Abstract
The concept that complex surgical procedures should be performed at high-volume centers to improve surgical morbidity and mortality is becoming widely accepted. We wanted to determine if there were differences in the treatment of patients with gastric cancer between community cancer centers and teaching hospitals in the United States. Data from the 2001 Gastric Cancer Patient Care Evaluation Study of the National Cancer Data Base comprising 6,047 patients with gastric adenocarcinoma treated at 691 hospitals were assessed. The mean number of patients treated was larger at teaching hospitals (14/year) when compared to community centers (5–9/year) (p < 0.05). The utilization of laparoscopy and endoscopic ultrasonography were significantly more common at teaching centers (p < 0.01). Pathologic assessment of greater than 15 nodes was documented in 31% of specimen at community hospitals and 38% at teaching hospitals (p < 0.01). Adjusted for cancer stage, chemotherapy and radiation therapy were utilized with equal frequency at all types of treatment centers. The 30-day postoperative mortality was lowest at teaching hospitals (5.5%) and highest at community hospitals (9.9%) (p < 0.01). These data support previous publications demonstrating that patients with diseases requiring specialized treatment have lower operative mortality when treated at high-volume centers.
Keywords: Gastric cancer, Operative mortality, Hospital volume, Survival, NCDB
Introduction
Approximately 22,000 patients in the United States (US) will be diagnosed with gastric carcinoma in 2006, a number that pales in comparison to more common malignancies such as colorectal (172,000 new cases) and breast cancer (211,000 new cases).1 Because of the relative infrequency of gastric cancer, most individual hospitals evaluate and treat a limited number of patients with stomach cancer, impairing the ability to develop expertise at many institutions. A lack of expertise may contribute to the dismal survival of gastric cancer in the US. In an earlier report from the National Cancer Data Base (NCDB), Hundahl et al.2 observed a 5-year survival rate of 78% for stage IA, 58% for stage IB, 34% for stage II, 20% for stage IIIA, 8% for stage IIIB, and 7% for stage IV disease.
A recent report from the World Health Organization3 shows a decline in the mortality of gastric cancer worldwide. However, the observed rate of decline in mortality is less in the US than what is observed for Japan. Factors that might contribute to the improved Japanese survival includes greater operative experience leading to more skilled surgeons, earlier diagnosis, different biologies of gastric cancer between countries, improved pathologic staging with stage migration (Will Rogers effect),4 and the frequency of neoadjuvant or adjuvant chemoradiation therapy use.
These data highlight the need for improved treatment of gastric cancer to increase patient survival. A controversial way to accomplish this might be to limit gastric cancer care to high-volume centers, if the results were better at such sites. Evidence-based hospital referral has been adopted by some insurance companies and consortiums of large health care purchasers, such as the Leapfrog group, based on studies showing better outcomes for surgical services at high-volume centers.5–7 Birkmeyer et al.8 reported that Medicare patients had a lower operative mortality if they had cancer-related procedures (gastrectomy, esophagectomy, colectomy, pancreatectomy, cystectomy, nephrectomy, and pulmonary resection) or cardiovascular disease at a high-volume hospital. The Japanese have also found a similar pattern of improved survival after the treatment of gastric carcinoma at high-volume centers. Nomura et al.9 reported 5-year survival rates to be significantly higher at centers with high surgical volumes (96–205/year), when compared to very-low-volume hospitals (1–28 cases/year).
To determine if patients treated for gastric cancer at high-volume and specialized centers in the US had better postoperative outcomes, we analyzed the preoperative evaluation and surgical treatment of gastric adenocarcinoma at three categories of hospitals, defined by the Commission on Cancer (CoC), namely, the Community Hospital Cancer Program (CHCP), Community Hospital Comprehensive Cancer Program (COMP), and Teaching Hospital Cancer Program (THCP). Data collected for the 2001 Gastric Cancer Patient Care Evaluation (PCE) by the NCDB were utilized.
Material and Methods
NCDB, Data Source, Case Selection, and Data Handling
The NCDB is a project of the American College of Surgeons (ACS) CoC. The NCDB was established in 1989 to serve as a comprehensive clinical surveillance resource for all forms of cancer diagnosed in the US and its operations have been supported in part by the American Cancer Society. In 2001, the database captured 73% of all newly diagnosed cancer cases in the US.
Data were submitted electronically in accordance with specified North American Association of Central Cancer Registries data transmission standards.10 Hospital cancer registrars abstracted each case according to a standardized set of data elements and definitions as described in the CoC’s Registry Operations and Data Standards, volume II.11 The NCDB elements include patient characteristics: sex, age or date of birth, and race/ethnicity; tumor characteristics: primary site, histology, behavior, grade, and American Joint Committee on Cancer (AJCC), fifth edition stage groups; and first course of treatment: surgery, radiation, chemotherapy, and others. In addition to the annual call for data, a call for participation in a 2001 PCE was issued and 711 of the 1,423 CoC-approved institutions in the US responded. Cancer registrars were also asked to provide information describing additional diagnostic and treatment information, 30-day mortality, and patient comorbidities. Data quality checks were conducted at the local and the depository level.
Patient Population
The 2001 Gastric PCE included data submitted from 711 CoC-approved institutions in the US. Participating institutions submitted data for consecutive hospital admissions and clinic visits between January 1, 2001 and December 31, 2001. Patients eligible for participation had a “microscopically confirmed neoplasm of the stomach that was either diagnosed or initially treated at the reporting facility.” Of 7,084 total patients, 6,099 (86%) were diagnosed with gastric adenocarcinoma. For this study, 52 patients were excluded because they were treated at nine institutions without a specialized hospital type, leaving 6,047 patients at 691 cancer program types for evaluation. This group comprises the study population.
Cancer Program Categories
Cancer programs were characterized as CHCP, COMP, or THCP. Community Hospital Cancer Programs diagnose and/or treat 100–650 cancer patients every year and will commonly refer patients to other institutions for diagnostic evaluation or treatment. A CHCP has neither a medical school affiliation nor residency programs but does posses a medical oncology unit or functional equivalent and infrequently participates in cancer research. Community Hospital Comprehensive Cancer Programs diagnose and/or treat more than 650 cancer cases per year, but are not associated with a medical school. A COMP may make outside patient referrals, has a medical oncology unit, and participates in cancer research. Teaching Hospital Cancer Programs are defined as facilities associated with a medical school that participates in the training of residents in at least four fields, two of which are medicine and surgery. A THCP offers a full range of diagnostic and therapeutic services on site and has an in-patient medical oncology unit. A THCP hospital also participates in cancer-related clinical research and has board-certified medical oncologists.
Statistical Analysis
All analyses were performed using the SPSS statistical software (SPSS for Windows, version 14.0; SPSS Inc, Chicago, IL, USA). Frequency distributions were calculated to get the mean number of cases by hospital category type. The chi-square (χ2) test was used for comparisons of proportions across levels of categorical variables. When the overall test was significant, pairwise comparisons among the three hospital categories were also calculated to asses which hospitals differed in rates. The p values reported were adjusted for multiple comparisons using the Bonferroni adjustments.12 Results were based on two-sided tests with a p = 0.01 significance level, except where indicated. Where specific values were unknown, these cases were excluded from the analysis when appropriate.
A forward stepwise binary logistic regression model was used to evaluate the impact of age, stage, and comorbid burden on determination of type of treatment, i.e., the odds of “surgery, with or without other treatment” compared to the odds of nonsurgical treatment (radiation and/or chemotherapy). The Wald statistic was used to test significance. Exponentiated estimates of the beta coefficients were interpreted as the estimates for the effect (odds ratio) of a particular variable, controlling for the other variables in the equation. A receiver operating characteristic (ROC) curve was created to examine the prediction results. The true-positive probability was calculated to define the sensitivity of the classification rule and the true-negative probability was calculated to determine the specificity to summarize how well the model performed.
Confidentiality
Data reported to the NCDB are retrospective in nature. No patient or physician identifiers were collected as part of the study. Case identification information (facility identification number and local registry accession number) was collected for administrative purposes only. Analyses were reported only at the aggregate level to assist hospital cancer programs with quality assurance, rather than used to make decisions about individuals and their care.
The ACS has executed a business associate agreement that includes a data-use agreement, with each of its CoC-approved hospitals. Results reported in this study were in compliance with the privacy requirements of the Health Insurance Portability and Accountability Act of 1996 as reported in the Standards for Privacy of Individually Identifiable Health Information; Final Rule (45 CFR Parts 160 and 164).
Results
Treatment Volumes
A total of 691 cancer programs were included in the study: 258 CHCP sites, 267 COMP sites, and 166 THCP sites. Although 37.3% of the programs were CHCPs, only 22% (n = 1329) of the cases were treated in this setting; 40.8% (n = 2,468) of the cases came from COMPs; and 37.2% (n = 2,250) of the patients came from THCPs. Community Hospital Cancer Programs saw on average of 5.2 cases/year; COMPs, 9.2; and THCPs 13.6. The THCPs treated more surgical patient on average (7.6 cases) than either the COMPs (5.3 cases) or the CHCPs (2.9 cases) (Table 1).
Table 1.
Number and Percent of Cancer Programs, Number and Percent of Patients by Cancer Program, Mean Number and Range of All Cases by Cancer Program Type, Number and Percent of Surgically Treated Cases, and Mean Number of Surgical Cases and Range by Cancer Program Type
| Community Cancer Centers | Comprehensive Community Cancer Centers | Teaching/Research Hospitals | Total | |
|---|---|---|---|---|
| Number (%) of cancer programs | 258 (37.3) | 267 (38.6) | 166 (24.0) | 691 |
| Number (%) of patients in study | 1,329 (22.0) | 2,468 (40.8) | 2,250 (37.2) | 6,047 |
| Mean number and range of all cases | 5.2 (1–39) | 9.2 (1–49) | 13.6 (1–55) | |
| Number (%) of surgical cases | 673 (20.5) | 1,369 (41.8) | 1,235 (37.7) | 3,277 |
| Mean number and range of surgical cases | 2.9 (1–16) | 5.3 (1–37) | 7.6 (1–40) |
Patient Demographics
Men, 3,751(62%), and women, 2,296 (38%), were equally distributed across hospital types. The mean age in years was 69.3 and the median age 71 (18–103 years). Significantly more 70 and older patients were seen in CHCPs when compared to COMPs or THCPs (p < 0.01). Significantly more 50 and younger patients were seen at THCPs when compared with CHCPs and COMPs (p < 0.01) (Table 2).
Table 2.
Patient Characteristics by Type Cancer Program
| Community Cancer Center | Comprehensive Cancer Center | Teaching/Research | Total | |
|---|---|---|---|---|
| n (%) | n (%) | n (%) | n (%) | |
| Gender | ||||
| Male | 805 (60.6) | 1,502 (60.9) | 1,444 (64.2) | 3,751 (62.0) |
| Female | 524 (39.4) | 966 (39.1) | 806 (35.8) | 2,296 (38.0) |
| Total | 1,329 | 2,468 | 2,250 | 6,047 |
| Age | ||||
| <50 | 106 (8.0) | 207 (8.4) | 264 (11.7) | 577 (9.5) |
| 50–69 | 402 (30.2) | 865 (35.0) | 836 (37.2) | 2,103 (34.8) |
| 70 and older | 821 (61.8) | 1,396 (56.6) | 1,150 (51.1) | 3,367 (55.7) |
| Total | 1,329 | 2,468 | 2,250 | 6,047 |
| Race/Ethnicity | ||||
| Caucasian | 966 (72.7) | 1,723 (69.8) | 1,387 (61.6) | 4,076 (67.4) |
| African American | 143 (10.8) | 278 (11.3) | 406 (18.0) | 827 (13.7) |
| Hispanic | 120 (9.0) | 206 (8.3) | 207 (9.2) | 533 (8.8) |
| Asian | 78 (5.9) | 218 (8.8) | 176 (7.8) | 472 (7.8) |
| Other | 22 (1.7) | 43 (1.7) | 74 (3.3) | 139 (2.3) |
| Total | 1,329 | 2,468 | 2,250 | 6,047 |
| AJCC stage | ||||
| O | 2 (0.2) | 8 (0.3) | 2 (0.1) | 12 (0.2) |
| I | 13 (1.0) | 28 (1.1) | 26 (1.2) | 67 (1.1) |
| IA | 105 (7.9) | 210 (8.5) | 182 (8.1) | 497 (8.2) |
| IB | 118 (8.9) | 218 (8.8) | 178 (7.9) | 514 (8.5) |
| II | 160 (12.0) | 286 (11.6) | 242 (10.8) | 688 (11.4) |
| III | 6 (0.3) | 12 (0.5) | 12 (0.5) | 30 (0.5) |
| IIIA | 156 (11.7) | 347 (14.1) | 337 (15.0) | 840 (13.9) |
| IIIB | 47 (3.5) | 96 (3.9) | 97 (4.3) | 240 (4.0) |
| IV | 427 (32.1) | 856 (34.7) | 835 (37.1) | 2,118 (35.0) |
| Unknown | 295 (22.2) | 407 (16.5) | 339 (15.1) | 1,041 (17.2) |
| Total | 1,329 | 2,468 | 2,250 | 6,047 |
The study population included 4,076 (67.4%) Caucasians, 827 (13.7%) African Americans, 533 (8.8%) Hispanic, 472 (7.8%) Asians, and 139 (2.3%) patients of other or unknown racial or ethnic background. Significantly fewer Caucasians were seen in THCPs when compared to CHCPs and COMPs (p < 0.01). THCPs saw significantly more African Americans than the other two types of cancer programs (p < 0.01). Significantly more Asians were seen at COMP hospitals when compared to CHCP hospitals (p < 0.01), but there was no significant difference when comparing the proportion of Asians in COMPs to the proportion found in THCPs.
A large percentage of this patient population did not have a documented AJCC Stage reported (n = 1,041, 17.2%). Of those patients with a documented stage, more than 64.5% had advanced stage (stage III or IV) at presentation. The largest subgroup had stage IV disease (n = 2,118, 35.0%). There were no significant differences in the stage at presentation between the different types of medical institutions. When specified, the most common location of the primary cancer was the cardia 27.4% (n = 1,656), followed by the antrum 18.3% (n = 1,107) and stomach, not otherwise specified (NOS) 17.9% (1,080) (Fig. 1). The distribution of tumor location was similar among all these categories of treatment institution.
Figure 1.
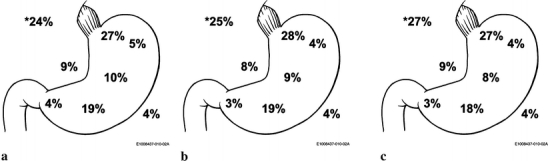
Location of cancer at presentation by cancer program types: A Community Hospital Cancer Program (CHCP), B Community Hospital Comprehensive Cancer Program (COMP), and C Teaching Hospital Cancer Program (THCP). Location of lesions: cardia, funds, body, antrum, pyloric, lesser curve, and greater curve by percent and others indicated by asterisk (C168, overlapping lesions and C169, stomach, NOS).
Diagnostic Testing
Diagnostic evaluation included computed tomography (CT) of the abdomen (n = 4,417, 73%) and pelvis (46.9%) over all hospital types. The abdominal CT suggested a diagnosis of cancer in 63.9% of patients at CHCP, 63.0% at COMP, and 68.2% at THCP. The CT of the abdomen was more likely to suggest cancer at the THCP when compared to the COMP (p < 0.01), but no significant difference was noted between the THCP and the CHCP. Computed tomography of the pelvis was suggestive for cancer in 36.9, 38.1, and 42.6% at CHCP, COMP, and THCP, respectively. The differences across cancer programs were not significant (p > 0.01). Only 15.6% (n = 946) of these cases were evaluated with endoscopic ultrasonography. That procedure was used more often at THCPs (33.5%) when compared to both CHCP (13.8%) and COMP (17.6%) (p < 0.01). 2-18F-Fluoro-2-deoxy-d-glucose positron emission tomography (F18-FDG-PET) was rarely used at any type of hospital (4.6%). When done, THCPs were more likely to use the F18-FDG-PET than CHCP for diagnostic testing (p < 0.01). No significant differences were noted between THCP and COMP use.
Intraoperative Assessment
Staging laparoscopy was performed significantly more often at THCPs (18.9%) than at COMPs (13.6%) or at CHCPs (10.5%) (p < 0.01). No significant differences were seen between COMPs and CHCPs. Peritoneal lavage with cytology for the assessment of occult peritoneal disease was rarely used at THCPs, COMPs, and CHCPs with rates of 5.7, 4.3, and 2.7%, respectively. Significantly fewer peritoneal lavages occurred at CHCPs when compared to THCPs (p < 0.01). No significant differences were found between THCPs and COMPs.
Surgical Treatment
Surgical intervention was undertaken for 54.2% (n = 3,277) of the all patients in the study. At CHCP hospitals, 673 out of 1,329 (50.6%) patients were surgically treated, 1,369 out of 2,468 (55.5%) were surgical patients at COMP, and 1,235 out of 2,250 (54.9%) patients underwent surgery at THCP. When surgeries were grouped as less extensive or more extensive, significantly more extensive surgeries were performed at THCPs than at CHCPs (p < 0.01); however, no significant difference was observed between COMPs and THCPs (p > 0.01). More specifically, the most frequently recorded type of surgical resection was gastrectomy, NOS (overall, 21.7%; CHCP, 26.0%; COMP, 20.5%; and THCP, 20.6%) followed by distal gastrectomy (overall, 16.2%; CHCP, 15.6%; COMP, 17.3%; and THCP, 15.2%), and partial or subtotal gastrectomy (overall, 13.3%; CHCP, 12.2%; COMP, 12.7%; and THCP, 14.6%). A near total or total gastrectomy was uncommonly performed (overall, 5.7%; CHCP, 4.3%; COMP 5.7%; and THCP, 6.4%). After pathologic analysis, 83.5% of the entire surgical patient population had a resection with curative intent (R0 resection), whereas 16.5% had a palliative resection (R1 resection). The frequencies of R0 and R1 resections were similar (CHCP, 81.2/18.8%, COMP, 84.6/15.4%, and THCP, 83.6/16.4%) among all institutions and not statistically different between different types of institution (p = 0.25).
Lymph node staging, a highly significant predictor of outcome, was evaluated using the number of lymph nodes examined after surgical resection. Analysis of at least 15 lymph nodes is required by the AJCC for accurate staging and exclusion of nodal metastases. Only 31.1% of patients at CHCPs, 31.0% at COMPs, and 38.4% at THCPs had more than 15 nodes examined (p < 0.01). A large number of patients from each hospital type had an unknown number of lymph nodes pathologically evaluated (26.6% CHCP, 22.6% COMP, and 15.4% THCP), but THCPs had significantly better documentation of lymph node evaluation (p < 0.05) (Fig. 2). A D1 lymphadenectomy with removal of only perigastric nodes was noted in 56.7% of surgically managed patients. Some patients had removal of some lymph nodes that are included in D2 or D3 nodes but most across all hospital types did not have a formal extended lymphadenectomy (Table 3).
Figure 2.
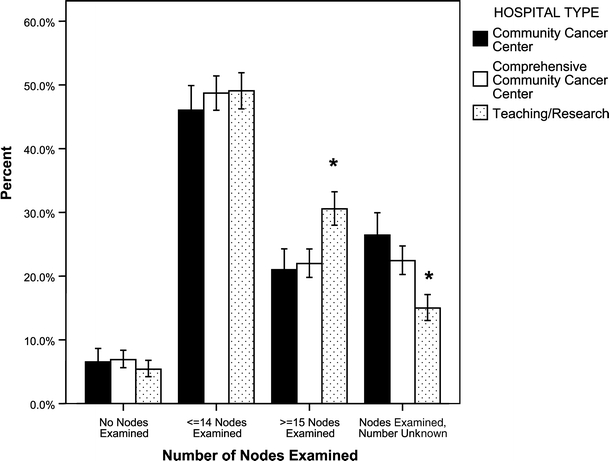
Percent of lymph nodes sampled at each hospital. CHCP = Community Cancer Center Program, COMP = Community Hospital Comprehensive Cancer Program, THCP = Teaching Hospital Cancer Program. p < 0.05 (asterisk). Error bars: 95% CI.
Table 3.
Lymph Nodes Sampled During Surgical Resection
| Resected | Community Cancer Center | Comprehensive Cancer Center | Teaching/Research |
|---|---|---|---|
| n (%) | n (%) | n (%) | |
| Perigastrica | |||
| Yes | 367 (71.1) | 775 (68.9) | 715 (70.4) |
| No | 149 (28.9) | 350 (31.1) | 301 (29.6) |
| Hepaticb | |||
| Yes | 21 (4.2) | 68 (6.5) | 70 (7.5) |
| No | 478 (95.8) | 979 (93.5) | 867 (92.5) |
| Celiacc | |||
| Yes | 40 (7.9) | 100 (9.5) | 96 (10.1) |
| No | 465 (92.1) | 952 (90.5) | 854 (89.9) |
| Splenicd | |||
| Yes | 14 (2.8) | 49 (4.7) | 52 (5.5) |
| No | 481 (97.2) | 988 (95.3) | 887 (94.5) |
| Paraaortice | |||
| Yes | 94 (18.8) | 170 (16.8) | 189 (20.2) |
| No | 405 (81.2) | 840 (83.2) | 745 (79.8) |
Comparison of column proportions did not include the unknown values in calculations.
aPerigastric unknown, n = 620
bHepatic unknown, n = 794
cCeliac unknown, n = 770
dSplenic unknown, n = 806
eParaaortic unknown, n = 834
Morbidity and Mortality
The prevalence of operative morbidity, specifically for postoperative hemorrhage, wound infection sepsis, and an anastomotic leak, was similar across all hospital categories (Table 4). Operative mortality, defined by 30-day mortality, was different among the institution types. Teaching Hospital Cancer Programs had the lowest 30-day mortality at 5.5%, compared to 7.9% at COMPs and 9.9% at CHCPs (Table 4). The proportional difference between THCPs and COMPs was not significant (p > 0.01), whereas, when compared to CHCPs, THCPs had significantly fewer deaths within 30 days of surgery (p < 0.01). For those that died within that 30-day period, no significant differences were found across all hospital types for stage (p > 0.01) or age (p > 0.01). In the logistic regression model stage was the most significant predictor of 30-day postoperative death (p = 0.0001), followed by age (p = 0.0001), and category of hospital (p = 0.004). Stage IV patients were 2.6 times (99% confidence interval [CI] = 1.6–4.3) more likely to die within 30 days of surgery; no other stage was significant (p > 0.01). Patients in CHCPs were almost twice as likely to die within 30 days when compared to THCPs (p = 0.001). Race and extent of surgery were not significant factors. The area under the ROC curve defined by the logistic model was 0.69 (99% CI = 0.64–0.74), where 0.5 represents a nondiscriminatory result. Long-term 5-year survival data are unavailable for this patient population and will not be until the fall of 2007.
Table 4.
Postoperative Mortality and Complications
| Community Cancer Center | Comprehensive Cancer Center | Teaching/Research | |
|---|---|---|---|
| n (%) | n (%) | n (%) | |
| 30-Day mortalitya | |||
| Yes | 55 (9.9) | 93 (7.9) | 59 (5.5) |
| No | 501 (90.1) | 1,080 (92.1) | 1,020 (94.5) |
| Bleedingb | |||
| Yes | 20 (4.2) | 40 (3.9) | 47 (5.1) |
| No | 451 (95.8) | 993 (96.1) | 881 (94.9) |
| Wound infectionc | |||
| Yes | 35 (7.4) | 54 (5.2) | 61 (6.6) |
| No | 440 (92.6) | 981 (94.8) | 868 (93.4) |
| Sepsisd | |||
| Yes | 34 (7.1) | 62 (6.0) | 43 (4.7) |
| No | 443 (92.9) | 973 (94.0) | 881 (95.3) |
| Anastomotic leake | |||
| Yes | 32 (6.7) | 68 (6.5) | 64 (6.6) |
| No | 447 (93.3) | 979 (93.5) | 884 (93.2) |
Comparison of column proportions did not include the unknown values in calculations.
a30-Day mortality unknown, n = 469
bBleeding unknown, n = 845
cWound infection unknown, n = 840
dSepsis unknown, n = 841
eAnastomotic Leak unknown, n = 803
Neoadjuvant and Adjuvant Therapy
Approximately 38.9% of the surgical patients received chemotherapy; 30.5% received radiation therapy. Neither neoadjuvant radiation therapy nor chemotherapy was frequently provided at all hospital types, but highest, although not significant (p > 0.01) at the THCPs when compared to CHCPs and COMPs (Table 5). Patients more frequently received adjuvant chemotherapy and radiation than neoadjuvant chemotherapy or radiation therapy at all the categories of hospitals. There were no statistically significant differences in the frequency of treatment with chemotherapy or radiation therapy by tumor stage between the hospital categories.
Table 5.
Radiation and Chemotherapy/Surgery Sequence
| Community Cancer Center | Comprehensive Cancer Center | Teaching/Research | |
|---|---|---|---|
| # (%) | # (%) | # (%) | |
| Radiationa | |||
| Neoadjuvant | 23 (12.1) | 53 (13.0) | 68 (19.1) |
| Adjuvant | 167 (87.9) | 356 (87.0) | 288 (80.9) |
| Chemotherapyb | |||
| Neoadjuvant | 36 (14.6) | 71 (15.0) | 87 (20.3) |
| Adjuvant | 210 (85.4) | 401 (85) | 342 (79.7) |
Comparison of column proportions did not include the unknown values in calculations.
aRadiation sequence unknown, n = 43
bChemotherapy sequence unknown, n = 128
Discussion
When examining the current status of gastric cancer patient care in the US and seeking ways to improve survival, looking to the East for guidance is a reasonable strategy. The standards of gastric cancer therapy have largely been set by the practices of Japanese physicians and surgeons, in a large part, because of their large experience with this disease. In Japan, there are 104,000 new cases annually in a population of 128 × 106,13 compared to the 22,000 cases seen annually in the US in a population of 296 × 106,1 an incidence that is more than 10 times greater than that of the US.
The high incidence seen in Japan has allowed the Japanese to develop surgical and medical strategies to improve mortality. However, several of the surgical principles practiced in Japan are difficult to incorporate into Western practices. Early detection programs in Japan have led to a significant decrease in mortality but this has not been replicated in the US because of the high cost to benefit ratio associated with the much lower incidence of gastric cancer in the US. A more controversial standard Japanese practice is the extended D2, D3, or even D4 lymphadenectomy performed for gastric cancer. Whereas extended lymphadenectomy has been associated with improved survival in Japan, with retrospective analysis, the value of this technique has not been proven in the West. Wanebo et al.,14 in a retrospective study from the US, as well as Bonenkamp et al.15 and Cuschieri et al.16, in randomized controlled trials from The Netherlands and Great Britain, respectively, have reported a lack of survival benefit with D2 lymphadenectomy. Both Bokenkamp et al.15 and Cuschieri et al.16 observed significantly higher mortality rates among those patients that had a D2 resection of 10 vs 4% and 13 vs 6.5%, respectively. The operative mortality after a D2 resection is much lower in Japan with reported rates below 2%.17,18 Kodera et al. from Japan reported an operative mortality rate of 0.8% from 523 patients in a D2 vs D3 study.19
Studies examining large national databases have found improved surgical mortality after gastrectomy for gastric cancer at high-volume centers. A study examined using the National Inpatient Sample reported on 23,690 hospitalized patients with a hospital discharge code of gastric cancer and any gastrectomy noted significant differences in mortality among hospital types. Very-low-volume hospitals (<4 cases/year) had a mortality rate of 8.9% compared with a rate of 6.4% seen at high-volume hospitals (>9 cases/year).20 In a study by Birkmeyer et al.21 of the Medicare population, the observed mortality rate was 13% at very-low-volume centers (<5 cases/year) compared to 8.7% at very-high-volume centers (>21 cases/year). Improved survival among high-volume hospitals was also reported in Swedish22 hospitals that treated >20 surgical patients a year and German23 hospitals that treated >50 patients/year.
The Japanese have also noted an association of lower patient survival rates among gastric cancer patients treated at low-volume centers when compared to high-volume centers. Nomura et al.9 analyzed a database of more than 55,000 patients and grouped them into the following time periods: 1975–1979, 1980–1984, 1985–1989, and 1990–1994. He found “positive relationships between hospital volume and 5-year survival” but over time the survival benefit seen at high-volume centers decreased and persisted in comparison with the very-low-volume centers.
Not all authors, however, have observed improved survival at high-volume centers. A Dutch study evaluated the impact of patient volume on operative mortality and found no differences. This study analyzed 1,987 gastric cancer patients treated at 22 hospitals between 1987 and 1997.24 A limitation of this study is the fact that only 1 of the 22 hospitals was a university hospital and the others were “general hospitals.” Many of the participating hospitals were considered to be low-volume centers.
Interpretation of the Results
Our study had similar results to the aforementioned American, Japanese, and European studies. In this study, there was a marked improvement (>50%) in operative mortality at higher volume centers (≥14 cases/year) when compared to lowest volume institutions (≤5 cases/year). The average 30-day postoperative mortality at the low-volume community centers was almost 10%. The annual volume of gastric surgeries performed seems to be inversely related to 30-day postoperative mortality. On average 2.9, 5.5, and 7.6 gastric surgeries were reported from CHCPs, COMPs, and THCPs, respectively. Corresponding 30-day mortality was 9.9, 7.9, and 5.5%, respectively. There were no significant differences seen in postoperative morbidities such as wound infection and hemorrhage by hospital category. The logistical model revealed three predictors of perioperative mortality: stage IV disease, advanced age, and institution type. Patients that had surgery at a CHCP were twice as likely to die postoperatively compared to patients treated surgically at THCPs. Among those that died, there were no significant differences of stage or age at the different institutions.
Because the absolute differences in surgical case number among the hospital types were not vastly different, this observed difference in outcomes may be a reflection of the infrastructure of the institution rather than individualized surgeon skill. Centers affiliated with a medical school might have more experience with caring for the critically ill in the form of larger and better equipped intensive care units, resident and fellow coverage, newer technology, and more subspecialized physicians to help manage patient care. Unfortunately, we could not analyze the infrastructure for each hospital type nor look at surgeon-specific experience in this study as this was not a part of the study.
Clinical staging is affected by the sensitivity and specificity of the diagnostic studies performed during the preoperative evaluation. An assessment of staging at the different institutions revealed some major differences. Teaching hospitals were more likely to detect malignancy on a CT of the abdomen and pelvis than the other two hospital types. This might be a reflection on the quality of CT scans obtained and the experience of the radiologists. Preoperative utilization of endoscopic ultrasound was higher at teaching hospitals, a predictable finding given the recent adaptation of this technology and the expertise required to interpret these images.
In terms of pathologic staging, Karpeh et al.25 previously reported that evaluation of more than 15 lymph nodes allows a better estimate of patient survival. In fact, a study by Smith et al.26 found that overall survival was largely dependent on the number of nodes examined and found an increase in survival when up to 40 lymph nodes was assessed. We found that teaching hospitals did a significantly better job meeting this recommendation; however, even at THCPs only 38.4% of patients had greater than 15 nodes assessed. The D1 lymphadenectomy was the most common operation for of nodal dissection, probably as a result of controlled Western surgical trials showing no survival benefit from a D2 dissection. Only a limited number of patients had any D2–D4 nodes resected and usually without a standardized extended lymphadenectomy. A notable observation was that the percentage of D2–D4 lymphadenectomy performed at THCPs was not higher than what was observed at COMPs or CHCPs.
To improve and obtain accurate surgical staging of gastric cancer patients, current practice will have to improve and will be dependent upon both the excision of nodes by the surgeon and their retrieval from the specimen by the pathologist. Given so few patients had 15 or more nodes removed at the time of surgery, regardless of hospital type, this practice could be improved by surgeons and pathologists working together to achieve the goal of identifying at least 15 nodes followed by the proper documentation of the microscopic evaluation of these nodes in the pathology report. If intraoperative pathologic evaluation of the surgical specimen is possible and the lymph node sampling is inadequate, the surgeon should excise additional lymph nodes.
A strength of this study is the large number of patients and hospitals included in the study. This broad sampling of hospitals leads to a close approximation of the current practice of gastric cancer treatment in the US. The main shortcoming is that many of the critical data categories had at least 20% “unknown” responses, and may have biased the reported results of this study. In addition, analysis of hospital infrastructure or surgeon volume was limited by lack data availability in the database. Another limitation is that the 5-year survival information has not yet been documented; these data will provide long-term outcome by type of treatment center.
Conclusion
Data from the Gastric PCE project suggests that there is significant room for improvement in the surgical management of gastric cancer in the US. Most hospitals, regardless of category, do not document the evaluation at least 15 lymph nodes necessary to meet AJCC standards. With a little over one third of all patients having more than 15 lymph nodes examined, pathologic staging is less accurate and results of surgical and adjuvant therapy are likely to be worse. Significantly more patients had appropriate staging with the recommended number of nodes included in the lymphadenectomy at teaching centers than at nonacademic centers. Thirty-day mortality rates after gastric cancer resection were significantly lower at teaching centers, further establishing the recommendation that complex oncologic operations should be performed at high-volume centers to obtain better patient outcomes, corroborating the results of several previous studies. Long-term survival data will provide more information on effectiveness of treatment at each of the different institution types.
Abbreviations
- AJCC
American Joint Committee on Cancer
- CoC
Commission on Cancer
- CHCP
Community Hospital Cancer Program
- COMP
Community Hospital Comprehensive Cancer Program
- NCDB
National Cancer Data Base
- PCE
Patient Care Evaluation
- THCP
Teaching Hospital Cancer Program
- US
United States
Footnotes
Presented at the 47th Annual Meeting of the Society for Surgery of the Alimentary Tract, Los Angeles, CA, May 20–25, 2006
The members of the Gastric Patient Care Evaluation Group from the Commission on Cancer are as follows:
Carol Conner Ph.D., M.D.; Thomas Habermann, M.D.; Scott A. Hundahl, M.D.; Martin S. Karpeh, Jr., M.D.; M. Margaret Kemeny, M.D.; Paul J. Kurtin, M.D.; Gregory Y. Lauwers, M.D.; Paul F. Mansfield, M.D.; Peter W. T. Pisters, M.D.; Andrew K. Stewart, M.A.; Zuo Feng Zhang, M.D.
DISCUSSION
Richard H. Bell, Jr. M. D. (Chicago Ill): Thank you, Dr. Reid-Lombardo. That was a nice presentation. This report deals with the thorny issue of where complex GI procedures are best done and by whom, and this is not an academic discussion, because in your data you suggest that there are about 500 preventable operative deaths per year in the United States for gastric cancer patients. The difficult question for me in all of this is identifying what is about large tertiary hospitals that results in better operative outcomes for complex GI cases. I think if we are going to argue for centralization, we need to understand the rationale for doing that. It is obviously a contentious issue.
So in this respect, I was disappointed a little bit with the way you examined your results, becaused you claimed that surgical volume predicts good outcomes, but this was really not a study of volume. You divided your hospitals based on structure and not on volume. You divided the hospitals based on their classification by the American College of Surgeons. And although you didn’t show it, there was data in the manuscript that showed there were actually significant volume overlaps between the three categories. It was certainly not a clean distinction by volume. Some of the less complex hospitals in your study did as many operations as the tertiary medical centers, although their average number was smaller. So I thought this study would have been more illuminating if, in addition to what you look at, you had looked at volume as an independent variable in addition to structure. You actually have an unusual study here in that you can look at both structure and volume. It may be that the structure of tertiary hospitals the better equipment, the higher performing ICUs, the resident coverage, all of these things are the primary driver of better outcomes.
From an educational point of view, this has implications in the sense that it would not do any good to do a better job of training community general surgeons to be more proficient in gastrectomy if the community hospitals is fundamentally a less capable environment for this type of operation. You have the data to lok at both structure and volume, and I wish you had done that perhaps in the future manuscript you will. Thank you.
Kaye Reid-Lombardo, M. D. (Rochester, Minn) Thank you, Dr. Bell. I definitely agree with you that it is less about volume, especially with gastric cancer, because, you are right, even at teaching hospitals the volumes are not that high. I think it has more to do with the infrastructure at each institution, as is being reported by many authors as well. I think factors such as ICU availability or the radiologic availability at each institution have more of an impact than the surgeon in and of him or herself. So I think this study will allow us to further examine things like infrastructure and see what the differences are and to make better recommendations based on that examination.
Michael A. Choti, M. D. (Baltimore, Md) Dr. Reid-Lombardo, I enjoyed your presentation and thank you for the opportunity to have reviewed the manuscript in advance. This is just another example of how the National Cancer Database can be a useful tool for analyzing a variety of cancer mangement question. This database is robust and different from SEER or other databases. I encourage others to utilize this resource.
I have two questions. The first, you report a surprisingly low number of palliative gastrectomies, yet you define this as margin positive resection and not based on symptomatic indications. While it is uncommon to have both proximal and distal positive margins, even in palliative resections, it is more common to have noncurative or R2 gastrectomy. In such cases the margins may be negative but residual disease is left behind as nodes or peritoneal implants. Were you able to see the difference between the margin positive and R2 resection?
The second question relates to the number of lymph nodes evaluated in this series. As you know, guidelines recommend histologic evaluation of at least 15 nodes. It was interesting how few gastrectomies in fact achieve greater 15 nodes. This number is significantly less than even colorectal cancer, where much attention has recently been given. Do you think in this case it is the pathologist diligence as much as the extent of lymphadenectomy that is the problem? Or are we setting the bar to high with the recommendation of 15 or more nodes? Thank you.
Dr. Reid-Lombardo: Thank you for questions. To answer your first question, it aws difficult to analyze R2, and so that is why we difficult to analyze R2, and so that is why we evaluated based on margin status. I would have expected a higher palliative rate as well. We could not analyze that from the database. I think to improve on the lymph node sampling , the pathologist and surgeon must work hand in hand. The pathologist should analyze the resected specimen at the time that the surgeon is still in the operating room and provide feedback. If there are less than 15 nodes, then more node sampling should be done by the surgeon. Now, I know the western studies have not shown improved survival among patients with D1 and D2 rescted nodes, but sampling more nodes would almost seem to me to assure that we are doing a more extended lymphadenectomy. One way that we can achieve that goal is to have better discussion, and not only discussion, but the pathology report should clearly indicate how many nodes have been examined and how many are positive or negative. Thank you.
Contributor Information
Kaye M. Reid-Lombardo, Phone: +1-507-284-2931, FAX: +1-507-284-5196, Email: reidlombardo.kaye@mayo.edu
Greer Gay, Phone: +1-312-202-5439, FAX: +1-312-202-5009, Email: ggay@facs.org.
Lina Patel-Parekh, Phone: +1-312-202-5085, FAX: +1-312-202-5009, Email: lpatel-parekh@facs.org.
Jaffer A. Ajani, Phone: +1-713-792-2828, FAX: +1-713-745-1163, Email: jajani@mdanderson.org
John H. Donohue, Phone: +1-507-284-2511, FAX: +1-507-284-5196, Email: donohue.john@mayo.edu
References
- 1.American Cancer Society. Cancer Facts and Figures 2006. Atlanta, GA: American Cancer Society, 2006.
- 2.Hundahl SA, Phillips JL, Menck HR. The National Cancer Data Base report on poor survival of U.S. gastric carcinoma patients treated with gastrectomy: Fifth edition American Joint Committee on Cancer staging, proximal disease, and the “different disease” hypothesis. Cancer 2000;88:921–932. [DOI] [PubMed]
- 3.Imamura Y, Yoshimi I. Comparison of cancer mortality (stomach cancer) in five countries: France, Italy, Japan, UK and USA from the WHO mortality database (1960–2000). Jpn J Clin Oncol 2005;35:103–105. [DOI] [PubMed]
- 4.Testa T, Nahum MA, Spinelli E, Carbone E, Flocchini GP, Motta G. The “Will Rogers effect” on stage grading. Ann Ital Chir 1999;70:829–830. [PubMed]
- 5.Birkmeyer JD, Warshaw Al, Finlayson SRG, Grove MR, Tosteson ANA. Relationship between hospital volume and late survival after pancreaticoduodectomy. Surgery 1999;126:78–83. [DOI] [PubMed]
- 6.Bach PB, Cramer LD, Schrag D, Downey RJ, Gelfand SE, Begg CB. The influence of hospital volume on survival after resection for lung cancer. N Engl J Med 2001;345:81–88. [DOI] [PubMed]
- 7.Schrag D, Cramer LD, Bach PB, Cohen AM, Warren JL, Begg CB. Influence of hospital procedure volume on outcomes following surgery for colon cancer. JAMA 2000;284:3028–3035. [DOI] [PubMed]
- 8.Birkmeyer JD, Siewers AE, Finlayson EV, Stukel TA, Lucas FL, Batista I, Welch HG, Wennberg DE. Hospital volume and surgical mortality in the United States. N Engl J Med 2002;346:1128–1137. [DOI] [PubMed]
- 9.Nomura E, Tsukuma H, Ajiki W, Oshima A. Population-based study of relationship between hospital surgical volume and 5-year-survival of stomach cancer patients in Osaka, Japan. Cancer Sci 2003;94:998–1002. [DOI] [PMC free article] [PubMed]
- 10.North American Association of Central Cancer Registries. Standards for cancer registries. In Hultstrom D, ed. Required Status Table, vol. II, version 10, Chap. IX. Springfield, IL: NAACCR, 2002, pp 107–118.
- 11.Commission on Cancer. Standards of the Commission on Cancer, vol. II: Registry Operations and Data Standards. Chicago, IL: American College of Surgeons, 1998.
- 12.Shaffer JP. Multiple hypothesis testing. Annu Rev Psychol 1995;46:561–584. [DOI]
- 13.The Research Group for Population-based Cancer Registration in Japan. Cancer incidence and incidence rates in Japan in 1999: Estimates based on data from 11 population-based cancer registries. Jpn J Clin Oncol 2004;34:352–356. [DOI] [PubMed]
- 14.Wanebo HJ, Kennedy BJ, Winchester DP, Fremgen A, Stewart AK. Gastric carcinoma: Does lymph node dissection alter survival? J Am Coll Surg 1996;183:616–624. [PubMed]
- 15.Bonenkamp JJ, Songun I, Hermans J, Sasako M, Welvaart K, Plukker JT, van Elk P, Obertop H, Gouma DJ, Taat CW, et al. Randomised comparison of morbidity after D1 and D2 dissection for gastric cancer in 996 Dutch patients. Lancet 1995;345:745–748. [DOI] [PubMed]
- 16.Cuschieri A, Fayers P, Fielding J, Craven J, Bancewicz J, Joypaul V, Cook P. Postoperative morbidity and mortality after D1 and D2 resections for gastric cancer: Preliminary results of the MRC randomised controlled surgical trial. The Surgical Cooperative Group. Lancet 1996;347:995–999. [DOI] [PubMed]
- 17.Fujii M, Sasaki J, Nakajima T. State of the art in the treatment of gastric cancer: From the 71st Japanese Gastric Cancer Congress. Gastric Cancer 1999;2:151–157. [DOI] [PubMed]
- 18.Sano T, Katai H, Sasako M, Maruyama K. One thousand consecutive gastrectomies without operative mortality. Br J Surg 2002;89:123. [PubMed]
- 19.Kodera Y, Sasako M, Yamamoto S, Sano T, Nashimoto A, Kurita A, Gastric Cancer Surgery Study Group of Japan Clinical Oncology Group. Identification of risk factors for the development of complications following extended and superextended lymphadenectomies for gastric cancer. Br J Surg 2005;92:1103–1109. [DOI] [PubMed]
- 20.Wainess RM, Dimick JB, Upchurch GR Jr, Cowan JA, Mulholland MW. Epidemiology of surgically treated gastric cancer in the United States, 1988–2000. J Gastrointest Surg 2003;7:879–883. [DOI] [PubMed]
- 21.Birkmeyer JD, Siewers AE, Finlayson EV, Stukel TA, Lucas FL, Batista I, Welch HG, Wennberg DE. Hospital volume and surgical mortality in the United States. N Engl J Med 2002;346:1128–1137. [DOI] [PubMed]
- 22.Jarhult J. The importance of volume for outcome in cancer surgery—an overview. Eur J Surg Oncol 1996;22:205–210. [DOI] [PubMed]
- 23.Bottcher K, Siewert JR, Roder JD, Busch R, Hermanek P, Meyer HJ. Risk of surgical therapy of stomach cancer in Germany. Results of the German 1992 Stomach Cancer Study. German Stomach Cancer Study Group (‘92). Chirurg 1994;65:298–306. [PubMed]
- 24.Damhuis RA, Meurs CJ, Dijkhuis CM, Stassen LP, Wiggers T. Hospital volume and post-operative mortality after resection for gastric cancer. Eur J Surg Oncol 2002;28:401–405. [DOI] [PubMed]
- 25.Karpeh MS, Leon L, Klimstra D, Brennan MF. Lymph node staging in gastric cancer: Is location more important than number? An analysis of 1,038 patients. Ann Surg 2000;232:362–371. [DOI] [PMC free article] [PubMed]
- 26.Smith DD, Schwarz RR, Schwarz RE. Impact of total lymph node count on staging and survival after gastrectomy for gastric cancer: data from a large US-population database. J Clin Oncol 2005;23:7114–7124. [DOI] [PubMed]


