Abstract
The homeobox gene CDX2 plays a major role in development, especially in the gut, and it also acts as a tumor suppressor in the adult colon. Using orthotopic and heterotopic xenografts of human primary colorectal tumor cells and cell lines in nude mice, we addressed the effect of the microenvironment on CDX2 expression. In cells expressing CDX2 at a high level in culture, this level was maintained in subcutaneous grafts but was reduced when implanted into the cecum wall. Reciprocally, in cells with low CDX2 expression in culture, the level remained low in grafts into the cecum wall but was stimulated subcutaneously. In vitro co-cultures showed that CDX2 expression was activated in cells grown on layers of skin fibroblasts but not on intestinal fibroblasts. The stimulation was transcriptional, as assessed by transfection experiments with reporter plasmids containing the murine Cdx2 promoter. Together, these data demonstrate experimentally that CDX2 expression is adaptable and strongly dependent on the microenvironment surrounding the tumor cells. We exclude a role of the Notch pathway in this regulation. The regulation of CDX2 by the microenvironment might be relevant during the process of metastatic dissemination when the gene is transiently turned down in invasive cells.
Colorectal cancer (CRC) is a major problem of public health because of its severity and high incidence. The severity is mainly attributable to the propensity of tumor cells to invade the stroma and eventually to disseminate into metastases. It is generally thought that the progression along the adenoma, carcinoma, metastasis sequence is accompanied by the accumulation of an increasing number of irreversible genomic alterations.1 In addition, reversible changes induced by the microenvironment facing malignant cells are also considered to be of prime importance for tumor cell invasion and metastatic dissemination.2 For instance, the epithelial-mesenchymal transition is a hallmark of invasive cells that escape the primary tumor, whereas the circulating tumor cells can undergo mesenchymal-epithelial transition at the homing site to form metastases.
The major reversible changes reported in invasive cells spreading out from primary tumors include the decrease of membranous E-cadherin, the massive nuclear translocation of β-catenin, the activation of the cell cycle inhibitor p16, and the intracellular accumulation of the basement membrane molecule laminin-γ2.2 Recently, a careful analysis of primary CRCs and liver metastases led us to show that invasive CRC cells are also characterized by a reversible decline of the intestine-specific homeoprotein CDX2.3 CDX2 is a transcription factor that plays a central role in embryos to define the organ-specific identity of the presumptive gut endoderm.4–7 At the adult stage, it belongs to the network that controls proliferation and differentiation of the continuously renewing gut epithelium.8,9 CDX2 also has a tumor suppressor function in the colon, because Cdx2+/− mice are hypersensitive to CRC progression in a mouse model of chemically induced cancers and in genetically predisposed APC+/− mice.10,11 A number of studies have reported that CDX2 expression is altered in CRCs, showing both inter- and intratumor heterogeneity.12–14 This observation is corroborated by the variable levels of CDX2 expression reported in cancer cell lines cultured in vitro.15 The CDX2 gene is rarely mutated in CRCs,14,16 suggesting that its altered expression in cancer cells depends on negative regulatory pathway(s). In vitro cell fusions have provided evidence for such an inhibitory pathway,15 but the molecular mechanism remains unknown.
We have previously reported that the mesenchyme can modulate homeobox gene expression, including CDX2, in the developing embryonic endoderm.17 The microenvironment has also been shown to modify the phenotype of pancreatic cancer cells in xenografts.18 For CDX2 in the context of CRCs, histopathological analyses have reported that gene expression is low in invasive cells but restored in metastases at the level corresponding to the primary tumors.3 However, there was still no experimental evidence for the dynamic nature and reversibility of these CDX2 changes. Using a model of orthotopic and heterotopic xenografts in nude mice, we demonstrate here that CDX2 expression is adaptable and strongly dependent on the microenvironment surrounding the tumor cells.
Materials and Methods
Human Primary Tumor Tissues and Tumor Cell Lines
Human primary colorectal tumors collected at the Department of Surgery in the University Hospital of Strasbourg were sequentially propagated as subcutaneous grafts in nude mice, without any step of culture in vitro.19 HT29, T84, and SW480 human CRC cell lines were cultured under standard conditions in Dulbecco’s modified Eagle’s medium containing 10% fetal bovine serum and 200 U/ml penicillin-streptomycin (Invitrogen, Cergy Pontoise, France). The γ-secretase inhibitor GSI (Calbiochem, Meudon, France) at 10 mmol/L in dimethyl sulfoxide (DMSO) was added to the culture medium at the indicated concentration. The cell line HT29-h11 was derived from HT29 by stable transfection with a pEGFP-based plasmid (Invitrogen) to constitutively express green fluorescent protein (GFP). These cells were selected for resistance to G418 (0.8 mg/ml). For co-cultures, tumor cells were seeded on top of confluent layers of human primary duodenal or skin fibroblasts. Fetal human skin and duodenal fibroblastic cells were prepared and frozen in 1985. Skin fibroblasts were derived from skin fragments of a premature 24-week-old fetus assigned for karyotypic examination (kindly provided by Dr. J.V. Ruch, University Hospital, Strasbourg, France). Fetal human duodenal fibroblasts were derived from the lamina propria and submucosa of a 28-week-old abortion. Both skin and duodenal fibroblasts were obtained by dissociation with 0.01% trypsin and 0.01% collagenase in Ca2+-Mg2+-free Ham’s F10 medium for 20 minutes at 37°C under gentle agitation, washed in Ham’s F10, and cultured in Dulbecco’s modified Eagle’s medium/Ham’s F12 with 15% fetal bovine serum. Subcultures of both types of fibroblasts were performed by conventional trypsinization. Skin fibroblasts were used at 11 to 18 passages, and duodenal fibroblasts at passages 2 to 6. In other experiments, tumor cells were co-cultured with the intestinal myofibroblastic cell lines MIC101 and MIC316 established from the lamina propria of the jejunum and of the colon of weaning rats, respectively.20 Immunolabeling with anti-α-smooth muscle actin antibody revealed ∼10% of myofibroblasts among human duodenal fibroblasts but only very rare labeled cells in the population of skin fibroblasts (data not shown); as previously reported,20 most of the MI101 and MIC316 cells were myofibroblasts.
Heterotopic and Orthotopic Xenografts in Nude Mice
Male athymic nu/nu mice, 6 to 8 weeks of age, were from Charles River (L’Arbresle, France) and housed under pathogen-free conditions. They were anesthetized with isoflurane gas (Forene; Abbott) at the time of surgery. For heterotopic or orthotopic implantation, primary tumor cells and tumor cell lines were dissociated mechanically and enzymatically. Fifty μl of cell suspension in Dulbecco’s modified Eagle’s medium (∼2 × 107cells/ml) were injected with a 30-gauge needle, under the back skin between the scapulae or into the cecum wall or into the liver. Each cell line was implanted in five animals subcutaneously, in five animals into the cecum wall, and in two animals in the liver. Tumor formation was checked twice a week and the mice were sacrificed by cervical dislocation 6 to 12 weeks after implantation to remove and analyze the tumors. Mice were injected with 10 mg/kg BrdU (Sigma) 4 hours before the sacrifice. When indicated, tumor fragments resulting from HT29-h11 cells injected and grown subcutaneously were cut into small pieces, and they were secondarily implanted into the cecum wall of nude mice. Alternatively, subcutaneous tumor HT29-h11 cells were mechanically dissociated and cultured in vitro in the presence of G418 (0.8 mg/ml) to prevent the growth of fibroblasts.
Immunohistochemistry and Western Blots
Immunohistochemistry used standard procedures. For a better comparison of the data, immunostaining of the various samples in a given experiment was performed in the same run. Briefly, tumors were fixed in 4% paraformaldehyde and embedded in paraffin. Sections (6 μm) were deparaffinized and heated for 10 minutes in 10 mmol/L citrate buffer, pH 6.2, for antigen retrieval. They were stained with Harris solution for histological examination or with Alcian blue solution for mucous detection or immunostained using the primary antibodies raised against CDX2 (392 mol/L, dilution 1:200; Biogenex, Plaisir, France), β-catenin (7D8, dilution 1:150; Upstate Biotechnology; Souffelweyersheim, France), Ki-67 (ab833, dilution 1:50; Abcam), or α-smooth muscle actin (clone 1A4, dilution 1:400; Sigma, St. Quentin, France). Secondary biotinylated anti-mouse or anti-rabbit antibodies (dilution 1:200; Vector Laboratories Inc., Burlingame, CA) were revealed using the Vectastain ABC kit (Vector Laboratories Inc.), as recommended by the supplier. For immunohistochemical detection of BrdU incorporated into nuclear DNA, slides were treated for 45 minutes in 2 N HCl after the step of antigen retrieval and then incubated with biotinylated anti-BrdU (ab2284, dilution 1:200; Abcam). Cell nuclei were counterstained with hematoxylin. HT29 cells grown on plastic dishes to confluence or co-cultures on top of fibroblasts were fixed in 4% paraformaldehyde and used for immunocytochemistry as described above.
SW480 cells were transfected with the appropriate plasmids using JetPEI, according to the recommendations of the manufacturer (PolyPlus Transfection, Illkirch, France). The plasmids pFlag-CBF1 encoding CBF1/CSL,21 pMyc-NICD1 encoding the intracellular domain of Notch1,22 and the reporter vector 4xwtCBF1Luc21 have been described. Transfected cells were visualized by indirect immunofluorescence as previously described,23 using mouse anti-Flag antibody (dilution 1:1000; Sigma) and Cy3-labeled anti-mouse antibody (dilution 1:3000; Interchim, Montluçon, France). CDX2 was detected using anti-CDX2 antibody (dilution 1:500; Biogenex) and Alexa-488-conjugated anti-mouse antibody (dilution 1:3000; Molecular Probes Invitrogen). All incubations were performed in 0.1% normal goat serum.
For Western blots, proteins were separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis, transferred to Immobilon-P membranes (Millipore, Molsheim, France), and analyzed using anti-CDX2 (dilution 1:5000; Biogenex) and anti-actin antibodies (dilution 1:15,000; Chemicon Int., Temecula, CA), as described.23 Peroxidase-conjugated sheep anti-mouse antiserum (dilution 1:5000; Amersham Pharmacia Biotech, Orsay, France) was used as secondary antibody and the membranes were revealed using ECL (Amersham Pharmacia Biotech).
Isolation of RNA and Semiquantitative Reverse Transcriptase-Polymerase Chain Reaction (RT-PCR)
RNA was extracted from cell lines and from grafted tumors using Tri Reagent (Sigma). One μg of RNA was used for semiquantitative RT-PCR as previously described8,24 using the human-specific primers indicated in Table 1. RT-PCR products were run on 2% agarose gels and stained with ethidium bromide.
Table 1.
Genes and Primers Used
| Gene | Primers |
|---|---|
| CDX2 | F: 5′-AAAGTGAGCTGGCTGCCACACTTG-3′ |
| R: 5′-TCCATCAGTAGATGCTGTTCGTGG-3′ | |
| CK19 | F: 5′-GGCTGAGAACGGGAAGCTTGTGATCAATGG-3′ |
| R: 5′-TTCCCAGGGGAGTCTCGCTGGTAGC-3′ | |
| GAPDH | F: 5′-GGCTGAGAACGGGAAGCTTGTGATCAATGG-3′ |
| R: 5′-GTCGCTGTTGAAGTCAGAGGAGACCACCTG-3′ | |
| Jagged2 | F: 5′-TCGAGCGGCCGGGGGGCCACAA-3′ |
| R: 5′-TCCCGGCCCAGCTGCAGCCGCCC-3′ | |
| Delta4 | F: 5′-CAGGGGGCTCCTTCCGGGGCTCCG-3′ |
| R: 5′-CAGTGGAATTTCCAACACACTGC-3′ | |
| Manic fringe | F: 5′-TCTCTCAGTCATTCCCCTGGGGCTC-3′ |
| R: 5′-AACCAGATGCTGGAGGGAATAGCTG-3′ | |
| Hes1 | F: 5′-AAGTGTGCTGGGGAAGTACCGAGC-3′ |
| R: 5′-TTGGGGAGTTTAGGAGGAGGGGT-3′ | |
| Math1 | F: 5′-AGGCTTCCGGAGGGAGCCAGCGG-3′ |
| R: 5′-GGGACCGAGGCGAAGTTTTGC-3′ | |
| E12 | F: 5′-GAAGCCGGACTCGAGACCCCGGGCTT-3′ |
| R: 5′-CTTGAGTGATATGTTTCCATTTCTC-3′ | |
| Master- mind3 | F: 5′-GGTGGGTGAGAAGCCCAGGCAGGGGCT-3′ |
| R: 5′-CAGTACTGAGCATTTGGAGGAAGCT-3′ |
Reporter Plasmids and Promoter Analyses
A BAC library of mouse genomic DNA was screened by hybridization with the 0.9-kb Cdx2 promoter fragment of the plasmid pCdx2–1Luc24 to isolate a large genomic segment containing the murine Cdx2 gene. Subfragments were inserted into the vector pGl3-Basic (Promega) to get reporter plasmids containing different lengths of the murine Cdx2 promoter. These fragments ended at position −33 with respect to the Cdx2 translation start site, and they extended up to position −1054 (pCdx2–1Luc24) and to position −9326 (pCdx2–9Luc). Plasmids pGl3-control and pRL-null were from Promega. pRL-null was used to standardize the transfection experiments.
For promoter analyses, HT29 cells were co-transfected for 4 hours under standard conditions23 using JetPEI (PolyPlus Transfection) with the reporter plasmids pGl3-control (Promega, Charbonnières, France), pCdx2–9Luc, or pCdx2–1Luc and the combination of pFlag-CBF1 and pMyc-NICD, together with pRL-null (Promega) for normalization. For co-cultures, epithelial cells were co-transfected for 4 hours with pGl3-control or pCdx2–9Luc or pCdx2–1Luc, together with pRL-null for normalization, and cells were then trypsinized and seeded on top of a confluent layer of human duodenal or skin fibroblasts. Firefly and Renilla luciferase activities were measured using a dual reporter luciferase assay (Promega) with a Lumistar luminometer (BMG, Offenburg, Germany). Each transfection experiment was repeated at least three times in triplicate. Firefly luciferase activity was normalized with Renilla luciferase activity, and average data were given ±SD.
Results
CDX2 Expression in Engrafted Primary Colorectal Tumor Cells Depends on the Site of Implantation in Nude Mice
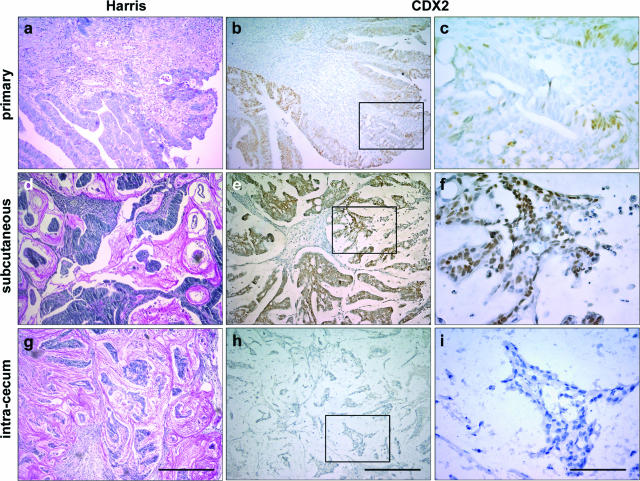
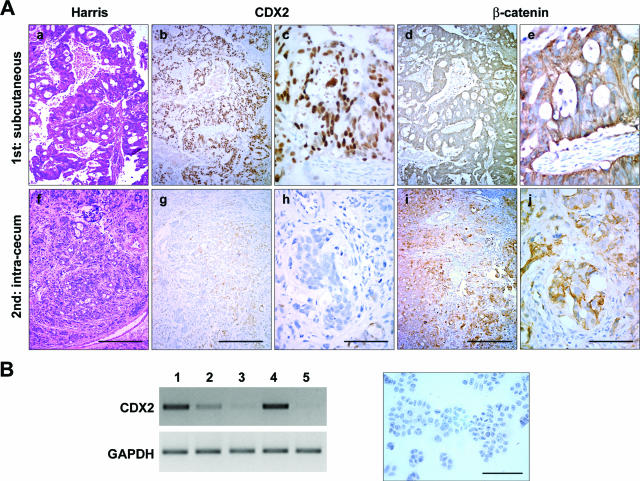
We have previously reported that human primary CRCs exhibit intratumor heterogeneity at the level of the genomic alterations present in malignant cells and that this heterogeneity is maintained when the tumor cells are propagated as subcutaneous xenografts in nude mice.19 On this basis, we investigated the expression pattern of CDX2 in xenografts because this transcription factor is known to be variably expressed in human primary CRCs. For this purpose, we compared by immunochemistry the expression of the CDX2 protein in 24 human primary CRCs and in the corresponding tumors grown in nude mice after subcutaneous injection of cancer cells. Among the 24 primary tumors, 10 showed strong and homogeneous CDX2 expression, 10 exhibited heterogeneous CDX2 expression, and 4 failed to express CDX2. A general description of the primary tumors and subcutaneous xenografts has already been reported.19 All subcutaneous tumors developed as compact structures without invasion of the host tissue. When primary tumors expressed CDX2 at a homogeneous and relatively high level, the high level was retained in the corresponding subcutaneous grafts (data not shown). However, when primary tumors exhibited a low and/or heterogeneous CDX2 expression (Figure 1, a–c), the subcutaneous xenografts showed an increased and homogeneous CDX2 expression (Figure 1, d–f). The heterotopic skin microenvironment is supposed to be different from the orthotopic intestinal environment. Thus, cancer cells from five of the above primary tumors were injected into the cecum wall of nude mice. In the cecum wall, tumors were less differentiated than those injected subcutaneously, and invasion of the mesenteric ganglia was observed in two of the five cases. Interestingly, the expression of CDX2 in the resulting tumors was very low in this environment (Figure 1, g–i), which strikingly contrasted with the pattern observed subcutaneously. Therefore, CDX2 expression changes in human colorectal tumor cells xenografted in nude mice, depending on the site of implantation.
Figure 1.
CDX2 expression in human colorectal tumors. a–c: An example of human colorectal tumor at the time of surgical resection is illustrated, showing heterogeneous CDX2 expression. d–i: The corresponding tumor cells were implanted subcutaneously (d–f) or in the cecum wall (g–i). Sections were stained with Harris solution for histopathological examination (a, d, g) or immunostained with anti-CDX2 antibody (b, c, e, f, h, i). The regions enclosed in b, e, and h are shown at higher magnification in c, f, and i, respectively. Scale bars = 200 μm (a, b, d, e, g, h); 40 μm (c, f, i).
CDX2 Is Down-Regulated in T84 and SW480 Colon Cancer Cells Grafted Orthotopically Versus Heterotopically
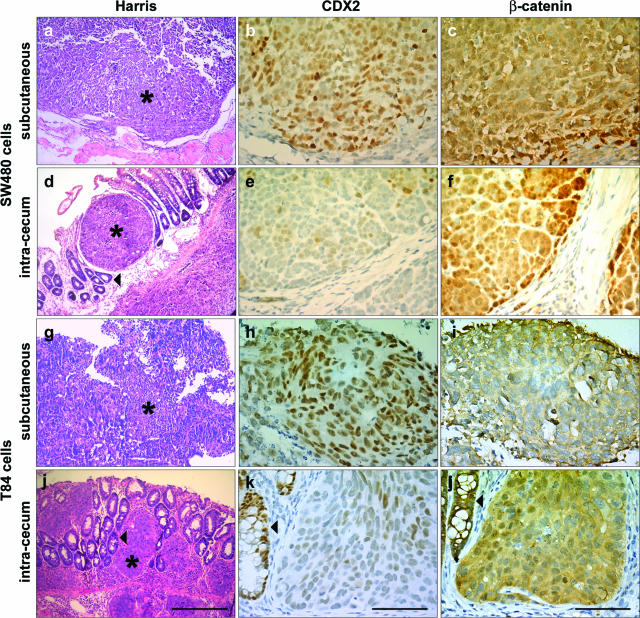
Because primary tumors are made of genetically heterogeneous clones, the different CDX2 patterns observed in orthotopic and heterotopic xenografts might result from a differential selection of CDX2-positive and -negative clones at the sites of implantation. An alternative possibility is that the level of CDX2 expression is not entirely dependent on characteristics intrinsic to the tumor clones but that the subcutaneous and cecum microenvironments differentially regulate the CDX2 gene in the tumor cells. To address this issue, we conducted orthotopic and heterotopic grafting experiments with a panel of well-characterized CRC cell lines established in culture. For this purpose, we first used SW480 and T84 cells, two human colon cancer cell lines known to express CDX2 at a significant level in culture.15 When SW480 and T84 cells were implanted subcutaneously, the tumors expressed CDX2 in most cell nuclei (Figure 2, b and h), consistent with its expression in cell culture. In contrast, CDX2 expression became low when the same cells were orthotopically grafted into the cecum wall (Figure 2, e and k). CDX2 protein was undetectable in a large proportion of nuclei, and only few cells retained a rather faint immunostaining, generally at the center of the tumors. This indicates that CDX2 can be down-regulated in established CRC cell lines when implanted in the intestinal microenvironment.
Figure 2.
Properties of SW40 and T84 cells implanted subcutaneously or in the cecum wall. Serial sections of tumors grown after subcutaneous (a–c, g–i) or cecum implantation (d–f, j–l) of SW480 (a–f) or T84 (g–l) cells were stained with Harris solution (a, d, g, j) or immunolabeled with anti-CDX2 (b, e, h, k) or anti-β-catenin antibodies (c, f, i, l). Asterisks indicate the tumors. Arrowheads point to the cecum epithelium of the mouse host. Scale bars = 200 μm (a, d, g, j); 40 μm (b, c, e, f, h, i, k, l).
Because the intracellular distribution of β-catenin can change during tumor progression2 and because CDX2 is an indirect target of β-catenin via SOX9 in intestinal crypts,25 we compared the β-catenin distribution in heterotopic and orthotopic xenografts of SW480 and T84 cells. In the subcutaneous tumors, β-catenin was diffuse in the cytoplasm and barely detected in cell nuclei (Figure 2, c and i). In the cecum grafts, it was also cytoplasmic, although some cell nuclei became immunolabeled, mostly at the periphery of the tumors (Figure 2, f and j).
CDX2 Is Up-Regulated in HT29 Colon Cancer Cells Grafted Heterotopically Versus Orthotopically
Grafting experiments were then conducted using HT29 cells that are characterized by a very low expression of CDX2 in culture.15 We also used two subclones of HT29 constitutively expressing green fluorescent protein (GFP) to visualize the tumor shape by fluorescence. Both subclones and the parental HT29 cells behaved identically in xenografts, so that only the data obtained with one clone, HT29-h11, are illustrated hereafter. Like parental HT29 cells, the HT29-h11 clone expressed CDX2 at a very faint level (see Figure 6a).
Figure 6.
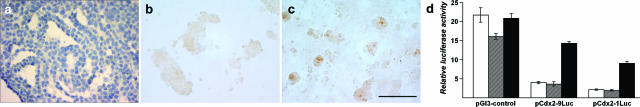
Differential regulation of CDX2 in HT29-h11 cells in co-culture with fibroblasts. a: HT29-h11 cells were cultured to confluence on standard plastic dishes; the cell sheet was collected as a Swiss roll and used for immunolabeling with anti-CDX2 antibody. Cell nuclei were counterstained with hematoxylin. Note the absence of CDX2 protein revealed by the absence of brown staining. Cells were co-cultured on top of duodenal (b) or skin fibroblasts (c) for 3 days and immunostained with anti-CDX2 antibody. No significant labeling was detected in the epithelial cells grown on duodenal fibroblasts, whereas cell nuclei expressed CDX2 when grown on top of skin fibroblasts. For a better visualization of the labeling, cell nuclei were not counterstained with hematoxylin in b and c. d: HT29-h11 cells were transfected with the reporter vector pGl3-control containing the SV40 promoter or with the plasmids pCdx2–9Luc or pCdx2–1Luc. The reporter plasmid pRL-null was co-transfected for normalization. Cells were subsequently cultured in standard plastic dishes (white boxes), on top of duodenal fibroblasts (hatched boxes), or on top of skin fibroblasts (black boxes). Relative luciferase activity is expressed as arbitrary units. The data are the results of three independent experiments conducted in triplicate and are given ±SD. Scale bar = 100 μm.
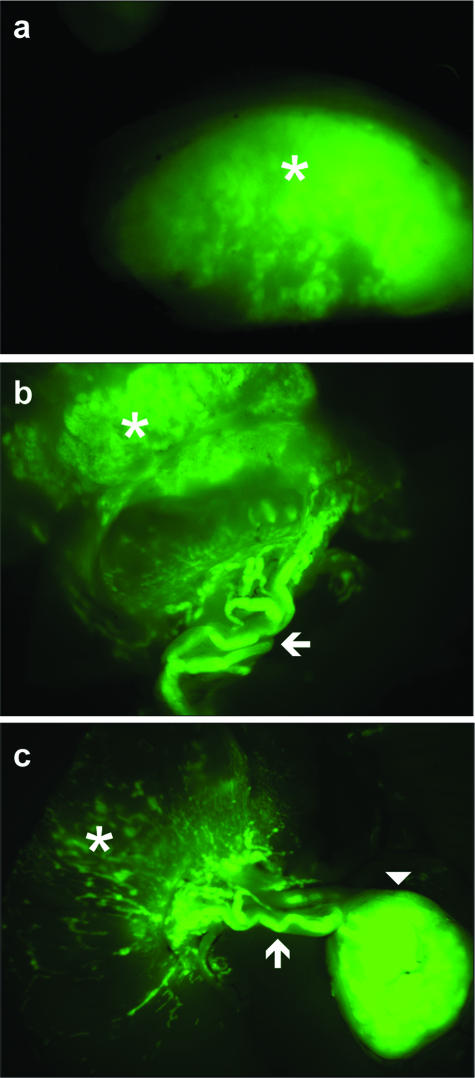
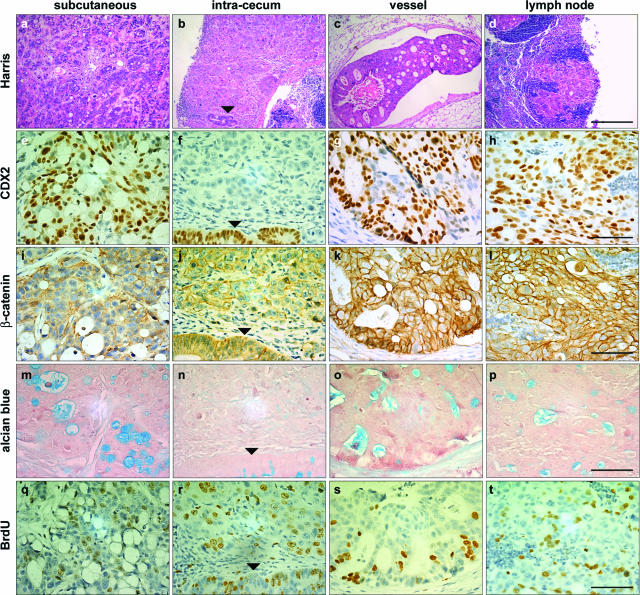
GFP fluorescence indicated that the subcutaneous tumors developed as compact structures showing a clear-cut boundary with the surrounding host tissue (Figure 3a). In contrast, the tumor shape was different when cells were orthotopically injected into the cecum wall, because GFP fluorescence revealed diffuse structures grown into the gut wall, from which emanated vessels (Figure 3b) that eventually reached lymph nodes (Figure 3c). The histology of these structures is illustrated in Figure 4, a–d. Although HT29-h11 cells are undifferentiated in culture, the tumors grown subcutaneously exhibited a differentiated phenotype assessed by the presence of glandular figures stained with Alcian blue and containing mucous-secreting cells (Figure 4m). Consistently, these cells expressed Muc2, a major component of intestinal mucous (not shown). Noteworthy, the subcutaneous tumors abundantly expressed nuclear CDX2 protein (Figure 4e), unlike cells in culture (see Figure 6a). Corroborating this results, HT29-h11 cells expressed the CDX2 mRNA when grown in subcutaneous tumors, whereas this transcript is almost undetectable in cells grown in vitro (Figure 5B, lanes 4 and 5). In contrast to the homogeneous histological pattern found in subcutaneous tumors, HT29-h11 cells generated various tumor structures when implanted in the cecum wall. Tumor structures grown into the lamina propria of the cecum were diffuse (Figure 4b) and undifferentiated because they failed to produce Alcian blue-positive mucous (Figure 4n). Importantly, they remained devoid of CDX2 protein like the cells in culture (Figure 4f). However, in striking contrast to the diffuse structures in the lamina propria, the tumors that colonized vessels and lymph nodes shared typical properties with the heterotopic subcutaneous xenografts. Indeed, they exhibited a differentiated phenotype (Figure 4, c and d), with mucous-rich glands (Figure 4, o and p), and they strongly expressed CDX2 (Figure 4, g and h).
Figure 3.
Macroscopic structure of HT29-h11 tumors visualized by GFP fluorescence. GFP fluorescence delineates the fate of the tumors of HT29-h11 cells implanted under the skin (a) or in the cecum wall of nude mice (b, c). Fluorescence was visualized from the luminal side (b) or the serosal side (c) of the cecum. Asterisks denote the bulk of the tumors, arrows point to tumor emboli within lymph vessels, and the arrowhead indicates a lymph node colonized by tumor cells.
Figure 4.
Properties of HT29-h11 cells implanted subcutaneously or in the cecum wall. Cells were injected subcutaneously (a, e, i, m, q) or in the cecum wall. In the cecum wall, they grew to form tumors in the lamina propria (b, f, j, n, r), emboli in lymph vessels (c, g, k, o, s), and metastases in lymph nodes (d, h, l, p, t). Serial tumor sections were stained with Harris solution (a–d), with anti-CDX2 antibody (e–h), with anti-β-catenin antibody (i–l), with Alcian blue solution (m–p), or with anti-BrdU antibody (q–t). Arrowheads point to the cecum epithelium of the mouse host. Scale bars = 200 μm (a–d); 40 μm (e–t).
Figure 5.
Reversible expression of CDX2 in xenografts. A: HT29-h11 cells were first implanted subcutaneously (a–e), and tumor fragments were then reimplanted in the cecum wall (f–j). Serial sections were stained with Harris solution (a, f) or immunostained with anti-CDX2 (b, c, g, h) or anti-β-catenin antibodies (d, e, i, j). c and h and e and j are higher magnifications of b and g and d and i, respectively. B: Tumors grown subcutaneously from HT29-h11 cells were dissociated, and cells were plated back in Petri dishes for in vitro cultures in the presence of G418 to prevent the growth of fibroblasts. In the left panel, cells were analyzed by RT-PCR for the expression of the CDX2 mRNA after one (lane 1), two (lane 2), and four (lane 3) passages. Controls included HT29-h11 cells continuously cultured in vitro (lane 5) or grown as subcutaneous tumors (lane 4). In the right panel, disappearance of CDX2 expression was assessed by immunochemistry using anti-CDX2 antibody in HT29-h11 cells grown as subcutaneous tumors and then plated back in vitro for four passages. Cell nuclei were counterstained with hematoxylin. Bar is 40 μm. Scale bars = 200 μm [A (a, b, d, f, g, i)]; 40 μm [A (c, e, h, j), B].
To look for a possible link between CDX2 expression and the distribution of β-catenin in engrafted HT29-h11 cells, tumors grown subcutaneously or in the cecum wall were immunostained with β-catenin antibody (Figure 4, i–l). In every case, β-catenin was mostly associated with cell membranes without any evidence for massive nuclear translocation. Therefore, in HT29 tumors the expression of CDX2 could not be correlated to the intracellular distribution of β-catenin. Immunohistochemical detection of cells having incorporated BrdU into the DNA identified ∼20% of cells in tumors grown either subcutaneously or in the cecum wall, which suggests a similar proliferation rate at both implantation sites (Figure 4, q–t). Similar results were obtained with Ki-67 labeling (data not shown).
Although the skin is a common grafting site in nude mice, it is not a major homing location of CRC metastases that generally develop in the liver. We injected HT29-h11 cells directly into the liver parenchyma of nude mice and the resulting tumors looked like those implanted subcutaneously (data not shown). They contained glandular structures stained with Alcian blue and expressed CDX2. Thus, CDX2 was up-regulated in the heterotopic environment of the liver, like in the skin.
Reversible Changes of CDX2 in HT29 Cells Grafted Heterotopically and Orthotopically
Because CDX2 is restored when HT29-h11 cells are grown under the skin of nude mice but not in the lamina propria of the cecum, we next investigated the dynamic nature of its expression. For this purpose, HT29-h11 cells were first grafted under the skin of nude mice for 8 weeks to induce a strong and homogeneous expression of CDX2, the subcutaneous tumors were then excised and cut into small pieces, and the fragments were implanted without cell dissociation into the cecum wall for an additional period of 8 weeks. As shown in Figure 5A, b, c, g, and h, high CDX2 expression in the subcutaneous xenografts returned to a low and heterogeneous pattern once implanted into the cecum wall. These data demonstrate the reversibility of CDX2 expression depending on the site of implantation. The overall distribution of β-catenin in the primary subcutaneous and secondary cecum tumors was not changed (Figure 5A, d, e, i, and j).
Turning down CDX2 expression in subcutaneous tumor fragments secondarily implanted into the cecum wall can result from spontaneous reversibility in HT29-h11 cells when they are taken away from the subcutaneous microenvironment or from the induced suppression of CDX2 by the cecum microenvironment. To address this point, subcutaneous tumors exhibiting high CDX2 expression were carefully excised and dissociated, and the tumor cells were plated for five passages in culture in vitro. In culture, cells kept their epithelial fate. As shown in Figure 5B by RT-PCR and immunochemistry, CDX2 expression progressively decreased to an almost undetectable level during consecutive passages in culture in vitro, favoring spontaneous reversibility.
CDX2 Is Differentially Expressed in HT29 Cells Co-Cultured with Fibroblasts
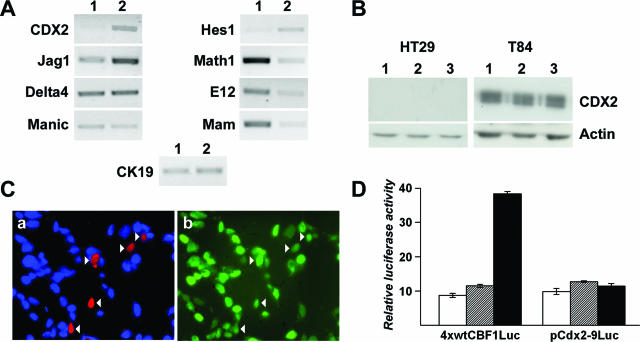
Based on the low expression of CDX2 in HT29-h11 in culture (Figure 6a) and on the different expression patterns when cells are grafted in subcutaneous or intestinal environment, we investigated the behavior of these cells in co-culture with human fibroblasts derived from human skin or duodenum or with rat myofibroblastic cell lines derived from the lamina propria of the jejunum or of the colon. Approximately 10% of the human duodenal fibroblasts exhibited myofibroblastic properties, as assessed by immunostaining of α-smooth muscle actin, whereas labeled cells were very rare among skin fibroblasts. After 3 days of co-culture, the epithelial cells grew as clusters that were larger on duodenal compared with skin fibroblasts (Figure 6, b and c). Immunocytochemistry failed to reveal any significant CDX2 expression in the nuclei of cells co-cultured with duodenal fibroblasts (Figure 6b). Rat jejunal and colonic myofibroblasts also failed to turn on CDX2 in co-cultured HT29 cells, indicating that the absence of inductive effect on CDX2 was independent of the longitudinal origin of the intestinal fibroblasts (data not shown). In contrast, clusters of cells co-cultured with skin fibroblasts, although smaller in size, clearly contained CDX2-positive nuclei (Figure 6c). Conditioned medium of skin fibroblasts did not induce CDX2 in HT29 cells (not shown), suggesting that soluble factors secreted by these fibroblasts are not sufficient to up-regulate CDX2 in the CRC cells.
To study the Cdx2 promoter in the co-culture conditions, HT29 cells were transfected with the reporter plasmids pCdx2–9Luc and pCdx2–1Luc containing, respectively, 9 kb and 1 kb of the murine Cdx2 promoter. After 4 hours in the transfection medium, cells were harvested, split into three batches, and plated for 2 days into cell culture dishes onto a confluent layer of duodenal fibroblasts or onto a confluent layer of skin fibroblasts. Figure 6d summarizes the results. The reporter plasmid pCdx2–9Luc produced a low luciferase activity in HT29 cells co-cultured with duodenal fibroblasts, like in cells directly seeded on the plastic support. On the contrary, promoter activity was stimulated by fourfold in HT29 cells co-cultured with skin fibroblasts. The plasmid pCdx2–1Luc gave a similar result as pCdx2–9Luc. The differential effect of the skin versus duodenal fibroblasts was specific to the Cdx2 promoter because it was not observed for the SV40 promoter in pGl3 control (Figure 6d). Rat MIC fibroblasts, like human duodenal fibroblasts, also failed to activate the Cdx2 promoter in co-cultured HT29 cells transfected with pCdx2–9Luc (data not shown). Taken together, these data indicate that co-cultures mimic the situation observed in heterotopic and orthotopic grafts because the CDX2 gene is up-regulated on skin fibroblasts such as in the tumors grown subcutaneously, but not on duodenal fibroblasts such as in the tumors grown in the cecum wall.
The Notch Pathway Does Not Control CDX2 Expression in Cultured Cancer Cells
Recently, the role of the Notch pathway connected to bHLH transcription factors has been uncovered in the gut epithelium, and the inhibition of this pathway in a mouse model of genetic predisposition to intestinal tumors partially restored the differentiation of mucous-secreting cells in the tumors.26,27 Because CDX2 is a regulator of Muc2,28,29 and because HT29-deriving tumors can differentially produce mucous-secreting structures and express CDX2 depending on the site of implantation, we investigated whether CDX2 could be regulated negatively by the Notch pathway in tumor cells.
A microarray comparison30 of the gene expression profile in HT29 cells in culture and xenografted under the skin of nude mice pointed to the up-regulation of CDX2 in the xenografts, and this was confirmed by RT-PCR (Figure 7A). Gene profiling also identified some elements of the Notch/bHLH pathway (data not shown). Figure 7A illustrates a confirmation of these results by RT-PCR. The transcripts of the ligand Jagged-1 and the bHLH transcription factor Hes1 increased in xenografts, whereas the mRNA of the bHLH proteins Math1 and E12 and of the co-activator Mastermind decreased. No change was found for the Notch ligand Delta4 and the modulator Manic Fringe. To investigate whether there could be a link between the changes in Notch signaling and CDX2 expression, HT29 cells were treated with increasing doses of the γ-secretase inhibitor GSI to repress Notch signaling. GSI at 10 nmol/L to 2 μmol/L did not turn on CDX2 protein expression in these cells (Figure 7B) either in the growth phase or at confluence; massive cell death occurred at higher doses of GSI. Conversely, GSI did not down-regulate CDX2 in T84 cells (Figure 7B). Next, we directly addressed the effect of Notch activation by transfecting SW480 cells with plasmids encoding the transcription factor targeted by Notch, CSL, together with the intracellular domain of Notch1 that activates CSL, NICD1. Immunofluorescence staining failed to reveal any obvious decline of endogenous CDX2 in the cells transfected with CSL and NICD1 compared with adjacent nontransfected cells (Figure 7C). Next, HT29, SW480, or T84 cells were transfected with the reporter plasmid pCdx2–9Luc alone or in combination with plasmids coding for CSL and NICD1. Although CSL and NICD1 stimulated the control plasmid 4xwtCBF1Luc containing binding sites for CSL, there was no effect on pCdx2–9Luc (Figure 7D). Finally, we addressed the effects of the transcription factor Hes1, which is a downstream target of CSL/NICD1. In co-transfection assays with or without the co-factors E12 and E47, Hes1 did not modify the transcriptional activity of pCdx2–9Luc (not shown). Taken together, these data suggest that Notch is not primarily involved in the dominant-negative pathway that down-regulates CDX2 in CRC cells.
Figure 7.
Absence of effect of the Notch/bHLH pathway on CDX2 expression. A: Expression of CDX2 and of several elements of the Notch/bHLH pathway in HT29 cells in culture (lane 1) and in subcutaneous xenografts (lane 2). RNA was analyzed by RT-PCR with primers for the indicated genes and compared with cytokeratin-19 (CK19). B: Effect of GSI on CDX2 expression. After 4 days in culture, HT29 and T84 cells were cultured for 3 days in control medium (lane 1), with the vehicle alone (lane 2, DMSO), or with 0.5 μmol/L GSI in DMSO (lane 3). CDX2 protein was detected by Western blot and related to actin. C: SW480 cells were transfected with the plasmids encoding Flag-CSL and Myc-NICD1. Transfected cells were identified by red fluorescence with anti-Flag antibody (a), and CDX2 expression was simultaneously detected in green using anti-CDX2 antibody (b). Arrowheads indicate the transfected cells. Cell nuclei were visualized by DAPI staining in a. D: SW480 cells were co-transfected with either the reporter plasmids pCdx2–9Luc or 4xwtCBF1Luc, alone (white boxes), with the plasmid encoding CSL (hatched boxes), or with the plasmids coding for CSL and NICD1 (black boxes). Cells were co-transfected with pRL-null for normalization. Relative luciferase activity is expressed as arbitrary units. The data are the results of three independent experiments conducted in triplicate and are given ±SD. Similar results were obtained in HT29 and T84 cells (not shown).
Discussion
In this study, we used the model of heterotopic and orthotopic xenografts in nude mice to address the effect of the microenvironment on CDX2 expression in human primary CRCs and in established colon cancer cell lines. The data show that the heterotopic subcutaneous microenvironment exerts a stimulatory effect on CDX2, whereas the orthotopic intestinal microenvironment has the opposite effect. Moreover, the successive subcutaneous and intracecum grafts demonstrate the reversibility of the CDX2 changes in tumor cells facing distinct microenvironments. In co-culture, CDX2 expression is stimulated on top of skin fibroblasts but not on duodenal fibroblasts, and this effect results from transcriptional activation of the Cdx2 promoter. Together, these data demonstrate experimentally that the microenvironment has a major regulatory effect on CDX2, a homeobox gene crucial for many developmental processes, and for slowing down tumor progression in the colon. In addition, these results validate the model of orthotopic versus heterotopic xenografts to investigate at the molecular level the adaptability and the reversible changes occurring in colon tumor cells facing different microenvironments.
We show here that the pattern of CDX2 expression changes in xenografts of human primary colorectal tumor cells depending on the site of implantation, although we have previously reported by allelotyping and CGH that these tumors are genetically stable in the grafting condition, especially for the CDX2 locus.19 Hence, the CDX2 changes are not attributable to the acquisition of new chromosomal rearrangement(s), but instead to a differential regulation of the gene by the microenvironment surrounding tumor cells. This conclusion is strengthened by the fact that CDX2 expression also changes with the implantation site in human colon cancer cell lines established several decades ago and considered therefore as genetically stable. Also in line with this conclusion are our findings 1) that the consecutive implantation of HT29 cells first under the skin and second in the cecum wall leads to the reversible up- and down-regulation of CDX2; 2) that the high CDX2 expression in HT29 cells grown as subcutaneous tumors is spontaneously reversed when cells are plated back in culture in vitro; and 3) that skin fibroblasts, but not duodenal or colonic fibroblasts, stimulate CDX2 expression in co-cultured HT29 cells. When extrapolated to human CRCs, these data collectively indicate that, besides the intrinsic heterogeneity attributable to the genetic divergence of subclones within a tumor, tumor heterogeneity can also result from reversible regulatory interactions between the tumor cells and their local microenvironment. Interestingly, the stimulatory effect exerted on CDX2 is not specific for the skin environment because other heterotopic microenvironments have the same effect, ie, the liver parenchyma and emboli within vessels and lymph nodes; however, the microenvironment of the intestinal lamina propria down-regulates CDX2 in intestinal cancer cells, as shown using T84 and SW480 cells. This mimics what happens during metastatic dissemination when invasive cells lose CDX2 expression and break the basement membrane to migrate into the intestinal lamina propria and what happens inversely when CDX2 expression is recovered at the homing site of metastases reorganized into glandular structures underlined by a basement membrane.
Using an experimental approach based on cell fusions, Hinoi and co-workers15 concluded to the existence of a regulatory pathway down-regulating CDX2 in HT29 cells and in other CRC cell lines. However, this pathway remains elusive. It comes out from our present results that the heterotopic microenvironment capable of restoring CDX2 expression in HT29 tumors might induce a signaling pathway that blunts and counteracts the dominant-negative cascade used to turn off the gene in cells in culture. An interesting observation is that HT29 tumors growing into the intestinal lamina propria fail to express CDX2 while remaining undifferentiated, whereas they arrange into glandular structures and express CDX2 when they grow in a heterotopic microenvironment. On this basis, we investigated whether signaling pathway(s) related to cell polarity and cell-cell interactions might control CDX2. One of these pathways uses β-catenin at the connection between adherens junctions and Wnt signaling. We found that β-catenin remains mainly associated to the plasma membrane in undifferentiated as in differentiated HT29 tumors, irrespective of the very different pattern of expression of CDX2, suggesting that β-catenin signaling is not the primary negative determinant of CDX2 in CRC cells. Tight junctions represent another type of junction complexes related to cell polarity and differentiation; they trap the transcription repressor ZONAB that can be released to move into the nucleus and to down-regulate a number of genes.31 By co-transfection experiments in several cell lines, we failed to identify any effect of ZONAB on the Cdx2 promoter (data not shown). Then we addressed the Notch/bHLH pathway on the basis of recent data describing its role in intestinal development and cancers.26,27 We found that several members of this signaling pathway are differently expressed in cultured cells and in subcutaneous grafts, suggesting that the pathway is modulated by epithelial-mesenchymal cell interactions. However, our experiments using an inhibitor of the pathway as well as overexpression of several elements of this pathway indicate that the Notch/bHLH pathway is not an important regulator of CDX2 in CRC cells in culture. Further in vivo investigations are needed. Inversely, elements of the Notch pathway have been proposed to be under the control of Cdx2.32 Thus, the mechanism(s) of control of Cdx2 by cell interactions remain elusive. We have previously shown that this gene is regulated by Ras and PI3K/Akt signaling,24,33 but the role of these pathways on Cdx2 expression has not been established so far in the context of cell interactions in vivo. Reciprocally, Cdx2 is regulated by extracellular matrix components, laminin-1 and collagen-1, but the downstream signaling pathways have not been elucidated.3,34
In conclusion, this study experimentally demonstrates that the expression of CDX2 in CRC cells is adaptable and strongly dependent on the tumor microenvironment, although the mechanistic elements of the regulatory pathway remain to be elucidated. Thus, CDX2 is likely to play an important role as an integrator of the reciprocal interactions between tumor cells and surrounding environment during CRC progression. This might be particularly important in the process of metastatic dissemination because the gene is transiently down-regulated in invasive cells.3 Investigations in the future will address the consequences of a transient drop of CDX2 on the spreading of invasive cells into metastases.
Acknowledgments
We thank Dr. T. Kadesch (Philadelphia, PA) for kindly providing the plasmids pFlag-CBF1, pMyc-NICD1, and 4xwtCBF1Luc.
Footnotes
Address reprint requests to Dr. Jean-Noël Freund, INSERM U682, 3 Avenue Molière, 67200 Strasbourg, France. E-mail: jean-noel.freund@inserm.u-strasbg.fr.
Supported by INSERM, the Association pour la Recherche Contre le Cancer (grant no. 3286), an Algerian-French fellowship (to F.B.), the Institut National du Cancer (grant no. ACI 2004), and the Consortium National des Génopôles (for microarrays).
I.G. was a fellow of the Association pour la Recherche Contre le Cancer and of the Institut National du Cancer.
References
- Diep CB, Kleivi K, Ribeiro FR, Teixeira MR, Lindgjaerde OC, Lothe RA. The order of genetic events associated with colorectal cancer progression inferred from meta-analysis of copy number changes. Genes Chromosom Cancer. 2006;45:31–41. doi: 10.1002/gcc.20261. [DOI] [PubMed] [Google Scholar]
- Brabletz T, Jung A, Spaderna S, Hlubek F, Kirchner T. Migrating cancer stem cells—an integrated concept of malignant tumour progression. Nat Rev Cancer. 2005;5:744–749. doi: 10.1038/nrc1694. [DOI] [PubMed] [Google Scholar]
- Brabletz T, Spaderna S, Kolb J, Hlubek F, Faller G, Bruns CJ, Jung A, Nentwich J, Duluc I, Domon-Dell C, Kirchner T, Freund JN. Down-regulation of the homeodomain factor cdx2 in colorectal cancer by collagen type I: an active role for the tumor environment in malignant tumor progression. Cancer Res. 2004;64:6973–6977. doi: 10.1158/0008-5472.CAN-04-1132. [DOI] [PubMed] [Google Scholar]
- Beck F, Chawengsaksophak K, Waring P, Playford RJ, Furness JB. Reprogramming of intestinal differentiation and intercalary regeneration in cdx2 mutant mice. Proc Natl Acad Sci USA. 1999;96:7318–7323. doi: 10.1073/pnas.96.13.7318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mutoh H, Hakamata Y, Sato K, Eda A, Yanaka I, Honda S, Osawa H, Kaneko Y, Sugano K. Conversion of gastric mucosa to intestinal metaplasia in Cdx2-expressing transgenic mice. Biochem Biophys Res Commun. 2002;294:470–479. doi: 10.1016/S0006-291X(02)00480-1. [DOI] [PubMed] [Google Scholar]
- Silberg DG, Sullivan J, Kang E, Swain GP, Moffett J, Sund NJ, Sackett SD, Kaestner KH. Cdx2 ectopic expression induces gastric intestinal metaplasia in transgenic mice. Gastroenterology. 2002;122:689–696. doi: 10.1053/gast.2002.31902. [DOI] [PubMed] [Google Scholar]
- Tamai Y, Nakajima R, Ishikawa T, Takaku K, Seldin MF, Taketo MM. Colonic hamartoma development by anomalous duplication in Cdx2 knockout mice. Cancer Res. 1999;59:2965–2970. [PubMed] [Google Scholar]
- Lorentz O, Duluc I, De Arcangelis A, Simon-Assmann P, Kedinger M, Freund JN. Key role of the Cdx2 homeobox gene in extracellular matrix-mediated intestinal cell differentiation. J Cell Biol. 1997;139:1553–1565. doi: 10.1083/jcb.139.6.1553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suh E, Traber PG. An intestine-specific homeobox gene regulates proliferation and differentiation. Mol Cell Biol. 1996;16:619–625. doi: 10.1128/mcb.16.2.619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aoki K, Tamai Y, Horiike S, Oshima M, Taketo MM. Colonic polyposis caused by mTOR-mediated chromosomal instability in Apc(+/Delta716) Cdx2(+/−) compound mutant mice. Nat Genet. 2003;35:323–330. doi: 10.1038/ng1265. [DOI] [PubMed] [Google Scholar]
- Bonhomme C, Duluc I, Martin E, Chawengsaksophak K, Chenard MP, Kedinger M, Beck F, Freund JN, Domon-Dell C. The Cdx2 homeobox gene has a tumour suppressor function in the distal colon in addition to a homeotic role during gut development. Gut. 2003;52:1465–1471. doi: 10.1136/gut.52.10.1465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ee HC, Erler T, Bhathal PS, Young GP, James RJ. Cdx-2 homeodomain protein expression in human and rat colorectal adenoma and carcinoma. Am J Pathol. 1995;147:586–592. [PMC free article] [PubMed] [Google Scholar]
- Hinoi T, Tani M, Lucas PC, Caca K, Dunn RL, Macri E, Loda M, Appelman HD, Cho KR, Fearon R. Loss of CDX2 expression and microsatellite instability are prominent features of large cell minimally differentiated carcinomas of the colon. Am J Pathol. 2001;159:2239–2248. doi: 10.1016/S0002-9440(10)63074-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Subtil C, Guérin E, Schneider A, Chenard MP, Martin E, Domon-Dell C, Duluc I, Brabletz T, Kedinger M, Duclos B, Gaub MP, Freund JN: Frequent rearrangements and amplification of the CDX2 homeobox gene in human sporadic colorectal cancers with chromosomal instability. Cancer Lett (in press) [DOI] [PubMed] [Google Scholar]
- Hinoi T, Loda M, Fearon ER. Silencing of CDX2 expression in colon cancer via a dominant repression pathway. J Biol Chem. 2003;278:44608–44616. doi: 10.1074/jbc.M307435200. [DOI] [PubMed] [Google Scholar]
- Yagi OK, Akiyama Y, Yuasa Y. Genomic structure and alterations of homeobox gene CDX2 in colorectal carcinomas. Br J Cancer. 1999;79:440–444. doi: 10.1038/sj.bjc.6690068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duluc I, Lorentz O, Fritsch C, Leberquier C, Kedinger M, Freund JN. Changing intestinal connective tissue interactions alters homeobox gene expression in epithelial cells. J Cell Sci. 1997;110:1317–1324. doi: 10.1242/jcs.110.11.1317. [DOI] [PubMed] [Google Scholar]
- Choudhury A, Moniaux N, Ulrich AB, Schmied BM, Standop J, Pour PM, Gendler SJ, Hollingsworth MA, Aubert JP, Batra SK. MUC4 mucin expression in human pancreatic tumours is affected by organ environment: the possible role of TGFbeta2. Br J Cancer. 2004;90:657–664. doi: 10.1038/sj.bjc.6601604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guenot D, Guerin E, Aguillon-Romain S, Pencreach E, Schneider A, Neuville A, Chenard MP, Duluc I, du Manoir S, Brigand C, Oudet P, Kedinger M, Gaub MP. Primary tumour genetic alterations and intra-tumoral heterogeneity are maintained in xenografts of human colon cancers showing chromosome instability. J Pathol. 2006;208:643–652. doi: 10.1002/path.1936. [DOI] [PubMed] [Google Scholar]
- Plateroti M, Rubin DC, Duluc I, Singh R, Foltzer-Jourdainne C, Freund JN, Kedinger M. Subepithelial fibroblast cell lines from different levels of gut axis display regional characteristics. Am J Physiol. 1998;274:G945–G954. doi: 10.1152/ajpgi.1998.274.5.G945. [DOI] [PubMed] [Google Scholar]
- Hsieh JJ, Henkel T, Salmon P, Robey E, Peterson MG, Hayward SD. Truncated mammalian Notch1 activates CBF1/RBPJk-repressed genes by a mechanism resembling that of Epstein-Barr virus EBNA2. Mol Cell Biol. 1996;16:952–959. doi: 10.1128/mcb.16.3.952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tani S, Kurooka H, Aoki T, Hashimoto N, Honjo T. The N- and C-terminal regions of RBP-J interact with the ankyrin repeats of Notch1 RAMIC to activate transcription. Nucleic Acids Res. 2001;29:1373–1380. doi: 10.1093/nar/29.6.1373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gross I, Lhermitte B, Domon-Dell C, Duluc I, Martin E, Gaiddon C, Kedinger M, Freund JN. Phosphorylation of the homeotic tumor suppressor Cdx2 mediates its ubiquitin-dependent proteasome degradation. Oncogene. 2005;24:7955–7963. doi: 10.1038/sj.onc.1208945. [DOI] [PubMed] [Google Scholar]
- Lorentz O, Cadoret A, Duluc I, Capeau J, Gespach C, Cherqui G, Freund JN. Downregulation of the colon tumour-suppressor homeobox gene Cdx-2 by oncogenic ras. Oncogene. 1999;18:87–92. doi: 10.1038/sj.onc.1202280. [DOI] [PubMed] [Google Scholar]
- Blache P, van de Wetering M, Duluc I, Domon C, Berta P, Freund JN, Clevers H, Jay P. SOX9 is an intestine crypt transcription factor, is regulated by the Wnt pathway, and represses the CDX2 and MUC2 genes. J Cell Biol. 2004;166:37–47. doi: 10.1083/jcb.200311021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fre S, Huyghe M, Mourikis P, Robine S, Louvard D, Artavanis-Tsakonas S. Notch signals control the fate of immature progenitor cells in the intestine. Nature. 2005;435:964–968. doi: 10.1038/nature03589. [DOI] [PubMed] [Google Scholar]
- van Es JH, van Gijn ME, Riccio O, van den BM, Vooijs M, Begthel H, Cozijnsen M, Robine S, Winton DJ, Radtke F, Clevers H. Notch/gamma-secretase inhibition turns proliferative cells in intestinal crypts and adenomas into goblet cells. Nature. 2005;435:959–963. doi: 10.1038/nature03659. [DOI] [PubMed] [Google Scholar]
- Mesquita P, Jonckheere N, Almeida R, Ducourouble MP, Serpa J, Silva E, Pigny P, Silva FS, Reis C, Silberg D, Van Seuningen I, David L. Human MUC2 mucin gene is transcriptionally regulated by cdx homeodomain proteins in gastrointestinal carcinoma cell lines. J Biol Chem. 2003;278:51549–51556. doi: 10.1074/jbc.M309019200. [DOI] [PubMed] [Google Scholar]
- Yamamoto H, Bai YQ, Yuasa Y. Homeodomain protein CDX2 regulates goblet-specific MUC2 gene expression. Biochem Biophys Res Commun. 2003;300:813–818. doi: 10.1016/s0006-291x(02)02935-2. [DOI] [PubMed] [Google Scholar]
- Le Brigand K, Russell R, Moreilhon C, Rouillard JM, Jost B, Amiot F, Magnone V, Bole-Feysot C, Rostagno P, Virolle V, Defamie V, Dessen P, Williams G, Lyons P, Rios G, Mari B, Gulari E, Kastner P, Gidrol X, Freeman TC, Barbry P: An open-access long oligonucleotide microarray resource for analysis of the human and mouse transcriptomes. Nucleic Acids Res 2006, 34: (in press) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Balda MS, Matter K. The tight junction protein ZO-1 and an interacting transcription factor regulate ErbB-2 expression. EMBO J. 2000;19:2024–2033. doi: 10.1093/emboj/19.9.2024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uesaka T, Kageyama N, Watanabe H. Identifying target genes regulated downstream of cdx2 by microarray analysis. J Mol Biol. 2004;337:647–660. doi: 10.1016/j.jmb.2004.01.061. [DOI] [PubMed] [Google Scholar]
- Kim S, Domon-Dell C, Wang Q, Chung DH, Di Cristofano A, Pandolfi PP, Freund JN, Evers BM. PTEN and TNF-α regulation of the intestinal-specific Cdx-2 homeobox gene through a PI3K, PKB/Akt, and NF-kappaB-dependent pathway. Gastroenterology. 2002;123:1163–1178. doi: 10.1053/gast.2002.36043. [DOI] [PubMed] [Google Scholar]
- Turck N, Gross I, Gendry P, Stutzmann J, Freund JN, Kedinger M, Simon-Assmann P, Launay JF. Laminin isoforms: biological roles and effects on the intracellular distribution of nuclear proteins in intestinal epithelial cells. Exp Cell Res. 2005;303:494–503. doi: 10.1016/j.yexcr.2004.10.025. [DOI] [PubMed] [Google Scholar]