Abstract
Osteolysis complicating arthroplasty reflects progressive generation of implant-derived wear particles, which prompt an inflammatory reaction attended by recruitment of osteoclasts to the prosthesis-bone interface. To identify a soluble mediator of peri-prosthetic osteolysis we first showed that implant particles induce c-src in murine bone marrow macrophages (BMMs), a protein specifically expressed when these cells commit to the osteoclast phenotype. The fact that tumor necrosis factor-α (TNF) is a potent osteoclastogenic agent while at the same time is the only soluble moiety known to be c-src inductive suggests that this cytokine may mediate implant particle-induced osteoclastogenesis. Consistent with this hypothesis, prosthesis-derived wear particles, recovered at revision arthroplasty, dose-dependently prompt TNF secretion by BMMs. Similarly, particulate polymemthylmethacrylate, the major component of orthopedic implant cement, induces BMM expression of TNF mRNA and protein in a time- and dose-dependent manner. Furthermore, failure of BMMs derived from mice deleted of both the p55 and p75 TNF receptors to express c-src in response to polymemthylmethacrylate indicates TNF is an essential mediator of particle induction of this osteoclast specific protein. To test the hypothesis that TNF mediates implant osteolysis, we established an in vivo murine model of this condition that histologically mirrors that of man. Verifying that TNF is essential to development of particle osteolysis, mice failing to express both the p55 and p75 TNF receptors are protected from the profound bone resorption attending polymemthylmethacrylate particle implantation on calvariae of wild-type animals. Finally, the protective effect of deletion of both TNF receptors is recapitulated in mice lacking only the p55 receptor. Thus, targeting TNF and/or its p55 receptor may arrest wear particle osteolysis.
Each year, 30,000 Americans undergo surgery to revise loosened orthopedic implants. In fact, periprosthetic osteolysis is the major complication following total joint replacement. As more than one million orthopedic prostheses are implanted annually worldwide, this problem promises to escalate. 1,2 Orthopedic implant loosening reflects generation of wear particles from articulating prosthetic joint surfaces and supporting cement, the latter consisting of polymethylmethacrylate (PMMA). 3 Particles so generated incite a chronic inflammatory response eventuating in osteoclastic bone resorption and mechanical failure of the implant. 4 The molecular mechanisms underlying this clinically important event, referred to as “wear debris-induced osteolysis,” are enigmatic.
The osteolytic process attending prosthetic loosening is associated with formation, at the implant-bone interface, of a cellular “membrane” containing abundant tissue macrophages, many of which have ingested implant-derived wear particles such as PMMA. 5-9 Several macrophage-produced osteoclastogenic cytokines, including interleukins-1 and -6 and tumor necrosis factor-α (TNF) are present in the tissue surrounding failed implants 6,10-12 and thus serve as potential mediators of wear particle-induced osteolysis. Hence, the most likely scenario holds that implant wear particles promote an inflammatory reaction consisting largely of fibroblasts and activated macrophages. Secretory molecules of the activated cells recruit osteoclasts, which are ultimately responsible for periprosthetic bone degradation. Thus, identification of proinflammatory cytokine(s) mediating the osteoclastogenesis of implant osteolysis is central to prevention of this complication.
Materials and Methods
Mice
Four- to 6-week-old lipopolysaccharide-resistant C3H/HeJ mice (Harlan Sprague-Dawley Inc., Indianapolis, IN) were used. Transgenic mice include: 1) mice in which the p55TNF receptor gene has been deleted 13 (provided by Dr. Warner Lesslauer (Hoffman-LaRoche)); 2) those in which both the p55TNF and p75TNF receptor genes have been deleted by interbreeding dominant negative p55TNF receptor knockout mice with their counterparts from p75TNF receptor knockout mice (provided by Dr. Mark Moore (Genentech, Inc., South San Francisco, CA)); and 3) their respective wild-type mice.
Particles
Wear particles were isolated from periprosthetic tissue obtained during revision surgery by a technique similar to that described by Campbell et al. 14 Briefly, tissue was minced, papain digested at 65°C for 24 hours, and centrifuged on a discontinuous sucrose gradient at 28,000 rpm at 4°C for 4 hours. The topmost layer was washed and placed under 10 ml each of 0.96 g/cm 3 and 0.90 g/cm 3 isopropanol and recentrifuged at 25°C for 2 hours. Particles recovered at the air/liquid interface were counted in a hemocytometer. FACS analysis documents 90% of recovered particles are 20 μm or less in diameter. Analysis of the particles by Fourier transform infrared spectroscopy demonstrates the presence of peaks coincident with those obtained with commercially prepared polyethylene (now shown). Commercially prepared PMMA particles, in the form of bone cement, were obtained from Wright Medical Technologies (Arlington, TN) with 50% of particles being 10 μm or less in diameter.
Particle Induction of c-src and TNF
Osteoclast precursors, in the form of bone marrow macrophages (BMMs), were obtained from long bone marrow. 15 Cells were grown to confluence in α-MEM/10% fetal calf serum containing 1000 U/ml M-CSF. TNF protein was measured in conditioned media using a commercial enzyme-linked immunosorbent assay (ELISA) (Genzyme, Cambridge, MA). Particle-exposed total BMM RNA was isolated, and TNF mRNA was detected by Northern analysis using a cDNA probe to bp 389-1180 primers. 18S ribosomal mRNA was used as a loading control. In a similar manner, BMMs were cultured with particles (10 8 particles/ml) for 24 hours, and cell associated c-src was determined by immunoblot using anti-c-src monoclonal antibody 327 (provided by Dr. Andrey Shaw, St. Louis, MO). 16
Transfection of J774A.1 Cells
The evening before transfection, 0.5 × 10 6 J774A.1 cells (ATCC T1B67) were added to each well of 6-well plates ± 2.0 mg/ml PMMA. 17 Cells were washed with 1 ml of OptiMEM (Life Technologies, Inc.), and medium was replaced with 0.8 ml of OptiMEM. The TNF promoter/reporter construct, TNF(−1059)chloramphenicol acetyl transferase (CAT) (provided by Dr. Bruce Beutler, Dallas, TX) was co-transfected with a luciferase reporter plasmid, pMB3–675Luc, as a control for transfection efficiency. pMB3–675Luc contains the first 675 bp of the murine integrin β3 promoter 18 and is not significantly affected by PMMA treatment. DNAs (TNF(−1059)CAT, 1.8 μg/well and pMB3–675Luc, 0.2 μg/well) were mixed in 0.1 ml of OptiMEM per well, and in a separate tube, lipofectAMINE (Life Technologies, Inc.) (5 μl/well) was mixed with 0.1 ml of OptiMEM per well. Diluted DNA samples and lipofectAMINE were mixed in equal volumes and incubated at R.T. for 30 minutes after which 0.2 ml of the mixture was added to each well. After 3 hours at 37°C +5% CO2, 1 ml of OptiMEM containing 20% fetal calf serum was added, and cells were incubated at 37°C +5% CO2 for an additional 20 hours. The wells were then washed with 1 ml of phosphate-buffered saline, scraped in 0.5 ml of phosphate-buffered saline, and transferred to 1.5 ml of microfuge tubes. Cells were pelleted in a microfuge at top speed for 30 seconds and resuspended in 50:1 reporter lysis buffer (Promega, Madison, WI). Cells were lysed twice for 20 seconds by vigorous vortexing. Cell debris was removed by microfuging for 1 minute. Twenty μl of extract were assayed for luciferase activity using the luciferase assay kit (Promega) with an Optocomp II luminometer (MGM Instruments). Twenty-five μl of extract were heat inactivated at 60°C for 10 minutes and mixed with 100 μl of 1× reporter lysis buffer containing 3 μl of 14C chloramphenicol (Amersham) and 0.25 mmol/L N-butryl CoA. After 16 hours of incubation at 37°C, CAT activity was determined by xylene extraction and liquid scintillation counting.
Murine Model of Particle-Induced Osteolysis
Fifteen to 20 g B6X127 p55−/−,p75−/−TNFr or p55−/−, p75+/+TNFr mice and wild-type littermates were anesthetized and the scalp incised longitudinally to expose the external cranial periosteum. The periosteum was elevated off the external cortex of the calvarium by sharp dissection. Thirty mg of commercially prepared PMMA particles were placed directly on the surface of the bone. Sham operation consisted of the entire procedure without particle implantation. The wound was then closed with small staples. Animals were sacrificed at 7 days, and the calvaria was excised, fixed, and decalcified in EDTA. Sagittal histological sections of temporal bones, approximately 2 to 3 mm lateral of the midsagittal suture, were stained for tartrate-resistant acid phosphatase (TRAP) activity. Both cortical porosity as an index of net osteolysis and the number of osteoclasts serve as our analytical target.
Statistics
Data are expressed as mean ± SD.
Results
Periprosthetic Wear Particles Prompt c-src Expression in Vitro
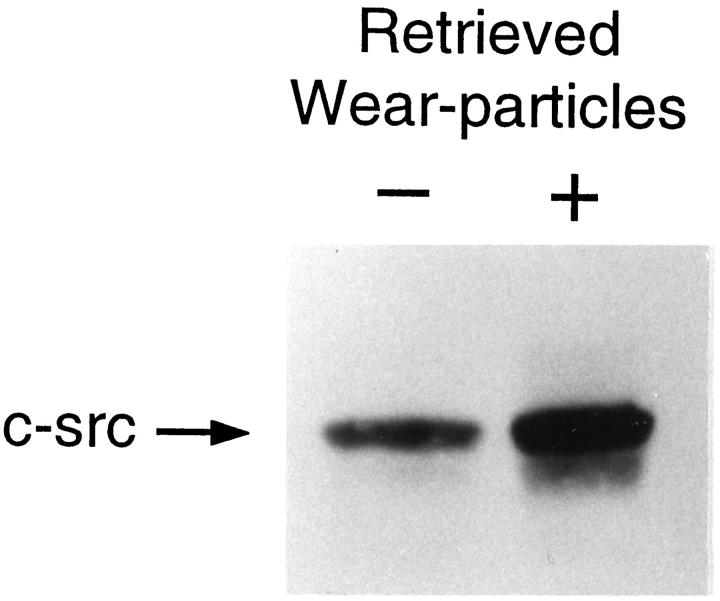
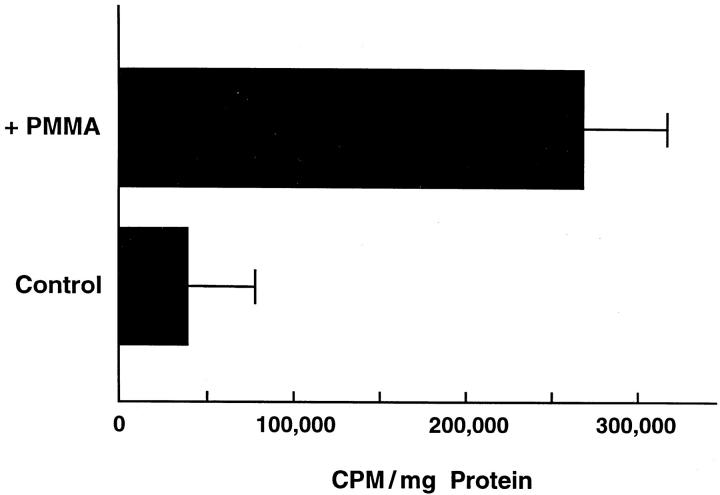
c-src expression is a specific marker of commitment to the osteoclast phenotype. 19 Thus, our first efforts to identify the cytokine mediating implant osteolysis were aimed at determining if prosthetic wear particles induce c-src in marrow macrophages, cells capable of acting as either mononuclear phagocytes or osteoclast precursors. 20 To this end, pure populations of murine BMMs derived from lipopolysaccharide resistant mice were isolated 15 and maintained with or without authentic wear particles extracted from periprosthetic tissue recovered from patients with failed implants undergoing revision surgery. 14 After 24 hours, the cells were lysed, and c-src content of equal amounts of protein were determined by immunoblot. BMMs exposed to patient-derived particles contain substantially more c-src protein than do nonparticle-exposed BMMs (Figure 1) ▶
Figure 1.
Periprosthetic wear particles induce c-src expression. BMMs, derived from endotoxin resistant C3H/HeJ mice and precultured 4 days, were maintained for 24 hrs ± particles (108/ml isolated from periprosthetic tissues obtained at revision arthroplasty). The cells were lysed and equal amounts of protein analyzed by immunoblot for c-src content.
Periprosthetic and PMMA Particles Induce TNF Protein and mRNA
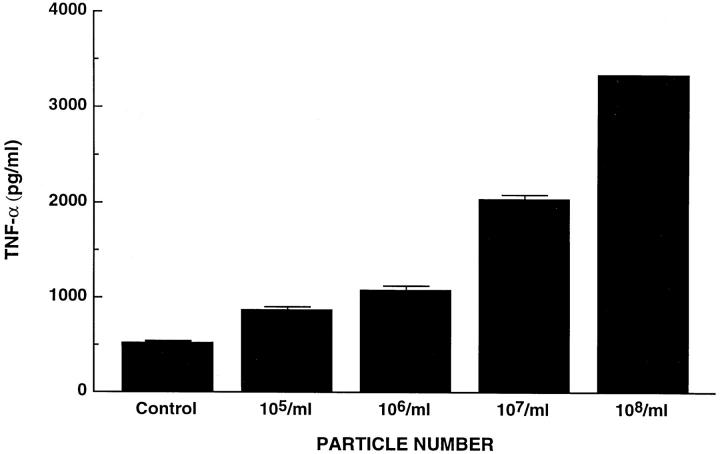
We have established that TNF is unique among a panel of osteoclastogenic cytokines in its capacity to induce c-src expression by mouse osteoclast precursors. 19 We also find that, like the human condition, 21 TNF prompts osteoclastogenesis in cultured murine marrow (data not shown). Because TNF and wear particles isolated from patients undergoing revision surgery both induce c-src expression, we reasoned particle-enhanced synthesis of the proto-oncogene might be a TNF-mediated event. Therefore, BMMs were exposed for 24 hours to increasing amounts of recovered wear debris. Measurement of TNF by ELISA (Genzyme, Cambridge, MA) within culture medium documents secreted cytokine parallels particle number (Figure 2) ▶ .
Figure 2.
Periprosthetic wear particles induce TNF expression. C3H/HeJ BMMs were precultured for 72 hours after which they were exposed to increasing numbers of retrieved wear particles. Secreted TNF was measured by ELISA 24 hours later.
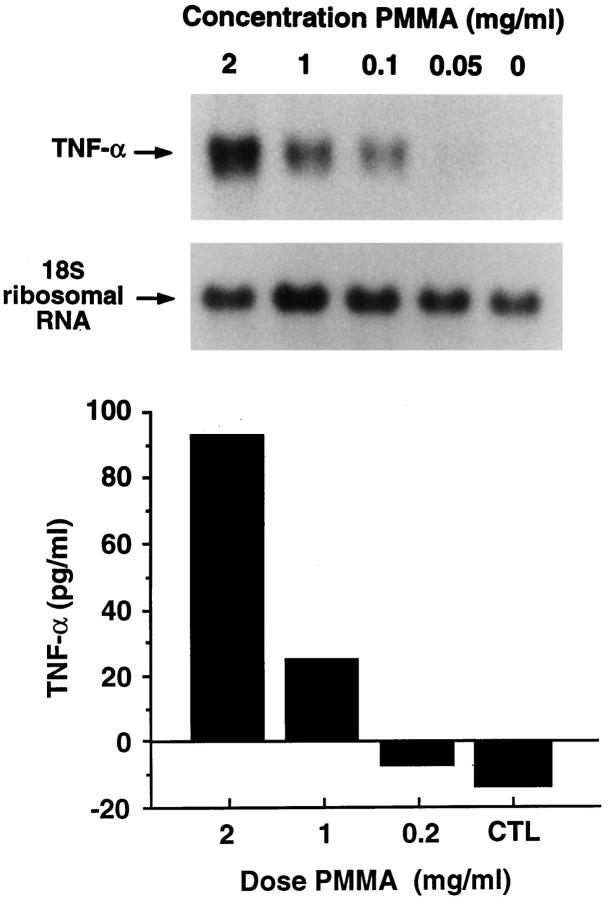
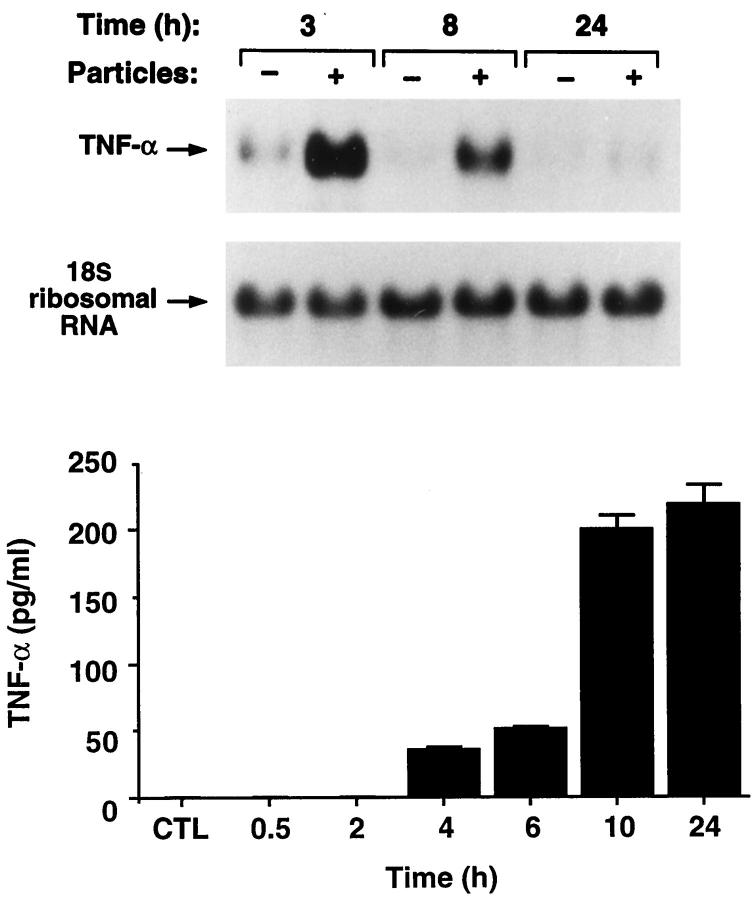
The amount of wear particles recoverable from periprosthetic tissues is limited. Thus, in the remainder of our experiments, we turned to a commercial source of particles. PMMA, in the form of bone cement, is a component of a large percentage of orthopedic implants. The polymer is abundant in wear debris 5-9 and is implicated as a major cause of osteolysis following total joint replacement surgery. 3 Therefore, we asked if commercial PMMA particles (50% <10 μm in diameter) (Wright Medical, Arlington TN), such as recovered wear debris, induce TNF. We find that TNF message and its secreted product increase with particle number (Figure 3) ▶ . In a similar experiment, performed with time, TNF mRNA substantially escalates within 3 hours of PMMA exposure after which it progressively declines and, by 24 hours, mirrors nonparticle-treated cells (Figure 4) ▶ . TNF, secreted under the influence of PMMA, is detectable by 4 hours and, in contrast to the short-lived message, remains elevated after 24 hours of particle exposure.
Figure 3.
PMMA particles induce TNF mRNA and protein in a dose-dependent manner. C3H/HeJ BMMs were precultured for 72 hours after which they were exposed to increasing amounts of PMMA particles. RNA was extracted and probed with a TNF cDNA (upper panel) 24 hours later. Secreted TNF was measured by ELISA (lower panel). The values represent the means of two assays. The negative values for control and 0.2 mg/ml PMMA reflect the sensitivity of the assay relative to the background.
Figure 4.
PMMA particles induce TNF mRNA and protein in a time-dependent manner. C3H/HeJ BMMs were precultured for 72 hours and exposed to 2 mg/ml PMMA. RNA was extracted with time and probed with a TNF cDNA (upper panel). Secreted TNF was measured by ELISA (lower panel).
PMMA Particles Transactivate the TNF Promoter
To determine whether particle-induced TNF reflects transactivation of the TNF gene, we maintained the murine macrophage line J774A.1 overnight with or without PMMA. The cells were then transfected with a CAT reporter construct (TNF(−1059)CAT) containing the full length murine TNF promoter 22 (provided by Dr. Bruce Beutler, Dallas TX) and normalized with co-transfected luciferase construct. After 23 hours, the transfectants were lysed and the lysate assayed for CAT activity. Pre-exposure of J774A.1 macrophages to PMMA particles induces the transfected TNF promoter almost fivefold (Figure 5) ▶ . Thus, PMMA particles enhance macrophage TNF expression by a mechanism involving gene transcription.
Figure 5.
PMMA transcriptionally activates the TNF gene. J774A.1 murine macrophages were exposed overnight to 2 mg/ml PMMA. The cells were washed and transfected with TNF(−1059)CAT reporter construct containing the full length (−1059 nt.) murine TNF promoter. After 23 hours they were lysed, and the lysate assayed for CAT activity. To control for transfection efficiency, all data were normalized to pMB3–675Luc, containing the first 675 nt. of the murine B3 integrin promoter.
BMMs from TNF Receptor-Deleted Mice Exposed to PMMA Particles Fail to Overexpress c-src
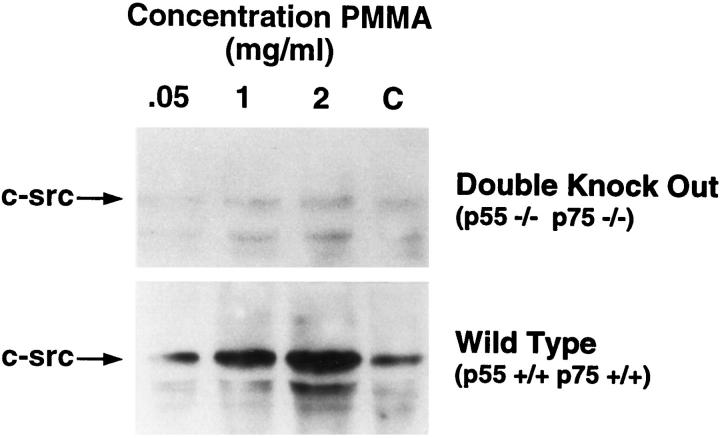
We hypothesized that particle induction of macrophage c-src is mediated by TNF. To further test the supposition, we turned to a genetically engineered mouse in which both the p55 and p75 TNF receptors are deleted (p55−/− p75−/−TNFr). 13 Wild type (p55+/+p75+/+TNFr) or TNF receptor deleted (p55−/−p75−/−TNFr) BMMs were cultured with PMMA particles. After 24 hours the cells were lysed, and equal amounts of protein probed by immunoblot for c-src content. BMMs from wild-type mice exposed to PMMA express c-src in a particle-dependent fashion, whereas the proto-oncogene is minimal and unaltered in p55−/−p75−/−TNFr BMMs regardless of particle number (Figure 6) ▶ . Because p55−/−p75−/−TNFr mice cannot respond to TNF, the cytokine is essential for PMMA-induced c-src expression in vitro.
Figure 6.
PMMA does not induce c-src in BMMs lacking TNF receptors. Wild type (p55+/+p75+/+TNFr) and TNF receptor-deleted (p55−/−p75−/−TNFr) BMMs were exposed for 24 hours to PMMA (2 mg/ml). Equal amounts of lysate protein were immunoblotted with anti-c-src monoclonal antibody.
TNF Receptor Deficient Mice Are Protected from PMMA Particle-Induced Osteolysis
Our in vitro findings suggest that TNF may be pivotal to implant osteolysis. To address this issue we developed an in vivo murine model involving placement of PMMA particles beneath the external calvarial periosteum and in direct contact with the sagittal aspects of both temporal bones. After 7 days, histological sections of temporal bones, 2 to 3 mm lateral to the midsagittal suture, were stained for TRAP activity, a feature characteristic of osteoclasts. Calvariae of wild-type mice (p55+/+,p75+/+TNFr) exposed to PMMA particles generate an inflammatory, supracranial response consisting of a fibrous membrane with abundant mononuclear cells, many of which contain phagocytosed cement particles (Figure 7) ▶ . Importantly, the PMMA-induced inflammatory membrane contains numerous juxtacortical osteoclasts, which within 1 week, have resorbed the width of the calvarial bone. Thus, the histological lesion induced by supracalvarial implantation of PMMA mimics tissue recovered at revision arthroplasty for prosthetic loosening. 23 Also, similar to the human situation, numerous cells within the murine lesion contain immunoreactive TNF (not shown).
Figure 7.
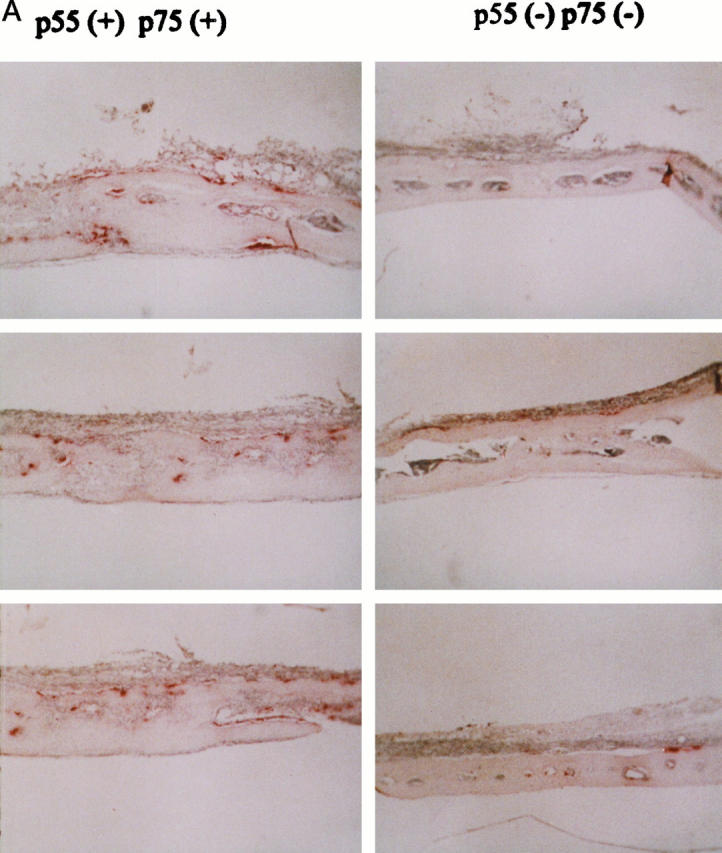
Mice lacking both TNFrs are protected from implant osteolysis. Original ×40 (A) and ×160 (B) magnification of calvaria of three pairs of littermates of p55+/+p75+/+ and p55−/−p75−/−TNFr mice in which 30 mg of PMMA was placed beneath the external periosteum. One week later the animals were sacrificed and histological sections, taken 2 to 3 mm lateral to the midsagittal suture, stained for TRAP activity. An inflammatory reaction with numerous osteoclasts (red-staining cells) that resorb though the calvarium occurs only in TNFr intact animals (p55+/+p75+/+TNFr) implanted with PMMA particles. Although double TNFr deleted mice (p55−/−/p75−/−TNFr) form an inflammatory membrane in response to PMMA particles, there is no evidence of enhanced osteoclastogenesis or bone resorption.
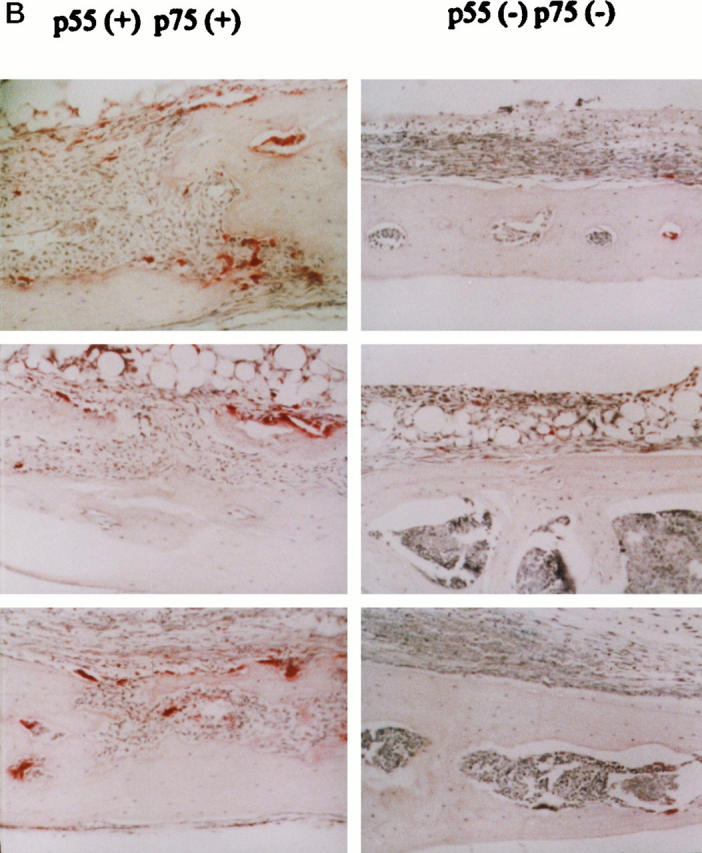
Next we implanted PMMA particles on the external calvaria of p55−/−p75−/−TNFr mice. Substantiating that TNF plays a pivotal role in the pathogenesis of implant osteolysis, the mutants similar to wild-type animals generate an inflammatory membrane, but are protected from particle-induced osteoclastogenesis and bone resorption. Finally, we repeated this experiment in animals deleted of only p55 TNFr. As seen in Figure 8 ▶ , the lesion developing in p55−/−p75+/+TNFr mice recapitulates that appearing in mice lacking both receptors.
Figure 8.
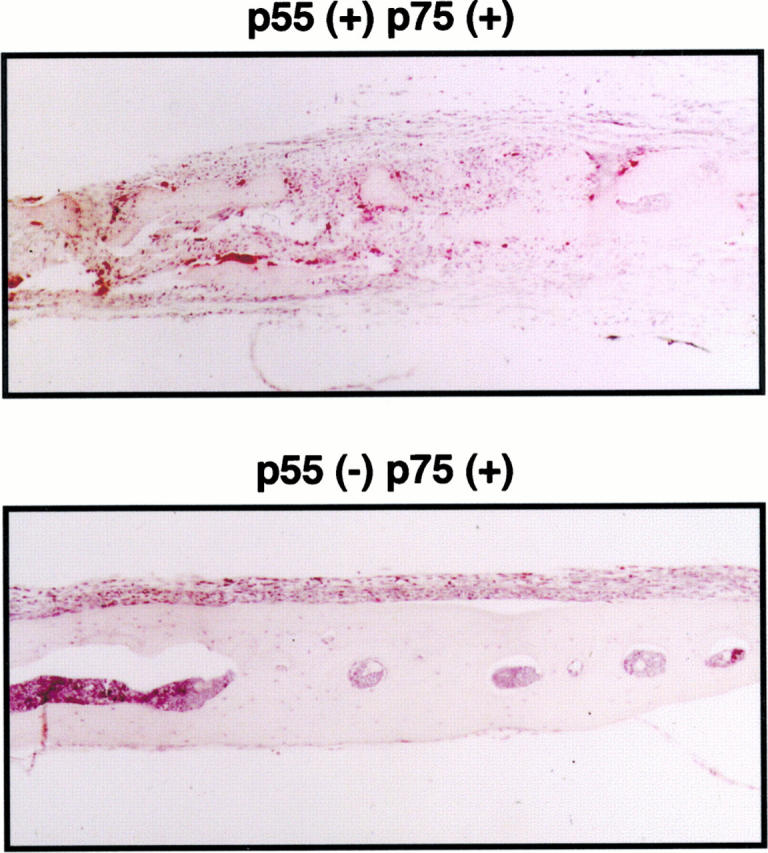
Particle-induced osteolysis is mediated via the p55TNFr. PMMA (30 mg) was placed beneath the external calvarial periosteum of p55+/+/p75+/+ and p55 −/−/p75+/+ TNFr mice. One week later the animals were sacrificed and histological sections, taken 2 to 3 mm lateral to the midsagital section, stained for TRAP activity. Although pp55−/−/p75+/+ mice develop an inflammatory reaction in response to PMMA like p55−/−/p75−/− mice, there is no evidence of osteoclastogenesis or bone resorption.
Discussion
As replacement of weight-bearing joints has yielded dramatic clinical results, loosening of these prostheses represents the major long-term complication effecting approximately 1% of patients each year. The inciting material consists of wear particles derived from various components of the implant including polyethylene, bone cement in the form of PMMA, or metal. 3 These particles insinuate between the prosthesis and bone where they induce a macrophage-rich membrane. 4 Ultimately, osteoclasts appear at the membrane-bone interface prompting focal resorption. As cytokines, such as interleukin-1, interleukin-6, and TNF, which are abundant in periprosthetic tissues, enhance resorptive activity, it is likely that one or more of these soluble agents modulate the osteoclastogenesis attending orthopedic implant loosening. 6,10-12
The osteoclast is a member of the monocyte/macrophage family. 24 Its ability to resorb bone depends on expression of the c-src proto-oncogene. 25 c-src−/− mice, although abundant in osteoclasts, develop osteopetrosis, a family of sclerotic diseases reflecting dysfunctional bone resorption. 26 These observations, taken with the rescue of c-src−/− animals by marrow transplantation 26 or overexpression of transfected c-src cDNA, 27 indicate that the proto-oncogene is essential for development of normal osteoclasts, specifically regulating their capacity to polarize and resorb bone. 25,26 The central role c-src plays in osteoclast function is consistent with the fact that its expression, by macrophages, parallels and is specific to osteoclast differentiation. 19,20
We find that particles derived from periprosthetic tissue obtained at revision arthroplasty, as well as PMMA, induce c-src in BMMs, indicating commitment of these cells to the osteoclast phenotype. This observation is in keeping with the capacity of PMMA to prompt osteoclast differentiation by mouse monocytes in vitro. 28 Furthermore, macrophages isolated from PMMA-induced foreign body granulomas become osteoclasts when cultured ex vivo. 29
Delineating the cytokines responsible for wear particle-induced osteolysis is central to defining the pathogenetic mechanisms of orthopedic implant loosening. We have shown that of a number of inflammatory cytokines, only TNF enhances c-src expression by BMMs. 19 In fact, TNF is the only soluble factor known to induce the proto-oncogene. Because TNF is a major product of activated macrophages, we reasoned that BMMs may play a dual role in implant osteolysis, namely as the host defense cell responding to the particle load and as osteoclast precursors. Taken with the capacity of PMMA particles to induce macrophage expression of TNF via transactivation of its gene, these data led us to hypothesize that particle induction of c-src, and ultimately osteoclastogenesis, is mediated by the cytokine. Our most compelling in vitro data supporting this contention is the failure of PMMA to induce c-src in BMMs lacking both TNFrs. Ultimate proof that TNF is central to implant osteolysis comes, however, from development of a in vivo murine model that mimics the human lesion. 23 Mice lacking both TNFrs develop an inflammatory response to implanted particles but fail to generate osteoclasts and thus are protected from osteolysis. This experiment establishes the central role played by TNF in the pathogenesis of implant osteolysis.
c-src induction by TNF in BMMs, as well as endotoxin-induced osteoclastogenesis, is mediated by the type-1 or p55 TNFr. 19 In contrast, our data indicate that the type-2 or p75 TNFr does not transmit osteoclastogenic signals (unpublished data). These observations prompted us to hypothesize that p55−/−p75+/+TNFr mice, like double receptor deleted animals, would be protected against particle-induced osteoclastogenesis and bone loss. The fact that such is the case indicates that therapy targeting TNF or its p55 receptor may be effective in preventing orthopedic implant loosening. Such strategies are extant in treating conditions such as AIDS and rheumatoid arthritis. 30,31 In this regard, we find thalidomide, which accelerates degradation of the mRNA of the cytokine, 30 blocks lipopolysaccharide induced c-src, an event mediated by TNF. 19 Furthermore, the anti-osteoclastogenic cytokine interleukin-4 blunts particle induction of the TNF gene. 32 Therefore, these two agents present themselves as possible inhibitors of implant osteolysis.
Acknowledgments
The authors thank Dr. Robert Schieber for helpful discussion.
Footnotes
Address reprint requests to Dr. Steven L. Teitelbaum, Department of Pathology, Washington University School of Medicine, Barnes-Jewish Hospital North, 216 South Kingshighway, St. Louis, MO 63110. E-mail: teitelbs@medicine.wustl.edu
Supported in part by a grant from the Orthopedic Research and Education Foundation (K. D. Merkel), the National Institutes of Health Grants #DE05413, #AR32788 (S. L. Teitelbaum), #AR42404 (F. P. Ross), #AR08335 (K. P. McHugh), #AR02035 (K. D. Merkel), and grants from the Barnes-Jewish Hospital Foundation (K. D. Merkel, Y. Abu-Amer).
References
- 1.Harris WH: The problem is osteolysis. Clin Orthop Relat Res 1995, 311:46-53 [PubMed] [Google Scholar]
- 2.Anonymous: Total hip replacement. The National Institutes of Health Consensus Statement 1994, 12 Sept:12–14
- 3.Jones LC, Hungerford DS: Cement disease. Clin Orthop 1987, 225:192-206 [PubMed] [Google Scholar]
- 4.Goldring SR, Clark CR, Wright TM: The problem in total joint arthroplasty: aseptic loosening. J Bone Jt Surg 1993, 75A:799-801 [DOI] [PubMed] [Google Scholar]
- 5.Jiranek WA, Machado M, Jasty M, Jevsevar D, Wolfe HJ, Goldring SR, Goldberg MJ, Harris WH: Production of cytokines around loosened cemented acetabular components. J Bone Jt Surg 1993, 75A:863-879 [DOI] [PubMed] [Google Scholar]
- 6.Kim KJ, Chiba J, Rubash HE: In vivo and in vitro analysis of membranes from hip prostheses inserted without cement. J Bone Jt Surg 1994, 76A:172-180 [DOI] [PubMed] [Google Scholar]
- 7.Linder L, Lindberg L, Carlsson A: Aseptic loosening of hip prostheses: a histologic and enzyme histochemical study. Clin Orthop 1983, 175:93-104 [PubMed] [Google Scholar]
- 8.Howie DW, Haynes DR, Hay S, Rogers SD, Pearcy MJ: The effect of titanium alloy and cobalt chrome alloy wear particles on production of inflammatory mediators IL-1, TNF, IL-6 and prostaglandin E2 by rodent macrophages in vitro. Trans Orthop Res Soc 1992, 17:344 [Google Scholar]
- 9.Horowitz SM, Doty SB, Lane JM, Burstein AH: Studies of the mechanism by which the mechanical failure of polymethylmethacrylate leads to bone resorption. J Bone Jt Surg 1993, 75:802-813 [DOI] [PubMed] [Google Scholar]
- 10.Andrew SM, Andrew JE, Hoyland JA, Wroblewski BM, Freemont AJ: Interleukin-6 in membrane from late loosening of the hip arthroplasties. Trans Orthop Res Soc 1993, 18:243 [Google Scholar]
- 11.Chiba J, Schwendeman LJ, Booth RE, Jr, Crossett LS, Rubash HE: A biochemical, histologic, and immunohistologic analysis of membranes obtained from failed cemented, and cementless total knee arthroplasty. Clin Orthop 1994, 299:114-124 [PubMed] [Google Scholar]
- 12.Appel AM, Sowder WG, Siverhus SW, Hopson CN, Herman JH: Prosthesis-associated pseudomembrane-induced bone resorption. Br J Rheum 1990, 29:32-36 [DOI] [PubMed] [Google Scholar]
- 13.Rothe J, Lesslauer W, Lotscher H, Lang Y, Koebel P, Kontgen F, Althage A, Zinkernagel R, Steinmetz M, Bluethmann H: Mice lacking the tumour necrosis factor receptor 1 are resistant to TNF-mediated toxicity but highly susceptible to infection by Listeria monocytogenes. Nature 1993, 364:798-800 [DOI] [PubMed] [Google Scholar]
- 14.Campbell P, Ma S, Schmalzried T, Amstutz H: Tissue digestion for wear debris particle isolation. J Biomed Mater Res 1994, 28:523-526 [DOI] [PubMed] [Google Scholar]
- 15.Clohisy DR, Chappel JC, Teitelbaum SL: Bone marrow-derived mononuclear phagocytes autoregulate mannose receptor expression. J Biol Chem 1989, 264:5370-5377 [PubMed] [Google Scholar]
- 16.Lipsich LA, Lewis AJ, Brugge JS: Isolation of monoclonal antibodies that recognize the transforming proteins of avian sarcoma viruses. J Virol 1983, 48:352-360 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Horowitz SM, Purdon MA: Mediator interactions in macrophage/particulate bone resorption. J Biomed Mater Res 1995, 29:477-484 [DOI] [PubMed] [Google Scholar]
- 18.McHugh K, Teitelbaum SL, Kitazawa S, et al: Cloning and characterization of the murine integrin β3 gene promoter [Abstract]. J Bone Miner Res 1994, 9(Suppl 1):S248 [Google Scholar]
- 19.Abu-Amer Y, Ross FP, Edwards J, Teitelbaum SL: Lipopolysaccharide-stimulated osteoclastogenesis is mediated by tumor necrosis factor via its p55 receptor. J Clin Invest 1997, 100:1557-1565 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tanaka S, Takahashi N, Udagawa N, Sasaki T, Fukui Y, Kurokawa T, Suda T: Osteoclasts express high levels of pp60c-src, preferentially on ruffled border membranes. FEBS Letters 1992, 313:85-89 [DOI] [PubMed] [Google Scholar]
- 21.Passeri G, Girasole G, Manolagas SC, Jilka RL: Endogenous production of tumor necrosis factor by primary cultures of murine calvarial cells: influence on IL-6 production and osteoclast development. Bone Miner 1994, 24:109-126 [DOI] [PubMed] [Google Scholar]
- 22.Beutler B, Brown T: A CAT reporter construct allows ultrasensitive estimation of TNF synthesis, and suggests that the TNF gene has been silenced in non-macrophage cell lines. J Clin Invest 1991, 87:1336-1344 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Goldring SR, Schiller AL, Roelke M, Rourke CM, O’Neill DA, Harris WH: The synovial-like membrane at the bone-cement interface in loose total hip replacements and its proposed role in bone lysis. J Bone Jt Surg 1983, 65A:575-584 [PubMed] [Google Scholar]
- 24.Shioi A, Ross FP, Teitelbaum SL: Enrichment of generated murine osteoclasts. Calcif Tissue Int 1994, 55:387-394 [DOI] [PubMed] [Google Scholar]
- 25.Boyce BF, Yoneda T, Lowe C, Soriano P, Mundy GR: Requirement of pp60c-src expression for osteoclasts to form ruffled borders and resorb bone in mice. J Clin Invest 1992, 90:1622-1627 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Soriano P, Montgomery C, Geske R, Bradley A: Targeted disruption of the c-src proto-oncogene leads to osteopetrosis in mice. Cell 1991, 64:693-702 [DOI] [PubMed] [Google Scholar]
- 27.Schwartzberg P, Xing L, Lowell CA, et al: Complementation of osteopetrosis in src−/− mice does not require src kinase activity [Abstract]. J Bone Miner Res 1996, 11:S135 [Google Scholar]
- 28.Sabokbar A, Fujikawa Y, Brett J, Murray DW, Athanasou NA: Increased osteoclastic differentiation by PMMA particle-associated macrophages: inhibitory effect by interleukin 4 and leukemia inhibitory factor. Acta Orthop Scand 1996, 67:593-598 [DOI] [PubMed] [Google Scholar]
- 29.Pandey R, Quinn J, Joyner C, Murray DW, Triffitt JT, Athanasou NA: Arthroplasty implant biomaterial particle associated macrphages differentiate into lacunar bone resorbing cells. Ann Rheum Dis 1996, 553:88-95 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Makonkawkeyoon S, Limson-Pobre RNR, Moreira AL, Schauf V, Kaplan G: Thalidomide inhibits the replication of human immunodeficiency virus type 1. Proc Natl Acad Sci USA 1993, 90:5974-5978 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Paterson DL, Georghiou PR, Allworth AM, Kemp RJ: Thalidomide as treatment of refractory aphthous ulceration related to human immunodeficiency virus infection. Clin Infect Dis 1995, 20:250-254 [DOI] [PubMed] [Google Scholar]
- 32.McHugh K, Teitelbaum SL, Merkel K, et al: Interleukin-4 blocks prosthesis wear particle-induced TNF transcription in mouse macrophages by inactivating NF-kB binding [Abstract]. J Bone Miner Res 1996, 11:S331 [Google Scholar]