Abstract
Hemangioblastomas, the most frequent manifestation of the hereditary von Hippel-Lindau disease (VHL), are highly vascularized tumors of the central nervous system. In previous studies, the endothelial-specific mitogen vascular endothelial growth factor (VEGF) was shown to be up-regulated in the stromal cells, the putative neoplastic cells in hemangioblastomas. Therefore, it was suggested that secretion of VEGF by stromal cells is the pathogenetic cause of the vascular lesions in hemangioblastomas. The novel basic helix loop helix transcription factor HRF/HIF-2α is a candidate regulator of VEGF expression during development. We therefore investigated expression of HRF/HIF-2α in hemangioblastomas and found the overexpression of VEGF mRNA in stromal cells to be highly correlated with elevated expression levels of HRF/HIF-2α mRNA. This finding is suggestive for a role of HRF in VEGF-dependent vascular growth in hemangioblastomas and could provide a link between transcriptional activation of the VEGF gene and loss of function of the VHL gene product. (Am J Pathol 1998, 153:25–29)
Hemangioblastomas, vascular tumors of the central nervous system, are the most frequent manifestation of the autosomal dominantly inherited von Hippel-Landau (VHL) disease. These tumors consist of a dense network of capillaries interspersed with stromal cells. The nature and the origin of the stromal cells is unclear up to now. It was hypothesized that stromal cells represent the neoplastic cell type in hemangioblastomas. 1 This view was substantiated by the finding that stromal cells produce high amounts of vascular endothelial growth factor (VEGF). 2 VEGF is a mitogen and survival factor specific for endothelial cells. 3 The specificity of VEGF for endothelial cells is due to the fact that the receptors for VEGF are almost exclusively expressed by endothelial cells. 4,5 There are several lines of evidence that the VEGF/VEGF receptor signal transduction system is the major physiological regulator of vascular growth in vivo. 6 In addition, VEGF appears to be a major regulator of tumor angiogenesis. In glioblastomas, which are malignant brain tumors of astrocytic origin, the expression level of VEGF has been shown to be correlated with vascular density of the tumors. However, unlike hemangioblastomas, which show a uniform up-regulation of VEGF in almost all stromal cells, glioblastomas show up-regulation only in those tumor cells that are adjacent to necroses. 7-9 Consistent with this finding, it was demonstrated that VEGF is up-regulated in glioma cells in response to hypoxia. 10 Recently, a heterodimeric transcription factor that was denominated hypoxia inducible factor 1 (HIF-1), was identified as a putative link between oxygen tension in a given tissue and expression of hypoxia-sensitive genes such as VEGF and erythropoietin. 11 Such genes share the presence of hypoxia-responsive elements in their 5′ and 3′ flanking regions, which are binding sites for HIF. 12 As this system was based on the reciprocal relationship of oxygen tension and growth factor expression, it was unclear whether it is involved in the high expression levels of VEGF in stromal cells of hemangioblastomas. More recently, a homologue of the α-subunit of HIF-1 was cloned by our and other groups and was denominated HRF/EPAS-1/HLF (which stands for HIF-related factor/endothelial PAS protein-1/HIF-like factor) 13-15 and recently renamed HIF-2α. 16 In the mouse embryo, this new factor is expressed by endothelial cells, neurons, smooth muscle cells, and a variety of epithelia. Endothelial expression of HRF could be correlated to the in vitro transactivation of the promoter of the receptor tyrosine kinase tie-2, 15 which is essential for signal transduction during embryonic angiogenesis. 17,18 In epithelia, the level of mRNA expression is highly correlated with the expression of VEGF. In addition, HRF acts as a transactivator of VEGF promoter constructs in vitro. 14 Thus, HRF is a novel candidate regulator of angiogenesis. To elucidate whether HRF could also be involved in the high VEGF expression observed in stromal cells of hemangioblastomas, we examined a series of cerebellar hemangioblastomas for the expression of VEGF and HRF and compared these data with normal brain and cerebellum. We found high expression levels of HRF mRNA in VEGF-producing stromal cells and the surrounding capillaries. This finding is suggestive for a role of this transcriptional regulator in VEGF-dependent vascular growth in hemangioblastomas.
Materials and Methods
Specimens
Tumor specimens were immediately snap-frozen after their removal and stored at −70°C until further use. Fourteen hemangioblastomas (from patients both with and without clinically confirmed VHL disease) and four glioblastomas were taken from the brain tumor bank of the Department of Neuropathology, Freiburg University Medical Center. For control purposes, normal cerebellum and normal cerebrum from two patients without neurological disease were obtained postmortem and snap-frozen in liquid nitrogen.
RNA Extraction and Northern Analysis
Total RNA was isolated using the guanidinium thiocyanate method. 19 Aliquots of 20 μg were separated on 1% agarose gels containing 0.66 mol/L formaldehyde in 1× MOPS buffer (40 mmol/L 3-(N-morpholino)propanesulfonic acid, 10 mmol/L sodium acetate, and 1 mmol/L EDTA (pH 7.2) and transferred to nylon membrane (Duralon-UV, Stratagene, La Jolla, CA) by standard procedures in 20× SSC (1× SSC is 150 mmol/L NaCl, 15 mmol/L sodium citrate). Probes for the mouse HRF cDNA (nt 1688-2344), human VEGF cDNA (kindly provided by Dr. H. Weich, Braunschweig), human HIF-1α cDNA (500-bp HindIII fragment, kindly provided by Dr. H. Marti, Bad Nauheim), and β-actin were labeled with [32P]dCTP using a random primer labeling kit (Stratagene). Hybridizations were carried out as described previously. 20
In Situ Hybridization
In situ hybridization was performed as described previously. 13 Single-stranded sense and antisense [35S]-labeled RNA probes were generated from a PstI-HindIII fragment of HRF subcloned in pBluescript and a hVEGF121 pBluescript plasmid using T3 and T7 polymerases (Stratagene) and [35S]UTP. Sections were hybridized with 2.5 × 10 4 cpm/μl probe overnight in hybridization buffer (50% formamide, 10% dextran sulfate, 10 mmol/L Tris/HCl, pH 7.5, 10 mmol/L sodium phosphate, pH 6.8, 5 mmol/L EDTA, 150 μg/ml Escherichia coli tRNA, 0.1 mmol/L UTP, 1 mmol/L ADPβS, 10 mmol/L γS-ATP, 1 mmol/L dithiothreitol, 1 mmol/L 2-mercaptoethanol) at 42°C. After washes at high stringency (2× SSC, 50% formamide, 10 mmol/L 2-mercaptoethanol, 48°C) slides were coated with Kodak NTB-2 emulsion and exposed for 2 to 4 weeks in the dark. Slides were developed in Kodak D-19 developer and counterstained with Giemsa’s solution (Merck, Darmstadt, Germany).
Immunohistochemistry
To identify endothelial cells, sectiones were incubated with anti-PECAM/CD31 mouse monoclonal antibody (BioGenex, San Ramon, CA), and binding of the antibody was visualized using the Peroxidase LSAB Kit (Dako, Glostrup, Denmark), according to the manufacturer’s instructions.
Results and Discussion
The high expression of VEGF in stromal cells of capillary hemangioblastomas is considered to be an important pathogenetic determinant for the formation of the vascular tumor phenotype. 2 HRF/EPAS-1, a novel member of the basic helix loop helix family of transcription factors, was recently shown to be a candidate regulator of VEGF gene expression in the mouse embryo. 14 We therefore examined the expression pattern of HRF in a series of seven hemangioblastomas and found striking correlation of VEGF expression with HRF expression in the stromal cells. Specimens were examined by in situ hybridization of parallel sections with HRF and VEGF antisense probes and sense probes as a control. Hybridization with sense probes did not show any specific signal (Figure 1D) ▶ . By hybridization with VEGF antisense, VEGF expression was seen in areas of clustered stromal cells as described previously (Figure 1, A and B) ▶ . These areas were overlapping with those expressing HRF in parallel sections (Figure 1C) ▶ . At higher magnification, HRF expression was detected in both stromal and endothelial cells (Figure 1F) ▶ . In contrast to previous studies in mouse embryos, in which HRF message was found in all capillary endothelia of the brain, no significant signal could be recorded on the tumor capillaries unless they were located in the vicinity of VEGF-producing stromal cells. The identity of endothelial cells and their discrimination from stromal cells could be confirmed by immunostaining of parallel sections with an anti-CD31 (PECAM) antibody (Figure 1G) ▶ . In the normal cerebellum, Purkinje cells exhibited noticeable hybridization with HRF antisense probe. The signal for HRF was less pronounced in endothelial cells and in neurons of the granular layer (not shown). In normal cerebrum, neurons, but not the glial cells of the gray matter, revealed hybridization signals for HRF. Less pronounced signals were seen on capillary endothelial cells as described previously for the mouse adult brain 13 (data not shown).
Figure 1.
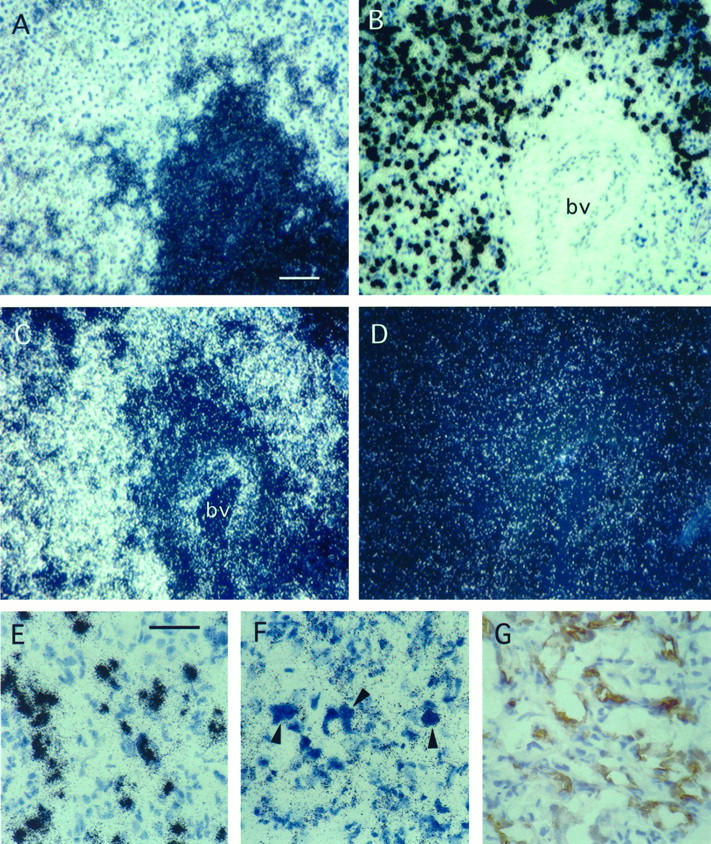
Expression of VEGF in stromal cells of capillary hemangioblastomas is correlated with expression of the transcription factor HRF/HIF-2α as shown by in situ hybridization. Parallel sections of a representative capillary hemangioblastoma were hybridized with 35S-labeled VEGF antisense RNA probe (A, dark-field illumination; B, bright-field view), 35S-labeled HRF/HIF-2α antisense RNA probe (C), or 35S-labeled sense control RNA probe (D). Whereas VEGF is expressed exclusively by stromal cells E, HRF/HIF-2α mRNA is detected on both stromal and endothelial cells (F). Endothelial cells are distinguished from stromal cells by the shape of their nuclei and positive staining for CD31 (G), bv, large blood vessel. Scale bars, 100 μm (A–D) and 50 μm (E–G).
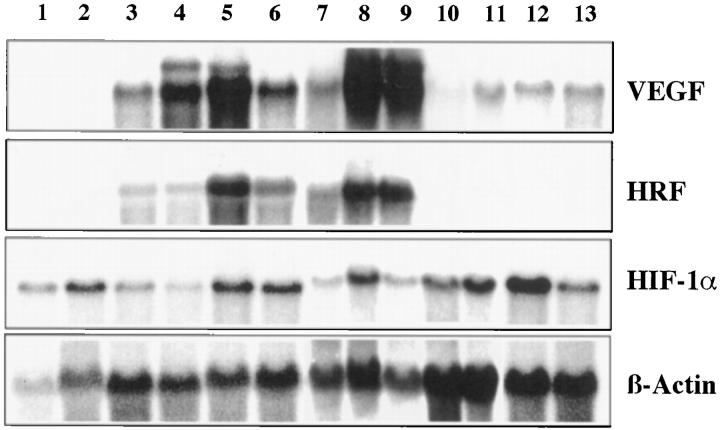
The results from Northern hybridization confirmed the data from in situ hybridization. Whereas in normal brain and cerebellum only low levels of HRF and VEGF mRNAs were detectable, strong hybridization signals were obtained in tissue from seven different hemangioblastomas (Figure 2) ▶ . Interestingly, the expression level of VEGF mRNA, which significantly varied among the different hemangioblastoma specimens, was highly correlated with the expression level of HRF. In contrast, the expression level of HIF-1α mRNA did not differ significantly between normal tissues and hemangioblastomas. In four glioblastomas that were analyzed for comparison, no elevated levels of HRF could be detected, although in three of these the VEGF mRNA was clearly up-regulated. The level of HIF-1α mRNA message was essentially the same in all tumors and normal tissues examined (Figure 2) ▶ .
Figure 2.
Northern blot analysis of total RNA from different human tissues. Lane 1, normal cerebrum; lane 2, normal cerebellum; lane 3 to 9, seven different capillary hemangioblastomas; lanes 10 to 13, four different glioblastomas. The membrane was hybridized with a 32P-labeled human VEGF cDNA and reprobed with a mouse HRF/HIF-2α probe, a human HIF-1α probe, and a β-actin cDNA as loading control.
These findings suggest that the mechanisms of VEGF up-regulation in hemangioblastomas differ from those in glioblastomas. In glioblastomas, high VEGF expression is seen in the so-called palisading cells in the vicinity of necrosis. It was therefore assumed that metabolic factors related to the necrosis play an important role for expression of the VEGF gene in these cells. 8,9 Hypoxia is believed to be the most important factor among these, and the transcription factor HIF-1α is the most likely mediator of gene activation in response to hypoxia. 21 In vitro, the HIF-1α transcription factor has been shown to transactivate the VEGF promoter via binding to a hypoxia-responsive element. 22 Several groups observed an increase in the protein level of HIF-1α via stabilization of the protein under hypoxic conditions whereas no induction of mRNA was observed. 12,23 In addition, all in situ data on HIF-1α mRNA that are available so far do not reveal a correlation between expression of HIF-1α mRNA and VEGF. In contrast, mRNA expression of HRF, the close relative of HIF-1α, is correlated with expression of VEGF mRNA in the lung and in other organs. 13,14 This also holds true for hemangioblastoma stromal cells. On the other hand, no such striking correlation between HRF and VEGF mRNA expression was observed in glioblastomas. Only a slight elevation of HRF mRNA level was observed in the palisading cells (data not shown), which is consistent with the data from Northern blot hybridization.
Hemangioblastoma, which may serve as a model for pathological angiogenesis in response to overexpression of VEGF, is the most frequent manifestation of VHL disease. 1 It may be speculated that loss-of-function mutations of the VHL gene result in increased gene expression of HRF in stromal cells of hemangioblastomas. VHL has recently been shown to control transcriptional elongation by interaction with elongin B and C. 24,25 One possibility, therefore, is that VHL controls transcriptional elongation of HRF in stromal cells. As the histogenesis of stromal cells is undefined, and no stromal cell line is available at present, this hypothesis requires additional evaluation. Whether this proposed dysregulation could hold true for other VHL-associated lesions, such as kidney and adrenal gland tumors, remains also to be examined. It has been shown that VHL mutations in certain cell lineages lead to an increased stability of VEGF mRNA. 26,27 This mechanism is different from transcriptional activation of VEGF, which is expected to occur in response to HRF. Additional experiments on the molecular interaction of VHL, HRF, and the VEGF gene are necessary.
The increased expression of HRF in capillary endothelial cells located in close vicinity of VEGF-overexpressing stromal cells in contrast to more distant endothelial cells may result from activation of endothelium by VEGF or other stromal-cell-derived factors. As HRF was found to be a transactivator of tie-2 promoter in vitro, it may be assumed that this transcription factor is involved in the transcriptional control of the tie genes, which are expressed in capillary endothelial cells in hemangioblastomas 28 (our own unpublished observations). Thus, it may be speculated that VEGF receptors that are up-regulated in hemangioblastoma capillaries 2 are also under the control of HRF.
In conclusion, our data show for the first time a correlation of HRF expression in a human tumor with overexpression of VEGF. The fact that this particular tumor is highly vascular underlines the central role this novel transcription factor may have for the regulation of vascular growth and differentiation.
Acknowledgments
We thank Dr. Werner Risau (Max-Planck-Institut Bad Nauheim) for continuous support and critical reading of the manuscript.
Footnotes
Address reprint requests to D. Ingo Flamme, Zentrum für Molekularbiologische Medizin der Universität zu Köln, Joseph-Stelzmann-Strasse 9, D 50924 Köln, Germany. E-mail: ingo.flamme@medizin.uni-koeln.de.
Supported by grant C4 from the Center for Clinical Research I, Freiburg University Medical School, and by grant PI158/3-1 from the Deutsche Forschungsgemeinschaft to K. H. Plate.
References
- 1.Wizigmann-Voos S, Plate KH: Pathology, genetics and cell biology of hemangioblastomas. Histol Histopathol 1996, 11:1049-1061 [PubMed] [Google Scholar]
- 2.Wizigmann-Voos S, Breier G, Risau W, Plate KH: Up-regulation of vascular endothelial growth factor and its receptors in von Hippel-Lindau disease-associated and sporadic hemangioblastomas. Cancer Res 1995, 55:1358-1364 [PubMed] [Google Scholar]
- 3.Ferrara N, Houck K, Jakeman L, Leung DW: Molecular and biological properties of the vascular endothelial growth factor family of proteins. Endocr Rev 1992, 13:18-32 [DOI] [PubMed] [Google Scholar]
- 4.Millauer B, Wizigmann Voos S, Schnürch H, Martinez R, Moller NP, Risau W, Ullrich A: High affinity VEGF binding and developmental expression suggest Flk-1 as a major regulator of vasculogenesis and angiogenesis. Cell 1993, 72:835-846 [DOI] [PubMed] [Google Scholar]
- 5.Peters KG, De Vries C, Williams LT: Vascular endothelial growth factor receptor expression during embryogenesis and tissue repair suggests a role in endothelial differentiation and blood vessel growth. Proc Natl Acad Sci USA 1993, 90:8915-8919 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Breier G, Risau W: The role of vascular endothelial growth factor in blood vessel formation. Trends Cell Biol 1996, 6:454-456 [DOI] [PubMed] [Google Scholar]
- 7.Plate KH, Breier G, Weich HA, Mennel HD, Risau W: Vascular Endothelial Growth Factor and Glioma Angiogenesis—Coordinate induction of VEGF receptors, distribution of VEGF protein and possible in vivo regulatory mechanisms. Int J Cancer 1994, 59:520-529 [DOI] [PubMed] [Google Scholar]
- 8.Plate KH, Breier G, Weich HA, Risau W: Vascular endothelial growth factor is a potential tumour angiogenesis factor in human gliomas in vivo. Nature 1992, 359:845-848 [DOI] [PubMed] [Google Scholar]
- 9.Shweiki D, Itin A, Soffer D, Keshet E: Vascular endothelial growth factor induced by hypoxia may mediate hypoxia-initiated angiogenesis. Nature 1992, 359:843-845 [DOI] [PubMed] [Google Scholar]
- 10.Damert A, Machein M, Breier G, Fujita MQ, Hanahan D, Risau W, Plate KH: Two distinct mechanisms contribute to upregulation of vascular endothelial growth factor in the palisading cells of rat glioma. Cancer Res 1997, 57:3860-3864 [PubMed] [Google Scholar]
- 11.Wang GL, Jiang BH, Rue EA, Semenza GL: Hypoxia inducible factor 1 is a basic helix loop helix pas heterodimer regulated by cellular O2 tension. Proc Natl Acad Sci USA 1995, 92:5510-5514 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wenger RH, Rolfs A, Marti HH, Guenet JL, Gassmann M: Nucleotide sequence, chromosomal assignment and mrna expression of mouse hypoxia inducible factor 1 alpha. Biochem Biophys Res Commun 1996, 223:54-59 [DOI] [PubMed] [Google Scholar]
- 13.Flamme I, Froehlich T, von Reutern M, Kappel A, Damert A, Risau W: HRF, a putative basic helix-loop-helix-PAS-domain transcription factor is closely related to hypoxia-inducible factor-1a and developmentally expressed in blood vessels. Mech Dev 1997, 63:51-60 [DOI] [PubMed] [Google Scholar]
- 14.Ema M, Taya S, Yokotani N, Sogawa K, Matsuda Y, Fujii-Kuriyama Y: A novel bHLH-PAS factor with close sequence similarity to hypoxia-inducible factor 1a regulates the VEGF expression and is potentially involved in lung and vascular development. Proc Natl Acad Sci USA 1997, 94:4273-4278 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tian H, McKnight SL, Russel DW: Endothelial PAS domain protein 1 (EPAS1), a transcription factor selectively expressed in endothelial cells. Genes Dev 1997, 11:72-82 [DOI] [PubMed] [Google Scholar]
- 16.Wenger RH, Gassmann M: Oxygen(es) and the hypoxia-inducible factor-1. Biol Chem 1997, 378:609-616 [PubMed] [Google Scholar]
- 17.Sato TN, Tozawa Y, Deutsch U, Wolburg-Buchholz K, Fujiwara Y, Gendronmaguire M, Gridley T, Wolburg H, Risau W, Qin Y: Distinct roles of the receptor tyrosine kinases Tie-1 and Tie-2 in blood vessel formation. Nature 1995, 376:70-74 [DOI] [PubMed] [Google Scholar]
- 18.Suri C, Jones PF, Patan S, Bartunkoya S, Maisonpierre PC, Davis S, Sato TN, Yancopoulos GD: Requisite role of angiopoietin-1, a ligand for the tie2 receptor, during early embryonic angiogenesis. Cell 1996, 87:1171-1180 [DOI] [PubMed] [Google Scholar]
- 19.Chomczynski P, Sacchi N: Single step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem 1987, 162:156-159 [DOI] [PubMed] [Google Scholar]
- 20.Stratmann R, Krieg M, Haas R, Plate KH: Putative control of angiogenesis in hemangioblastomas by the von Hippel-Lindau tumor suppressor gene. J Neuropathol Exp Neurol 1997, 56:1242-1252 [DOI] [PubMed] [Google Scholar]
- 21.Semenza GL: Transcriptional regulation by hypoxia-inducible factor 1: molecular mechanisms of oxygen homeostasis. Trends Cardiovasc Med 1996, 6:151-157 [DOI] [PubMed] [Google Scholar]
- 22.Forsythe JA, Jiang B, Iyer NV, Agani F, Leung SW, Koos RD, Semenza GL: Activation of vascular endothelial growth factor gene transcription by hypoxia-inducible factor 1. Mol Cell Biol 1996, 16:4604-4613 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Huang LE, Arany Z, Livingston DM, Bunn HF: Activation of hypoxia-inducible transcription factor depends primarily upon redox-sensitive stabilization of its alpha subunit. J Biol Chem 1996, 271:32253-32259 [DOI] [PubMed] [Google Scholar]
- 24.Kibel A, Iliopoulos O, DeCaprio JA, Kaelin WGJ: Binding of the von Hippel-Lindau tumor suppressor gene to elongin B and C. Science 1995, 269:1444-1446 [DOI] [PubMed] [Google Scholar]
- 25.Duan DR, Pause A, Burgess WH, Aso T, Chen DYT, Garrett KP, Conaway RC, Conaway JW, Linehan WM, Klausner RD: Inhibition of transcription elongation by the VHL tumor suppressor protein. Science 1995, 269:1402-1406 [DOI] [PubMed] [Google Scholar]
- 26.Gnarra JR, Zhou S, Merrill MJ, Wagner JR, Krumm A, Papavassiliou E, Oldfield EH, Klausner RD, Linehan M: Post-transcriptional regulation of vascular endothelial growth factor mRNA by the product of the VHL tumor suppressor gene. Proc Natl Acad Sci USA 1996, 93:10589-10594 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Iliopoulos O, Levy AP, Jiang C, Kaelin WGJ, Goldberg MA: Negative regulation of hypoxia-inducible genes by the von Hippel-Lindau protein. Proc Natl Acad Sci USA 1996, 93:10595-10599 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hatva E, Böhling T, Jääskeläinen J, Persico MG, Haltia M, Alitalo K: Vascular growth factors and receptors in capillary hemangioblastomas and hemangiopericytomas. Am J Pathol 1996, 148:763-775 [PMC free article] [PubMed] [Google Scholar]