Abstract
Posttransplant lymphoproliferative disease can be treated by the infusion of Epstein-Barr virus-specific cytotoxic T lymphocytes, which were raised against lymphocytes immortalized with a laboratory strain of Epstein-Barr virus (B95.8). Whether the immunodominant epitopes in B95.8 are shared in virus from tumors will affect the general applicability of this therapy. We have characterized the viral strain and the sequence of commonly recognized cytotoxic T-lymphocyte epitopes in 25 posttransplant lymphoproliferative disease specimens from 19 patients. Type A virus was present in 24 of 25 specimens. No variation in two LMP2A epitopes and a few variations mostly outside the targeted epitopes or silent in three EBNA3C epitopes were found, with one variation (Arg to Lys) detected in an EBNA3C epitope in 12 of 24 tumors. However, cytotoxic T lymphocytes to B95-8-derived EBNA3C peptides specifically lysed both B95-8 and the Lys-variant peptide-loaded target cells, although with less efficiency. These results suggest that adoptive immunotherapy using cytotoxic T lymphocytes expanded with B95.8 stimulators or vaccine strategies using B95.8-derived sequence will generally target Epstein-Barr virus strains present in posttransplant lymphoproliferative disease tumors.
Posttransplant lymphoproliferative disorders (PTLDs) are clinically and histologically heterogeneous lymphoid proliferations occurring in organ or bone marrow transplant recipients. 1-3 Generally these are B-cell proliferations and are associated with Epstein-Barr virus (EBV). 4,5 Particular kinds of immune manipulations aimed at preventing or controlling organ rejection or graft-versus-host disease such as the use of anti-CD3 antibodies or high doses of cyclosporine are associated with increased risk of PTLDs. 6,7 Withdrawal or reduction of immunosuppressive therapy is sometimes associated with regression of these tumors. 8 In bone marrow transplant recipients, infusion of lymphocytes from the patient’s bone marrow donor often leads to disease regression but may be associated with graft-versus-host disease. 9 EBV-specific cytotoxic T lymphocytes expanded in vitro in response to irradiated EBV-transformed B lymphocytes have also been reported to induce regression of PTLDs in some cases and to prevent the development of PTLDs in the bone marrow transplant setting without graft-versus-host disease. 10,11 The virus used to transform stimulator cells in these studies was a laboratory strain of EBV referred to as B95.8. 12 An important question with regard to the general application of this approach is how closely B95.8 corresponds with viral genomes present in PTLD lesions with regard to epitopes recognized by EBV cytotoxic T lymphocytes (CTLs).
Over a wide spectrum of HLA alleles, the immunodominant EBV latency antigens are present in a few proteins: EBNA3A, EBNA3B, and EBNA3C. 13,14 Sequence variations among these immunodominant antigens are well recognized. Two EBV subtypes, A and B, that differ mainly in the coding sequences for EBNA2, EBNA3A, EBNA3B, and EBNA3C have been characterized. 15-17 These differences are such that CTLs have been identified that recognize type A virus but not type B virus and vice versa. 18,19 A subdominant response to LMP2 has been consistently detected among individuals who are HLA A0201. 20 Strain differences have also been recognized in LMP2 and these occur independently of subtypes A and B. 21
To determine whether variations in the sequence of viral proteins are likely to limit the applicability of CTLs raised in response to B95.8-infected lymphocytes as anti-tumor CTLs, we analyzed a series of PTLD lesions and determined the impact of a common variant on CTL killing.
Materials and Methods
PTLD Tumor Samples
DNA samples from 25 PTLD specimens from 19 organ transplant patients were obtained from the Johns Hopkins Hospital Lymphoma Bank and the Southwest Oncology Group study tumor bank. More than one tumor specimen was available for patients 3, 4, 7, and 9. Some of these cases have been reported previously (Table 1) ▶ . 22-24 We also studied a tumor specimen from a patient with B-cell lymphoproliferative disease arising in the setting of aplastic anemia treated with immunosuppression (patient 13). 25
Table 1.
PTLD Cases Cited in Other Reports
Genotyping for EBV Subtypes
Major EBV subtypes (A and B) were distinguished by polymerase chain reaction (PCR) amplification using primers spanning the EBNA3C region. 17 The B95.8 cell line was used as a positive control for type A, whereas AG876 was used as a positive control for type B virus. The B-cell lymphoma line BJAB was used as a negative control.
Sequence Analysis of EBV CTL Epitopes
For sequence analyses of EBV CTL epitopes, PCR primers were designed to amplify a 195-bp region of EBNA3C that includes three CTL epitopes (HLA A2.1, B44, and B27-restricted) 26,27 and a 148-bp region of LMP2A containing HLA A2.1- and A24.2-restricted epitopes. 28 The primers were: EBNA3C, 5′-TACGCTTCCTTCGTGGTAAA, 5′-CGATTGTCTTGTGAAACCAG; LMP2A, 5′-CTTGCTATCCTGACCGAATG, 5′-CTGCTGTAAGAATCCAGGCA. The High Fidelity Taq enzyme Platinum Pfx DNA polymerase system (Life Technologies, Inc., Gaithersburg, MD) was used to generate the desired PCR products. PCR products were electrophoresed on a 1.8% agarose gel, excised, purified using the Qiaex II Gel Extraction kit (Qiagen, Chatsworth, CA), and sequenced directly with the SequiTherm Excel DNA PCR sequencing kit (Epicentre Technologies, Madison, WI), or the ABI Prism BigDye Terminator Cycle Sequencing kit and the PE ABI 377 sequencer.
For cases with multiple tumors, sequence analyses of EBV CTL epitopes were also performed by cloning sequencing. Briefly, PCR products were electrophoresed and purified by using Spin-X tubes (Costar, Corning, NY), and then cloned into the pCR2.1-TOPO cloning vector (Invitrogen, Carlsbad, CA). Approximately 10 bacterium colonies were analyzed for each DNA sample. Plasmid DNA was extracted and sequenced by using the ABI 377 sequencer.
Peripheral Blood Mononuclear Cells (PBMCs) and Synthetic Peptides
PBMCs were from HLA-B27 EBV-seropositive healthy platelet donors. PBMCs were isolated by density gradient centrifugation using Ficoll-Hypaque 1.077 (Biochrom, Berlin, Germany) and cryopreserved immediately.
Peptides corresponding to viral epitopes recognized by CTLs were synthesized by Macromolecular Resources (Ft. Collins, CO). RRIYDLIEL (referred to as Arg peptide) and its variant RKIYDLIEL (Lys peptide) corresponding to EBNA3C residues 258 to 266. 26 The peptide CLGGLLTMV corresponding to LMP2 residues 426 to 434 and restricted through HLA-A2 was used as a control. 28 Peptides were dissolved in 10% dimethyl sulfoxide (Sigma, St. Louis, MO) to a final concentration of 1 mg/ml, and stored in aliquots at −80°C. This peptide stock solution was further diluted in appropriate assay media for individual experiments.
Establishment of Peptide-Specific CTL Lines
PBMCs were incubated at 107/ml in 50 μg/ml of peptide (diluted in RPMI 1640) for 2 hours at 37°C. The peptide-loaded PBMCs were irradiated (2500 rads), washed once, and resuspended in lymphocyte expansion medium, LyEM [45% RPMI 1640, 45% Click’s media, 2 mmol/L glutamine, 10 mmol/L HEPES, 100 IU/ml penicillin, 100 μg/ml streptomycin, 10% v/v fetal bovine serum (FBS) at 0.5 × 106/ml]. One ml was added to each well of a 24-well plate. PBMCs were resuspended in LyEM at 2 × 106/ml and 1 ml added to each well to achieve a responder:stimulator ratio of 4:1. The CTL lines were restimulated on day 7 with irradiated peptide-loaded PBMCs at the same responder:stimulator ratio. Peptide-specific CTL lines were harvested on day 14 and tested in chromium-release assay.
Chromium-Release Assay
Phytohemaglutinin (PHA) blasts for chromium-release assays were generated by culturing PBMCs at a concentration of 2 × 106/ml in the presence of 5 μg/ml of PHA (Sigma) in RPMI-FBS (RPMI 1640, 2 mmol/L l-glutamine, 10 mmol/L HEPES, 100 IU/ml penicillin, 100 μg/ml streptomycin, 10% v/v FBS) for 3 days. PHA blasts were washed four times and then cultured for at least 4 more days in RPMI-FBS containing 100 U/ml interleukin-2 before used in chromium-release assays. Autologous PHA blasts were incubated with 51CrO4 for 90 minutes then washed four times. Labeled cells (1 × 105/ml, 50 μl/well) were added to 96 V-bottom plates containing 50 μl/well of medium with or without 40 μg/ml of peptide and incubated for 2 hours at 37°C. Peptide-specific CTLs were added to each well (1.5 × 106/ml, 100 μl/well; E:T ratio, 30:1) for the subsequent 5-hour incubation. The concentration of peptides in the final assay volume (200 μl) was 10 μg/ml. Medium for CTL assays was RPMI-FBS. Thirty μl of culture supernatant was harvested and measured for 51CrO4 level by using a γ counter (TopCount NXT; Packard Instrument Company, Meriden, CT).
Results
EBV Subtypes in PTLD
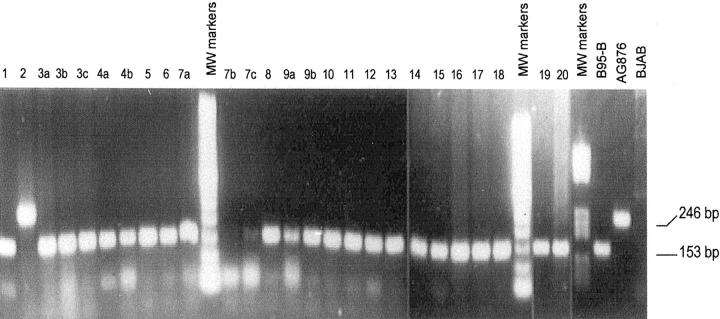
Among 25 PTLD tumor specimens from 19 patients, PCR typing of the EBNA3C open reading frame (ORF) showed that 24 specimens from 18 patients harbored type A virus as indicated by the presence of a 153-bp amplification product, whereas only one case (case 2) harbored type B virus as indicated by the presence of a 246-bp product (Figure 1) ▶ . In the four patients with tumor specimens obtained at different times, the specimens consistently showed the same viral type.
Figure 1.
PCR typing for EBV subtypes. A 153-bp band for type A EBV and a 246-bp band for type B virus are shown. Only one PTLD tumor (case 2) is associated with type B EBV infection.
Sequence Conservation of EBV CTL Epitopes
We then performed targeted sequencing to determine whether there was variation in commonly targeted CTL epitopes. Two regions were targeted: part of EBNA3C containing three CTL epitopes (A2.1-, B44-, and B27-restricted) and a part of LMP-2 containing two epitopes (A2.1- and A24.2-restricted). These regions were amplified and directly sequenced in 24 PTLD specimens from 18 patients including one patient with type B EBV. Two hot spots of variation were identified in EBNA3C in patients with type A virus. A missense G to A mutation (RRIYDLIEL to RKIYDLIEL) in the HLA B27-restricted epitope (residues 258 to 266) was detected in 12 specimens (Figures 2 and 3A) ▶ ▶ . A silent C to T mutation in a region of overlapping HLA B44- and A2.1-restricted epitope was present in 14 specimens. A missense T to A mutation (Leu to Met) occurring outside any recognized CTL epitopes was detected in the two type A and the type B specimens. Four other variations that are type B-specific were also recognized, including two A to T changes (Tyr to Phe) with one in the HLA B27-restricted epitope, and two other mutations occurring outside recognized CTL epitopes (A to G, C to G) were also detected (Figures 2 and 3A) ▶ ▶ .
Figure 2.
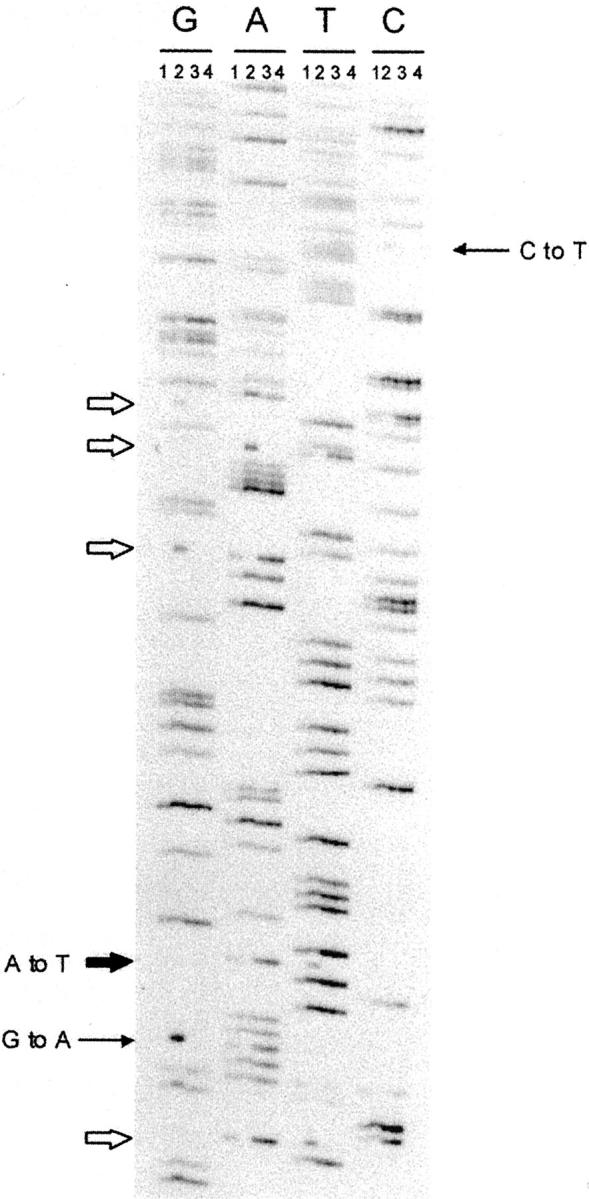
Sequencing for EBNA3C CTL epitopes. Lane 1, case 8; lane 2, case 2, type B EBV; lane 3, case 3a; lane 4, case 3b. The variations between type A and B EBV are shown by thick arrows including one missense mutation (A to T) within a CTL epitope (filled thick arrow). Two mutations (one silent C to T, one missense G to A) among type A EBV isolates are shown by thin arrows.
Figure 3.

Direct PCR sequencing analyses of EBV CTL epitopes in PTLD. A: The HLA A2.1-restricted EBNA3C epitope (underlined) is overlapped with the HLA B44 epitope (italics). B: The HLA A2.1-restricted LMP2A epitope (underlined) is overlapped with the HLA A24.2 epitope (italics). Tumor samples 3a, 3b, 3c, 4a, 4b, 7a, 7b, 7c, 9a, or 9b are from a single patient at different times.
Sequencing of the LMP2A region showed one missense G to C mutation (Cys to Ser) and a silent mutation (T to C) detected in the B-cell lymphoproliferative disease specimen from the patient with aplastic anemia. This silent mutation was also detected in two PTLD tumors from a single patient (case 4). Two missense mutations (both G to A) occurring outside CTL epitopes were also detected in four other PTLD specimens, including three tumors from a single patient (case 7) (Figure 3B) ▶ .
By direct PCR sequencing, the four patients (cases 3, 4, 7, and 9) with tumor specimens obtained at different times showed no intrapatient sequence variation in either the EBNA3C or LMP2A regions sequenced. Because direct sequencing might miss some small percentage of variants, we also performed sequenced cloned PCR products to check whether there was any evidence of intrapatient variation throughout time. Sequencing of the cloned products confirmed the results of direct sequencing, but identified additional variations in 1 of 10 or 2 of 10 plasmid clones (Figure 4) ▶ . These variations are likely to represent artifacts of Taq amplification. In any case, only the two variations detected in case 4a would affect the epitope sequence.
Figure 4.
Cloning sequencing for EBV CTL epitopes in PTLD patients (cases 3, 4, 7, and 9) with multiple tumors at different times. A: The EBNA3C epitopes. B: The LMP2A epitope. The bolded variations were only detected by cloning sequencing, but the possibility that these changes represent PCR artifacts cannot be excluded.
CTL Recognition to the EBNA3C Epitope Variant
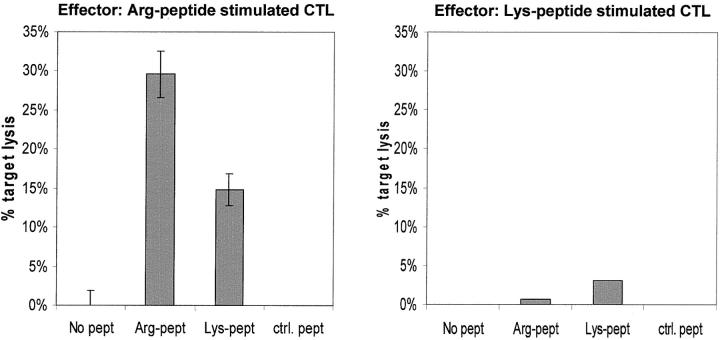
The missense mutation in EBNA3C resulting in an Arg to Lys substitution has been reported in 40% of virus isolates from healthy Australian Caucasians, but not in any isolates from Africa, Southeast Asia, or Papua New Guinea. 29 To determine whether the presence of this variant would affect CTL-mediated killing, we stimulated PBMCs from a healthy HLA-B27 EBV-seropositive donor with peptide corresponding to the B27 epitope. CTLs were harvested on day 14 and lytic specificity assessed by chromium-release assay. CTLs generated in response to stimulation with the Arg peptide lysed Arg peptide loaded PHA blasts and variant Lys peptide loaded blasts, although somewhat less efficiently (Figure 5) ▶ . The killing was peptide-specific as neither PHA blasts alone nor PHA blasts loaded with an irrelevant control peptide were killed. In the complementary experiment, CTLs generated by stimulation with the variant Lys peptide were not able to kill any of the targets (Figure 5) ▶ .
Figure 5.
CTL killing of B95.8-derived peptide (Arg peptide) (left) and Lys-variant peptide (right). CTL clones were raised against these peptide-loaded autologous PBMCs, and tested for CTL recognition to peptide-loaded autologous PHA blasts labeled with 51CrO4. Arg peptide, Lys peptide, and ctrl peptide are autologous PHA blasts loaded with different peptides.
Discussion
The investigations reported here show that type A virus predominates in PTLD lesions and that the general specificity of T cells expanded in response to B95.8 cells is appropriate for the great majority of these tumors. Early studies had suggested that type B virus might be much more common in immunocompromised patients. 30,31 However, it now seems that the relative increase of type B virus in HIV patients with lymphoma reflects the prevalence of type B virus in the gay male community rather than an effect of immunocompromise. 32 With regard to PTLD lesions, ours is the third series to demonstrate an overwhelming preponderance of type A virus. 33,34 A similar predominance of type A virus has been reported in Hodgkin’s disease, nasopharyngeal carcinoma, and healthy Caucasians in England. 35-38
There remains the possibility that although laboratory strain virus type and PTLD virus type (A versus B) generally match, more subtle variations in the genome might limit the effectiveness of T cells expanded ex vivo in response to laboratory strain virus. EBV strain variations have been reported from populations in Borneo where HLA A11 predominates such that the A11-restricted epitope of EBNA-3B is absent. 39 Genetic instability in tumors might make the occurrence of such strain variants more likely through mutation. Our sequence analysis of EBV CTL epitopes in EBNA3C and LMP2A showed that with the exception of the Arg to Lys variant in the HLA B27-restricted EBNA3C epitope in 12 of 24 tumors, EBNA3C epitopes and LMP2A epitopes were well conserved in PTLD tumors. Furthermore, in the serial tumor specimens from a single PTLD patient there was no evidence of ongoing mutation of viral CTL epitopes, as analyzed by direct PCR sequencing. The results of cloning sequencing for these serial tumors were inconclusive. In addition, our approach would have missed viral genome deletions of the sort recently been reported. 40
The Arg to Lys mutation (Lys variant) of the HLA B27 epitope of EBNA3C has been reported in 40% of virus isolates from healthy Australian Caucasians. 29 We detected this variation in 50% of specimens from American patients. The impact of this epitope sequence variation difference on CTL recognition was further tested. Killing of the Lys variant peptide-loaded target cells was observed, although at a lower efficiency than the Arg form. The lack of killing in the Lys peptide-stimulated CTL lines from the same donor may reflect poor immunogenicity of Lys variant virus. Responses to the Arg and Lys variants have recently been analyzed in detail by Brooks and colleagues 41 who found responses to more than one strain in some healthy individuals and inferred infection by co-resident viral strains. They reported that both Arg and Lys variants could be recognized by Arg-specific CTL clones although the Lys variant required a 10-fold higher target peptide concentration to achieve 50% maximum lysis. The only substitution of amino acid found in the HLA A2.1-restricted LMP2A epitope in the B-cell lymphoproliferative disease has previously been reported not to interfere with CTL recognition and killing. 20
Immunotherapeutic approaches to the prevention or treatment of neoplasia are promising alternatives to conventional cytotoxic therapy with drugs or radiation. Graft-versus-leukemia effects have proven to be very potent in the treatment of some leukemias. 42-44 However, in general the cell populations that are associated with anti-tumor effects have been difficult to differentiate from those associated with graft-versus-host disease. The use of EBV-specific CTLs expanded ex vivo is a striking exception to this generalization. EBV CTLs have been proven effective at prophylaxis in the allogeneic marrow transplant setting and have not been associated with graft-versus-host disease. 11 These dramatic successes have kindled interest in the more general application of EBV CTL therapy to PTLD occurring in the recipients of solid organ transplants. Although we fully expect that exceptions will be noted with time, this first detailed investigation of epitopes in PTLD lesions suggests that CTL lines expanded in response to B95.8-transformed B-cell lines will generally have appropriate specificities for treatment of solid organ transplant patients in the United States.
Footnotes
Address reprint requests to R. F. Ambinder, M.D., Ph.D., Viral Oncology Program, Cancer Research Building, Rm 389, 1650 Orleans St., Baltimore, MD 21231-1000. E-mail: ambinri@jhmi.edu.
Supported by PO1 CA15396 and Johns Hopkins Singapore.
References
- 1.Knowles DM, Cesarman E, Chadburn A, Frizzera G, Chen J, Rose EA, Michler RE: Correlative morphologic and molecular genetic analysis demonstrates three distinct categories of posttransplantation lymphoproliferative disorders. Blood 1995, 85:552-565 [PubMed] [Google Scholar]
- 2.Frizzera G, Hanto DW, Gajl-Peczalska KJ, Rosai J, McKenna RW, Sibley RK, Holahan KP, Lindquist LL: Polymorphic diffuse B-cell hyperplasias and lymphomas in renal transplant recipients. Cancer Res 1981, 41:4262-4279 [PubMed] [Google Scholar]
- 3.Nalesnik MA: Posttransplantation lymphoproliferative disorders (PTLD): current perspectives. Semin Thorac Cardiovasc Surg 1996, 8:139-148 [PubMed] [Google Scholar]
- 4.Chadburn A, Cesarman E, Knowles DM: Molecular pathology of posttransplantation lymphoproliferative disorders. Semin Diagn Pathol 1997, 14:15-26 [PubMed] [Google Scholar]
- 5.Hanto DW, Gajl-Peczalska J, Frizzera G, Arthur DC, Balfour HH, McClain K, Simmons RL, Najarian JS: Epstein-Barr virus (EBV) induced polyclonal and monoclonal B-cell lymphoproliferative diseases occurring after renal transplantation. Ann Surg 1983, 198:356-369 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Swinnen LJ, Costanzo-Nordin MR, Fisher SG, O’Sullivan EJ, Johnson MR, Heroux AL, Dizikes GJ, Pifarre R, Fisher RI: Increased incidence of lymphoproliferative disorder after immunosuppression with the monoclonal antibody OKT3 in cardiac-transplant recipients. N Engl J Med 1990, 323:1723-1728 [DOI] [PubMed] [Google Scholar]
- 7.Craig FE, Gulley ML, Banks PM: Posttransplantation lymphoproliferative disorders. Am J Clin Pathol 1993, 99:265-276 [DOI] [PubMed] [Google Scholar]
- 8.Starzl TE, Porter KA, Iwatsuki S, Rosenthal JT, Shaw BW, Jr, Atchison RW, Nalesnik MA, Ho M, Griffith BP, Hakala TR, Hardesty RL, Jaffe R, Bahnson HT: Reversibility of lymphomas and lymphoproliferative lesions developing under cyclosporin-steroid therapy. Lancet 1984, 1:583-587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.O’Reilly RJ, Small TN, Papadopoulos E, Lucas K, Lacerda J, Koulova L: Biology and adoptive cell therapy of Epstein-Barr virus-associated lymphoproliferative disorders in recipients of marrow allografts. Immunol Rev 1997, 157:195-216 [DOI] [PubMed] [Google Scholar]
- 10.Khanna R, Bell S, Sherritt M, Galbraith A, Burrows SR, Rafter L, Clarke B, Slaughter R, Falk MC, Douglass J, Williams T, Elliott SL, Moss DJ: Activation and adoptive transfer of Epstein-Barr virus-specific cytotoxic T cells in solid organ transplant patients with posttransplant lymphoproliferative disease. Proc Natl Acad Sci USA 1999, 96:10391-10396 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rooney CM, Smith CA, Ng CY, Loftin SK, Sixbey JW, Gan Y, Srivastava DK, Bowman LC, Krance RA, Brenner MK, Heslop HE: Infusion of cytotoxic T cells for the prevention and treatment of Epstein-Barr virus-induced lymphoma in allogeneic transplant recipients. Blood 1998, 92:1549-1555 [PubMed] [Google Scholar]
- 12.Miller G, Lipman M: Release of infectious Epstein-Barr virus by transformed marmoset leukocytes. Proc Natl Acad Sci USA 1973, 70:190-194 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Khanna R, Burrows SR, Kurilla MG, Jacob CA, Misko IS, Sculley TB, Kieff E, Moss DJ: Localization of Epstein-Barr virus cytotoxic T cell epitopes using recombinant vaccinia: implications for vaccine development. J Exp Med 1992, 176:169-176 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Murray RJ, Kurilla MG, Brooks JM, Thomas WA, Rowe M, Kieff E, Rickinson AB: Identification of target antigens for the human cytotoxic T cell response to Epstein-Barr virus (EBV): implications for the immune control of EBV-positive malignancies. J Exp Med 1992, 176:157-168 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dambaugh T, Hennessy K, Chamnankit L, Kieff E: U2 region of Epstein-Barr virus DNA may encode Epstein-Barr nuclear antigen 2. Proc Natl Acad Sci USA 1984, 81:7632-7636 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zimber U, Aldinger HK, Lenoir GM, Vuillaume M, Knebel-Doeberitz MV, Laux G, Desgranges C, Wittmann P, Freese UK, Schneider U, Freese U-K, Bornkamm GW: Geographical prevalence of two types of Epstein-Barr virus. Virology 1986, 154:56-66 [DOI] [PubMed] [Google Scholar]
- 17.Sample J, Young L, Martin B, Chatman T, Kieff E, Rickinson AB: Epstein-Barr virus types 1 and 2 differ in their EBNA-3A, EBNA-3B, and EBNA-3C genes. J Virol 1990, 64:4084-4092 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Moss DJ, Misko IS, Burrows SR, Burman K, McCarthy R, Sculley TB: Cytotoxic T-cell clones discriminate between A- and B-type Epstein-Barr virus transformants. Nature 1988, 331:719-721 [DOI] [PubMed] [Google Scholar]
- 19.Murray RJ, Young LS, Calender A, Gregory CD, Rowe M, Lenoir GM, Rickinson AB: Different patterns of Epstein-Barr virus gene expression and of cytotoxic T-cell recognition in B-cell lines infected with transforming (B95.8) or nontransforming (P3HR1) virus strains. J Virol 1988, 62:894-901 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lee SP, Tierney RJ, Thomas WA, Brooks JM, Rickinson AB: Conserved CTL epitopes within EBV latent membrane protein 2: a potential target for CTL-based tumor therapy. J Immunol 1997, 158:3325-3334 [PubMed] [Google Scholar]
- 21.Busson P, Edwards RH, Tursz T, Raab-Traub N: Sequence polymorphism in the Epstein-Barr virus latent membrane protein (LMP)-2 gene. J Gen Virol 1995, 76:139-145 [DOI] [PubMed] [Google Scholar]
- 22.Murray PG, Swinnen LJ, Constandinou CM, Pyle JM, Carr TJ, Hardwick JM, Ambinder RF: BCL-2 but not its Epstein-Barr virus-encoded homologue, BHRF1, is commonly expressed in posttransplantation lymphoproliferative disorders. Blood 1996, 87:706-711 [PubMed] [Google Scholar]
- 23.Tao Q, Robertson KD, Manns A, Hildesheim A, Ambinder RF: Epstein-Barr virus (EBV) in endemic Burkitt’s lymphoma: molecular analysis of primary tumor tissue. Blood 1998, 91:1373-1381 [PubMed] [Google Scholar]
- 24.Tao Q, Swinnen LJ, Yang J, Srivastava G, Robertson KD, Ambinder RF: Methylation status of the Epstein-Barr virus major latent promoter C in iatrogenic B cell lymphoproliferative disease. Application of PCR-based analysis. Am J Pathol 1999, 155:619-625 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Robertson KD, Manns A, Swinnen LJ, Zong JC, Gulley ML, Ambinder RF: CpG methylation of the major Epstein-Barr virus latency promoter in Burkitt’s lymphoma and Hodgkin’s disease. Blood 1996, 88:3129-3136 [PubMed] [Google Scholar]
- 26.Brooks JM, Murray RJ, Thomas WA, Kurilla MG, Rickinson AB: Different HLA-B27 subtypes present the same immunodominant Epstein-Barr virus peptide. J Exp Med 1993, 178:879-887 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Burrows SR, Sculley TB, Misko IS, Schmidt C, Moss DJ: An Epstein-Barr virus-specific cytotoxic T cell epitope in EBV nuclear antigen 3 (EBNA 3). J Exp Med 1990, 171:345-349 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lee SP, Thomas WA, Murray RJ, Khanim F, Kaur S, Young LS, Rowe M, Kurilla M, Rickinson AB: HLA A2.1-restricted cytotoxic T cells recognizing a range of Epstein-Barr virus isolates through a defined epitope in latent membrane protein LMP2. J Virol 1993, 67:7428-7435 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Khanna R, Slade RW, Poulsen L, Moss DJ, Burrows SR, Nicholls J, Burrows JM: Evolutionary dynamics of genetic variation in Epstein-Barr virus isolates of diverse geographical origins: evidence for immune pressure-independent genetic drift. J Virol 1997, 71:8340-8346 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Boyle MJ, Sculley TB, Penny R, Sewell WA, Tschuchnigg M, Berger MF, Cooper DA: The role of Epstein-Barr virus subtypes in human immunodeficiency virus-associated lymphoma. Leuk Lymphoma 1993, 10:17-23 [DOI] [PubMed] [Google Scholar]
- 31.Kyaw-Tanner MT, Esmore D, Burrows SR, Benson EM, Sculley TB: Epstein-Barr virus-specific cytotoxic T cell response in cardiac transplant recipients. Transplantation 1994, 57:1611-1617 [PubMed] [Google Scholar]
- 32.Yao QY, Croom-Carter DS, Tierney RJ, Habeshaw G, Wilde JT, Hill FG, Conlon C, Rickinson AB: Epidemiology of infection with Epstein-Barr virus types 1 and 2: lessons from the study of a T-cell-immunocompromised hemophilic cohort. J Virol 1998, 72:4352-4363 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Frank D, Cesarman E, Liu YF, Michler RE, Knowles DM: Posttransplantation lymphoproliferative disorders frequently contain type A and not type B Epstein-Barr virus. Blood 1995, 85:1396-1403 [PubMed] [Google Scholar]
- 34.Smir BN, Hauke RJ, Bierman PJ, Gross TG, d’Amore F, Anderson JR, Greiner TC: Molecular epidemiology of deletions and mutations of the latent membrane protein 1 oncogene of the Epstein-Barr virus in posttransplant lymphoproliferative disorders. Lab Invest 1996, 75:575-588 [PubMed] [Google Scholar]
- 35.Gledhill S, Gallagher A, Jones DB, Krajewski AS, Alexander FE, Klee E, Wright DH, O’Brien C, Onions DE, Jarrett RF: Viral involvement in Hodgkin’s disease: detection of clonal type A Epstein-Barr virus genomes in tumour samples. Br J Cancer 1991, 64:227-232 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ambinder RF, Browning PJ, Lorenzana I, Leventhal BG, Cosenza H, Mann RB, MacMahon EME, Medina R, Cardona V, Grufferman S, Olshan A, Levin A, Petersen EA, Blattner W, Levine PH: Epstein-Barr virus and childhood Hodgkin’s disease in Honduras and the United States. Blood 1993, 81:462-467 [PubMed] [Google Scholar]
- 37.Jenkins PJ, Farrell PJ: Are particular Epstein-Barr virus strains linked to disease? Semin Cancer Biol 1996, 7:209-215 [DOI] [PubMed] [Google Scholar]
- 38.Yao QY, Rowe M, Martin B, Young LS, Rickinson AB: The EBV carrier state: dominance of a single growth transforming isolate in the blood and in the oropharynx of healthy virus carriers. J Gen Virol 1991, 72:1579-1590 [DOI] [PubMed] [Google Scholar]
- 39.Campos-Lima PO, Levitsky V, Brooks J, Lee SP, Hu LF, Rickinson AB, Masucci MG: T cell responses and virus evolution: loss of HLA A11-restricted CTL epitopes in Epstein-Barr virus isolates from highly A11-positive populations by selective mutation of anchor residues. J Exp Med 1994, 179:1297-1305 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Gottschalk S, Ng CY, Perez M, Smith CA, Sample C, Brenner MK, Heslop HE, Rooney CM: An Epstein-Barr virus deletion mutant associated with fatal lymphoproliferative disease unresponsive to therapy with virus-specific CTLs. Blood 2001, 97:835-843 [DOI] [PubMed] [Google Scholar]
- 41.Brooks JM, Croom-Carter DS, Leese AM, Tierney RJ, Habeshaw G, Rickinson AB: Cytotoxic T-lymphocyte responses to a polymorphic Epstein-Barr virus epitope identify healthy carriers with coresident viral strains. J Virol 2000, 74:1801-1809 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kolb HJ, Schattenberg A, Goldman JM, Hertenstein B, Jacobsen N, Arcese W, Ljungman P, Ferrant A, Verdonck L, Niederwieser D: Graft-versus-leukemia effect of donor lymphocyte transfusions in marrow grafted patients. European Group for Blood and Marrow Transplantation Working Party Chronic Leukemia. Blood 1995, 86:2041-2050 [PubMed] [Google Scholar]
- 43.Mackinnon S, Papadopoulos EB, Carabasi MH, Reich L, Collins NH, Boulad F, Castro-Malaspina H, Childs BH, Gillio AP, Kernan NA: Adoptive immunotherapy evaluating escalating doses of donor leukocytes for relapse of chronic myeloid leukemia after bone marrow transplantation: separation of graft-versus-leukemia responses from graft-versus-host disease. Blood 1995, 86:1261-1268 [PubMed] [Google Scholar]
- 44.Slavin S, Nagler A, Naparstek E, Kapelushnik Y, Aker M, Cividalli G, Varadi G, Kirschbaum M, Ackerstein A, Samuel S, Amar A, Brautbar C, Ben-Tal O, Eldor A, Or R: Nonmyeloablative stem cell transplantation and cell therapy as an alternative to conventional bone marrow transplantation with lethal cytoreduction for the treatment of malignant and nonmalignant hematologic diseases. Blood 1998, 91:756-763 [PubMed] [Google Scholar]