Abstract
Background
Human muscular dystrophies are a heterogeneous group of genetic disorders which cause decreased muscle strength and often result in premature death. There is no known cure for muscular dystrophy, nor have all causative genes been identified. Recent work in the small vertebrate zebrafish Danio rerio suggests that mutation or misregulation of zebrafish dystrophy orthologs can also cause muscular degeneration phenotypes in fish. To aid in the identification of new causative genes, this study identifies and maps zebrafish orthologs for all known human muscular dystrophy genes.
Results
Zebrafish sequence databases were queried for transcripts orthologous to human dystrophy-causing genes, identifying transcripts for 28 out of 29 genes of interest. In addition, the genomic locations of all 29 genes have been found, allowing rapid candidate gene discovery during genetic mapping of zebrafish dystrophy mutants. 19 genes show conservation of syntenic relationships with humans and at least two genes appear to be duplicated in zebrafish. Significant sequence coverage on one or more BAC clone(s) was also identified for 24 of the genes to provide better local sequence information and easy updating of genomic locations as the zebrafish genome assembly continues to evolve.
Conclusion
This resource supports zebrafish as a dystrophy model, suggesting maintenance of all known dystrophy-associated genes in the zebrafish genome. Coupled with the ability to conduct genetic screens and small molecule screens, zebrafish are thus an attractive model organism for isolating new dystrophy-causing genes/pathways and for use in high-throughput therapeutic discovery.
Background
Muscular dystrophies are a heterogeneous group of genetic disorders characterized by loss of muscle strength and integrity. Common pathological hallmarks of the mammalian muscular dystrophies include the presence of necrotic muscle fibers, fiber size variation, centralized nuclei indicating fiber regeneration, inflammatory infiltrates, and replacement of muscle fibers by fat and connective tissue to varying degrees. However, muscular dystrophies differ in their age of onset, severity, the muscle groups affected, additional non-muscle phenotypes (such as reduced average IQ) and the genetic mode of inheritance (reviewed in [1]).
To date, 31 distinct muscular dystrophies have been described and 25 distinct genes have been causatively linked to these muscular dystophies [2]. The most common, Duchenne Muscular Dystrophy (DMD), accounts for the majority of dystrophy patients. DMD affects 1 in 3500 males and typically results in death by the third or fourth decade. The mutated gene in DMD, dystrophin, was identified in the 1980's [3] and its characterization has led to methods of genetic testing and a better understanding of dystrophic pathology. However, no cure has yet been identified. In addition, the causative genes remain unknown in several dystrophies and additional patients with unclassified dystrophy phenotypes. Finally, while several dystrophy-associated genes encode proteins that directly or indirectly interact, others, including the nuclear proteins (lamin A/C and emerin), and cytoplasmic proteins (TRIM32) have not yet been linked in a common pathway that would make apparent the cause of their dystrophic phenotype.
The small freshwater zebrafish, Danio rerio, has recently emerged as a promising model organism for the study of muscular dystrophies and other human diseases. Due to its small size, large numbers of offspring (50–350 per week), rapid development of the skeletal musculature, and transparency in embryonic/juvenile stages, zebrafish provide an excellent system for genetic screens to identify novel muscular dystrophy-causing genes and pathways. More recent experiments have also proven zebrafish a useful organism for drug screens using whole vertebrates, suggesting that identification of dystrophic zebrafish mutants may allow drug screens for muscular dystrophy therapeutics. (See [4-8] for zebrafish drug screens.)
The zebrafish sapje mutant was identified in 1996 with grossly normal muscle at 2 days post-fertilization (dpf) but decreased muscle organization and motility at 5 dpf [9]. The causative gene mutation was mapped to a nonsense mutation in dystrophin, suggesting conservation of this dystrophy-associated molecular pathway in fish [10]. Other studies have employed anti-sense morpholino or RNAi knockdowns to show similar dystrophy-like pathology and phenotypes when dystrophin (DMD/BMD), caveolin-3 (LGMD 1C), δ-sarcoglycan (LGMD 2F), or laminin α2 (MDC 1A) proteins are reduced, suggesting that this conservation may extend to other orthologs of human dystrophy-associated genes [10-16].
To aid in the further identification of zebrafish dystrophy mutants, we have interrogated current sequence databases to identify zebrafish orthologs of the known human dystrophy genes. Positioning these genes allows rapid candidate identification during genetic mapping of dystrophic zebrafish mutants and may allow prioritization of novel mutants – those with linkage to a genomic region containing no known dystrophy-associated ortholog. Due to the evolving nature of the Sanger Centre Zebrafish Genome assembly, we have also identified the BAC clone location of these genes. BAC sequences should allow more consistent local sequence information and easy updating when future genome alignments are released. This study has identified orthologous zebrafish transcripts for 24 out of 25 of the known human dystrophy-associated genes and 4 additional myopathy-related genes. Genomic positions have been identified for all 29 of these genes and BAC locations for 24. This genomic data suggests that at least two dystrophy genes are duplicated in the zebrafish genome. Localization of the closest mammalian gene neighbors also shows that syntenic relationships are conserved for 19 dystrophy- and myopathy-causing genes.
Results and discussion
Mutations in 25 genes have been linked to 27 distinct forms of human muscular dystrophy (MD). In humans, these genes are distributed across 17 of the 23 chromosomes. Protein products of these genes position throughout the muscle fiber – from the extracellular matrix and sarcolemmal membrane to the sarcomere, the golgi, the cytoplasm, and the nucleus.
We surveyed the zebrafish GenBank database to identify putative orthologs of the 25 human muscular dystrophy-associated genes and four additional myopathy-associated genes. Results with a high degree of similarity and significant sequence coverage were confirmed by reciprocal blast into Genbank mammalian databases. This in silico approach identified orthologous transcripts for 23 out of 25 muscular dystrophy-associated genes and all 4 myopathy-associated genes within the Genbank database (Tables 1 and 2).
Table 1.
Zebrafish orthologs of human muscular dystrophy genes and their genomic locations.
| Fish Genomic Location | Fish BAC Loc | |||||||||||
| Gene | Symbol | Associated Disease | Fish EST | Notes | Scaffold location | Clone Location | Gene location | Notes | BAC Name | Notes | Synteny | Notes |
| Calpain-3 | CAPN3 | LGMD2A | NM_001004571 | 2601 | NA-BX322589 | Chr17 62.6 Mb and 63.1 Mb | By blasts and synteny, appears split,. The first ~ 1550 nt has no corresponding BAC. | N/A – zC283F6 | Human CAPN3 matches well with zK12H9 | Yes | Syntenic with GANC on genome and ZFP106 on genome and BAC (zC283F6) | |
| Caveolin 3 | CAV3 | LGMD1C, hyperCKemia, Rippling muscle disease | NM_205738 | 879 | BX664752 | Chr6 33.2 Mb | Organized in 2 exons, similar to human CAV3 | zKp111E5 | On one side | Syntenic with OXTR on BAC and genome. | ||
| Dystrophin | DMD | Duchenne MD, Becker MD | XM_678461, XM_678362, XM_678552, partial | All three are partial transcripts, but in order, cover most of the DMD coding region | 42 | BX004756, CT033808 | Chr1 9.6 Mb-9.4 Mb | Duplications within gene likely incorrect. Additional partial sequence located on Chr1 scaffold 49 at 10.5 Mb | zC59A4, zC274B7 | Transcripts span these two overlapping BACs | No | |
| Dysferlin | DYSF | LGMD2B, Miyoshi Myopathy, Distal myopathy with anterior tibial onset | XM_684324 | Many transcripts are similar to dysferlin, but this is the only one that aligns closer to dysferlin (rather than myoferlin or otoferlin) on reciprocal blast | 1155 | CR847843 | Chr7 83.3 Mb-83.5 Mb | Human dysferlin also identifies Chr12 (BC063743, likely fish myoferlin) and Chr13 (XM_682373, similar to myoferlin, dysferlin, and otoferlin) | zKp78E10 – bZ50C18 | The first BAC contains the 5' ~ 1/7th while the second BAC overlaps and gives coverage to the transcript end | No | Flanking genes are on Chr7 but not in the same region or on the same scaffold as dysferlin |
| Emerin | EMD | Emery-Dreifuss MD | XM_685843, XM_549369 | Identical except for a single 7nt internal fragment. Likely alternatively spliced variants. Poor homology to mammalian emerin. | 3352 | No data | Chr23 18.96 MB | Duplication of last 4/5 of transcript on Chr7 39.54 Mb, scaffold 1085. No synteny with Chr7. | zC133L21 | Identical matches on unfinished BACs zK233H12, zK181F15, and zK93L1. | Yes | RPL10 and FLNA are at 19.1 Mb and 19.0 Mb, on Chr23 Both are syntenic on BACs. |
| Fukutin related protein | FKRP | MDC1C, LGMD2I | XM_695011, partial | 2206 | No data | Chr15 26.6 Mb | zK31C13 | Yes | STRN4 and SLC1A5 are syntenic on the genome and the BAC. | |||
| Lamin A/C | LMNA | Emery-Dreifuss MD, LGMD1B, CMD1A, etc. | NM_152971 | 2371 | CR848742 | Chr16 37.8 Mb | zK181C1 | No | Flanking genes are not syntenic with each other, either | |||
| Myotilin | TTID | LGMD1A, myofibrillar myopathy | None found | Closest match is Zv5 predicted transcript ENSDARG015348 | 1999 | CT573287 | Chr14 12.2 Mb & 10.8 Mb | ENSDARG015348 split between two loci in Zv6. Genome incorrect. | zK101K8 | Complete and contiguous BAC coverage | No | Flanking genes are not syntenic with each other, either |
| Sarcoglycan alpha | SGCA | LGMD2D | XM_680178 | Close homology with SCGE | 1664 | BX548040 | Chr12 870 Kb | zC190L11 | On one side | Syntenic with Col1A1 on both genome and BAC | ||
| Sarcoglycan beta | SGCB | LGMD2E | NM_001034973 | First half of transcript aligns with full length of human SGCB | 2974 | CT583700 | Chr20 59.8 Mb | Second half of EST is located on Chr25 1.3 Mb (Scaffold 3566) | zC253J24 | Yes | Both genes syntenic on genome and BAC | |
| Sarcoglycan delta | SGCD | LGMD2F, CMD1L | NM_001001816 | 3106 | BX294656 | Chr21 39.0–38.7 Mb | zC238M13 | First 300nt located on zK189O20 | On one side | Syntenic with MRPL22 | ||
| Sarcoglycan gamma | SGCG | LGMD2C | NM_001003748 | 2184 | BX927291 | Chr15 20.2 Mb | Incomplete coverage. Duplicate exon also on scaffold 2184 but not on BAC. | zC261A10 | Complete coverage | On one side | Syntenic with SACS on both genome and BAC | |
| Telethonin | TCAP | LGMD2G, Dilated cardio-myopathy (CMD1N) | XM_679011 | 371 | CR387996 | Chr3 19.9 Mb | Human TCAP also identifies a locus on Chr16 scaffold 2377 but coverage is less complete and exons are not contiguous | zK183N6 | No | |||
| Tripartite Motif-containing protein 32 | TRIM32 | LGMD2H | XM_686142 | Human TRIM32 only has one coding exon. | NA688 | No data | No data | Coding sequence on scaffold NA688. Putative 5' UTR exons are located in duplicate on Chr8, scaffold 1244 | None | Putative 5' UTR exons are located on zK72L14 & zK65O14 | Yes | ASTN2 spans the both the human and the zebrafish TRIM32 loci on Scaffold NA688 |
| Titin | TTN | LGMD2J, Tibial MD, Hereditary Myopathy with early respiratory failure | XM_679005 (TTN2, partial), XM_678144 (TTN1, partial) | Locus is duplicated. Only partial transcripts available | 3186 | BX640499, BX571737, BX640465 | Chr9 41.8 Mb-42.2 Mb | Locus duplications are in tandem. Duplicate genes are divergent in sequence and likely to be true duplicates. | zKp67D2, dZ258D18, zK190I10, dZ249N21, zC198B21 | BACs overlap to cover the entire titin locus. | Yes | Syntenic with FLJ39502 and FKBP7 on genome and BAC. |
MD-Muscular Dystrophy, LGMD-Limb Girdle Muscular Dystrophy, CMD-Congenital Muscular Dystrophy, nt-nucleotides.
Table 2.
Zebrafish orthologs of human congenital muscular dystrophy and selected myopathy genes and their genomic locations. Genes associated with both non-congenital and congenital muscular dystrophies are in Table 2-1.
| Fish Genomic Location | Fish BAC Loc | |||||||||||
| Gene | Symbol | Associated Disease | Fish EST | Notes | Scaffold location | Clone Location | Gene location | Notes | BAC Name | Notes | Synteny | Notes |
| Collagen 6A1 | Col6A1 | Bethlem myopathy, Ullrich CMD | XM_693161, partial | 1607 | CR925698 | Chr11 35.3 Mb | zK287I12 | On one side | Syntenic with Col6A2 on genome and BAC. | |||
| Collagen 6A2 | Col6A2 | Bethlem myopathy, Ullrich CMD | XM_691072 | The first 320 bases are likely not part of this transcript. | 1607 | CR925698-BX323597 | Chr11 35.2 Mb | The first 320 bases are located in multiple places on other chromosomes | zK287I12 – zC227N13 | The first 320 bases are located on zC184B9. | On one side | Syntenic with Col6A1 on genome and BAC. |
| Collagen 6A3 | Col6A3 | Bethlem myopathy, Ullrich CMD | XM_679796 | XM_687365 is also orthologous to mammalian Col6A3, but is more similar to a second predicted Col6A3 mammalian locus. | 1361-1360 | No data | Chr9 19.0 Mb and 15.0 Mb | The beginning is located on scaffold 1361, the repeating middle elements are on both scaffolds, and the end is on 1360. Note that the genomic locus may be misorganized. | zC5M6 | Unfinished BAC covers entire transcript on various fragments | Yes | Syntenic with MLPH on Chr9 and with COPS8 on Chr9 and clone zC5M6. |
| Desmin | DES | DCM1, CMD1I, several skeletal and/or cardio-myopathies | NM_130963 | 1342 | No data | Chr9 7.3 Mb | Several loci are orthologous to human desmin. Most ruled out due to closer homology with other proteins. Additional loci on Chr20 (scaffold 2945), and Chr13 (scaffold 1885) could not be ruled out and may be duplications. | None | Homologous sequences were found, but none were near-exact matches to the zebrafish transcript sequence. | No | Chr9 locus is not syntenic with the other desmin-like genes, either. | |
| Fukutin | FCMD | Fukuyama CMD | XM_686729, partial | 792, 793 | CR753888, CT027618, BX072578 | Chr5 78.4–79.0 Mb | Full match on the first two clones, partial match on the third. Likely a genomic misalignment. | zC286A10, zC154E10 | Full coverage of the partial transcript on both | On one side | FSD1CL is syntenic on both genome and BAC. | |
| Filamin C | FLNC | Myofibrillar myopathy | XM_693754, XM_687344, partial | Duplicated. Divergent nucleotide sequences. First contains the Human FLNC unique region. Second transcript is only partial. | 505, 3643 | AL954190, No data | Chr4 7.5 Mb, Chr25 32.9 Mb | Human FLNC unique region is not part of XM_687344, but is located immediately after it on Chr25. | zC284B12, zK3006 | Both BACs match XM_693754. No BACs for XM_687344 | On one side | Chr4 locus not syntenic, though flanking genes are elsewhere on Chr4. Partial NAG6 matches on Chr25. |
| Integrin Alpha 7 | ITGA7 | CMD with integrin deficiency | None found | Closest EST is a closer match to mammalian ITGA6 | 1560 | No data | Chr11 2.5 Mb | Location identified using human ITGA7 only | zC245G15 | Used human ITGA7 | No | Flanking genes are not syntenic with each other, either |
| Acetyl-glucosaminyl-transferase-like protein | LARGE | MDC1D | NM_001004537 | LARGE1B (NM_001004538) is highly orthologous. | 570 | No data | Chr4 39.4 Mb | LARGE1B located on Chr18, scaffold 2725, clone BX908385. | None | LARGE1B located on both zC282N12 & zC206G24 | No | The closest flanking genes are predictions |
| Laminin alpha 2 | LAMA2 | Merosin-deficient CMD | XM_694983 | Partial, predicted | 2875 | No data | Chr20 3.8 Mb | Aligns with LAMA2 predicted transcripts GenScan01065 and FGENESH78171 | None | On one side | Syntenic with ARHGAP18, but NOT the highly similar LAMA1 locus (on Chr24) | |
| Polyadenylate-binding protein, nuclear 1 | PABPN1 | Oculo-pharyngeal MD | BC079522 | NM_213259 also matches but diverges over the 3' non-coding end. NM_213259 3' end is discontinuous with its 5' end on the genome and BACs and may not represent a real transcript. | 3471 | BX294113 and CT583644 | Chr24 21.4 Mb and 21.6 Mb | Duplication on Chr24 clones is likely due to genomic misalignment since clones overlap in the Sanger fingerprinted contigs. | zKp73G8 | No | SLC22A17 is located on Chr24, but not in the same region. | |
| Protein O-Mannose Beta-1,2-N-Acetyl-glucosaminyl-transferase | POMGNT1 | Muscle-eye-brain (MEB) | BC097123 | 985 | No data | Chr6 69.0 Mb | zK170G13, zC156B18 | Sequencing of first BAC is unfinished | On one side | Syntenic with TSPAN1 on both genome and BAC | ||
| Protein-O-mannosyl-transferase 1 | POMT1 | LGMD2K, Walker Warburg syndrome | XM_693177 | 723 | BX511209 and No data | Chr5 56.2 Mb & 56.3 Mb | Split between 3 loci. Exons 1–3 at first location, exons 3–17 at second location. Exons 17–22 potentially on Chr17 at 37.47 Mb. | zC129A6 | Covers only first 3 exons. No matches for other exons. | No | ||
| Sarcoglycan epsilon | SGCE | Myoclonic dystonia | NM_001002594 | Close homology with SCGA | 2827 | BX640469 | Chr19 41.07 Mb | zK104M9 | On one side | Syntenic with CASD1 on both genome and BAC | ||
| Selenoprotein N, 1 | SEPN1 | Rigid spine MD1 (RSMD1), Multiminicore disease | NM_001004294 | 2451 | BX323794 & R626962 | Chr17 1.8 Mb & 2.3 Mb | Duplication likely due to genome misalignment since the BACs overlap. Both clones have full transcript coverage. | zC247C16, zC15D5 | BACs overlap, suggesting that the genomic duplication is a misalignment. | On one side | Syntenic with FAM54B on genome and BAC | |
CMD/DCM-Congenital Muscular Dystrophy, MD-Muscular Dystrophy, nt-nucleotides.
No orthologous transcript sequence was identified in the zebrafish Genbank database for the non-congenital MD gene, myotilin, or the congenital MD (CMD) gene, integrin alpha 7 (ITGA7). However, interrogation of Version 5 of the Sanger Centre Zebrafish Genome with human myotilin protein sequence identified a highly conserved ENSEMBL-predicted zebrafish myotilin transcript. Complete and contiguous sequence for this ENSEMBL-predicted myotilin was also found on a single BAC clone. However, the predicted transcript is no longer contiguous within Version 6 of the genome, suggesting that the current genomic alignment through this region may be incorrect. For integrin alpha 7 (ITGA7), a putative genomic and BAC location was identified by similarity to human ITGA7 over other integrins, though no transcript sequence has yet been identified. These data suggest that zebrafish have gene orthologs for all known human MD genes. In combination with mutant and morpholino data demonstrating zebrafish dystrophy phenotypes upon down-regulation of several MD gene orthologs, these data recommend the zebrafish as an excellent model organism for genetic screens to identify additional vertebrate MD-causing genes and pathogenic pathways.
Genomic positions of zebrafish dystrophy orthologs
Genomic loci of zebrafish orthologs were identified in version 6 of the Sanger Centre Zebrafish Genome using the blastn algorithm with zebrafish RNA sequences. Locations were independently confirmed using the tblastn algorithm with human protein sequences. Human protein sequences were also used in case gene duplications were present but not reported in the EST database. Human sequences often returned several locations, sometimes correlating with related genes within a gene family. Additional loci were ruled out where possible by performing similar analyses with paralogs and/or by synteny with paralogs.
All 29 genes could be placed in whole or in part on Version 6 of the Sanger Centre Zebrafish Genome. TRIM32, responsible for Limb Girdle Muscular Dystrophy 2H (LGMD 2H), resides on an orphan scaffold that has not yet been integrated into the chromosomal organization of the genome. The remaining 28 genes are scattered across 18 chromosomes with the majority of chromosomes having only one dystrophy ortholog (Fig. 1). Only Chr 9 (Collagen 6A3, desmin, and duplicate titin genes) and Chr 11 (ITGA7 and two syntenic collagen genes) contain more than two dystrophy orthologs. It is interesting to note that there is currently no identified sex chromosome in zebrafish. Indeed, dystrophin and emerin, genes that reside on the human X chromosome, are found on different chromosomes in zebrafish, and characterization of the zebrafish dystrophin mutant, sapje, has demonstrated an autosomal recessive inheritance pattern.
Figure 1.
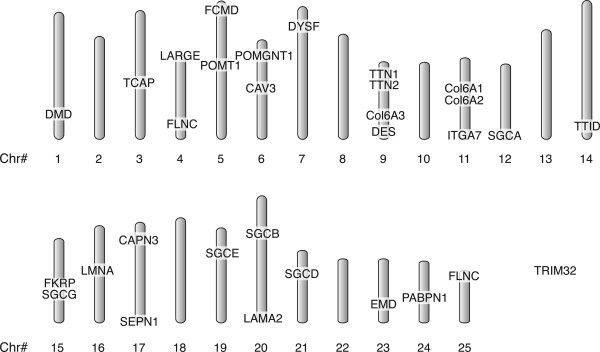
Distribution of zebrafish muscular dystrophy orthologs. Orthologs of the 25 muscular dystrophy-associated genes and 4 additional myopathy-associated genes were identified on 20 of the 25 zebrafish chromosomes by computer searches of the Sanger Centre Zebrafish Genome. Duplicate loci were found for FLNC and TTN orthologs. TRIM32 is located on an orphan scaffold that has not yet been integrated within the genome.
Genomic loci identified in the Sanger Centre Database frequently showed non-contiguous organization of transcript sequences, suggesting that the genome is not yet correctly organized in these regions. Thus, BAC clone locations were identified within the Sanger Centre Zebrafish Clone Database to allow rapid updating of dystrophy ortholog positions as the genome assembly continues to evolve. Clone data was also used where possible to distinguish duplications due to genomic misalignments versus real duplications by determining if the associated clones overlapped. Using both zebrafish nucleotide and human protein sequences, at least partial BAC coverage was identified for 24 out of the 29 genes of interest.
Genome loci verification
Version 6 of the Sanger Centre Zebrafish Genome contains better sequence coverage of the dystrophy-associated genes than the previous version, due in large part to use of physical data to integrate the clone sequence and whole shotgun method sequence (data not shown). Genomic positions could be found for all 29 genes, and 19 of these genes show conservation with humans of syntenic relationships with at least one neighboring gene, including TRIM32.
Though more complete than previous versions, Version 6 of the zebrafish genome is not yet entirely correct, since several transcripts appear split between distant genomic loci or have portions (usually corresponding to exons found singly on a BAC) multiply identified in close proximity on the genome. In particular, genes with repeat-rich or modular elements, like dystrophin and collagen 6A3, may be more difficult to align electronically, resulting in genomic sequences that do not agree with BAC sequence data. However, 24 of the transcripts were found with nearly complete coverage spanning one or more BAC clones which should provide better local sequence coverage until complete clone information has been incorporated into the genome assembly.
To test these data, we compared the in silico identification of genomic loci with those previously identified. To date, only four dystrophy-associated orthologs have been physically localized in the zebrafish genome. By radiation hybrid (RH) mapping using the T51 RH panel, researchers have mapped dystrophin to Chr 1 [13,17] and delta sarcoglycan to Chr 21 [12]. Similarly, caveolin 3 has been localized to Chr 6 [14]. Finally, a BAC walk between two genetic markers on Chr 9 identified and positioned titin in the interval [18]. All four positions agree with the data presented here from Version 6 of the Sanger Centre Zebrafish Genome. In addition, genetic mapping using polymorphic microsatellites within and flanking zebrafish titin has confirmed the duplication of zebrafish titin that was found in silico (data not shown).
To expand the set of genes for which we have physical position information, calpain-3 was located using radiation hybrid analysis with the T51 RH panel. In silico mapping places calpain-3 on Chr 17. However, RH mapping of calpain-3 places the orthologous zebrafish transcript on Chr 22 nearest to marker fa11a04.s1. To reconfirm computer-based findings, an independent analysis was performed and again returned Chr 17 as the most likely calpain-3 locus with synteny to neighboring genes. While BLAST analysis did identify other loci with some similarity to human calpains, none were located on Chr 22.
These data, the appearance of genomic duplications of genes in whole or in part, and the identification of non-contiguous transcripts in the genome suggest that the current Sanger Centre Zebrafish Genome still contains regions of misassembly, especially where continuity and singleness of transcripts is confirmed within the clone database. Nonetheless, locations for a greatly increased number of gene orthologs could be identified in Version 6 of the genome as compared with Version 5, suggesting improvement of the genome assembly over time (data not shown). In combination with the 80% success rate in the 5 genes with physical mapping data, this suggests a strong correlation between rough physical gene location and the current genome assembly.
Gene duplications
Multiple distinct zebrafish transcripts were identified for each of four genes: Filamin C, emerin, dystrophin, and titin. For emerin, the two identified transcripts differ only by a single 7 basepair internal fragment, suggesting differential splicing, or mis-prediction of one of the transcripts. Both transcripts identified the same genomic and BAC locus, further suggesting a single gene locus. In the case of dystrophin, the multiple transcripts all appear to be partial sequences of the large dystrophin mRNA (> 14 kb in humans) [19], and position to Chr 1.
Two putative zebrafish FLNC transcripts were identified in Genbank that position to different genomic loci. Of these, only the FLNC predicted-transcript XM_693754 contains an exon highly orthologous to human exon 47, the conserved FLNC-unique region in mammals. A second FLNC transcript, XM_687344, is only a partial transcript and does not contain this exon. However, comparison of human FLNC exon 47 with the zebrafish genome identified a second locus for this exon immediately following the genomic locus of XM_687344, suggesting that a full transcript sequence would identify this gene as FLNC. Filamin C (FLNC) appears to be a true duplication, with transcripts divergent at the nucleotide and protein levels.
Titin, which codes for an enormous mRNA in humans (> 82 kb) [20], shows multiple transcripts due to its length, as well as an apparent gene duplication event. Duplicated loci were found in head-to-tail juxtaposition with genes divergent at both the nucleotide and protein levels. Due to the large number of titin transcripts, only two transcripts from each gene locus have been listed. Two additional genes, dysferlin and desmin, may also have genomic duplications, identified by multiple zebrafish genomic loci orthologous to the human protein sequences. While many of these loci were ruled out due to closer homology with other human proteins within the gene families, not all additional loci could be eliminated.
Studies of the Hox gene clusters in fish suggest a full genome duplication event in ancestral teleost lineages after the divergence of ray-finned fish (from which zebrafish derive) and lobe-finned fish (from which mammals derive) [21]. Further comparative genomics studies report that at least 20% of gene duplicates have been maintained in zebrafish, often by divergence of regulation between the duplicate loci that imposes an evolutionary constraint on both genes [22]. Of the zebrafish gene orthologs in this study, however, we find that only two genes show strong evidence of duplicate gene maintenance – titin and FLNC – with at most four gene duplications suggested by the genomic sequence (including dysferlin and desmin). Further, the juxtaposition of duplicate titin loci strongly suggests a tandem gene duplication event after the teleost ancestral genome duplication.
Thus, at least one and at most 3 of the 29 genes studied (3–10%) show evolutionary maintenance of duplicate gene sequences from the whole genome duplication event, below the 20% previously reported [22]. Given the widespread distribution of these genes (Fig. 1), it is unlikely that the absence of dystrophy gene duplications is due to lack of duplication of a specific chromosomal region, or to secondary loss of a specific chromosomal region after polyploidization. It is also unlikely that the low number of dystrophy gene duplications in zebrafish is the result of an overall detrimental affect of duplicate copies of these genes since paralogs of many dystrophy genes are found in both mammals and fish. While it is quite possible that all existing duplicates were not identified in this study, it is also possible that these genes may evolve more slowly, preventing divergence of duplicate loci that would subject both to evolutionary constraint.
Conclusion
To aid in the development of zebrafish as a suitable candidate for genetic screens for dystrophy-causing mutations and to create a genomic map of dystrophy-associated zebrafish genes, we searched existing zebrafish sequence databases to identify zebrafish orthologs of dystrophy-causing genes. Using Genbank and Sanger Centre databases, 28 out of 29 genes studied showed identifiable ortholog transcripts. These data suggest that zebrafish may express muscle genes orthologous to those previously shown in mammals to be required for normal muscle maintenance and/or regeneration. Genomic loci were also identified for all 29 genes (though one, TRIM32, is currently located on an orphan scaffold). Comparison of in silico ortholog mapping with published physical mapping confirms that the current genome and in silico techniques were able to identify correct chromosomal locations for at least 4 genes out of 5 genes with available positional information. Only 3–10% of dystrophy gene duplicates appear to have been maintained since the teleost genome duplication, fewer than other gene groups studied in fish, indicating that the dystrophy-related genes may be slow to evolve independent functions or regulation. These data should aid in the genetic mapping of zebrafish dystrophy mutants, creation of mutant lines for high-throughput testing of dystrophy therapies, and identification of novel dystrophy-causing genes.
Methods
Computer identification of orthologous zebrafish ESTs
For each gene (Table 1 and Table 2), human transcript sequences (starting with NM) and human protein sequences (starting with NP) were identified in NCBI databases [23]. Zebrafish ESTs orthologous to the human protein sequences were identified by BLAST into the NCBI zebrafish nr database using the tblastn algorithm. Responses were prioritized by percentage similarity and amount of coverage. Where more than one reasonable candidate EST was returned, all such ESTs were reciprocally compared with mammalian sequences in NCBI (nr database) using the tblastx algorithm to determine which one was most similar to the mammalian gene being studied. Sequences starting with NM representing known EST sequences were preferred. Predicted sequences (starting with XM) were used only when they showed high percentage similarity to mammalian sequences and when no other highly correlated zebrafish ESTs were returned. In some cases, zebrafish ortholog candidates could still not be distinguished and all such candidates were noted and pursued for the following identification steps. Where more than one human isoform is known for a given gene, all isoforms were independently queried against zebrafish databases as above. In no case, however, did different isoforms of a single human gene identify disparate zebrafish genes.
Computer identification of genomic location
Zebrafish ESTs were then compared with the current zebrafish genome assembly, Zv6, in the Wellcome Trust Sanger Institute databases as of April 2006. To identify genomic locations of zebrafish ortholog ESTs, the Ensembl blast program [24] was used with the blastn algorithm and "Near exact match" parameters. Returned hits were ranked by e value and assessed for transcript coverage. Note that percentage of sequence identity was typically > 95% over short stretches (likely corresponding to exons). Where more than one location had similar levels of coverage and sequence identity, all such locations are noted. To confirm genomic loci (or if no zebrafish EST was identified), human protein sequences were compared with the genome using a tblastx algorithm and the parameter "Allow some local mismatch". Multiple loci were frequently identified with the human protein, but could often be ruled out based on a closer orthology to other genes within a gene group (using the analyses methods herein).
Computer identification of position on sequenced clones
Because the genome assembly is still not complete and certain regions may be misaligned, we also identified the clone locations of zebrafish genes where possible. Zebrafish ESTs were compared with finished and unfinished clone sequences using the Sanger D. rerio Blast Server [25]. The blastn algorithm was used with a filter for low complexity regions and Repeatmasker to mask short repeat sequences. Returned sequences were ordered by e value and analyzed for coverage and exon breaks corresponding to those seen in genomic locations. All finished clone sequences with complete coverage are listed. Unfinished (incompletely sequenced) clones are noted only where there was no reasonable alignment with a finished clone. In the case where a gene spans more than one clone, clones are noted with plusses between them. Again, loci were confirmed by homology to human protein sequences using the tblastn algorithm (without Repeatmasker).
Determination of synteny
Neighboring genes and their orientations with respect to the human gene of interest were determined using NCBI Entrez GeneView. In many cases, closest neighbors were predicted or non-coding RNAs. Non-coding RNAs were not used. Some predicted genes did retain syntenic relationships and are listed. Where neighboring predicted genes were not found in the zebrafish genome, the closest known coding gene was used instead. Genomic loci for neighboring genes were determined as above, using the human protein sequence and tblastn algorithm in either the Zebrafish Genome or in the Clone Database.
Radiation hybrid mapping
Primers were designed to calpain-3 sequence NM_001004571 and used in PCR reactions with the zebrafish T51 radiation hybrid panel as previously described [26,27]. SAMapper was used to obtain LOD scores and map distances to known zebrafish markers [28]. Primers used were:
CAPN3 (Forward): 5'- CACTAGTGTCACAGGCAGCGTTTC-3'
CAPN3 (Reverse): 5'- GTTGCCGTCCATCATGAGCTTTGAG-3'
Authors' contributions
LSS identified ortholog sequences and genomic map locations and drafted the manuscript. JRG and LMK conceived of the project and assisted in drafting the manuscript. JRG also performed initial sequence searches. EDV and TJP participated in initial sequence searches. RB and YZ performed the radiation hybrid mapping of calpain-3 and assisted in project design. All authors read and approved the final manuscript.
Acknowledgments
Acknowledgements
LSS, JRG, EDV, TJP, and LMK are supported by a grant from the Bernard F. and Alva B. Gimbel Foundation. JRG is also supported by a grant from the Muscular Dystrophy Association. RB, YZ, and LIZ are supported by a genome grant from the NIDDK. LIZ and LMK are investigators with Howard Hughes Medical Institute.
Contributor Information
Leta S Steffen, Email: lsteffen@gmail.com.
Jeffrey R Guyon, Email: jguyon@enders.tch.harvard.edu.
Emily D Vogel, Email: emvogel@umich.edu.
Rosanna Beltre, Email: rbeltre@enders.tch.harvard.edu.
Timothy J Pusack, Email: tpusack@enders.tch.harvard.edu.
Yi Zhou, Email: yzhou@enders.tch.harvard.edu.
Leonard I Zon, Email: zon@enders.tch.harvard.edu.
Louis M Kunkel, Email: kunkel@enders.tch.harvard.edu.
References
- Dalkilic I, Kunkel LM. Muscular dystrophies: genes to pathogenesis. Curr Opin Genet Dev. 2003;13:231–238. doi: 10.1016/S0959-437X(03)00048-0. [DOI] [PubMed] [Google Scholar]
- Neuromuscular disorders: gene location. Neuromuscul Disord. 2006:64–90. [PubMed] [Google Scholar]
- Monaco AP, Neve RL, Colletti-Feener C, Bertelson CJ, Kurnit DM, Kunkel LM. Isolation of candidate cDNAs for portions of the Duchenne muscular dystrophy gene. Nature. 1986;323:646–650. doi: 10.1038/323646a0. [DOI] [PubMed] [Google Scholar]
- Khersonsky SM, Jung DW, Kang TW, Walsh DP, Moon HS, Jo H, Jacobson EM, Shetty V, Neubert TA, Chang YT. Facilitated forward chemical genetics using a tagged triazine library and zebrafish embryo screening. J Am Chem Soc. 2003;125:11804–11805. doi: 10.1021/ja035334d. [DOI] [PubMed] [Google Scholar]
- Milan DJ, Peterson TA, Ruskin JN, Peterson RT, MacRae CA. Drugs that induce repolarization abnormalities cause bradycardia in zebrafish. Circulation. 2003;107:1355–1358. doi: 10.1161/01.CIR.0000061912.88753.87. [DOI] [PubMed] [Google Scholar]
- Peterson RT, Shaw SY, Peterson TA, Milan DJ, Zhong TP, Schreiber SL, MacRae CA, Fishman MC. Chemical suppression of a genetic mutation in a zebrafish model of aortic coarctation. Nat Biotechnol. 2004;22:595–599. doi: 10.1038/nbt963. [DOI] [PubMed] [Google Scholar]
- Stern HM, Murphey RD, Shepard JL, Amatruda JF, Straub CT, Pfaff KL, Weber G, Tallarico JA, King RW, Zon LI. Small molecules that delay S phase suppress a zebrafish bmyb mutant. Nat Chem Biol. 2005;1:366–370. doi: 10.1038/nchembio749. [DOI] [PubMed] [Google Scholar]
- Ton C, Parng C. The use of zebrafish for assessing ototoxic and otoprotective agents. Hear Res. 2005;208:79–88. doi: 10.1016/j.heares.2005.05.005. [DOI] [PubMed] [Google Scholar]
- Granato M, van Eeden FJ, Schach U, Trowe T, Brand M, Furutani-Seiki M, Haffter P, Hammerschmidt M, Heisenberg CP, Jiang YJ, Kane DA, Kelsh RN, Mullins MC, Odenthal J, Nusslein-Volhard C. Genes controlling and mediating locomotion behavior of the zebrafish embryo and larva. Development. 1996;123:399–413. doi: 10.1242/dev.123.1.399. [DOI] [PubMed] [Google Scholar]
- Bassett DI, Bryson-Richardson RJ, Daggett DF, Gautier P, Keenan DG, Currie PD. Dystrophin is required for the formation of stable muscle attachments in the zebrafish embryo. Development. 2003;130:5851–5860. doi: 10.1242/dev.00799. [DOI] [PubMed] [Google Scholar]
- Dodd A, Chambers SP, Love DR. Short interfering RNA-mediated gene targeting in the zebrafish. FEBS Lett. 2004;561:89–93. doi: 10.1016/S0014-5793(04)00129-2. [DOI] [PubMed] [Google Scholar]
- Guyon JR, Mosley AN, Jun SJ, Montanaro F, Steffen LS, Zhou Y, Nigro V, Zon LI, Kunkel LM. Delta-sarcoglycan is required for early zebrafish muscle organization. Exp Cell Res. 2005;304:105–115. doi: 10.1016/j.yexcr.2004.10.032. [DOI] [PubMed] [Google Scholar]
- Guyon JR, Mosley AN, Zhou Y, O'Brien KF, Sheng X, Chiang K, Davidson AJ, Volinski JM, Zon LI, Kunkel LM. The dystrophin associated protein complex in zebrafish. Hum Mol Genet. 2003;12:601–615. doi: 10.1093/hmg/12.6.601. [DOI] [PubMed] [Google Scholar]
- Nixon SJ, Wegner J, Ferguson C, Mery PF, Hancock JF, Currie PD, Key B, Westerfield M, Parton RG. Zebrafish as a model for caveolin-associated muscle disease; caveolin-3 is required for myofibril organization and muscle cell patterning. Hum Mol Genet. 2005;14:1727–1743. doi: 10.1093/hmg/ddi179. [DOI] [PubMed] [Google Scholar]
- Chiang AP, Beck JS, Yen HJ, Tayeh MK, Scheetz TE, Swiderski RE, Nishimura DY, Braun TA, Kim KY, Huang J, Elbedour K, Carmi R, Slusarski DC, Casavant TL, Stone EM, Sheffield VC. Homozygosity mapping with SNP arrays identifies TRIM32, an E3 ubiquitin ligase, as a Bardet-Biedl syndrome gene (BBS11) Proc Natl Acad Sci U S A. 2006;103:6287–6292. doi: 10.1073/pnas.0600158103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pollard SM, Parsons MJ, Kamei M, Kettleborough RN, Thomas KA, Pham VN, Bae MK, Scott A, Weinstein BM, Stemple DL. Essential and overlapping roles for laminin alpha chains in notochord and blood vessel formation. Dev Biol. 2006;289:64–76. doi: 10.1016/j.ydbio.2005.10.006. [DOI] [PubMed] [Google Scholar]
- Bolanos-Jimenez F, Bordais A, Behra M, Strahle U, Mornet D, Sahel J, Rendon A. Molecular cloning and characterization of dystrophin and Dp71, two products of the Duchenne Muscular Dystrophy gene, in zebrafish. Gene. 2001;274:217–226. doi: 10.1016/S0378-1119(01)00606-0. [DOI] [PubMed] [Google Scholar]
- Xu X, Meiler SE, Zhong TP, Mohideen M, Crossley DA, Burggren WW, Fishman MC. Cardiomyopathy in zebrafish due to mutation in an alternatively spliced exon of titin. Nat Genet. 2002;30:205–209. doi: 10.1038/ng816. [DOI] [PubMed] [Google Scholar]
- Koenig M, Hoffman EP, Bertelson CJ, Monaco AP, Feener C, Kunkel LM. Complete cloning of the Duchenne muscular dystrophy (DMD) cDNA and preliminary genomic organization of the DMD gene in normal and affected individuals. Cell. 1987;50:509–517. doi: 10.1016/0092-8674(87)90504-6. [DOI] [PubMed] [Google Scholar]
- Labeit S, Kolmerer B. Titins: giant proteins in charge of muscle ultrastructure and elasticity. Science. 1995;270:293–296. doi: 10.1126/science.270.5234.293. [DOI] [PubMed] [Google Scholar]
- Amores A, Force A, Yan YL, Joly L, Amemiya C, Fritz A, Ho RK, Langeland J, Prince V, Wang YL, Westerfield M, Ekker M, Postlethwait JH. Zebrafish hox clusters and vertebrate genome evolution. Science. 1998;282:1711–1714. doi: 10.1126/science.282.5394.1711. [DOI] [PubMed] [Google Scholar]
- Postlethwait JH, Woods IG, Ngo-Hazelett P, Yan YL, Kelly PD, Chu F, Huang H, Hill-Force A, Talbot WS. Zebrafish comparative genomics and the origins of vertebrate chromosomes. Genome Res. 2000;10:1890–1902. doi: 10.1101/gr.164800. [DOI] [PubMed] [Google Scholar]
- National Center for Biotechnology Information http://www.ncbi.nlm.nih.gov/
- Ensembl Multi BlastView http://www.ensembl.org/Multi/blastview?species=Danio_rerio
- Danio rerio Blast Server http://www.sanger.ac.uk/cgi-bin/blast/submitblast/d_rerio
- Jagadeeswaran P, Gregory M, Zhou Y, Zon L, Padmanabhan K, Hanumanthaiah R. Characterization of zebrafish full-length prothrombin cDNA and linkage group mapping. Blood Cells Mol Dis. 2000;26:479–489. doi: 10.1006/bcmd.2000.0330. [DOI] [PubMed] [Google Scholar]
- Kwok C, Critcher R, Schmitt K. Construction and characterization of zebrafish whole genome radiation hybrids. Methods Cell Biol. 1999;60:287–302. doi: 10.1016/s0091-679x(08)61906-8. [DOI] [PubMed] [Google Scholar]
- Stewart EA, McKusick KB, Aggarwal A, Bajorek E, Brady S, Chu A, Fang N, Hadley D, Harris M, Hussain S, Lee R, Maratukulam A, O'Connor K, Perkins S, Piercy M, Qin F, Reif T, Sanders C, She X, Sun WL, Tabar P, Voyticky S, Cowles S, Fan JB, Cox DR, et al. An STS-based radiation hybrid map of the human genome. Genome Res. 1997;7:422–433. doi: 10.1101/gr.7.5.422. [DOI] [PubMed] [Google Scholar]


