Abstract
Dendritic cells (DCs) and natural killer (NK) cells are essential components of the innate immune system and have a central role in initiation and regulation of adaptive immune responses. During the early critical immune activities, DCs and NK cells interact and reciprocally regulate each other via cell-cell contact. The molecular mediators of the DC–NK-cell crosstalk are largely undefined. In the present study, we show in mice that DC stimulation of NK-cell IFN-γ secretion requires DC membranebound but not secreted products; is increased by augmenting the expression of DC transmembrane tumor necrosis factor (tmTNF) and NK-cell transmembrane TNF receptor type 2 (tmTNFR2); is inhibited by blocking TNF or TNFR2 but not TNFR1; is impaired by knocking out DC Tnf or NK-cell Tnfr2 but not DC Tnfr1 or Tnfr2 and NK-cell Tnf or Tnfr1; and is restored in TNF-deficient DCs by reconstituting tmTNF, but cannot be mimicked by soluble TNF. We also demonstrate that DC TNF and NK-cell TNFR2 are required for DC-mediated NK-cell proliferation and amplification of cytotoxic activity. These novel findings provide the first evidence that DC–NK-cell crosstalk mediates enhancement of NK-cell functions via triggering NK-cell tmTNFR2 by DC tmTNF.
Introduction
Dendritic cells (DCs) and natural killer (NK) cells are essential effector cells of the innate immune system that rapidly recognize and directly eliminate microbial pathogens and transformed cells, induce inflammation, and initiate, polarize, and regulate adaptive immune responses.1–9 Recent studies have shown that DCs and NK cells enhance and regulate their functions via interaction and reciprocal stimulation.10–20 NK cells induce in DCs maturation, and increases in expression of the major histocompatibility complex (MHC) and costimulatory molecules, and secretion of IL-12p70. Reciprocally, DCs induce in NK cells activation, and proliferation, augmentation of perforin-mediated cytotoxicity and IFN-γ secretion. The specifically amplified DC and NK-cell functions empower the innate immune system with increased abilities to eliminate infected and transformed cells, induce Th1-type adaptive immune responses and, consequently, efficiently contain infections and cancer. In vivo, DCs and NK cells colocalize and possibly interact at the sites of inflammation and in lymph nodes.6,21–23
The mechanisms and mediators that govern DC–NK-cell interaction are obscure. Both soluble and transmembrane molecules have been implicated in DC–NK-cell interaction. However, while secreted cytokines such as IL-12, IL-18, IL-2, and type I interferons have been examined and inconsistently found to be involved in DC–NK-cell interaction, the transmembrane molecules have not been explored so far.10,14,20,24–28 If secreted cytokines are major mediators of DC–NK-cell interaction, the interaction should efficiently occur both in cell-cell contact and following their physical separation, which enables the cell communication via secreted molecules and excludes the involvement of transmembrane molecules. Contrarily, it has been consistently shown that the interaction only occurs efficiently when DCs and NK cells are in direct contact but not physically separated.10,14–16 These findings indicate that transmembrane molecules might be initiators and early mediators of DC–NK-cell interaction. However, the secreted cytokines might be induced during the early process and could contribute to the interaction later during the process.
Both DCs and NK cells express transmembrane TNF (tmTNF),29,30 which is a homotrimeric 26 kDa type II protomer protein. tmTNF can be cleaved by the specific membranebound metalloprotease TNF-alpha-converting enzyme ([TACE] ADAM-17) and secreted as a 17 kDa soluble homotrimeric cytokine (soluble TNF [sTNF]). TNF mediates vitally important cell contact–dependent functions such as structural and functional organization of secondary lymphoid organs, apoptosis, inflammation, and immunoregulation.31,32 The diverse functions are induced via cognate interactions between TNF and its 2 transmembrane receptors, TNF receptor type 1 (TNFR1, TNFRSF1A, p55/60, CD120a) and TNF receptor type 2 (TNFR2, TNFRSF1B, p75/80, CD120b), which have structurally distinct intracellular domains and therefore activate different signaling pathways. TNFR1 contains a C-terminal region of 80 amino acids named “death domain” that binds TNFR1-associated protein with death domain (TRADD) and signals activation of both caspases/apoptosis and NF-κB. In contrast, TNFR2 has within the intracellular domain a C-terminal region of 78 amino acids that binds TNFR-associated factor 2 (TRAF2) and an N-terminal RING-finger motif and signals activation of NF-κB and expression of cytokine genes. Interestingly, while TNFR1 can be activated by both tmTNF and sTNF, TNFR2 can be only activated by tmTNF.31,32
TNF mediates DC maturation and NK-cell activation,1,2,4,5,33,34 the 2 important biologic responses that are reciprocally induced in DC–NK-cell interaction. In humans, not only tmTNF is expressed on both DCs and NK cells, but also TNFR2 is expressed on NK cells.29,30,35 In addition, anti-TNF neutralizing antibodies have been shown to decrease the ability of NK cells to stimulate the increase in DC CD86 expression in humans, indicating that NK-cell TNF could contribute to DC maturation stimulated in DC–NK-cell interaction.14,15 Together, the evidence suggests that tmTNF could be a mediator of DC–NK-cell contact interaction and both NK cell–induced maturation of DCs and DC-induced activation of NK cells. In the present study, we examined the role of TNF and its cognate receptors in DC-induced activation of NK cells in mice. We defined a novel mechanism of DC–NK-cell crosstalk, which is nonsecretory and mediated via cell-cell contact and cognate interaction of DC tmTNF and NK-cell tmTNFR2.
Materials and methods
Mice
Eight-week-old wild-type female C57BL/6 (H-2kb/I-Ab) mice were obtained from Taconic (Germantown, NY). TNF-, TNFR1-, and TNFR2-deficient (TNF−/−, TNFR1−/−, and TNFR2−/−, respectively) female mice with C57BL/6 background were purchased from The Jackson Laboratory (Bar Harbor, ME). Mice were housed at the University of Pittsburgh Cancer Institute's Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) International–accredited animal facility. The animal studies were performed in accordance with the protocols approved by the University of Pittsburgh Institutional Animal Care and Use Committee.
Reagents
The following reagents were used: low-toxic rabbit complement (Cedarlane Laboratories, Hornby, ON, Canada); mouse recombinant GM-CSF and IL-4 (R&D Systems, Minneapolis, MN); human recombinant IL-2 (Chiron, Emeryville, CA); mouse recombinant sTNF (Alexis Biochemicals, San Diego, CA); Escherichia coli lipopolysaccharide (LPS) (Sigma Chemical, St Louis, MO); hybridomas producing rat anti–mouse CD4, CD8, and CD45/B220 monoclonal antibodies (mAbs) (American Type Culture Collection [ATCC], Rockville, MD); MACS NK-Cell and DC Isolation Kits (Miltenyi Biotec, Auburn, CA); fluorochrome-conjugated anti–mouse NK1.1, DX5, CD69, CD3, CD14, CD19, CD11c, CD11b, CD205, CD80, CD86, CD40, H-2kb, and I-Ab mAb and corresponding isotype control mAb (BD PharMingen, San Diego, CA); rat anti–mouse TNF mAb (Pierce Endogen, Rockford, IL); rabbit anti–mouse TNFR1 and TNFR2 antibodies and control Ig (Santa Cruz Biotechnology, Santa Cruz, CA); Armenian hamster anti–mouse TNFR1 and TNFR2 and isotype control mAb (BD PharMingen); mouse anti–β-actin mAb (Sigma Chemical); biotin- and peroxidase-conjugated goat anti–rat and goat anti–rabbit IgG antibodies (Pierce Endogen); R-phycoerythrin–conjugated streptavidin (Jackson ImmunoResearch Laboratories, West Grove, PA); mouse Quantikine TNF, TNFR2, and IFN-γ enzyme-linked immunosorbent assay (ELISA) kits (R&D Systems); and adenoviral vectors containing the green fluorescence protein (GFP) and mouse TNF genes (2 × 1010 plaque-forming units (pfu)/mL) (University of Pittsburgh Vector Core Facility).
Generation of noncleavable TNF cDNA
TNF cDNA lacking the TACE cleavage site was generated by deletion of the nucleotides coding for the amino acids 77 to 92 in the mouse wild-type Tnf cDNA (InvivoGen, San Diego, CA) using QuickChange II Site-Directed Mutagenesis Kit (Stratagene, La Jolla, CA). The oligonucleotides primers used for deletion mutagenesis were 5′-CATCAGTTCTATGGCCCAGGCCCACGTCGTAGCAAACC-3′ (sense), and 5′-GGTTTGCTACGACGTGGGCCTG GGCCATAGAACTGATG-3′ (antisense).
Purification of fresh DCs
CD11c+ DCs were purified from spleen-cell suspensions by positive immunoselection using MACS DC Isolation Kit as recommended by the manufacturer. The purified DCs were more than 90% CD11c+CD3−NK1.1−CD14−CD19− and expressed low levels of CD80, CD86, CD40, and MHC class I and MHC class II molecules and were referred as in vivo–generated, freshly isolated immature DCs (fiDCs).
Generation of DCs in vitro
Myeloid immature DCs were produced by 5-day culturing of mature cell–depleted bone marrow cells (0.1 × 106/mL) in the presence of recombinant mouse GM-CSF and IL-4 (15 ng/mL each). Thus, generated DCs were 95% CD11c+CD205+/−lineage marker−. They expressed the CD80, CD86, CD40, and MHC class I and MHC class II molecules at low to intermediate levels. These DCs did not secrete IL-12p70 and IL-18 either unstimulated or following stimulations with LPS, TNF, or NK cells and only secreted IL-12p70 following stimulation with the cocktail IL-4, LPS, IFN-γ, and CD40L. They were referred as in vitro–generated, cytokine-stimulated immature DCs (ciDCs). DCs gained the ability to secrete IL-12p70 in response to LPS alone following 7 days of culture. Mature DCs (mDCs) were produced by 6- to 24-hour LPS stimulation of fiDCs or ciDC and were referred to as fmDCs and cmDCs, respectively. TNF−/−, TNFR1−/−, and TNFR2−/− ciDCs showed normal growth and expression of the costimulatory and MHC molecules.
Purification of fresh NK cells
NK1.1+CD3− cells were purified from splenocytes by negative immunoselection using MACS NK Cell Isolation Kit as recommended by the manufacturer. These cells were more than 75% NK1.1+CD3−CD69− and were referred to as in vivo–generated freshly isolated resting NK (fNK) cells.
Activation and expansion of NK cells
Purified fNK cells (0.1 × 106/mL) were supplemented with 6000 IU/mL IL-2 and cultured for 6 days. In this culture, NK cells expanded 10- to 20-fold and were more than 97% NK1.1+CD3−CD69+. They were referred to as cultured IL-2–activated NK (caNK) cells. fNK cells were also cultured with IL-2 for 1 to 24 hours and referred to as fresh aNK (faNK) cells.
DC–NK-cell cocultures, gene transfer, and functional testing
fiDCs and fNK cells, as well as ciDCs and caNK cells, were seeded individually or combined in a 1:1 ratio (0.5 × 106/mL each) in 96-well round bottom plates. In some experiments, DCs and NK cells were cocultured in 24-well plates equipped with Transwell inserts having semiporous membrane (0.2 μm pore diameter; Corning, Corning, NY). To inhibit DC secretory activity and preserve cell membrane–bound molecules, ciDCs and cmDCs were fixed with 1% (wt/vol) paraformaldehyde for 30 minutes on ice. The cells were additionally cultured for 24 hours or various periods of time in the absence or presence of the 200 ng/mL LPS and/or 6000 IU/mL IL-2. In the Tnf gene overexpression or restoration experiments, wild-type or TNF−/− ciDCs were transduced with the GFP or mouse wild-type Tnf by their coincubation with 60 pfu per cell of the adenoviral vectors containing the genes at 37°C for 1 hour. In addition, TNF−/− ciDCs were transduced with recombinant plasmids containing either the wild-type or noncleavable Tnf using lipofectamine transfection reagent and Opti-MEM I reduced serum medium (Invitrogen, Carslbad, CA) following the manufacturer's protocol. Transduced DCs were seeded into 24-well plates (0.5 × 106/mL) and cultured for 24 hours to allow expression of the transduced gene–encoded proteins and then assessed in the 24-hour cocultures. In blocking experiments, wild-type mouse ciDCs and caNK cells were cocultured for 24 hours in the absence or presence of isotype-control mAb or anti-TNF, anti-TNFR1, and/or anti-TNFR2 mAb (20 μg/mL). Following the cell cultures, cell-culture–conditioned media were collected and assessed for TNF and/or IFN-γ using ELISA. In addition, cell lysates were obtained by 30 minutes of incubation of DCs and NK cells on ice in radioimmunoprecipitation assay (RIPA) lysis buffer (Upstate, Lake Placid, NY). The cell lysates were assessed for the presence of TNF and TNFR2 using ELISA. Data were presented as means of triplicates ± SD of means of TNF, TNFR2, and IFN-γ nanograms per 0.5 × 106 cells per milliliter. NK-cell proliferation and cytotoxicity were measured using standard 18-hour [3H]thymidine incorporation and 4-hour 51Cr release assays, respectively.36,37
Flow cytometry
Phenotype of DCs and NK cells was determined using direct 2-color flow cytometry. Cell-surface expression of TNF, TNFR1, and TNFR2 by DCs and NK cells was examined using the previously described indirect 1-color flow cytometry assays.29,30
Western blot
Western blot was performed as previously described.36
Statistics
The statistical significances of differences between the results were tested using the exact 1-sided Wilcoxon test. The results were considered significantly different with P values less than .05.
Results
Cell membrane–bound but not secreted molecules are required for DC stimulation of NK cells
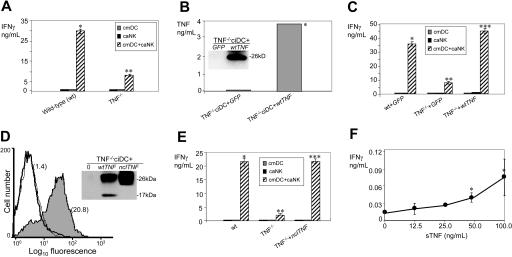
Previous studies have provided compelling evidence that DCs only effectively mediate NK-cell activation in direct cell-cell contact but not physically separated.10,14 This indicates that an activity of locally secreted or cell-membrane–expressed molecules in the cleft of the DC–NK-cell contact area mediates DC stimulation of NK cells. To assess these possibilities, we compared the abilities of viable (secretion and transmembrane molecule competent) and paraformaldehyde-fixed (secretion incompetent, transmembrane molecule competent)37 ciDCs and cmDCs to stimulate caNK cells to secrete IFN-γ. We found that fixed ciDCs and cmDCs mediated perfectly normal stimulation of IFN-γ secretion by caNK cells (Figure 1A). This finding strongly suggests that DC stimulation of NK cells is initiated and mediated mainly by cell membrane–bound molecules and that secreted molecules participate little if at all in the early activity.
Figure 1.
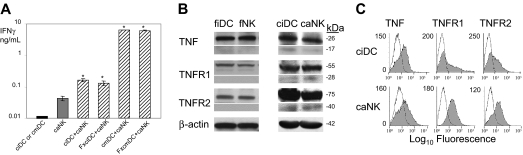
DCs stimulate NK-cell IFN-γ secretion via transmembrane but not secreted molecules, and DCs and NK cells express on cell-surface transmembrane TNF and TNFRs. (A) Secretion-deficient DCs mediate NK-cell IFN-γ secretion. Viable or paraformaldehyde-fixed ciDCs (ciDC and FxciDC, respectively) and 6-hour LPS-prestimulated viable or formaldehyde-fixed ciDCs (cmDC and FxcmDC, respectively) and viable caNK cells (caNK) were plated into 96-well plates either alone or together in a 1:1 ratio and incubated for 24 hours. Following the culture, cell-culture–conditioned media were assessed by IFN-γ ELISA. Data are from a representative experiment of 2 similar experiments performed. The results are means ± SD of triplicates IFN-γ nanograms per 0.5 × 106 cells per milliliter. Asterisks indicate significant increases in IFN-γ secretion in DC+NK-cell cocultures versus DC or NK-cell cultures alone. (B) Transmembrane but not soluble TNF, TNFR1, and TNFR2 are expressed in fiDCs, fNK cells, ciDCs, and caNK cells. Western blot analyses of the DC and NK-cell lysates were performed using 120 μg cell proteins per lane. Relative quantities of the proteins in the Western blotting bands were determined by densitometry. Protein bands are as follows: 26 kDa, tmTNF; 17 kDa, sTNF; 55 kDa, tmTNFR1; 28 kDa, sTNFR1; 75 kDa, tmTNFR2; 40 kDa, sTNFR2; and 42 kDa, β-actin. (C) TNF, TNFR1, and TNFR2 are expressed on cell surface of ciDCs and caNK cells. Flow cytometry analyses of viable ciDCs and caNK cells were performed. The results are log10 fluorescence intensity obtained with isotype-matched nonreactive control antibodies (open histograms) and specific antibodies (filled histograms).
DCs express tmTNF, while NK cells express tmTNFRs
Next, we examined whether DCs and NK cells express tmTNF and tmTNFRs that are potentially capable of mediating this cellular interaction via cell-cell contact. We found a substantial presence of the 26 kDa tmTNF, 55 kDa tmTNFR1, and 75 kDa tmTNFR2 proteins in fiDCs and ciDCs and fNK and caNK cells. In contrast, the 17 kDa sTNF, 28 kDa sTNFR1, or 40 kDa sTNFR2 proteins were not found in the fresh cells and were found in the cultured cells in 10- to 15-fold smaller quantity than their transmembrane forms. tmTNF and tmTNFRs were at 3- to 7-fold higher levels in the cultured than fresh cells (Figure 1B). In addition, fiDCs and fNK cells (see data in “Expression of tmTNF on DCs and TNFRs on NK cells directly correlates with DC and NK-cell abilities to crosstalk”) and ciDCs and caNK cells (Figure 1C) expressed these molecules on the cell surface, but fresh cells did this at lower levels. These findings show that DCs and NK cells express significant levels of tmTNF, tmTNFR1, and tmTNFR2 on the cell surface but contain, if at all, a small amount of the secreted molecules. Therefore, both in vivo– and in vitro–generated DCs and NK cells are potentially able to interact upon cell-cell contact by engaging tmTNF and tmTNFRs. Specifically, tmTNF expressed on DCs might engage tmTNFRs expressed on NK cells and induce biologic responses of NK cells such as IFN-γ secretion.
Expression of tmTNF on DCs and tmTNFRs on NK cells is associated with the ability of DCs and NK cells to reciprocally stimulate IFN-γ secretion
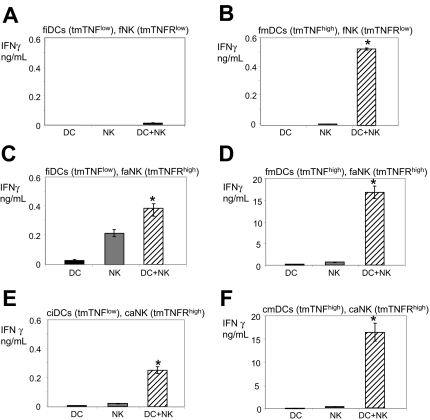
We noticed that ciDCs express more tmTNF than fiDCs, fmDCs and cmDCs express more tmTNF than fiDCs and ciDCs, and faNK and caNK cells express more tmTNFR1 and tmTNFR2 than fNK cells (Figure 1 and “Expression of tmTNF on DCs and TNFRs on NK cells directly correlates with DC and NK-cell abilities to crosstalk”). We initially assessed the association of expression levels of DC tmTNF and NK-cell tmTNFRs with the ability of these cells to reciprocally stimulate IFN-γ secretion. We found that fiDCs (low tmTNF) did not stimulate significant IFN-γ secretion in fNK cells (low TNFRs) (Figure 2A); fmDCs (high tmTNF) induced medium-level IFN-γ secretion in fNK cells (low TNFRs) (Figure 2B); fiDCs (low tmTNF) stimulated medium-level IFN-γ secretion in faNK cells (high TNFRs) (Figure 2C); fmDCs (high tmTNF) induced high-level IFN-γ secretion in faNK cells (high tmTNFRs) (Figure 2D); ciDCs (low tmTNF) induced medium-level IFN-γ secretion in caNK cells (high tmTNFRs) (Figure 2E); and cmDCs (high tmTNF) induced high-level IFN-γ secretion in caNK cells (high tmTNFRs) (Figure 2F), which was similar to that induced by fmDCs in faNK cells (Figure 2D). These data show an association between the expression of DC tmTNF and NK-cell tmTNFRs with the ability of DCs and NK cells to reciprocally stimulate IFN-γ secretion. They also demonstrate that both in vivo– and in vitro–generated DCs and NK cells can reciprocally stimulate each other and that this activity can be rapidly enhanced by stimuli that induce increases in expression of tmTNF on DCs and tmTNFRs on NK cells.
Figure 2.
DC-mediated stimulation of IFN-γ secretion by NK cells corresponds to the expression levels of DC tmTNF and NK-cell tmTNFRs. (A) Unstimulated fiDCs and fNK cells. (B) LPS-stimulated fmDCs and fNK cells. (C) IL-2–stimulated fiDCs and faNK cells. (D) LPS- and IL-2–stimulated fmDCs and faNK cells. (E) Unstimulated ciDCs and caNK cells. (F) LPS-stimulated cmDCs and caNK cells. The fiDCs and fNK cells (A-D) and ciDC and caNK cells (E-F) were incubated either alone (DC, NK) or together (DC+NK) in a 1:1 ratio without (A,C,E) or with (B,D,F) 200 ng/mL LPS and/or without (A-B,E-F) or with (C-D) 6000 IU/mL IL-2 for 24 hours. At the end of these cultures, cell-culture–conditioned media were assessed by IFN-γ ELISA. Data are from 1 representative experiment of 2 to 12 similar experiments performed. The results are means ± SD of triplicates IFN-γ nanograms per 0.5 × 106 cells per milliliter. Asterisks indicate significant increases in IFN-γ secretion in DC+NK-cell cocultures versus DC or NK-cell cultures alone.
Expression of tmTNF on DCs and TNFRs on NK cells directly correlates with DC and NK-cell abilities to crosstalk
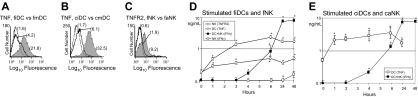
To more directly assess the relationships between the expression of tmTNF on DCs and tmTNFRs on NK cells and the abilities of DCs and NK cells to reciprocally stimulate IFN-γ secretion, we comparatively examined time courses of the LPS-induced increases in expression of tmTNF on fiDCs and ciDCs, IL-2–induced increases in expression of TNFR1 and TNFR2 on fNK cells, and LPS- and/or IL-2–induced IFN-γ secretion in fiDC–fNK-cell and ciDC–caNK-cell cocultures (Figure 3). We found that LPS rapidly induced high increases in tmTNF expression on the cell surface of both fiDCs (Figure 3A,D) and ciDCs (Figure 3B,E). The responses of fiDC and ciDC to LPS were very similar in dynamics and magnitude. Similarly, IL-2 rapidly induced high increases in cell-surface expression of tmTNFR1 (data not shown) and tmTNFR2 (Figure 3C–D) on fNK cells. Importantly, fmDCs or cmDCs, respectively, induced high and similar increases in IFN-γ secretion in faNK cells (Figure 3D) and caNK cells (Figure 3E). The increases in secretion of IFN-γ coincided with the picks (4 to 24 hours of stimulation) of increases in tmTNF expression on fmDCs and cmDCs and in tmTNFR2 expression on faNK cells.
Figure 3.
Expressions of tmTNF by DCs and tmTNFRs by NK cells correlate with DC and NK-cell ability to cross-stimulate IFN-γ secretion. (A) LPS rapidly induces increases in cell-surface expression of TNF on fiDCs. fiDCs were either untreated (fiDC) or 6-hour LPS stimulated (fmDC). (B) LPS rapidly induces increases in cell-surface expression of TNF on ciDCs. ciDCs were either untreated (ciDC) or 6-hour LPS induced (cmDC). (C) IL-2 rapidly stimulates increases in cell-surface expression of TNFR2 on fNK cells. fNK cells were either untreated (fNK) or 6-hour IL-2 activated (faNK). Panels A-C present flow cytometry analyses of the cell-surface molecules (thin-line open histograms, isotype control mAb; thick-line open histograms, nonstimulated cells stained with specific mAb; filled histograms, stimulated cells stained with specific mAb). The numbers in parentheses are mean fluorescence intensity (MFI). (D) LPS-stimulated fiDCs and IL-2–stimulated fNK cells rapidly increase the expression of cell-associated TNF and TNFR2, respectively, and enhance their abilities to cross-stimulate IFN-γ secretion. fiDCs or fNK cells were cultured for the indicated periods of time either unstimulated (data not shown, no changes observed) or stimulated with LPS or IL-2, respectively. Following these cultures, the cell-associated TNF was measured in the cell lysates of DCs (DC (TNF)), and cell-associated TNFR2 was measured in cell lysates of NK cells (NK (TNFR2)) using ELISA. In parallel, fiDCs and fNK cells were incubated alone (NK-cell data are shown [NK (IFN-γ)]; DC data are negative, not shown) or together (DC+NK (IFN-γ)) in the presence of LPS and IL-2 for the indicated periods of time. The cell-culture–conditioned media were assessed by IFN-γ ELISA. Data are from an experiment. The results are means ± SD of triplicates TNF, TNFR2, and IFN-γ nanograms per 0.5 × 106 cells per milliliter. (E) LPS-stimulated ciDCs rapidly increase the expression of cell-associated TNF and acquire increased ability to stimulate caNK-cell IFN-γ secretion. ciDCs were cultured for the indicated periods of time either unstimulated (data not shown, no changes observed) or stimulated with LPS. Following these cultures, cell-associated TNF was measured in the cell lysates using ELISA (DC (TNF)). In parallel, ciDC and caNK cells were incubated alone (negative data, not shown) or together (DC+NK (IFN-γ)) in the presence of LPS for the indicated periods of time. The cell-culture–conditioned media were assessed by IFN-γ ELISA. Data are from a representative experiment of 3 similar experiments performed. The results are means ± SD of triplicates TNF and IFN-γ nanograms per 0.5 × 106 cells per milliliter. Asterisks in panels D-E indicate significant increases in the cell-associated proteins in stimulated (1 to 48 hours) versus unstimulated (0 hours) NK cells and/or DCs (NK (TNFR2); DC (TNF)) or in IFN-γ secretion in DC+NK-cell cocultures (DC+NK (IFN-γ)) versus NK-cell cultures alone.
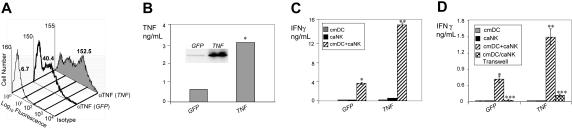
To more directly correlate DC tmTNF expression and DC ability to mediate NK-cell IFN-γ secretion, we examined the effects of Tnf transfer on ciDC tmTNF expression and stimulation of NK-cell IFN-γ secretion. We found that transfer of Tnf produced in ciDCs 4- to 5-fold increases in cell-surface expression of 26 kDa tmTNF (Figure 4A–B) and ability to stimulate caNK-cell IFN-γ secretion (Figure 4C). In addition, similar to nontransduced cmDCs,10,14 TNF-transduced cmDCs strongly stimulated IFN-γ secretion in caNK cells only when DCs and NK cells were coincubated in cell-cell contact but not physically separated (Figure 4D). Therefore, the increased stimulation of NK cells by TNF-transduced DCs is mediated preferentially by cell-membrane–associated molecules, similar to that of nontransduced DCs.
Figure 4.
Increases in expression of tmTNF induced by TNF gene transfer augment the ability of DCs to induce IFN-γ secretion by NK cells. (A) Transfer of Tnf results in increases in cell-surface expression of TNF on DCs. ciDCs were transduced with adenoviral vectors containing GFP (GFP) or wild-type Tnf (TNF) and stimulated with LPS for 6 hours, stained with isotype control mAb (isotype) and anti-TNF mAb (αTNF), and examined by flow cytometry. The numbers adjacent to histograms are MFI. (B) Transfer of Tnf results in increases in expression of tmTNF in ciDCs. Lysates of the GFP- or Tnf-transduced ciDCs were assessed for TNF using Western blot (inset) and ELISA (columns). Data are from one experiment. Western blot shows the 26 kDa tmTNF. The 17 kDa sTNF was 15-fold less expressed than tmTNF (data not shown). ELISA data are means of triplicates TNF nanograms per 0.5 × 106 cells per milliliter. (C) Transfer of Tnf leads to high increases in the ability of ciDCs to induce IFN-γ secretion by caNK cells. GFP- or Tnf-transduced ciDCs (cmDC) and untreated caNK cells (caNK) were cultured either alone or together (cmDC+caNK) in the presence of LPS for 24 hours. After this incubation, the cell-culture–conditioned media were assessed using IFN-γ ELISA. Data are from one experiment and represent means ± SD of triplicates IFN-γ nanograms per 0.5 × 106 cells per milliliter. (D) Tnf- transduced wild-type ciDCs are only capable of efficiently inducing secretion of IFN-γ in wild-type caNK cells in cell-cell contact but not physically separated. GFP- or Tnf-transduced ciDCs (cmDC) and untreated caNK cells (caNK) were cultured in the presence of 200 ng/mL LPS for 24 hours either alone or together in standard wells (cmDC+caNK) or physically separated in Transwells (cmDC/caNK Transwell). After this incubation, the cell-culture–conditioned media were assessed using IFN-γ ELISA. Data are from an experiment and represent means ± SD of triplicates IFN-γ nanograms per 0.5 × 106 cells per milliliter. In panel B, the single asterisk indicates a significant increase of cell-associated TNF in ciDCs transduced with Tnf compared with ciDCs transduced with GFP. In panels C and D, single asterisks indicate a significant increase in IFN-γ in cocultures of GFP-transduced cmDCs+caNK cells versus cultures of cmDCs or caNK cells alone. Double asterisks indicate a significant enhancement of IFN-γ in cocultures of TNF-transduced cmDCs+caNK cells versus cocultures of GFP-transduced cmDCs+caNK cells. Triple asterisks indicate significant decreases of IFN-γ in cocultures of cmDC/caNK cells separated by Transwell membrane versus cmDC+caNK-cell cocultures without separation.
These findings strongly suggest that DC tmTNF and NK-cell tmTNFRs might be involved in DC induction of IFN-γ secretion by NK cells.
tmTNF, but not sTNF, is an important mediator of DC–NK-cell crosstalk
To directly assess the role of TNF in DC stimulation of NK-cell IFN-γ secretion, we compared the abilities of wild-type and TNF−/− cmDCs to stimulate IFN-γ secretion by wild-type and TNF−/− caNK cells, respectively. We consistently found that cocultures of TNF−/− cmDCs and TNF−/− caNK cells produced 5- to 10-fold less IFN-γ than cocultures of wild-type cmDCs and wild-type caNK cells (Figure 5A). This profound defect of TNF−/− DCs and NK cells to cross-stimulate IFN-γ secretion shows that TNF is critically important for DC–NK-cell crosstalk.
Figure 5.
TNF−/− DCs and NK cells are impaired in the ability to reciprocally stimulate IFN-γ secretion, and the impairment can be fully repaired by restoring TNF−/− DCs with tmTNF but not sTNF. (A) TNF−/− DCs and TNF−/− NK cells have a remarkably decreased ability to reciprocally stimulate IFN-γ secretion. Wild-type cmDCs and wild-type caNK cells or TNF−/− cmDCs and TNF−/− caNK cells were cultured either alone or together for 24 hours. cmDCs were generated by LPS stimulation of ciDCs. Following this culture, the cell-culture–conditioned media were assessed for IFN-γ using ELISA. Data are from a representative experiment of 7 similar experiments performed. They are means ± SD of triplicates IFN-γ nanograms per 0.5 × 106 cells per milliliter. (B) Transfer of wild-type Tnf restores cell-associated tmTNF in TNF−/− DCs. TNF−/− ciDCs were transduced with adenoviral vectors containing GFP or wild-type Tnf (wtTNF). Cell lysates of transfected cells were assessed using Western blot assays (inset) and ELISA (columns). Data are from an experiment. Western blot was assessed with anti-TNF antibody. ELISA data are means of triplicates TNF nanograms per 0.5 × 106 cells per milliliter (SD is 0.2 ng). (C) Transfer of wild-type Tnf into TNF−/− DCs restores DC ability to stimulate IFN-γ secretion by TNF−/− NK cells. Wild-type ciDCs were transduced with adenoviral vector containing GFP (wt+GFP), and TNF−/− ciDCs were transduced with adenoviral vectors containing either GFP (TNF−/−+GFP) or wild-type Tnf (TNF−/−+wtTNF). The transduced wild-type or TNF−/− DCs and respective wild-type or TNF−/− untreated caNK cells were cultured either alone or together, in the presence of LPS, for 24 hours. Following this culture, the cell-culture–conditioned media were assessed by IFN-γ ELISA. Presented data are from one experiment and represent means ± SD of triplicates IFN-γ nanograms per 0.5 × 106 cells per milliliter. (D) Transfer of noncleavable Tnf restores cell-surface expression of tmTNF in TNF−/− DCs. TNF−/− ciDCs were transduced with the plasmid DNA containing either wtTnf or nclTnf. Transduced DCs were compared with mock-transduced TNF−/− ciDCs for the expression of tmTNF on cell surface using flow cytometry and Western blot (insert). Flow cytometry presents log10 fluorescence intensity of TNF−/− ciDCs either mock transduced or transduced with nclTnf and stained with isotype-matched control antibody (thin-line open histogram) or anti-TNF antibody (thick-line open histogram, mock transduced; filled histogram, nclTnf transduced). Numbers in parentheses are MFI. Western blot presents TNF forms in lysates of TNF−/− DCs either mock transduced (0) or transduced with wtTNF or nclTnf. Presented data are from one experiment. (E) Transfer of nclTnf fully restores the ability of TNF−/− DCs to stimulate IFN-γ secretion by TNF−/− NK cells. TNF−/− cmDCs mock transduced (TNF−/−) or transduced with nclTnf (TNF−/−+nclTNF) and nontransduced TNF−/− caNK cells were cultured alone or together for 24 hours. For comparison, the ability of nontransduced LPS-stimulated wild-type cmDCs to stimulate wild-type caNK cells was assessed (wt). Following this culture, the cell-culture–conditioned media were assessed by IFN-γ ELISA. Presented data are from an experiment and are means ± SD of triplicates IFN-γ nanograms per 0.5 × 106 cells per milliliter. (F) sTNF is a poor stimulator of NK-cell IFN-γ secretion. Wild-type caNK cells were incubated for 24 hours in the absence or presence of grading concentrations of mouse recombinant sTNF. Following the incubation, the cell-culture–conditioned media were collected and assessed by IFN-γ ELISA. Presented data are from one experiment and represent means ± SD of triplicates IFN-γ nanograms per 0.5 × 106 cells per milliliter. In panels A, C, and E, single asterisks indicate significant increases of IFN-γ in wild-type cmDC+wild-type caNK-cell cocultures in comparison with wild-type cmDC or caNK-cell cultures alone. In panel B, the single asterisk indicates a significant increase of cell-associated TNF in TNF−/− ciDCs transduced with wtTnf compared with TNF−/− ciDCs transduced with GFP. In panel F, single asterisks indicate significant increases of IFN-γ in sTNF-stimulated versus unstimulated caNK-cell cultures. Double asterisks indicate significant decreases of IFN-γ in cocultures of untreated, GFP-transduced, or mock-transduced TNF−/− cmDCs with TNF−/− untreated caNK cells in comparison with cocultures of untreated or GFP-transduced wild-type cmDCs with untreated wild-type caNK cells. Triple asterisks indicate significant increases of IFN-γ in cocultures of wtTnf- or nclTnf-transduced TNF−/− cmDCs with untreated TNF−/− caNK cells in comparison with cocultures of mock-transduced or GFP-transduced TNF−/− cmDCs with untreated TNF−/− caNK cells.
Because mDCs produce both tmTNF and sTNF, we examined which form of TNF is a mediator of the crosstalk. We reconstituted TNF−/− ciDCs either with both tmTNF and sTNF by transferring wild-type Tnf (wtTnf), or with only tmTNF by transferring noncleavable Tnf (nclTnf), and examined the generation of tmTNF and sTNF and the ability of the engineered DCs to stimulate IFN-γ secretion by TNF−/− caNK cells (Figure 5B–E). The TNF−/− ciDCs transduced with either wtTnf or nclTnf rapidly expressed on cell-surface 26-kDa tmTNF (Figure 5B,D and data not shown). However, while nclTnf-transduced TNF−/− ciDCs did not generate 17 kDa sTNF (Figure 5D, inset, and data not shown), wtTnf-transduced TNF−/− ciDCs did so (Figure 5D, inset; 0.4 to 2.0 ng/0.5 × 106 cells/mL). Similar to wtTnf-transduced TNF−/− DCs, nontransduced and especially wtTnf-transduced wild-type DCs produced 17 kDa sTNF (Figure 1B; 0.2 to 4.0 ng/0.5 × 106 cells/mL). Remarkably, the defect of TNF−/− DCs and NK cells to cross-stimulate increases in secretion of IFN-γ could be fully repaired by restoring in TNF−/− DCs either wtTnf (Figure 5B–C) or nclTnf (Figure 5D–E). These findings show that tmTNF is a critically important mediator of DC–NK-cell reciprocal stimulation of IFN-γ secretion and indicate that tmTNF of DCs but not of NK cells might be mediator of the function. They also suggest that sTNF might not play a significant role in DC–NK-cell crosstalk.
To directly assess the role of sTNF in DC-mediated stimulation of NK cells, we examined the effect of recombinant sTNF on wild-type caNK-cell IFN-γ secretion. We found that sTNF, even at concentration of 100 ng/mL, very poorly stimulated NK cells to secrete IFN-γ (Figure 5F). This stimulation was more than 300-fold lower than that induced by wild-type cmDCs or TNF−/− ciDCs transduced with wtTnf or nclTnf, which contained less than 4 ng tmTNF per 0.5 × 106 cells (Figures 3D–E, 4B–C, 5B–E and data not shown). Remarkably, the lowest tested concentration of sTNF (12.5 ng/mL) was 3- to 6-fold greater than the maximal concentration of sTNF found in our cmDC cultures but was ineffective in stimulating caNK-cell IFN-γ secretion. These findings demonstrate that sTNF does not play a significant role in DC–NK-cell crosstalk.
TNF-TNFR2 engagement mediates DC–NK-cell crosstalk
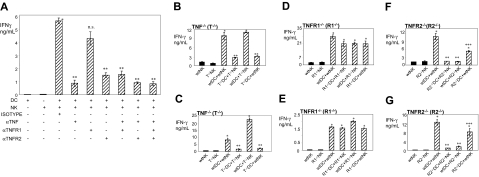
To further asses the importance of TNF and to determine the contribution of the signaling via TNFR1 and/or TNFR2 in DC-mediated stimulation of NK-cell IFN-γ secretion, we performed blocking experiments using antibodies to TNF, TNFR1, and TNFR2. We found that individual mAb to TNF and TNFR2 significantly (6.4- and 3.8-fold, respectively) suppressed IFN-γ secretion in wild-type cmDC–caNK-cell cocultures (Figure 6A). In contrast, mAb to TNFR1 did not significantly change this function, and combined mAb to TNF, TNFR2, and/or TNFR1 similarly suppressed DC–NK-cell crosstalk as individual mAb to TNF or TNFR2. These data confirm that TNF is a significant mediator of DC stimulation of NK-cell IFN-γ secretion and suggest that TNF-TNFR2 engagement and signaling via TNFR2 might be the main pathway of DC–NK-cell crosstalk and that the TNF-TNFR1 pathway does not play a significant role in this activity.
Figure 6.
Interaction between DC-TNF and NK-cell TNFR2 mediates increased secretion of IFN-γ by NK cells in DC–NK-cell crosstalk. (A) Disruption of TNF-TNFR2 but not TNF-TNFR1 interplay impairs DC–NK-cell cross-stimulation of IFN-γ secretion. cmDCs and caNK cells were cultured either alone or together in the presence of isotype control mAb (isotype), anti-TNF (αTNF), anti-TNFR1 (αTNFR1), and/or anti-TNFR2 (αTNFR2) mAb for 24 hours. Following these incubations, cell-culture–conditioned media were assessed by IFN-γ ELISA. Presented data are from 1 representative experiment of 4 similar experiments performed. They are means ± SD of triplicates IFN-γ nanograms per 0.5 × 106 cells per milliliter. Single asterisk indicates significant increase of IFN-γ secretion in DC+NK-cell cocultures treated with isotype control mAbs in comparison with DC or NK-cell cultures alone. Double asterisks indicate significant decrease of IFN-γ secretion in mAb-treated DC–NK-cell cocultures in comparison with isotype control or anti-TNFR1 mAb-treated DC–NK-cell cocultures. ns indicates nonsignificant difference between anti-TNFR1– and isotype control mAb-treated DC+NK-cell cocultures. (B-C) DC TNF, but not NK-cell TNF, is required for DC stimulation of IFN-γ secretion by NK cells. (D-E) Neither NK-cell nor DC TNFR1 is required for DC induction of IFN-γ secretion by NK cells. (F-G) NK-cell TNFR2, but not DC TNFR2, is required for most DC induction of IFN-γ secretion by NK cells. Wild-type (wt) or TNF−/− (B-C), TNFR1−/− (D-E), and TNFR2−/− (F-G) fiDCs and fNK cells (B,D,F) or ciDCs and caNK cells (C,E,G) were cultured for 24 hours in the presence of LPS (both fresh and cultured cells) and/or IL-2 (only fresh cells) either alone or in the following combinations: wild-type+wild-type, gene-deficient+gene-deficient, wild-type+gene-deficient, and gene-deficient+wild-type. After this incubation, cell-culture–conditioned media were assessed by IFN-γ ELISA. The presented data of fresh cells are from one experiment and of the cultured cells from a representative experiment of 2 to 3 similar experiments performed. They represent means ± SD of triplicates IFN-γ nanograms per 0.5 × 106 cells per milliliter. Single asterisks indicate significant increases of IFN-γ in DC+NK-cell cocultures in comparison with DC or NK-cell cultures alone. Double asterisks indicate significant decreases of IFN-γ in TNF−/−DC+TNF−/−NK-cell or TNF−/−DC+wtNK-cell and TNFR2−/−DC+TNFR2−/−NK-cell or wtDC+TNFR2−/−NK-cell cocultures in comparison with wtDC+wtNK-cell or wtDC+TNF−/−NK-cell and wtDC+wtNK-cell or TNFR2−/−DC+wtNK-cell cocultures, respectively. Triple asterisks indicate statistical significance of the small decrease of IFN-γ in TNFR2−/−DC+wtNK-cell cocultures in comparison with wtDC+wtNK-cell cocultures.
Cognate interaction of DC TNF and NK-cell TNFR2 mediates DC–NK-cell crosstalk
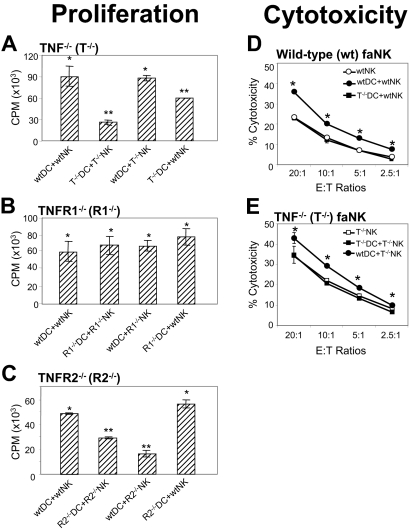
To precisely define the contributions of DC and NK-cell TNF, TNFR1, and TNFR2 in DC stimulation of NK cells and biologic relevance of this pathway of DC–NK-cell crosstalk, we examined the ability of wild-type or TNF−/−, TNFR1−/−, and TNFR2−/− fmDCs or cmDCs to respectively stimulate IFN-γ secretion by wild-type or TNF−/−, TNFR1−/−, and TNFR2−/− fNK and faNK or caNK cells (Figures S1 [available on the Blood website; see the Supplemental Figures link at the top of the online article] and 6B-G). We found that wild-type fmDCs or cmDCs induced high IFN-γ secretion by wild-type or TNF-deficient fNK and faNK or caNK cells, respectively. In sharp contrast, TNF-deficient fmDCs or cmDCs poorly stimulated IFN-γ secretion by TNF-deficient or wild-type fNK and faNK or caNK cells, respectively (Figures S1A and 6B–C). Complementarily, TNFR2-deficient fNK or faNK and caNK cells poorly secreted IFN-γ in response to either TNFR2-deficient or wild-type fmDCs and cmDCs, while wild-type fNK or faNK and caNK cells secreted high levels of IFN-γ in response to either wild-type or TNFR2-deficient fmDCs and cmDCs, respectively (Figures S1C and 6F–G). In sharp contrast, TNFR1-deficient or wild-type fNK or faNK and caNK cells secreted high levels of IFN-γ in response to either TNFR1-deficient or wild-type fmDCs and cmDCs, respectively (Figures S1B and 6D–E). Similarly, wild-type or TNFR1- and TNFR2-deficient fmDCs, but not TNF-deficient fmDCs, optimally stimulated proliferation of wild-type or TNF- and TNFR1-deficient fNK cells but not TNFR2-deficient fNK cells (Figure 7A–C), and wild-type ciDCs, but not TNF-deficient ciDCs, stimulated cytotoxic activity of wild-type or TNF-deficient fNK cells (Figure 7D–E). These data demonstrate that TNF-TNFR2 but not TNF-TNFR1 interaction mediates DC-induced enhancement of NK-cell functions and that this process occurs via selective interplay of DC TNF and NK-cell TNFR2. They also show that this mechanism could be rapidly activated by microbial products in in vivo–generated DCs and NK cells and might be therefore operative in vivo and biologically relevant.
Figure 7.
Interaction between DC-TNF and NK cell–TNFR2 mediates proliferation and increased cytotoxic activity of NK cells in DC–NK-cell crosstalk. (A) DC TNF, but not NK-cell TNF, is required for DC stimulation of NK-cell proliferation. (B) Neither NK-cell nor DC TNFR1 is required for DC induction of NK-cell proliferation. (C) NK-cell TNFR2, but not DC TNFR2, is required for DC induction of NK-cell proliferation. Wild-type (wt) or TNF−/− (A), TNFR1−/− (B), and TNFR2−/− (C) fiDCs (irradiated with 30 Gy [3000 rad]) and fNK cells were cultured for 4 days in the presence of 200 ng/mL LPS and 300 IU/mL IL-2 either alone or in the following combinations: wild-type+wild-type, gene-deficient+gene-deficient, wild-type+gene-deficient, and gene-deficient+wild-type. Following this incubation, the cell cultures were supplemented with [3H]thymidine (1 μCi/200μL [0.037 MBq/200 μL]), incubated for additional 18 hours, harvested, and the DNA-incorporated radioactivity measured in a β-scintillation counter. Data are from a representative experiment of 2 to 5 similar experiments performed. They represent means ± SD of triplicates counts per minute of cocultured DC+NK cells following subtraction of counts per minute of DCs and NK cells cultured alone. Single asterisks indicate significant increases of counts per minute in cocultured DCs+NK cells in comparison with counts per minute of alone-cultured DCs or NK cells. Double asterisks indicate significant decreases of counts per minute in cocultured TNF−/−DCs+TNF−/−NK cells or TNF−/−DCs+wtNK cells and TNFR2−/−DCs+TNFR2−/−NK cells or wtDCs+TNFR2−/−NK cells in comparison with wtDCs+wtNK cells or wtDCs+TNF−/−NK cells and wtDCs+wtNK cells or TNFR2−/−DC+wtNK cells, respectively. Similar findings to those presented in panel C were obtained in 2 independent experiments with wild-type and TNFR2−/− fiDC and fNK cells stimulated only by LPS (data not shown). (D-E) DC TNF, but not NK-cell TNF, is required for DC stimulation of NK-cell cytotoxic activity. Wild-type or TNF−/− ciDCs and 24-hour IL-2–activated faNK cells were cultured for 24 hours in the absence of cytokines or microbial products either alone or in the following combinations and 1:2 ratio: wild-type+wild-type, TNF−/−+TNF−/−, wild-type+TNF−/−, and TNF−/−+wild-type. Following this incubation, the cells were assessed for their NK cytotoxicity using the standard 4-hour 51Cr-release assays against YAC-1 tumor-cell targets. Data are from a representative experiment of 3 similar experiments performed. They represent means ± SD of triplicates percentages of killing by alone-cultured NK cells and cocultured DCs+NK cells. Single asterisks indicate significant increases in cytotoxicity of DCs+NK cells in comparison with those of NK cells alone.
Discussion
DCs stimulate the critically important NK-cell functions proliferation, cytotoxicity, and secretion of the central Th1-type cytokine IFN-γ and induce polarized Th1 adaptive immune responses to pathogens and transformed cells.3,4,8–10,14,16 The governing mechanisms of DC regulation of NK-cell functions are largely elusive. It has been suggested that DCs regulate NK cells via both transmembrane and secreted cytokines.10–20,24–28 This implies that the crosstalk occurs in the cleft of DC–NK-cell contact, is mediated by the molecules that are locally concentrated on the cell membrane and/or secreted, and is restricted on the cells in direct contact. Here, we demonstrate for the first time that DC–NK-cell contact–dependent crosstalk stimulating NK-cell IFN-γ secretion, proliferation, and cytotoxicity is mediated by a novel nonsecretory mechanism via selective engagement of cell-surface–expressed DC tmTNF and NK-cell tmTNFR2. Both in vivo– and in vitro–generated DCs and NK cells have this ability, indicating that the mechanism might be operative in vivo and therefore biologically important.
We show that while TNF−/− DCs are impaired and sTNF is unable to stimulate NK-cell IFN-γ secretion, either secretion-sufficient or secretion-deficient wild-type DCs or nclTNF-restored TNF−/− DCs are fully capable to do so, strongly suggesting that cell membrane tmTNF is a major mediator of DC–NK-cell interaction and arguing against a significant role for conventionally secreted cytokines such as sTNF and IL-12p70 at the onset of DC–NK-cell crosstalk. However, the secretion of cytokines might be induced during the initial, cell-contact–dependent phase of DC–NK-cell crosstalk via transmembrane molecules, and the secreted cytokines might be significantly involved during the late phase of the interaction.
Pathogen- and cytokine-stimulated DCs secrete IL-12p70, IL-23, IL-18, IL-15, and IL-2, which are potent activators of NK cells.38–40 Therefore, these cytokines have been studied as mediators of DC-induced NK-cell activation. These studies have provided conflicting findings.10,14,20,24–27 Thus, it has been shown in humans and mice that IL-12 is not required for DC-mediated stimulation of NK-cell CD69 expression, IFN-γ secretion, cytotoxicity, or antitumor activity.10,14,25 In contrast, it has been demonstrated in mice that cytomegalovirus-induced expansion of antiviral LY49H+ NK cells is mediated by CD8α+ lymphoid DCs via IL-12 20 and that myeloid DCs engineered to produce IL-12 mediate increases in NK-cell IFN-γ secretion and antitumor activity.26 A more recent study has shown that both human and mouse myeloid DCs mediate enhancement of NK-cell IFN-γ secretion via IL-12p70.27 Interestingly, in this study, as in ours, wild-type mouse bone marrow–derived myeloid DCs were day 5 of culture, at an early stage of development, and did not secrete detectable amount of IL-12p70. The authors suggested that the preassembled stores of IL-12 are polarized and secreted by DCs in the localized space of DC–NK-cell contact and that formation and activity of hypothetical immune synapses between the interacting cells might be a molecular mechanism of the localized secretion of IL-12 and its induction of IFN-γ secretion by NK cells. We have found that wild-type caNK cells expressing high levels of tmTNF, but neither wild-type fNK cells expressing low levels of tmTNF nor TNF-deficient caNK cells, mediate in day 5 culture ciDC maturation and increased expression of CD80, CD86, and CD40 costimulatory molecules and tmTNF (Figure S2 and data not shown), and in day 9 culture cmDCs enhanced secretion of IL-12p70 (Figure S3). The findings indicate that elusive critical signaling molecules that form the immune synapses and trigger polarized secretion of IL-12 by DCs might be tmTNF and tmTNFRs of both DCs and NK cells. This could occur via conventional or reverse signal transduction through DC tmTNFRs or tmTNF, respectively. Our finding that DC-mediated stimulation of IFN-γ secretion by NK cells requires expression of TNF on DCs, but not on NK cells, and expression of TNFR2 on NK cells, but not on DCs, supports the reverse signaling model. However, our findings of a low but significant decrease of the ability of TNFR2−/− DCs to stimulate IFN-γ secretion by wild-type NK-cells (Figures S1C and 7E–F) could indicate that a fraction of DC-mediated stimulation of NK cells is induced by DC-secreted IL-12p70 conventionally triggered via engaging DC-TNFR2 with NK-cell TNF. It is also possible that NK cell–mediated triggering of KARAP/DAP12- and TREM2-activating receptors on IL-4–propagated DCs coparticipates with DC-TNFR2 triggering in the induction of DC IL-12p70 secretion and increased expression of tmTNF and the amplification of DC-mediated NK-cell activation.41 Our main data strongly support the possibility that the synaptic interplay of DCtmTNF-NKtmTNFR2 directly induces most DC-mediated NK-cell activation.
Our recent study has provided indications that DC–NK-cell cross-stimulation of IFN-γ secretion by NK cells and IL-12p70 secretion by DCs could occur via orchestrated interplays between multiple TNF superfamily ligands and their cognate receptors.42 Ferlazzo et al have demonstrated that human DCs can activate NK cells via triggering NKp30 and possibly some other NK cell–activating receptors.16 These findings suggest that DC–NK-cell cross-stimulation might be mediated by synergistic interactions of several transmembrane ligand-receptor pairs, including those of TNF superfamily and activating NK-cell and DC receptors. Therefore, it is plausible that the bidirectional synapses of multiple transmembrane ligands, including tmTNF, and multiple transmembrane receptors, including tmTNFR2, mediate DC–NK-cell interaction and reciprocal stimulation. The signals generated in the synapses could directly induce DC–NK-cell polarization and increases in secretion of IL-12p70 and IFN-γ.
DCs and NK cells are present in infected, inflamed tumor tissues and in peripheral lymphoid organs, and DC–NK-cell colocalization and contact-dependent interaction have been demonstrated in inflamed tissues and lymph nodes.21–23,42 Therefore, DCs and NK cells may interact via transmembrane molecules in these tissues. We show here that splenic fiDCs and fNK cells express low levels of tmTNF and tmTNFR2 and crosstalk poorly. However, bacterial LPS rapidly induces increases in expression of tmTNF on fiDCs, and IL-2, which could be provided by pathogen-stimulated DCs,24 rapidly induces increases in expression of tmTNFR2 on fNK cells. Thus, stimulated fmDCs and/or faNK cells acquire remarkable ability to crosstalk via tmTNF-tmTNFR2 interplay upon their cell-cell contact. These findings indicate that DC–NK-cell contact–dependent crosstalk via tmTNF-tmTNFR2 interplay could occur in vivo at the periphery (sites of infection/inflammation) and in lymph nodes. For example, at the periphery, the invading pathogens can directly induce fiDC maturation and increases in expression of tmTNF and ability to stimulate NK cells. These fmDCs can interact with fNK cells via tmTNF-tmTNFR2 interplay and promote NK-cell proinflammatory functions at the entry of pathogens and/or migrate into regional lymph nodes and stimulate there resident NK cells to secrete IFN-γ and induce Th1 immune responses. IL-2 and other NK cell–activating cytokines secreted by pathogen-stimulated fmDCs can activate fNK cells and induce their increases in expression of tmTNFR2 and, consequently, amplification of fmDC-tmTNF–mediated activation and IFN-γ secretion. The resulting faNK cells express increased levels of both tmTNFR2 and tmTNF and can interact with enhanced ability with DCs that also express both tmTNF and tmTNFR2 by respective interplays of the transmembrane molecules, induce further increases of DC maturation and IL-12p70 secretion, and lead to enhanced stimulation of proinflammatory responses at the periphery. These faNK cells could also migrate to and interact similarly with resident DCs in regional lymph nodes and enhance polarization of Th1 responses.
In conclusion, we have defined a novel essential mechanism of DC–NK-cell crosstalk that is nonsecretory and is mediated via cell-cell contact and cognate interplay of tmTNF and tmTNFR2. This finding coupled with the previous demonstration of an important role for secreted cytokines in DC–NK-cell crosstalk suggests that DC–NK-cell crosstalk is mediated and regulated by 2 interdependent mechanisms/signals, the nonsecretory signal 1 and the secretory signal 2. The signal 1 is induced in cell-cell contact by synaptic interplay of tmTNF and tmTNFR2 and might also involve several other transmembrane ligands and receptors, including those of TNF superfamily. It rapidly induces reciprocal activation and Th1 polarization of interacting DCs and NK cells, leading to DC-mediated localized secretion of NK cell–stimulating cytokine IL-12p70 and NK cell–mediated secretion of DC-stimulating cytokine IFN-γ. The secreted central Th1 cytokines provide the signal 2 that further reciprocally stimulates and amplifies the effector functions of interacting DCs and NK cells, and both signals lead sequentially to graded increases of inflammatory reactions, direct elimination of pathogens and transformed cells at the periphery, and polarization of Th1 adaptive immune responses in the lymph nodes.
Supplementary Material
Acknowledgments
This work was supported by research funding from National Institutes of Health grants I-PO DE13059 and RO1 DE14775 (N.L.V.).
The authors thank to Dr Giorgio Trinchieri for critical review of the manuscript.
Footnotes
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Authorship
Contribution: J.X., A.K.C., and J.L.T. performed experiments and analyzed data; L.G. generated noncleavable TNF; A.G. provided adenoviral vectors and contributed in transfection experiments; and N.L.V. was accountable for the conception, design of research, and writing of the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Nikola L. Vujanovic, University of Pittsburgh Cancer Institute, Hillman Cancer Center, G.17d, 5117 Centre Ave, Pittsburgh, PA; e-mail: vujanovicnl@msx.upmc.edu.
References
- 1.Banchereau J, Steinman RM. Dendritic cells and the control of immunity. Nature. 1998;392:245–252. doi: 10.1038/32588. [DOI] [PubMed] [Google Scholar]
- 2.Kapsenberg ML. Dendritic-cell control of pathogen-driven T-cell polarization. Nat Rev Immunol. 2003;3:984–993. doi: 10.1038/nri1246. [DOI] [PubMed] [Google Scholar]
- 3.Trinchieri G. Biology of natural killer cells. Adv Immunol. 1989;47:187–376. doi: 10.1016/S0065-2776(08)60664-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Robertson MJ, Ritz J. Biology and clinical relevance of human NK cells. Blood. 1990;76:2421–1990. [PubMed] [Google Scholar]
- 5.Lanier LL, Phillips JH. Natural killer cells. Curr Opin Immunol. 1992;4:38–42. doi: 10.1016/0952-7915(92)90121-t. [DOI] [PubMed] [Google Scholar]
- 6.Moretta A. Natural killer cells and dendritic cells: rendezvous in abused tissues. Nat Rev Immunol. 2002;2:957–964. doi: 10.1038/nri956. [DOI] [PubMed] [Google Scholar]
- 7.Ruggeri L, Capanni M, Urbani E, et al. Effectiveness of donor natural killer cell alloreactivity in mismatched hematopietic transplants. Science. 2002;295:2097–2100. doi: 10.1126/science.1068440. [DOI] [PubMed] [Google Scholar]
- 8.Moretta L, Bottino C, Ferlazzo G, et al. Surface receptors and functional interactions of human natural killer cells: from bench to the clinic. Cell Mol Life Sci. 2003;60:2139–2146. doi: 10.1007/s00018-003-3102-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Farag SS, Fehniger TA, Ruggeri L, Velardi A, Caligiuri MA. Natural killer cell receptors: new biology and insights into the graft-versus-leukemia effect. Blood. 2002;100:1935–1947. doi: 10.1182/blood-2002-02-0350. [DOI] [PubMed] [Google Scholar]
- 10.Fernandez NC, Lozier A, Flament C, Ricciardi-Castagnoli P, Bellet D, Suter M. Dendritic cells directly trigger NK cell functions: cross-talk relevant in innate anti-tumor immune responses in vivo. Nat Med. 1999;5:405–411. doi: 10.1038/7403. [DOI] [PubMed] [Google Scholar]
- 11.Goodier M, Londei M. Lipopolysaccharide stimulates the proliferation of human CD56+CD3- NK cells: a regulatory role of monocytes and IL-10. J Immunol. 2000;165:139–147. doi: 10.4049/jimmunol.165.1.139. [DOI] [PubMed] [Google Scholar]
- 12.Osada T, Nagawa H, Kitayama J, et al. Peripheral blood dendritic cells, but not monocyte-derived dendritic cells, can augment human NK cell function. Cell Immunol. 2001;213:14–23. doi: 10.1006/cimm.2001.1858. [DOI] [PubMed] [Google Scholar]
- 13.Yu Y, Hagihara M, Ando K, et al. Enhancment of human cord blood CD34+cell-derived NK cell cytotoxicity by dendritic cells. J Immunol. 2001;166:1590–1600. doi: 10.4049/jimmunol.166.3.1590. [DOI] [PubMed] [Google Scholar]
- 14.Gerosa F, Baldani-Guerra B, Nisii C, Marchesini V, Carra G, Trinchieri G. Reciprocal activating interaction between natural killer cells and dendritic cells. J Exp Med. 2002;195:327–333. doi: 10.1084/jem.20010938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Piccioli D, Sbrana S, Melandri E, Valiante NM. Contact-dependent stimulation and inhibition of dendritic cells by natural killer cells. J Exp Med. 2002;195:335–341. doi: 10.1084/jem.20010934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ferlazzo G, Tsang ML, Morretta L, Melioli G, Steinman RM, Munz C. Human dendritic cells activate resting natural killer (NK) cells and are recognized via NKp30 receptor by activated NK cells. J Exp Med. 2002;195:343–351. doi: 10.1084/jem.20011149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Poggi A, Carosio R, Spaggiari GM, et al. NK cell activation of dendritic cells is dependent on LFA-1-mediated induction of calcium-calmodulin kinase II: inhibition by HIV-1 Tar C-terminal domain. J Immunol. 2002;168:95–101. doi: 10.4049/jimmunol.168.1.95. [DOI] [PubMed] [Google Scholar]
- 18.Jinushi M, Takehara T, Kanto T, et al. Critical role of MHC class I-related chain A and B expression on IFN-alpha-stimulated dendritic cells in NK cell activation: impairment in chronic hepatitis C virus infection. J Immunol. 2003;170:1249–1256. doi: 10.4049/jimmunol.170.3.1249. [DOI] [PubMed] [Google Scholar]
- 19.Maillard RB, Son Y-I, Redlinger R, et al. Dendritic cells mediate NK cell help for Th1 and CTL responses: two-signal requirement for the induction of NK cell helper function. J Immunol. 2003;171:2366–2373. doi: 10.4049/jimmunol.171.5.2366. [DOI] [PubMed] [Google Scholar]
- 20.Andrews DM, Scalzo AA, Yokoyama WM, Smyth MJ, Degli-Esposti MA. Functional interactions between dendritic cells and NK cells during viral infection. Nat Immunol. 2003;4:175–181. doi: 10.1038/ni880. [DOI] [PubMed] [Google Scholar]
- 21.Moretta A, Marcenaro E, Sivori S, Della Chiesa M, Vitale M, Moretta L. Early liaisons between cells of the innate immune system in inflamed peripheral tissues. Trends Immunol. 2005;26:668–675. doi: 10.1016/j.it.2005.09.008. [DOI] [PubMed] [Google Scholar]
- 22.Ferlazzo G, Munz C. NK cell compartments and their activation by dendritic cells. J Immunol. 2004;172:1333–1339. doi: 10.4049/jimmunol.172.3.1333. [DOI] [PubMed] [Google Scholar]
- 23.Bajenoff M, Breart B, Huang AYC, et al. Natural killer cell behavior in lymph nodes revealed by static and real-time imaging. J Exp Med. 2006;203:619–631. doi: 10.1084/jem.20051474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Granucci F, Zanoni I, Pavelka N, et al. A contribution of mouse dendritic cell-derived IL-2 for NK cell activation. J Exp Med. 2004;200:287–295. doi: 10.1084/jem.20040370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.van den Broeke LT, Daschbach E, Thomas EK, Berzofsky AG. Dendritic cell-induced activation of adaptive and innate immunity. J Immunol. 2003;171:5842–5852. doi: 10.4049/jimmunol.171.11.5842. [DOI] [PubMed] [Google Scholar]
- 26.Rodriguez-Calvillo M, Duarte M, Tirapu I, et al. Upregulation of natural killer cells functions underlies the efficacy of intratumorally injected dendritic cells engineered to produce interleukin-12. Exp Hematol. 2002;30:195–204. doi: 10.1016/s0301-472x(01)00792-5. [DOI] [PubMed] [Google Scholar]
- 27.Borg C, Jalil A, Laderach D, et al. NK cell activation by dendritic cells (DCs) requires the formation of a synapse leading to IL-12 polarization in DCs. Blood. 2004;104:3267–3275. doi: 10.1182/blood-2004-01-0380. [DOI] [PubMed] [Google Scholar]
- 28.Walzer T, Dalod M, Robbins SH, Zitvogel L, Vivier E. Natural killer cells and dendritic cells: “l'union fait la force”. Blood. 2005;106:2252–2258. doi: 10.1182/blood-2005-03-1154. [DOI] [PubMed] [Google Scholar]
- 29.Kashii Y, Giorda R, Herberman RB, Whiteside TL, Vujanovic NL. Constitutive expression and role of the tumor necrosis factor family ligands in apoptotic killing by human natural killer cells. J Immunol. 1999;163:5358–5366. [PubMed] [Google Scholar]
- 30.Lu G, Janjic BM, Janjic J, Whiteside TL, Storkus WJ, Vujanovic NL. Innate direct anticancer effector function of human immature dendritic cells. II. Role of TNF, LT-α1β2, Fas ligand and TRAIL. J Immunol. 2002;168:1831–1839. doi: 10.4049/jimmunol.168.4.1831. [DOI] [PubMed] [Google Scholar]
- 31.Wallach D., Varfolmeev EE, Malinin NL, Goltsev YV, Kovalenko AV, Bildin MP. Tumor necrosis factor receptor and Fas signaling mechanisms. Annu Rev Immunol. 1999;17:331–367. doi: 10.1146/annurev.immunol.17.1.331. [DOI] [PubMed] [Google Scholar]
- 32.Wajant H, Pfitzenmaier K, Scheurich P. Tumor necrosis factor signaling. Cell Death Differ. 2003;10:45–65. doi: 10.1038/sj.cdd.4401189. [DOI] [PubMed] [Google Scholar]
- 33.Cella M, Engering A, Pinet V, Pieters J, Lanzavecchia A. Inflammatory stimuli induce accumulation of MHC class II complexes on dendritic cells. Nature. 1997;388:782–787. doi: 10.1038/42030. [DOI] [PubMed] [Google Scholar]
- 34.Jewett A, Bonavida B. Pivotal role of endogenous TNF-alpha in the IL-2-driven activation and proliferation of the functionally immature NK free subset. Cell Immunol. 1993;151:257–269. doi: 10.1006/cimm.1993.1236. [DOI] [PubMed] [Google Scholar]
- 35.Masson AT, McVicar WW, Smith CA, Toung HA, War CF, Ortaldo JR. Regulation of NK cells through the 80-kDa TNFR (CD120b). J Leukoc Biol. 1995;58:249–255. doi: 10.1002/jlb.58.2.249. [DOI] [PubMed] [Google Scholar]
- 36.Li S, Xu J, Makarenkova VP, et al. A novel epitope of N-CAM defines precursors of human adherent NK cells. J Leukoc Biol. 2004;76:1187–1199. doi: 10.1189/jlb.0802386. [DOI] [PubMed] [Google Scholar]
- 37.Vujanovic NL, Nagashima S, Herberman RB, Whiteside TL. Non-secretory apoptotic killing by human natural killer cells. J Immunol. 1966;157:1117–1126. [PubMed] [Google Scholar]
- 38.Wan Y, Bramson J. Role of dendritic cell-derived cytokines in immune regulation. Curr Pharm Des. 2001;7:977–992. doi: 10.2174/1381612013397627. [DOI] [PubMed] [Google Scholar]
- 39.Stager S, Kaye PM. CD8+ T-cell priming regulated by cytokines of the innate immune system. Trends Mol Med. 2004;10:366–371. doi: 10.1016/j.molmed.2004.06.003. [DOI] [PubMed] [Google Scholar]
- 40.Trinchieri G, Pflanz S, Kastelein RA. The IL-12 family of heterodimeric cytokines: new players in the regulation of T cell responses. Immunity. 2003;19:641–644. doi: 10.1016/s1074-7613(03)00296-6. [DOI] [PubMed] [Google Scholar]
- 41.Terme M, Tomasello E, Maruyama K, et al. IL-4 confers NK stimulatory capacity to murine dendritic cells: a signaling pathway involving KARAP/DAP12-triggering receptor expressed on myeloid cell 2 molecules. J Immunol. 2004;172:5957–5966. doi: 10.4049/jimmunol.172.10.5957. [DOI] [PubMed] [Google Scholar]
- 42.Makarenkova V, Chakrabarti AK, Liberatore JA, et al. Dendritic cells and natural killer cells interact via multiple TNF family molecules. J Leukoc Biol. 2005;77:408–413. doi: 10.1189/jlb.1104675. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.