linear correlation between potential rates of SeO42− reduction and salinity, pH, concentrations of total Se, porosity, or organic carbon in the sediments. However, potential selenate reduction was correlated with apparent Km for selenate and with potential rates of denitrification (r = 0.92 and 0.81, respectively). NO3−, NO2−, MoO42−, and WO42− inhibited selenate reduction activity to different extents in sediments from both Hunter Drain and Massie Slough, Nev. Sulfate partially inhibited activity in sediment from freshwater (salinity = 1 g/liter) Massie Slough samples but not from the saline (salinity = 60 g/liter) Hunter Drain samples. We conclude that dissimilatory selenate reduction in sediments is widespread in nature. In addition, in situ selenate reduction is a first-order reaction, because the ambient concentrations of selenium oxyanions in the sediments were orders of magnitude less than their Kms.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Balderston W. L., Sherr B., Payne W. J. Blockage by acetylene of nitrous oxide reduction in Pseudomonas perfectomarinus. Appl Environ Microbiol. 1976 Apr;31(4):504–508. doi: 10.1128/aem.31.4.504-508.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bilous P. T., Weiner J. H. Dimethyl sulfoxide reductase activity by anaerobically grown Escherichia coli HB101. J Bacteriol. 1985 Jun;162(3):1151–1155. doi: 10.1128/jb.162.3.1151-1155.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan Y. K., Knowles R. Measurement of denitrification in two freshwater sediments by an in situ acetylene inhibition method. Appl Environ Microbiol. 1979 Jun;37(6):1067–1072. doi: 10.1128/aem.37.6.1067-1072.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Culbertson C. W., Zehnder A. J., Oremland R. S. Anaerobic oxidation of acetylene by estuarine sediments and enrichment cultures. Appl Environ Microbiol. 1981 Feb;41(2):396–403. doi: 10.1128/aem.41.2.396-403.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaspar H. F. Denitrification in marine sediment: measurement of capacity and estimate of in situ rate. Appl Environ Microbiol. 1982 Mar;43(3):522–527. doi: 10.1128/aem.43.3.522-527.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Macy J. M., Michel T. A., Kirsch D. G. Selenate reduction by a Pseudomonas species: a new mode of anaerobic respiration. FEMS Microbiol Lett. 1989 Oct 1;52(1-2):195–198. doi: 10.1016/0378-1097(89)90195-x. [DOI] [PubMed] [Google Scholar]
- Oremland R. S., Hollibaugh J. T., Maest A. S., Presser T. S., Miller L. G., Culbertson C. W. Selenate reduction to elemental selenium by anaerobic bacteria in sediments and culture: biogeochemical significance of a novel, sulfate-independent respiration. Appl Environ Microbiol. 1989 Sep;55(9):2333–2343. doi: 10.1128/aem.55.9.2333-2343.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oremland R. S., Umberger C., Culbertson C. W., Smith R. L. Denitrification in san francisco bay intertidal sediments. Appl Environ Microbiol. 1984 May;47(5):1106–1112. doi: 10.1128/aem.47.5.1106-1112.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith R. L., Oremland R. S. Anaerobic oxalate degradation: widespread natural occurrence in aquatic sediments. Appl Environ Microbiol. 1983 Jul;46(1):106–113. doi: 10.1128/aem.46.1.106-113.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiner J. H., MacIsaac D. P., Bishop R. E., Bilous P. T. Purification and properties of Escherichia coli dimethyl sulfoxide reductase, an iron-sulfur molybdoenzyme with broad substrate specificity. J Bacteriol. 1988 Apr;170(4):1505–1510. doi: 10.1128/jb.170.4.1505-1510.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zehr J. P., Oremland R. S. Reduction of selenate to selenide by sulfate-respiring bacteria: experiments with cell suspensions and estuarine sediments. Appl Environ Microbiol. 1987 Jun;53(6):1365–1369. doi: 10.1128/aem.53.6.1365-1369.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]



