Abstract
The human immunodeficiency virus (HIV) causes impairment of the immune system in part by targeting CD4+ T cells for infection and dysfunction. HIV envelope (Env) present on free virions and infected cells causes dysfunction of uninfected bystander CD4+ T cells via interaction with both CD4 and coreceptors. Env is commonly used as part of a cocktail of HIV antigens in current vaccines. In DNA and viral vector vaccine approaches, antigen-presenting cells (APCs) and non-APCs in the vicinity of the vaccine delivery site and draining lymph node express vaccine-derived antigens. The studies here demonstrate that cell-surface expression of Env on APCs and non-APCs as part of the vaccine action causes an inhibition of antigen-induced CD4+ T-cell activation and proliferation mediated by CD4 binding and suggests a potential mechanism for reduced activity of Env-containing HIV vaccines. Similar studies using a functional Env lacking CD4 binding circumvented suppression, suggesting an alternative and potentially superior approach to HIV vaccine design.
Introduction
It is estimated that 4.9 million new HIV infections occurred in 20051 mostly in regions of the world with limited access to health care. The development of a vaccine is the most effective approach to combat this epidemic. The envelope (Env) protein of the virus is a key component of an effective vaccine because it exclusively induces neutralizing antibodies, which are protective in the macaque model.2 However, Env has been demonstrated to suppress CD4+ T cells by multiple mechanisms. Inhibition has been characterized by a lack of proliferation,3,4 the induction of anergy,5,6 and various apoptotic mechanisms.7,8 The role of Env interactions with CD4 and coreceptors in mediating the various forms of suppression is under debate with studies demonstrating a role for each.9–12 R5 Env caused apoptosis of CD4+ T cells from a CCR5Δ32 homozygous donor,9 whereas, in another study, Env interaction with CXCR4 and a nonsignaling CD4 induced CD4+ T-cell apoptosis.13 The mechanism of suppression observed and the receptors involved varied according to the nature of Env used and the cell culture system studied. Env is a trimeric protein with each of its monomers possessing a CD4 binding site. Thus, an Env trimer4 or aggregates created by antibodies against monomeric gp120 subunits8 can mediate cross-linking of CD4 and/or coreceptor leading to signaling. In patients infected with HIV, trimeric Env capable of suppression is found on the surface of infected cells as well as on the virus itself. The use of Env in HIV vaccines, especially in commonly used DNA and viral vectors, could be envisioned to deliver similar CD4+ T-cell suppressive signals
Although antigen-presenting cells (APCs) such as dendritic cells (DCs) are prime targets for vaccines, other cell types at the site of vaccine delivery, and potentially the major vaccine antigen-expressing cell types, are non-APCs such as myocytes, fibroblasts, subepithelial, and epithelial cells for muscle, dermal, subcutaneous, and mucosal delivery, respectively, as well as cells in the draining lymph nodes.14,15 CD4+ T cells that are recruited to the site of vaccine delivery as well as to the draining lymph node in response to vaccine-induced inflammation may then interact with and be suppressed by interaction with Env expressed on the surface of all of these cells.
We designed an in vitro experimental model to mimic in vivo Env vaccine delivery. Our results demonstrate that APCs and non-APCs expressing Env of both R5 and X4 coreceptor usage mediated suppression of CD4+ T-cell activation and proliferation in response to antigen. In addition, we identify model X4 and R5 nonsuppressive Envs, which mediate fusion and infection, thus preserving important neutralizing epitopes, and present a new approach for more effective Env immunogens in vaccine design.
Materials and methods
Cell culture
Peripheral blood mononuclear cells (PBMCs) from HIV seronegative donors were obtained through an IRB-approved protocol. Approval was obtained from the University of Pennsylvania institutional review board for these studies. Informed consent was provided according to the Declaration of Helsinki. CD4+ T cells were purified by negative selection using the Human T-cell Subset Column kit (R & D Systems, Minneapolis, MN). Human embryonic kidney 293 cells (ATCC, Gaithersburg, MD) were propagated in DMEM supplemented with glutamine (Invitrogen, Carlsbad, CA) and 10% FCS (Hyclone, Ogden, UT) (complete medium). DCs were produced as described previously and treated with GM-CSF (50 ng/mL) + IL-4 (100 ng/mL) (R & D Systems) in AIM V medium (Invitrogen).16,17 In certain experiments, PBMCs or purified CD4+ T cells were stained with 3.33 μM CFSE (Molecular Probes, Carlsburg, CA) according to the manufacturer's protocol.
Plasmid constructs and RNA synthesis
Env genes from plasmids encoding IIIB, 8X, 8XD, 8XV3Bal, and 8XDV3Bal Envs18–20 were cloned into the RNA expression plasmid T7TS.21 Plasmids PCR3-89.6, pcDNA3-SF162, PCI-YU-2, and pcDNA3-JR-Fl containing upstream T7 promoter sites were obtained from the AIDS Reference and Reagent Program (Bethesda, MD). Plasmids were linearized at unique sites downstream of the 3′ coding sequence and in vitro transcribed with T7 polymerase to synthesize m7GpppG-capped mRNA with additional poly (A) tail using the mMessage T7 ultra kit (Ambion, Austin, TX). RNA was complexed to lipofectin and delivered to 293T cells as described.22
Cocultures of Env-expressing 293T cells and CFSE-labeled cells
293T cells in 6- or 12-well plates were transfected with Env or luciferase encoding mRNA or poly(AC) RNA (Sigma, St Louis, MO), incubated for 4 to 5 hours, and cocultured with 4 or 2 × 106 CFSE-labeled PBMCs or negatively selected purified CD4+ T cells with 0.1 μg/mL superantigen TSST-1 (toxic shock syndrome toxin-1; Sigma) or anti-CD3 mAb (0.5-1 μg/mL; Muromonab-CD3; Ortho Biotech, Bridgewater, NJ). In certain experiments, 2 μg/mL monomeric IIIB or SF162 gp-120, 20 to 80 U/mL IL-2 (AIDS Reference and Reagent Program), and/or 10 μg/mL sCD4 (Progenics, Tarrytown, NY) were added. Proliferation of CD4+ T cells by CFSE dilution was assayed by flow cytometry 4 to 5 days after initiation of coculture. CD4+ T cells were identified using anti-CD4 antibody conjugated to allophycocyanin (BD Pharmingen, San Jose, CA) or mouse anti–human CD4 (Clone OKT4) and goat anti–mouse Ig-allophycocyanin as a secondary antibody.
Detection of apoptosis and activation in CD4+ T cells exposed to Env
PBMCs were cocultured with Env-expressing 293T cells or 700 μM cycloheximide (CHX; Sigma) and activated with 0.1 μg/mL TSST-1 or 1 μg/mL anti-CD3 mAb. Apoptosis was examined 6, 17, and 36 hours after coculture and identified as annexin V–positive (BD Pharmingen, San Diego, CA) and 7-AAD–negative (Sigma) cells. Total cell death was measured by positive staining for 7-AAD in ungated cells. For studies measuring up-regulation of activation markers in Env-treated PBMCs, CD69 peridinin chlorophyll protein (day 1), HLA-DR fluorescein isothiocyanate and CD25 phycoerythrin cyanine 7 (BD Immunocytometry Systems, San Jose, CA) (day 2) were used.
Purified CD4+ T cells were exposed to IIIB Env-expressing 293T cells for 1 day, stimulated with 1 μg/mL anti-CD3 antibody for 10 minutes for p38 or 2 days followed by 20 U/mL IL-2 for 2 to 20 minutes for STAT5a, fixed, and analyzed by flow cytometry using anti–phospho-MAPK (pT180/pY182) Alexa Fluor 647 or anti-STAT5 (pY694) Alexa Fluor 647 (BD Pharmingen), respectively. For AKT phosphorylation, PBMCs were cocultured with IIIB Env-expressing 293T cells for 2 hours in complete DMEM and then for 18 hours in DMEM 0.5% serum, stimulated with 1 μg/mL anti-CD3 antibody for 10 minutes, fixed, and stained with anti–phospho-AKT (ser 473) Alexa Fluor488 (Cell Signaling Technologies, Danvers, MA) and anti-CD4 v4 (clone L120) phycoerythrin (BD Immunocytometry Systems).
DC infection with vaccinia virus
DCs were suspended at a concentration of 2 million/mL and infected with vaccinia-encoding IIIB, MN, or 92UG037.8 Env (NIH AIDS Research and Reference Reagent Program), T7 Polymerase (VTF7.3),23 or with wild-type WR strain for 2 hours, washed, and incubated for 4 hours. Infected DCs were cocultured with CFSE-labeled PBMCs at a ratio of 1:10 and activated with 0.1 ng/mL TSST-1.
Protein expression by fusion assay and Western blot
The protocol previously used by Nussbaum et al24 and Rucker et al25 was used for fusion assays between mRNA-encoding Env-transfected 293T cells and QT6 cells expressing CD4 and/or CXCR4 or CCR5. For Western blot analysis, 25 μg protein in lysates from Env- or luciferase-transfected 293T cells were analyzed using rabbit polyclonal anti-IIIB Env serum.26
Results
Env induces suppression of CD4+ T-cell proliferation
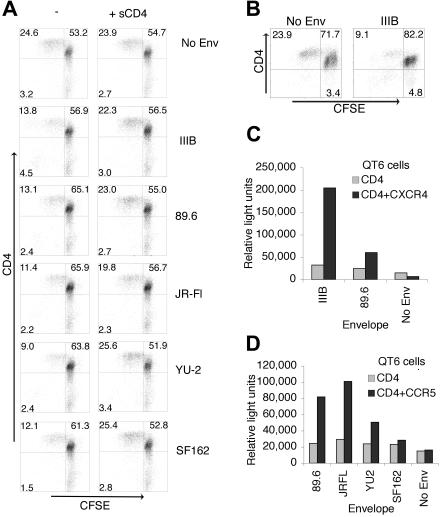
Following vaccine delivery, a number of cell types other than APCs take up and express vaccine-derived antigens. Expression by these cells presents Env in the absence of costimulatory molecules. 293T cells were transfected with Env-encoding mRNA as consistent, and low-level protein expression is obtained in most cells,17 allowing uniform exposure of CD4+ T cells to Env. PBMCs were used as a source of CD4+ T cells and cocultured with 293T cells expressing X4-tropic IIIB, dual tropic 89.6, and M-tropic JR-Fl, YU-2, and SF162 Envs. Cells were stimulated with a concentration of anti-CD3 mAb that typically stimulates one quarter of the CD4+ T cells. To monitor proliferation, PBMCs were stained with CFSE (5-and 6-carboxyfluorescein diacetate, succinimidyl ester).27 CD4+ T cells that encountered any of the Envs had lower proliferation levels compared with cultures exposed to control RNA-transfected 293T cells (Figure 1A). Similar inhibition was observed when cells were activated by the superantigen TSST-1, a model antigen for activation via the T-cell receptor (TCR), which requires the same costimulatory and adhesion molecules as conventional antigens.28,29 This suggested that suppression of proliferation was not dependent on a particular mode of activation. In addition, negatively selected purified CD4+ T cells exposed to Env also failed to proliferate, arguing against a role for CD8+ T cells and natural killer cells in mediating suppression (Figure 1B).
Figure 1.
HIV Env suppresses anti–CD3-activated CD4+ T-cell proliferation. (A) 293T cells expressing IIIB, 89.6, YU-2, SF162, and JR-Fl Envs or no Env (poly(A, C) RNA transfected) were cocultured with CFSE-labeled PBMCs with or without sCD4. CD4+ T cells were activated with anti-CD3 mAb (0.5 μg/mL), stained with anti-CD4 mAb, and monitored for proliferation 4 days after coculture by flow cytometry. (B) Negatively selected purified CD4+ T cells were cocultured with 293T cells expressing IIIB Env or no Env and activated using anti-CD3 mAb (0.5 μg/mL). Fusion assays demonstrated surface expression of functional Env. Encoding mRNA-transfected 293T cells were mixed with QT6 cells expressing CD4 and CXCR4 (C) or CCR5 (D), and fusion of the 2 cell populations was measured by luciferase production. Quadrants are labeled with the percent of events in each. Experiments shown are representative of 3 independent experiments.
To characterize the signal delivered by Env, we examined whether suppression could be overcome by blocking Env interaction with CD4. Soluble (s)CD4 almost completely abrogated Env-induced suppression (Figure 1A). P values were calculated using results from 3 independent experiments. A paired Student t test with 2 tails was performed using the percentage of change from control across experiments (no Env) or sCD4 treatment and is shown in Table 1.
Table 1.
Statistical analysis of suppression of CD4+ T cells proliferation by Env
| Env strain | P of suppression compared with no Env control | P of suppression compared with sCD4 treatment |
|---|---|---|
| IIIB | < .005 | < .005 |
| 89.6 | .01 | .02 |
| JR-F1 | .02 | .04 |
| Yu-2 | .01 | .02 |
P values were calculated using results from 3 independent experiments. Percentage of change from control (no Env) or sCD4 treatment was used in a paired Student t test with 2 tails.
Expression of functional trimeric Env on 293T cells was confirmed in fusion assays demonstrating that mRNA delivery resulted in surface expression of fusion-competent Env (Figure 1C-D). Delivery of Env-containing vaccines in vivo results in a variety of expression levels that are governed by the cellular vaccine target and the vaccine delivery method. In our experimental system that mimics vaccination with Env, we chose delivery of Env by encoding mRNA to 293T cells, which results in expression of Env in nearly 100% of cells at much lower levels compared with other methods of delivery, such as DNA. This was important as lymphocyte trafficking does not occur in our system. Thus, if only 25% of the cells expressed Env, then only about 25% of the CD4+ T cells added would be exposed. This low level of expression was sufficient to mediate suppression, and, in fact, a 5-fold reduction in the quantity of Env encoding mRNA delivered which results in a 5-fold lower level of protein expressed resulted in similar levels of suppression (data not shown). Thus, at the range of expression levels seen in our culture system significantly large variations in the amount of Env expressed did not alter the degree of suppression exerted. This could explain the lack of correlation observed between the degree of suppression mediated by an Env strain (Figure 1A) and the amount of fusion mediated (Figure 1C-D).
Several reports have found that trimeric or aggregated forms of Env are required to mediate suppression.5,8 However, others have found that monomeric gp120 could exert suppressive effects.3,7 In our system, CD4+ T cells were exposed to trimeric cell-surface–expressed Env as well as monomeric gp120 released into the supernatant, likely from dissociation of cell-surface gp160. PBMCs exposed exclusively to exogenous monomeric gp120 derived from IIIB or SF162 failed to suppress CD4+ T-cell proliferation (Table 2). The added gp120 retained the ability to block binding of CD4 mAb (leu-3a) demonstrating functional binding to CD4. This confirmed that trimeric surface expressed Env-mediated suppression in our system. Thus, transfection of 293T cells with encoding mRNA resulted in surface expression of functional Env, which mediated inhibition of antigen-specific CD4+ T-cell proliferation in a CD4-dependent manner.
Table 2.
Effect of monomeric gp120 on CD4+ T-cell proliferation
| No Env | IIIB gp120 | SF162 gp120 | |
|---|---|---|---|
| CD4+ cells undergoing proliferation, % | 15.3 | 14.4 | 12.5 |
| CD4− cells undergoing proliferation, % | 7.7 | 7.4 | 7.3 |
Anti-CD3 mAb (0.5 μg/mL) stimulated CFSE-labeled PBMCs were exposed to 2 μg/mL IIIB or SF162 gp120 or left untreated. CD4+ T cells were monitored for proliferation 4 days after coculture by flow cytometry and expressed as percentage of total CD4+ T cells.
DCs expressing Env suppress CD4+ T-cell proliferation
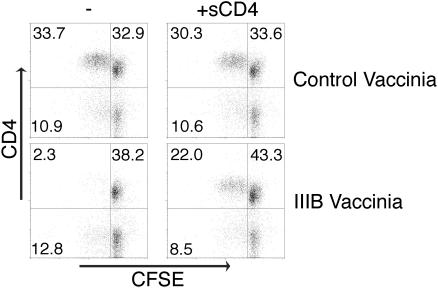
Presentation of vaccine-derived antigens by APCs activates immune responses and is the basis of an effective vaccine. APCs, such as DCs in their mature form, express a variety of costimulatory molecules on their surface and provide a strong activation signal for T and B cells.30 Certain studies have shown that activation, including potent costimulation, could overcome suppression induced by CD4 cross-linking.31 Other studies have not supported this, demonstrating that HIV-infected DCs were able to suppress CD4+ T cells in an Env-dependent fashion.3 Currently, viral vector or nucleic acid delivery of encoding antigen to DCs (either directly in vivo or after in vitro culture and loading of DCs) is in human trials and represents a potent vaccine approach.32,33 We infected DCs with vaccinia virus encoding various Envs34,35 and tested their ability to affect CD4+ T-cell proliferation in response to superantigen. DCs expressing the X4 Envs IIIB and MN and the R5 Env 92UG037.8 (a Clade A primary isolate) were found to inhibit CD4+ T-cell proliferation to antigen, which was overcome by sCD4 (Figure 2; data not shown). The P values comparing IIIB Env to no Env or sCD4 treatment were less than .005. Expression of Env by these types of vectors in vitro has been previously demonstrated.34–36 Very low multiplicities of infection (MOIs) of vaccinia were used as T-cell toxicity was observed at MOIs typically used in fusion assays. However, even at low MOIs (0.3-0.6) a minority of the suppression could not be overcome with sCD4. This Env-independent suppression of T-cell proliferation was also observed with 2 control viruses, vTF7-323 encoding T7 Polymerase and WR, the strain used to create all of the Env-expressing and control viruses. We concluded that vaccinia viruses had a small but significant level of toxicity in our system, which accounts for the inability of sCD4 to completely overcome the suppression of proliferation. In addition, the ability of Env to interact with CD4 molecules on DCs and suppress their maturation and function cannot be ruled out in our system as it has been observed in other systems.37,38 Expression of functional Env was confirmed in fusion assays between DCs and Quail cells (data not shown). Thus, mature DCs expressing Env mediate suppression of antigen-specific CD4+ T-cell proliferation despite expressing multiple costimulatory molecules.
Figure 2.
DCs expressing Env and presenting antigen suppress CD4+ T-cell proliferation. DCs were infected with vaccinia virus encoding IIIB Env or control vaccinia encoding T7 polymerase and cocultured with CFSE-labeled PBMCs with 0.1 ng/mL TSST-1 and with or without sCD4. This concentration of superantigen requires presentation by DCs as no proliferation is observed in their absence. Proliferation of CD4+ T cells was measured 5 days later by flow cytometry. Quadrants are labeled with the percent of events in each. Experiments shown are representative of 3 independent experiments.
Neither apoptosis nor IL-2–dependent anergy mediate CD4+ T-cell suppression
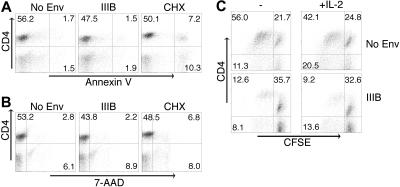
Env-induced CD4+ T-cell dysfunction has been described as anergy, apoptosis, cell death, or a failure to proliferate.3–8,39 We observed that Env suppresses CD4+ T cells in our system by hindering proliferation. We next investigated whether anergy or apoptosis occurred. PBMCs were cocultured with Env-expressing 293T cells and were activated using TSST-1 superantigen. Six, 18, and 36 hours later, CD4+ T cells were analyzed for early apoptosis using annexin V staining of 7-AAD–negative cells. CHX is a protein synthesis inhibitor that induces apoptosis of PBMCs and was used as a positive control. Env failed to induce apoptosis of CD4+ T cells above the level seen in cells unexposed to Env (Figure 3A). In addition, an increase in cell death (7-AAD uptake in ungated cells) in Env-exposed CD4+ T cells was not observed (Figure 3B). Thus, apoptosis and cell death are not major contributors to the suppression of proliferation induced by Env in our system.
Figure 3.
Neither apoptosis nor IL-2–dependent anergy mediate Env-induced CD4+ T-cell suppression. PBMCs were cocultured with IIIB Env or luciferase-expressing 293T cells or 700 μM cycloheximide (CHX) and activated with 0.1 μg/mL TSST-1. Thirty-six hours later, apoptotic CD4+ T cells were analyzed by flow cytometry and defined as Annexin V positive and 7-AAD negative for early apoptotic cells (A) and 7-AAD positive for dead cells (B). (C) CFSE-labeled PBMCs were cocultured with IIIB Env-expressing 293T cells in the presence of 80 U/mL IL-2 and activated with 0.1 μg/mL TSST-1. CD4+ T-cell proliferation was measured 5 days later by flow cytometry. Quadrants labeled with percent of events in each. Experiments shown are representative of 3 independent experiments.
T-cell anergy is defined as a state of nonresponsiveness to activation with antigen. The addition of exogenous IL-2 overcomes most but not all types of anergy.40 Selliah and Finkel39 observed that CD4+ T-cell suppression induced by Env could be overcome with exogenous IL-2. To investigate this mechanism, CFSE-stained PBMCs were cultured with control or Env-expressing 293T cells and activated with TSST-1 in the presence of increasing amounts of exogenous IL-2 (20, 40, or 80 U/mL). IL-2 was unable to prevent suppression of proliferation of CD4+ T cells by Env (Figure 3C; data not shown), arguing against this form of anergy as the mechanism of suppression.
CD4+ T cells activated in the presence of Env phosphorylate AKT, MAPK, and STAT5a but fail to completely up-regulate activation markers
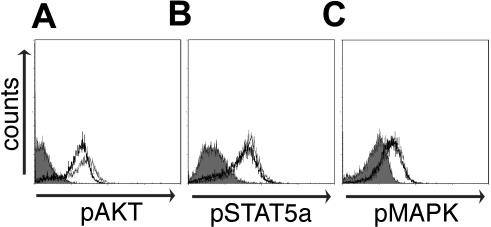
CD4+ T cells exposed to Env failed to proliferate in response to antigen but did not apoptose, anergize, or die. We next sought to determine the location of the block in the activation cascade. In response to T-cell activation, phosphorylation of key signaling molecules p38 MAPK and AKT relay the TCR signal to downstream mediators.41,42 CD4+ T cells exposed to Env and activated by anti–CD3-phosphorylated AKT and MAPK similar to control-treated cells (Figure 4). IL-2 is an important cytokine involved in T-cell proliferation, and its binding to both the low- and high-affinity IL-2Rs leads to the phosphorylation of STAT5a.43 Exogenous IL-2 was unable to circumvent the suppression of proliferation induced by Env (Figure 3C), but cells exposed to and suppressed by Env were able to phosphorylate STAT5a in response to IL-2 (Figure 4), suggesting that early signaling mediators in the IL-2 cascade also were not impaired.
Figure 4.
CD4+ T cells activated in the presence of Env phosphorylate AKT, p38 MAPK, and STAT5a but fail to up-regulate the activation markers CD69, CD25, and HLA-DR. PBMCs were cocultured with Env or control RNA-expressing 293T cells for 24 hours. Phosphorylation of AKT or MAPK 10 minutes after anti-CD3 (1.0 μg/mL) stimulation was measured using flow cytometry with gating on CD4+ T cells. Purified CD4+ T cells were exposed to control RNA or Env-expressing 293T cells and activated using anti-CD3 in the absence of IL-2. Two days later, cells were stimulated with 20 U/mL IL-2 and analyzed for STAT5a phosphorylation 7 minutes later. CD4+ T cells exposed to IIIB Env-transfected 293T cells and stained with isotype control mAb (shaded gray) or specific phosphoprotein mAb (thin line) or specific phosphoprotein mAb-stained control-transfected 293T cells (thick line) are shown for each signaling molecule. Experiments shown are representative of 3 independent experiments.
On activation, CD4+ T cells go through a series of events prior to proliferation, including up-regulation of markers of activation such as CD69, HLA-DR, and CD25.44 PBMCs were exposed to IIIB En-expressing 293T cells and activated using anti-CD3 or superantigen. Up-regulation of CD69 (day 1) and HLA-DR and CD25 (day 2) was monitored using flow cytometry. CD4+ T cells exposed to Env were unable to completely up-regulate their activation markers (Table 3; data not shown). Thus, although exposure of CD4+ T cells to Env did not suppress the phosphorylation of immediate signaling mediators, it suppressed the up-regulation of early and late cell-surface markers of activation and proliferation.
Table 3.
Effect of HIV Env on activation marker expression
| No Env | IIIB Env | |
|---|---|---|
| CD4+ cells expressing, % | ||
| CD69 | 78.9 | 56.6 |
| HLA-DR | 23.5 | 12.9 |
| CD25 (IL2R-α) | 60.4 | 31.6 |
| CD4− cells expressing, % | ||
| CD69 | 59.2 | 61.8 |
| HLA-DR | 43.7 | 41.6 |
| CD25 (IL2R-α) | 33.3 | 22.2 |
PBMCs were exposed to control or IIIB Env expressing 293T cells and activated with anti-CD3. Expression of CD69 on day 1 and HLA-DR and CD25 on day 2 on CD4+ and CD4− T cells was determined using flow cytometry.
Functional Envs lacking CD4 binding do not suppress antigen-induced CD4+ T cells
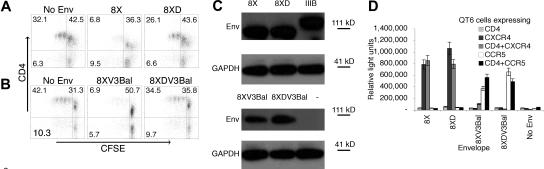
Env-induced suppression could be overcome by preventing its interaction with CD4 (Figure 1A). We hypothesized that an Env lacking the ability to bind CD4 could serve as a better immunogen by circumventing CD4+ T-cell suppression. The chemokine receptor binding site is an important neutralization region.45 Thus, mutations that abrogate coreceptor binding, such as a loss of CD4binding that is required to expose these regions, might adversely affect generation of neutralizing antibodies.46–48 The 8X Env is a CD4-independent derivative of IIIB, which stably exposes its chemokine receptor binding site and mediates fusion and infection in the absence of CD4 but still binds CD4.18–20,49 A single amino acid mutation in the CD4 binding site of Env (368D to R) prevents binding.20 We investigated whether this mutation in 8X would avoid suppression. The 8XD Env is identical to 8X except for a single amino acid loss of function mutation in its CD4 binding site.20 Switching their V3 regions with the V3 region of Ba-L to generate R5 using Envs developed 8XV3Bal and 8XDV3Bal. The CD4 binding Envs 8X and 8XV3Bal suppressed proliferation, whereas the non-CD4 binding but otherwise identical 8XD and 8XDV3Bal Envs completely circumvented Env-induced inhibition of proliferation (Figure 5). Thus, a single loss of function mutation in the CD4 binding site was sufficient to overcome suppression. Similar amounts of Env protein in 8X and 8XD and 8XV3Bal and 8XDV3Bal transfected 293T cells were confirmed by Western blot (Figure 5C). These Envs possess fewer glycosylation sites and hence have a lower molecular weight compared with IIIB. In addition, surface expression of functional Envs was confirmed in a fusion assay (Figure 5D). Although the fusion assay is not quantitative, the observation that similar levels of fusion as well as similar amounts of total Env protein are produced after mRNA transfection (Figure 5C) in comparing 8XV3Bal with 8XDV3Bal or 8X with 8XD, suggests that similar levels of surface expression were present. The X4 8XD Env and R5 8XDV3Bal Env could serve as superior model immunogens by circumventing CD4+ T-cell suppression while retaining important neutralization epitopes.
Figure 5.
Functional Envs lacking CD4 binding do not suppress antigen-induced CD4+ T-cell proliferation. 293T cells were transfected with 8X or 8XD Env-encoding mRNA; (A) 8XV3Bal or (B) 8XDV3Bal Env-encoding mRNA, and control poly(AC) RNA and cocultured with CFSE-labeled PBMCs. CD4+ T cells were activated with 0.1 μg/mL TSST-1, and proliferation was measured 4 days later using flow cytometry. P values were .01 (A) and less than .005 (B) for comparison to sCD4 treatment. P values were less than .005 in comparing 8XD and 8XDV3Bal with no Env control. Quadrants are labeled with the percent of events in each. (C) Expression of Env protein by mRNA-transfected 293T cells was confirmed by Western blot analysis. (D) Surface expression of functional Env on mRNA-transfected 293T cells was determined using fusion assays. Error bars represent the standard error of the mean. Experiments shown are representative of 3 independent experiments.
Discussion
The quest for an effective HIV vaccine faces many hurdles, including difficulty in developing strong and broad immune responses and the ability of the virus to evade immune recognition. The system studied here models the encounter of CD4+ T cells with both APCs and non-APCs expressing Env delivered as a vaccine. Both X4 and R5 Envs suppressed the activation and proliferation of CD4+ T cells via specific interaction with CD4. Suppression was characterized by a lack of proliferation despite normal activation of the main signaling pathways involved in T-cell activation and proliferation, AKT, MAPK, and STAT5a but a deficient up-regulation of early and late cell-surface activation markers. The suppression of antigen-induced proliferation was not due to IL-2–dependent anergy, apoptosis, or cell death.
The role of CD4 versus the coreceptors CXCR4 and CCR5 in mediating Env-induced suppression is controversial.9–12 Some reports have suggested a role for coreceptors in mediating suppression in conjunction with CD4.10,12 Others have suggested that Env can mediate suppression solely via CD4 or coreceptors.9,11,13 In this study, we show that Env delivered as a vaccine mediates a suppression of activation and proliferation regardless of its coreceptor usage. Although CD4 interaction is required for this suppression, both coreceptors could have played a role. To test this, we examined suppression of X4 Envs in the presence of AMD3100, which blocks Env interaction with CXCR4.50 AMD3100 caused a nonspecific increase in proliferation in both Env- and control-transfected 293T-exposed CD4+ T cells, which may be attributed to blocking inhibitory chemokine signaling through CXCR4.51 Similar studies with TAK-779,52 which blocks R5 Env interaction with CCR5, did not overcome suppression. This suggests that in our system studying vaccine-delivered Env in the absence of HIV infection, inhibition of CD4+ T-cell proliferation occurred in a CD4-dependent, chemokine receptor-independent manner.
Previous studies demonstrating the suppressive effects of Env have focused on a single aspect of CD4+ T-cell suppression such as apoptosis or anergy and used systems to model interactions that occur during HIV infection. In this report, we present a comprehensive study examining multiple facets of Env-induced suppression when it is used as an immunogen. DCs that acquire the vaccine will express protein from all the delivered antigens. When the protein is processed and presented to antigen-specific CD4+ T cells by the APCs, suppression by surface-expressed Env would occur, thus hindering immune responses to all antigens in the cocktail. This is the first study demonstrating that Env when used as a vaccine could suppress immune responses, making the findings highly relevant to the field of vaccine design. Because Env is an essential component in an HIV vaccine that aims to generate neutralizing antibodies, inclusion of nonsuppressive forms of Env will enhance immune responses.
DCs are prime targets for vaccines in vivo. Studies have shown a good correlation between the uptake of vaccine by DCs and the magnitude of the immune response induced.14 The studies demonstrated here suggest that DCs that acquire an Env-containing vaccine could actually hinder the immune response. This hypothesis is supported by a nonhuman primate study where animals were immunized with identical Gag and Pol antigens in the presence or absence of Env53 using a vaccine regimen of plasmid DNA prime and replication-incompetent adenoviral vector boost. Inclusion of Env in the cocktail led to a decrease in Gag- and Pol-specific IFN-γ responses in PBMCs.
We describe a modified Env that circumvents this suppression without losing important neutralizing epitopes. The X4 Env 8XD and the R5 8XDV3Bal have 2 key advantages as vaccine models. First, they have a single amino acid loss of function mutation in their CD4 binding site,20 thereby preventing suppression. Second, they have a comparatively open conformation with fewer glycosylation sites, thereby stably exposing their chemokine receptor binding site. Exposure of this important neutralization site could induce a better directed neutralizing antibody response against this conserved site.45 These specific Envs would not become a component of a new vaccine approach, because effective vaccines would include primary isolate Envs of suitable Clade and coreceptor specificity. The production of such an Env that shares these properties can be easily accomplished. HIV strains possessing the appropriate Env can be serially passaged in cell lines expressing decreasing amounts of CD4 to generate a CD4-independent variant.39 Env can then be cloned from the CD4-independent strain and mutated at a single amino acid (368 D to R) to abrogate CD4 binding. This results in an Env of suitable Clade and coreceptor specificity which is nonsuppressive because it lacks CD4 binding and should also stably expose its coreceptor binding site by virtue of its CD4 independence, possibly leading to enhanced neutralizing antibody responses.
HIV is an immunosuppressive virus and mediates its immune destructive activities in a number of manners. This includes direct killing of T helper cells, dysfunction of APCs and bystander T cells, and direct and indirect effects on almost all arms of the innate and acquired immune system. One major difficulty in developing an effective HIV vaccine is the lack of immune correlates of protection. A second that we demonstrate in this study is that immunogens, including Env used in HIV vaccines, are intrinsically immunosuppressive. We demonstrate that HIV Env, the only antigen capable of inducing neutralizing antibodies, inhibits the proliferation of CD4+ T cells in response to antigen. This inhibition to T helper cell proliferation is independent of the antigen stimulating the CD4+ T cells but occurs when the APCs or other cells in the region of the delivered vaccine take up the vaccine and express Env. Using an Env that is mutated to prevent CD4 binding but stably expresses the coreceptor binding site avoided this T-cell dysfunction. Model animal studies to determine whether this vaccine approach results in more potent immune responses are needed.
Acknowledgments
HIV-1 IIIB gp120; HIV envelope plasmids PCR3-89.6, pcDNA3-SF162, PCI-YU-2, and pcDNA3-JR-Fl; and vaccinia viruses vP1198, vP1489, and vP1174 (Virogenics Corp) were obtained through the NIH AIDS Research and Reference Reagent Program, Division of AIDS, NIAID, NIH. The polyclonal anti-IIIB Env rabbit serum and the vaccinia vTF7-3 were kind gifts from Dr Robert Doms and Dr Stuart Isaacs, University of Pennsylvania.
This work was supported by the National Institutes of Health (grants AI-50484 and AI-060505).
Footnotes
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Authorship
Contribution: K.F. designed and performed the research, analyzed the data, and wrote the paper; H.H. performed the research and analyzed the data; H.N. performed the research; J.A.H. designed the research and contributed vital new reagents; D.W. designed the research, analyzed the data, and wrote the paper.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Drew Weissman, Division of Infectious Diseases, 3610 Hamilton Walk, Philadelphia, PA 19104; e-mail: dreww@mail.med.upenn.edu.
References
- 1.UNAIDS/WHO. AIDS epidemic update. [Accessed on February 3, 2006]; Available at http://www.unaids.org/epi/2005/doc/report_pdf.asp.
- 2.Mascola JR, Lewis MG, Stiegler G, et al. Protection of Macaques against pathogenic simian/human immunodeficiency virus 89.6PD by passive transfer of neutralizing antibodies. J Virol. 1999;73:4009–4018. doi: 10.1128/jvi.73.5.4009-4018.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kawamura T, Gatanaga H, Borris DL, Connors M, Mitsuya H, Blauvelt A. Decreased stimulation of CD4+ T cell proliferation and IL-2 production by highly enriched populations of HIV-infected dendritic cells. J Immunol. 2003;170:4260–4266. doi: 10.4049/jimmunol.170.8.4260. [DOI] [PubMed] [Google Scholar]
- 4.Schwartz O, Alizon M, Heard JM, Danos O. Impairment of T cell receptor-dependent stimulation in CD4+ lymphocytes after contact with membrane-bound HIV-1 envelope glycoprotein. Virology. 1994;198:360–365. doi: 10.1006/viro.1994.1042. [DOI] [PubMed] [Google Scholar]
- 5.Liegler TJ, Stites DP. HIV-1 gp120 and anti-gp120 induce reversible unresponsiveness in peripheral CD4 T lymphocytes. J Acquir Immune Defic Syndr. 1994;7:340–348. [PubMed] [Google Scholar]
- 6.Masci AM, Galgani M, Cassano S, et al. HIV-1 gp120 induces anergy in naive T lymphocytes through CD4-independent protein kinase-A-mediated signaling. J Leukoc Biol. 2003;74:1117–1124. doi: 10.1189/jlb.0503239. [DOI] [PubMed] [Google Scholar]
- 7.Westendorp MO, Frank R, Ochsenbauer C, et al. Sensitization of T cells to CD95-mediated apoptosis by HIV-1 Tat and gp120. Nature. 1995;375:497–500. doi: 10.1038/375497a0. [DOI] [PubMed] [Google Scholar]
- 8.Banda NK, Bernier J, Kurahara DK, et al. Crosslinking CD4 by human immunodeficiency virus gp120 primes T cells for activation-induced apoptosis. J Exp Med. 1992;176:1099–1106. doi: 10.1084/jem.176.4.1099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Arthos J, Cicala C, Selig SM, et al. The role of the CD4 receptor versus HIV coreceptors in envelope-mediated apoptosis in peripheral blood mononuclear cells. Virology. 2002;292:98–106. doi: 10.1006/viro.2001.1266. [DOI] [PubMed] [Google Scholar]
- 10.Berndt C, Mopps B, Angermuller S, Gierschik P, Krammer PH. CXCR4 and CD4 mediate a rapid CD95-independent cell death in CD4(+) T cells. Proc Natl Acad Sci U S A. 1998;95:12556–12561. doi: 10.1073/pnas.95.21.12556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Holm GH, Gabuzda D. Distinct mechanisms of CD4+ and CD8+ T-cell activation and bystander apoptosis induced by human immunodeficiency virus type 1 virions. J Virol. 2005;79:6299–6311. doi: 10.1128/JVI.79.10.6299-6311.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Vlahakis SR, Algeciras-Schimnich A, Bou G, et al. Chemokine-receptor activation by env determines the mechanism of death in HIV-infected and uninfected T lymphocytes. J Clin Invest. 2001;107:207–215. doi: 10.1172/JCI11109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Roggero R, Robert-Hebmann V, Harrington S, et al. Binding of human immunodeficiency virus type 1 gp120 to CXCR4 induces mitochondrial transmembrane depolarization and cytochrome c-mediated apoptosis independently of Fas signaling. J Virol. 2001;75:7637–7650. doi: 10.1128/JVI.75.16.7637-7650.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chattergoon MA, Robinson TM, Boyer JD, Weiner DB. Specific immune induction following DNA-based immunization through in vivo transfection and activation of macrophages/antigen-presenting cells. J Immunol. 1998;160:5707–5718. [PubMed] [Google Scholar]
- 15.Wolff JA, Malone RW, Williams P, et al. Direct gene transfer into mouse muscle in vivo. Science. 1990;247:1465–1468. doi: 10.1126/science.1690918. [DOI] [PubMed] [Google Scholar]
- 16.Sallusto F, Lanzavecchia A. Efficient presentation of soluble antigen by cultured human dendritic cells is maintained by granulocyte/macrophage colony-stimulating factor plus interleukin 4 and downregulated by tumor necrosis factor alpha. J Exp Med. 1994;179:1109–1118. doi: 10.1084/jem.179.4.1109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Weissman D, Ni H, Scales D, et al. HIV gag mRNA transfection of dendritic cells (DC) delivers encoded antigen to MHC class I and II molecules, causes DC maturation, and induces a potent human in vitro primary immune response. J Immunol. 2000;165:4710–4717. doi: 10.4049/jimmunol.165.8.4710. [DOI] [PubMed] [Google Scholar]
- 18.Edwards TG, Hoffman TL, Baribaud F, et al. Relationships between CD4 independence, neutralization sensitivity, and exposure of a CD4-induced epitope in a human immunodeficiency virus type 1 envelope protein. J Virol. 2001;75:5230–5239. doi: 10.1128/JVI.75.11.5230-5239.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hoffman TL, LaBranche CC, Zhang W, et al. Stable exposure of the coreceptor-binding site in a CD4-independent HIV-1 envelope protein. Proc Natl Acad Sci U S A. 1999;96:6359–6364. doi: 10.1073/pnas.96.11.6359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.LaBranche CC, Hoffman TL, Romano J, et al. Determinants of CD4 independence for a human immunodeficiency virus type 1 variant map outside regions required for coreceptor specificity. J Virol. 1999;73:10310–10319. doi: 10.1128/jvi.73.12.10310-10319.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kariko K, Kuo A, Barnathan E. Overexpression of urokinase receptor in mammalian cells following administration of the in vitro transcribed encoding mRNA. Gene Ther. 1999;6:1092–1100. doi: 10.1038/sj.gt.3300930. [DOI] [PubMed] [Google Scholar]
- 22.Kariko K, Kuo A, Barnathan ES, Langer DJ. Phosphate-enhanced transfection of cationic lipid-complexed mRNA and plasmid DNA. Biochim Biophys Acta. 1998;1369:320–334. doi: 10.1016/s0005-2736(97)00238-1. [DOI] [PubMed] [Google Scholar]
- 23.Elroy-Stein O, Moss B. Cytoplasmic expression system based on constitutive synthesis of bacteriophage T7 RNA polymerase in mammalian cells. Proc Natl Acad Sci U S A. 1990;87:6743–6747. doi: 10.1073/pnas.87.17.6743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Nussbaum O, Broder CC, Berger EA. Fusogenic mechanisms of enveloped-virus glycoproteins analyzed by a novel recombinant vaccinia virus-based assay quantitating cell fusion-dependent reporter gene activation. J Virol. 1994;68:5411–5422. doi: 10.1128/jvi.68.9.5411-5422.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Rucker J, Doranz BJ, Edinger AL, Long D, Berson JF, Doms RW. Cell-cell fusion assay to study role of chemokine receptors in human immunodeficiency virus type 1 entry. Methods Enzymol. 1997;288:118–133. doi: 10.1016/s0076-6879(97)88011-1. [DOI] [PubMed] [Google Scholar]
- 26.Lorenzo MM, Herrera E, Blasco R, Isaacs SN. Functional analysis of vaccinia virus B5R protein: role of the cytoplasmic tail. Virology. 1998;252:450–457. doi: 10.1006/viro.1998.9483. [DOI] [PubMed] [Google Scholar]
- 27.Lyons AB, Parish CR. Determination of lymphocyte division by flow cytometry. J Immunol Methods. 1994;171:131–137. doi: 10.1016/0022-1759(94)90236-4. [DOI] [PubMed] [Google Scholar]
- 28.Scales D, Ni H, Shaheen F, Capodici J, Cannon G, Weissman D. Nonproliferating bystander CD4+ T cells lacking activation markers support HIV replication during immune activation. J Immunol. 2001;166:6437–6443. doi: 10.4049/jimmunol.166.10.6437. [DOI] [PubMed] [Google Scholar]
- 29.Shoukry NH, Lavoie PM, Thibodeau J, D'Souza S, Sekaly RP. MHC class II-dependent peptide antigen versus superantigen presentation to T cells. Hum Immunol. 1997;54:194–201. doi: 10.1016/s0198-8859(97)00074-8. [DOI] [PubMed] [Google Scholar]
- 30.Steinman RM. The dendritic cell system and its role in immunogenicity. Annu Rev Immunol. 1991;9:271–296. doi: 10.1146/annurev.iy.09.040191.001415. [DOI] [PubMed] [Google Scholar]
- 31.Tateyama M, Oyaizu N, McCloskey TW, Than S, Pahwa S. CD4 T lymphocytes are primed to express Fas ligand by CD4 cross-linking and to contribute to CD8 T-cell apoptosis via Fas/FasL death signaling pathway. Blood. 2000;96:195–202. [PubMed] [Google Scholar]
- 32.Goepfert PA, Horton H, McElrath MJ, et al. High-dose recombinant Canarypox vaccine expressing HIV-1 protein, in seronegative human subjects. J Infect Dis. 2005;192:1249–1259. doi: 10.1086/432915. [DOI] [PubMed] [Google Scholar]
- 33.Mwau M, Cebere I, Sutton J, et al. A human immunodeficiency virus 1 (HIV-1) clade A vaccine in clinical trials: stimulation of HIV-specific T-cell responses by DNA and recombinant modified vaccinia virus Ankara (MVA) vaccines in humans. J Gen Virol. 2004;85:911–919. doi: 10.1099/vir.0.19701-0. [DOI] [PubMed] [Google Scholar]
- 34.Gao F, Morrison SG, Robertson DL, et al. Molecular cloning and analysis of functional envelope genes from human immunodeficiency virus type 1 sequence subtypes A through G. The WHO and NIAID Networks for HIV Isolation and Characterization. J Virol. 1996;70:1651–1667. doi: 10.1128/jvi.70.3.1651-1667.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Perkus ME, Limbach K, Paoletti E. Cloning and expression of foreign genes in vaccinia virus, using a host range selection system. J Virol. 1989;63:3829–3836. doi: 10.1128/jvi.63.9.3829-3836.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Jacotot E, Callebaut C, Blanco J, Riviere Y, Krust B, Hovanessian AG. HIV envelope glycoprotein-induced cell killing by apoptosis is enhanced with increased expression of CD26 in CD4+ T cells. Virology. 1996;223:318–330. doi: 10.1006/viro.1996.0483. [DOI] [PubMed] [Google Scholar]
- 37.Fantuzzi L, Purificato C, Donato K, Belardelli F, Gessani S. Human immunodeficiency virus type 1 gp120 induces abnormal maturation and functional alterations of dendritic cells: a novel mechanism for AIDS pathogenesis. J Virol. 2004;78:9763–9772. doi: 10.1128/JVI.78.18.9763-9772.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Williams MA, Trout R, Spector SA. HIV-1 gp120 modulates the immunological function and expression of accessory and co-stimulatory molecules of monocyte-derived dendritic cells. J Hematother Stem Cell Res. 2002;11:829–847. doi: 10.1089/152581602760404630. [DOI] [PubMed] [Google Scholar]
- 39.Selliah N, Finkel TH. Cutting edge: JAK3 activation and rescue of T cells from HIV gp120-induced unresponsiveness. J Immunol. 1998;160:5697–5701. [PubMed] [Google Scholar]
- 40.Schwartz RH. T cell anergy. Annu Rev Immunol. 2003;21:305–334. doi: 10.1146/annurev.immunol.21.120601.141110. [DOI] [PubMed] [Google Scholar]
- 41.Bauer B, Baier G. Protein kinase C and AKT/protein kinase B in CD4+ T-lymphocytes: new partners in TCR/CD28 signal integration. Mol Immunol. 2002;38:1087–1099. doi: 10.1016/s0161-5890(02)00011-1. [DOI] [PubMed] [Google Scholar]
- 42.Dong C, Davis RJ, Flavell RA. MAP kinases in the immune response. Annu Rev Immunol. 2002;20:55–72. doi: 10.1146/annurev.immunol.20.091301.131133. [DOI] [PubMed] [Google Scholar]
- 43.Johnston JA, Bacon CM, Finbloom DS, et al. Tyrosine phosphorylation and activation of STAT5, STAT3, and Janus kinases by interleukins 2 and 15. Proc Natl Acad Sci U S A. 1995;92:8705–8709. doi: 10.1073/pnas.92.19.8705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Dannecker G, Mahlknecht U, Schultz H, Hoffmann MK, Niethammer D. Activation of human T cells by the superantigen Staphylococcus enterotoxin B: analysis on a cellular level. Immunobiology. 1994;190:116–126. doi: 10.1016/S0171-2985(11)80287-1. [DOI] [PubMed] [Google Scholar]
- 45.Decker JM, Bibollet-Ruche F, Wei X, et al. Antigenic conservation and immunogenicity of the HIV coreceptor binding site. J Exp Med. 2005;201:1407–1419. doi: 10.1084/jem.20042510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Lapham CK, Ouyang J, Chandrasekhar B, Nguyen NY, Dimitrov DS, Golding H. Evidence for cell-surface association between fusin and the CD4-gp120 complex in human cell lines. Science. 1996;274:602–605. doi: 10.1126/science.274.5287.602. [DOI] [PubMed] [Google Scholar]
- 47.Trkola A, Dragic T, Arthos J, et al. CD4-dependent, antibody-sensitive interactions between HIV-1 and its co-receptor CCR-5. Nature. 1996;384:184–187. doi: 10.1038/384184a0. [DOI] [PubMed] [Google Scholar]
- 48.Wu L, Gerard NP, Wyatt R, et al. CD4-induced interaction of primary HIV-1 gp120 glycoproteins with the chemokine receptor CCR-5. Nature. 1996;384:179–183. doi: 10.1038/384179a0. [DOI] [PubMed] [Google Scholar]
- 49.Matthias LJ, Yam PT, Jiang XM, et al. Disulfide exchange in domain 2 of CD4 is required for entry of HIV-1. Nat Immunol. 2002;3:727–732. doi: 10.1038/ni815. [DOI] [PubMed] [Google Scholar]
- 50.Donzella GA, Schols D, Lin SW, et al. AMD3100, a small molecule inhibitor of HIV-1 entry via the CXCR4 co-receptor. Nat Med. 1998;4:72–77. doi: 10.1038/nm0198-072. [DOI] [PubMed] [Google Scholar]
- 51.Vlahakis SR, Villasis-Keever A, Gomez T, Vanegas M, Vlahakis N, Paya CV. G protein-coupled chemokine receptors induce both survival and apoptotic signaling pathways. J Immunol. 2002;169:5546–5554. doi: 10.4049/jimmunol.169.10.5546. [DOI] [PubMed] [Google Scholar]
- 52.Baba M, Takashima K, Miyake H, et al. TAK-652 inhibits CCR5-mediated human immunodeficiency virus type 1 infection in vitro and has favorable pharmacokinetics in humans. Antimicrob Agents Chemother. 2005;49:4584–4591. doi: 10.1128/AAC.49.11.4584-4591.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Letvin NL, Huang Y, Chakrabarti BK, et al. Heterologous envelope immunogens contribute to AIDS vaccine protection in rhesus monkeys. J Virol. 2004;78:7490–7497. doi: 10.1128/JVI.78.14.7490-7497.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]