Abstract
Pancreatic and colorectal carcinomas frequently express oncogenic/mutant K-Ras that contributes to both tumorigenesis and clinically observed resistance to radiation treatment. We have previously shown that farnesyltransferase inhibitors (FTI) radiosensitize many pancreatic and colorectal cancer cell lines that express oncogenic K-ras at doses that inhibit the prenylation and activation of H-Ras but not K-Ras. In the present study, we have examined the mechanism of FTI-mediated radiosensitization in cell lines that express oncogenic K-Ras and found that wild-type H-Ras is a contributor to radiation survival in tumor cells that express oncogenic K-Ras. In these experiments, inhibiting the expression of oncogenic K-Ras, wild-type H-Ras, or epidermal growth factor receptor (EGFR) led to similar levels of radiosensitization as treatment with the FTI tipifarnib. Treatment with the EGFR inhibitor gefitinib led to similar levels of radiosensitization, and the combinations of tipifarnib or gefitinib plus inhibition of K-Ras, H-Ras, or EGFR expression did not provide additional radiosensitization compared with tipifarnib or gefitinib alone. Finally, supplementing culture medium with the EGFR ligand transforming growth factor α was able to reverse the radiosensitizing effect of inhibiting K-ras expression. Taken together, these findings suggest that EGFR-activated H-Ras signaling is initiated by oncogenic K-Ras to promote radiation survival in pancreatic and colorectal cancers.
Keywords: Ras, EGFR, radiosensitivity, signal transduction, cancer
Introduction
Oncogenic (mutationally activated) Ras isoforms are found in approximately 30% of human malignancies and play a critical role in tumorigenesis and resistance of malignant neoplasms to standard therapies such as chemotherapy and radiation (reviewed in Ref. [1]). In addition, wild-type Ras can be activated to high levels through the overexpression/activation of Ras-activating signaling partners, such as the epidermal growth factor receptor (EGFR). Thus, there has been great interest and excitement in the development of farnesyltransferase inhibitors (FTIs) as a means to block Ras membrane localization and inhibit Ras-mediated signals in tumor cells. However, the clinical results obtained in the implementation of these compounds have been generally disappointing (reviewed in Refs. [2,3]).
One potential reason may be that the identity of the relevant clinical targets of FTI and the mechanism(s) of antitumor action of these compounds remain unclear. Despite being initially developed as inhibitors of Ras function, clinical and preclinical data suggest that although activated Ras may be a target for some of the effects of FTI on cell growth and survival, there are likely other targets as well [4]. The impact of FTI on Ras-mediated radioresistance is similarly complex. In H-ras-transformed REF cells as well as human tumor cell lines that express oncogenic H-ras, inhibition of H-Ras processing by FTI results in radiosensitization [5]. However, we have recently found that FTI can radiosensitize pancreatic and colorectal cancer cell lines that express oncogenic K-Ras at concentrations that inhibited posttranslational processing and activation of wild-type H-Ras, but did not appear to alter K-Ras posttranslational processing or activation [6,7]. The observation that FTI could radiosensitize tumor cell lines that express oncogenic K-Ras could be interpreted to mean that oncogenic K-Ras is not a relevant target or that it is uninvolved in the mechanism of FTI-mediated radiosensitization in pancreatic carcinoma cells. However, the oncogenic K-Ras-expressing pancreatic carcinoma cell lines that were radiosensitized by FTI alone were also radiosensitized to a similar extent by knockdown of K-Ras expression by small interfering RNA (siRNA) [6,7]. Moreover, neither K-Ras knockdown nor FTI radiosensitized pancreatic carcinoma cell lines that expressed only wild-type K-Ras [6,7].
Because activating mutations at the K-ras locus have been postulated to contribute to tumorigenesis and radiotherapy resistance in both pancreatic and colorectal carcinomas, it is critically important to better define the mechanism(s) by which potential cytotoxic and/or radiosensitizing treatments are effected by oncogenic K-Ras signaling. In addition, the relative contribution of the H- and N-Ras isoforms to radiation resistance in cancer cells that express oncogenic K-Ras remains unclear. Therefore, the current study seeks to further investigate the target and mechanism of FTI-mediated radiosensitization in oncogenic K-Ras-expressing cancer cells and to better define the role of other Ras isoforms in these cells.
Materials and Methods
Cells
PSN-1 cells were obtained through a material transfer agreement with Merck & Co Inc. (West Point, PA). DLD-1 and SG-5 cells were a kind gift of Eric Stanbridge (Irvine, CA). All other cell lines were obtained from the American Type Culture Collection (Manassas, VA). Cells were cultured in DMEM (Life Technologies, Rockville, MD) supplemented with 10% FBS (HyClone, Logan, UT), penicillin (100 U/ml), and streptomycin (100 mg/ml; Life Technologies, Inc., Rockville, MD) and maintained at 37°C in water-saturated 5% carbon dioxide-95% air. Cells were regularly tested by polymerase chain reaction (PCR) to ensure the absence of Mycoplasma contamination.
Inhibitors and siRNA
Tipifarnib (R115777) was obtained from Janssen Research (Johnson and Johnson, Springhouse, PA). Gefitinib was obtained from AstraZeneca (Wilmington, DE). Tipifarnib and gefitinib were dissolved as concentrated stock solutions in DMSO and diluted at the time of treatment with medium. Control cells were treated with medium containing an equal concentration of DMSO or nonspecific IgG, as indicated. SMARTpool siRNA directed against control (nonspecific) and specific (K-Ras, cat no. M-005069-00; H-Ras, cat no. M-004142-00; N-Ras, cat no. M-003919-00; EGFR, cat no. M-003114-01) targets were purchased from Dharmacon (Lafayette, CO).
Treatment of Cells before Clonogenic Cell Survival Assays
For experiments with siRNA, cells were seeded at 25% confluence and cultured overnight. The next morning, cells were transfected with 0.6 pmol of the indicated siRNA SMARTpool duplexes (Dharmacon RNA Technologies) in Optimem transfection medium using Oligofectamine (Life Technologies, Rockville, MD). After 12 hours, the transfection medium was replaced with fresh, prewarmed culture medium. Cultures were replated as single cells 24 hours after transfection to assure log phase growth and to facilitate harvest of single cells for survival plating. For experiments with inhibitors, the indicated inhibitor was added to mid log phase cell cultures at least 24 hours after transfection for 1 to 24 hours (as indicated) before plating for survival/irradiation and continued for 24 hours after irradiation. At that time, the medium was replaced with drug-free medium. Control cultures were also refed to control for this manipulation.
Clonogenic Cell Survival after Irradiation
Cells were treated as described above and plated as single cells before irradiation. Cells were irradiated with a Mark 1 cesium irradiator (J.L. Shepherd, San Fernando, CA) at a dose rate of 1.7 Gy/min. Colonies were stained and counted 10 to 30 days after irradiation. The surviving fraction was calculated as follows: [numbers of colonies formed] ÷ [number of cells plated (irradiated) × plating efficiency (unirradiated)]. Each point on the survival curve represents the mean surviving fraction from at least three dishes.
Whole Cell Lysate Preparation
Cells were lysed on culture dishes with reducing Laemmli sample buffer after rinsing once with PBS. Samples were boiled, sheared, clarified by centrifugation at 14,000 RPM and stored at -20°C. Protein concentrations of lysates were determined by amido black staining.
Immunoprecipitation
Immunoprecipitation was carried out as previously described [8]. Cells were treated as indicated, washed twice with ice-cold PBS, and lysed in 1 ml of ice-cold lysis buffer (1% Nonidet P-40, 100 mol/l NaCl, 50 mol/l NaF, 1 mol/l dithiothreitol, 25 mol/l benzamidine, 1 mol/l phenylmethylsulfonyl, fluoride, 2 mg/ml aprotinin, 2 mg/ml leupeptin, 2 mol/l sodium orthovanadate and 50 mol/l Tris, pH 7.4). Proteins concentrations were determined using the Bio-Rad Protein Assay Reagent according to manufacturer's instructions. One milligram of total cell protein was immunoprecipitated from each lysate using 4 µg of anti-EGFR (1 mg/ml) for 15 hours at 4°C. The resultant immune complexes were washed four times in ice-cold lysis buffer and resuspended in reducing Laemmli sample buffer for Western blot analysis.
SDS-PAGE and Western Blot Analysis
For total cell proteins, equal amounts of protein were separated by SDS-PAGE under denaturing conditions and blotted onto nitrocellulose membranes (Bio-Rad Laboratories, Hercules, CA). Membranes were blocked in PBS containing 0.1% Tween 20 and 5% powdered milk before the addition of primary antibody. N-Ras and H-Ras antibodies (Oncogene Research products, La Jolla, CA) were used at a dilution of 1:5000; K-Ras antibody (Santa Cruz Biotechnology, Santa Cruz, CA) was used at a dilution of 1:100; β-actin clone AC-15 (Sigma, St. Louis, MO) was used at 1:5000 dilution; and EGFR antibody (Cell Signaling, Danvers, MA) was used at a dilution of 1:1000. Antibody binding was detected by using the ECL chemiluminescence kit and Hyperfilm (Amersham, Arlington Heights, IL). Hyperfilm images were digitized using an AGFA Arcus II scanner (AGFA, Ridgefield Park, NJ), and figures were assembled using Adobe Photoshop and Microsoft PowerPoint.
Results
Our prior studies have shown that signaling through oncogenic H-, K-, and N-Ras can all contribute to tumor cell radiation survival. Wild-type H-Ras can also promote cellular radioresistance when overexpressed or activated by receptor signaling. Therefore, we examined the contribution of the three main Ras isoforms (N, H, and K) to radiosensitivity and FTI-mediated radiosensitization in pancreatic and colorectal cancer cells that have K-Ras oncogenic mutations. In these experiments, we have used tipifarnib, a compound that is highly selective for FTI over geranylgeranyltransferase inhibitor activity, in combination with genetic deletion and siRNA expression knockdown to separate the contributions of the Ras isoforms to radiation survival.
Tipifarnib and other FTIs when used alone can radiosensitize most, but not all, pancreatic and colorectal carcinoma cell lines that express oncogenic K-Ras, despite having no detectable effect on K-Ras processing or activation at concentrations that produce radiosensitzation. In contrast, at these concentrations H-Ras farnesylation and activity are inhibited [6,7]. These observations raised questions regarding the mechanism of radiation resistance in cells with oncogenic K-Ras. To evaluate the contribution of oncogenic K-Ras signaling to radiation survival, DLD-1 colorectal carcinoma cells and the isogenic K-Ras knockout cell line SG-5 were treated with 2.5 µmol/l tipifarnib for 24 hours before irradiation, and clonogenic cell survival after radiation was determined. DLD-1 cells express one oncogenic and one wild-type copy of K-Ras. In SG-5 cells, the oncogenic copy has been deleted by homologous recombination. Tipifarnib radiosensitized DLD-1 cells and tipifarnib-treated DLD-1 cells showed a similar level of radiosensitivity compared with radiosensitivity of the K-Ras knockout SG-5 cell line (Figure 1A). However, tipifarnib did not radiosensitize SG-5 cells (Figure 1A). Analogous results were obtained using siRNA to specifically inhibit K-Ras expression in MiaPaCa-2 cells (homozygous oncogenic K-RasG12C) in which loss of oncogenic K-Ras expression resulted in decreased radiation survival, and tipifarnib treatment did not cause additional radiosensitzation after K-Ras knockdown (Figure 1, B and C). We have also verified the isoform specificity of K-Ras knockdown in these cells (Figure 1D).
Figure 1.
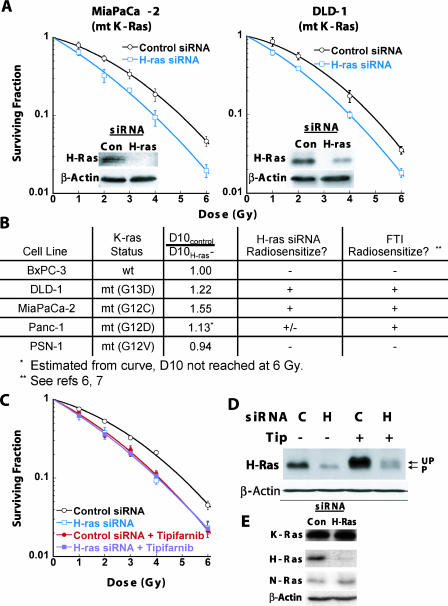
Oncogenic K-Ras expression is necessary for FTI-mediated radiosensitization. (A) DLD-1 and SG-5 cells were pretreated for 24 hours with 2.5 µmol/l tipifarnib and clonogenic cell survival assays were performed as described in Materials and Methods. DLD-1 cells express one oncogenic and one wild-type copy of K-Ras. In SG-5 cells, the oncogenic copy has been deleted by homologous recombination. (B) Clonogenic cell survival assays were performed on MiaPaCa-2 cells that were transfected with either nonspecific siRNA (control) or siRNA directed against K-Ras 48 hours before irradiation and then treated with or without 2.5 µmol/l tipifarnib 24 hours before irradiation as described in Materials and Methods. (C) Cells were treated as in (B) (without irradiation) and whole-cell lysates were analyzed by Western blot with the indicated antibody as described in Materials and Methods. C, control; K, K-Ras siRNA. (D) Western blot analysis of whole-cell lysates 48 hours after transfection with either control (con) or K-ras-specific (K-Ras) siRNA probed with antibodies specific for the K, H or N-Ras isoforms, as indicated.
We next evaluated the impact of wild-type H-Ras on radiation survival in carcinoma cell lines that express oncogenic K-Ras. In the cell lines sensitized by FTI treatment, H-Ras knockdown resulted in radiosensitization (Figure 2, A and B). This radiosensitization is similar in magnitude to the radiosensitization provided by FTI (Figure 1) [6,7]. Importantly, H-Ras knockdown failed to radiosensitize PSN-1, a pancreatic carcinoma cell line that is radiosensitized by K-Ras knockdown but is not sensitized by FTI alone (Figure 2B) [6,7]. Moreover, H-Ras knockdown did not radiosensitize BxPC-3, a pancreatic cancer cell line that expresses only wild-type K-ras (Figure 2B). The combination of tipifarnib + H-Ras knockdown did not produce a greater degree of radiosensitization than either agent alone (Figure 2, C and D). We have also verified the isoform specificity of H-Ras knockdown in these cells (Figure 2E).
Figure 2.
FTI and knockdown of wild-type H-Ras expression lead to nonadditive radiosensitization of cells that express mutant K-Ras. (A) Clonogenic cell survival assays were performed using MiaPaCa-2 and DLD-1 cells that were transfected with either nonspecific siRNA (control) or siRNA directed against H-Ras 48 hours before irradiation as described in Materials and Methods. (B) Summary of the results from clonogenic cell survival assays comparing survival after transfection of pancreatic and colorectal carcinoma cells with nonspecific siRNA to survival after transfection with H-ras specific siRNA. Radiosensitization was scored positive (indicated by “+”) if the survival ratio for control/H-ras siRNA exceeded 1.1 at both 2 Gy and at 10% clonogenic cell survival. For comparison, the ability of FTI to radiosensitize these cell lines in previous studies is similarly indicated. (C) Clonogenic cell survival assays were performed on MiaPaCa-2 cells that were transfected with either nonspecific siRNA (control) or siRNA directed against H-Ras 48 hours before irradiation and then treated with or without 2.5 µmol/l tipifarnib 24 hours before irradiation as described in Materials and Methods. (D) Cells were treated as in (C) (without irradiation) and whole cell lysates were analyzed by Western blot with the indicated antibody as described in Materials and Methods. Effects of FTI on H-Ras processing are noted as unprocessed (UP) and processed (P) H-Ras proteins. C, control siRNA; H, H-ras-specific siRNA. (E) Western blot analysis of whole-cell lysates 48 hours after transfection with either control (con) or H-ras-specific (H-Ras) siRNA probed with antibodies specific for the K, H, or N-Ras isoforms, as indicated.
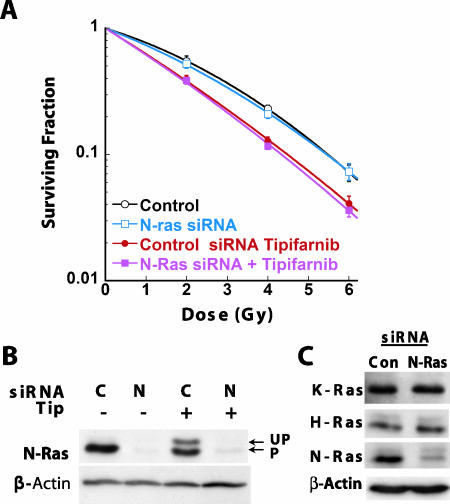
To determine whether N-Ras similarly contributes to radiosensitivity in cells that express oncogenic K-Ras, we examined the impact of N-Ras knockdown on MiaPaCa-2 cells. These experiments showed that N-Ras knockdown did not promote radiosensitization and did not interfere with tipifarnib-mediated radiosensitization (Figure 3, A and B). As a control for knockdown specificity, we have verified that knockdown with the N-Ras siRNA pool was specific for the desired Ras isoform (Figure 3C). In addition, whereas the experiments in Figures 1 to 3 use Dharmacon SMARTpool siRNAs to achieve isoform-specific knockdown, we have obtained similar results using self-designed siRNA that are complimentary to different areas of the K- and H-Ras primary sequences (data not shown). Together these data indicate that wild-type H-Ras, but not N-Ras, has a role in determining the radiosensitivity of cells expressing oncogenic K-Ras.
Figure 3.
Expression of N-Ras is not necessary for FTI-mediated radiosensitization. (A) Clonogenic cell survival assays were performed on MiaPaCa-2 cells after transfection with control or N-Ras-specific siRNA 48 hours before irradiation followed by treatment with or without 2.5 µmol/l tipifarnib 24 hours before irradiation. (B) Cells were treated as in (A) (without irradiation) and whole-cell lysates were analyzed by Western blot with the indicated antibody as described in Materials and Methods. C, control siRNA; N, N-ras-specific siRNA. Effects of FTI on N-Ras processing are noted as unprocessed (UP) and processed (P) N-Ras proteins. (C) Western blot analysis of whole-cell lysates 48 hours after transfection with either control (con) or N-ras-specific (N-Ras) siRNA probed with antibodies specific for the K, H, or N-Ras isoforms, as indicated.
Autocrine activation of EGFR by transforming growth factor (TGF)α occurs in response to transfection of rat intestinal epithelial cells transfected with oncogenic K-ras [9]. In addition, TGFα has been shown to activate EGFR-dependent H-Ras signaling in pancreatic cancer cells that express mutant K-ras [10] and antibodies to TGFα have been shown to radiosensitize oncogenic K-Ras-expressing A549 cells [11]. Therefore, we hypothesized that wild-type H-Ras that is stimulated by EGFR activation is the target of FTI-mediated radiosensitization in cells that express oncogenic K-Ras. In initial experiments, gefitinib inhibition of EGFR activation was used to test this model. Gefitinib-mediated inhibition of EGFR activation radiosensitized oncogenic K-Ras-expressing pancreatic carcinoma cells and radiosensitization by gefitinib was not additive to K-Ras knockdown (Figure 4A). These results are in agreement with data that EGFR inhibitors can radiosensitize other cells that express oncogenic K-Ras, but implicate oncogenic K-Ras as a contributor to EGFR-mediated radioresistance [11–13]. We have also found that cetuximab, an immunoglobulin-derived EGFR inhibitor, radiosensitizes DLD-1 cells (data not shown).
Figure 4.
Signaling though EGFR determines radiation survival in cells that express oncogenic K-Ras. (A) Clonogenic cell survival assays were performed on MiaPaCa-2 cells that were transfected with or without 10 µmol/l gefitinib 24 hours before irradiation as described in Materials and Methods. In parallel samples (without irradiation), EGFR was immunoprecipitated and Western blot analysis with an antiphosphotyrosine antibody was performed as described in Materials and Methods. (B) Clonogenic cell survival assays were performed on MiaPaCa-2 cells after transfection with control or EGFR specific siRNA 48 hours before irradiation followed by treatment with or without 2.5 µmol/l tipifarnib 24 hours before irradiation. (C) Cells were treated as in (B) (without irradiation) and whole-cell lysates were analyzed by Western blot with the indicated antibody as described in Materials and Methods. C, control siRNA; E, EGFR-specific siRNA. (D) Clonogenic cell survival assays were performed on MiaPaCa-2 cells after transfection with control or K-Ras specific siRNA 48 hours before irradiation followed by treatment with or without 200 pmol/l TGFα 24 hours before irradiation. (E) Clonogenic cell survival assays were performed on MiaPaCa-2 cells after transfection with control or H-Ras-specific siRNA 48 hours before irradiation followed by treatment with or without 200 pmol/l TGFα 24 hours before irradiation.
One potential concern with these EGFR inhibitor studies is that there may be off-target effects, such as inhibition of other HER family members that complicate experimental interpretation. To more specifically assess the contribution of the EGFR signaling to radiation survival in oncogenic K-Ras-expressing cells, an siRNA approach was used. In these experiments, EGFR knockdown radiosensitized MiaPaCa-2 cells and the combination of EGFR knockdown with tipifarnib treatment did not radiosensitize cells more than either agent used alone (Figure 4, B and C). These results confirm the experiments with gefitinib and suggest that tipifarnib inhibition blocks cell signaling initiated at the EGFR. Therefore, we tested whether activation of EGFR by the addition of exogenous TGFα could reverse the radiosensitive phenotype of K-Ras knockdown cells. In these experiments, TGFα added to cell cultures 24 hours after transfection with control or K-Ras-specific siRNA (24 hours before irradiation) was able to completely reverse the ability of K-Ras knockdown to radiosensitize pancreatic cancer cells (Figure 4D). Taken together, these findings suggest that oncogenic K-Ras promotes H-Ras activation by EGFR, leading to enhanced radiation survival.
Discussion
We and others have shown that the activation of H-Ras, either by mutation or EGFR activation, leads to increased phosphatidyl inositol 3′-kinase (PI3K)/Akt-mediated radio-resistance in a variety of human and rodent cell lines (reviewed in Refs. [2,14]). In cells that express oncogenic K-ras, inhibiting K-Ras function with a combination of FTI/geranylgeranyltransferase inhibitor, siRNA knockdown, or genetic deletion leads to increased radiosensitivity of cancer cells [6,15–20]. Recently, we found that FTI alone can radiosensitize some, but not all, oncogenic K-Ras-expressing colorectal and pancreatic cancer cell lines [6,7], raising questions about the role of K-Ras activity in radiation survival. In the present study, we have shown that the mechanism for this radiosensitization involves FTI-mediated H-Ras inhibition. We have further demonstrated that oncogenic K-Ras promotes radiation survival through EGFR signaling.
In our studies, inhibition of EGFR signaling through either gefitinib or siRNA led to increased radiosensitivity in oncogenic K-Ras-expressing cells. These data are in agreement with studies from several other investigators showing that EGFR inhibitors can radiosensitize cells that express oncogenic K-Ras [11–13]. Moreover, promising results have been obtained from preliminary clinical trials of combined EGFR inhibitor plus chemoradiotherapy for patients with pancreatic cancer [21]. However, it had been previously unclear how a cell line with a mutation that is supposedly “downstream” can be sensitized by inhibiting the “upstream” receptor. Our data suggest that the mechanism that underlies this phenomenon is the ability of oncogenic K-Ras to activate EGFR-mediated signaling through wild-type H-Ras.
This mechanism helps to explain the previously observed discrepancies between Ras mutation status and FTI sensitivity in cancer cells. However, it is important to point out that the target and mechanism of FTI-mediated cellular radiosensitization are not necessarily the same as the target and mechanism of FTI-mediated cellular cytotoxicity or growth inhibition. In this respect, it is interesting to note that addition of an EGFR inhibitor to gemcitabine chemotherapy produced a relative increase of 25% in 1-year survival in patients with pancreatic cancer (absolute increase from 17% to 24%) [22,23]. It is unclear whether K-Ras-mediated activation of EGFR alone is sufficient to promote EGFR inhibitor-mediated direct cytotoxicity. In one study of a panel of EGFR-expressing pancreatic cancer cell lines, coexpression of Erb-B3 was found to be a better predictor of EGFR inhibitor-mediated cytotoxicity than Ras status or EGFR expression level [24]. Thus, the potential contribution of oncogenic K-Ras-mediated EGFR activation to pancreatic and colorectal cancer biology and response to therapy requires further investigation.
Despite our current findings, the role of the various Ras isoforms in cellular radiosensitivity similarly requires further study. Some oncogenic K-Ras-expressing cell lines are clearly radiosensitized by inhibiting either K-Ras or PI3K/Akt activation [6,18,19,25]. However, other oncogenic K-Ras-expressing cell lines appear to be less sensitive to PI3K inhibition [20,26]. In addition, transfection of oncogenic K-Ras into Rat-2 cells results in increased radiosensitivity, possibly through a p38 mitogen-activated protein kinase (MAPK)-dependent mechanism [27]. One potential explanation for this phenomenon lies in genetic heterogeneity of these systems. It seems likely that the phenotype of cells that express activated Ras isoforms will depend, at least to some extent, on the levels of coexpression of different Ras interacting signaling partners. Our results predict that EGFR would be one such partner. Another possibility lies in the differential subcellular localization of Ras isoforms when expressed at different levels. At low expression levels, activated H- and K-Ras localize to distinct, non-cholesterol-containing plasma membrane microdomains, and this may be one mechanism of the observed preference of H-Ras for activating PI3K > MAPK, whereas K-Ras activates MAPK > PI3K [28]. However, at higher H- or K-Ras expression levels this confined localization begins to break down [28,29]. Thus, at different expression levels or in different cellular contexts, H- and K-Ras activation may lead to differential activation of signaling partners such as PI3K and MAPK. In this context, it is interesting to note that the oncogenic K-Ras-expressing cell line PSN-1 that was not radiosensitized by FTI alone has a very high level of oncogenic K-Ras expression (unpublished observations).
In conclusion, our data are consistent with a model in which oncogenic K-Ras leads to activation of EGFR and H-Ras to promote cellular radioresistance. In addition, these results indicate that H-Ras is a target of FTI-mediated radiosensitization in cells that express oncogenic K-Ras. These results have significant implications for targeting cellular survival signals in tumors expressing oncogenic K-Ras.
Abbreviations
- EGFR
epidermal growth factor receptor
- FTI
farnesyltransferase inhibitor
Footnotes
This study was supported by grants RO1 CA73820 (E.J.B.) and PO1 CA75138 (W.G.M.) and an Eastern Cooperative Oncology Group/Aventis/Ortho Biotech Young Investigator Award (K.A.C.).
References
- 1.Shaw RJ, Cantley LC. Ras, PI(3)K and mTOR signalling controls tumour cell growth. Nature. 2006;441:424–430. doi: 10.1038/nature04869. [DOI] [PubMed] [Google Scholar]
- 2.Cengel KA, McKenna WG. Molecular targets for altering radiosensitivity: lessons from Ras as a pre-clinical and clinical model. Crit Rev Oncol Hematol. 2005;55:103–116. doi: 10.1016/j.critrevonc.2005.02.001. [DOI] [PubMed] [Google Scholar]
- 3.Brunner TB, Hahn SM, Gupta AK, Muschel RJ, McKenna WG, Bernhard EJ. Farnesyltransferase inhibitors: an overview of the results of preclinical and clinical investigations. Cancer Res. 2003;63:5656–5668. [PubMed] [Google Scholar]
- 4.Sebti SM, Der CJ. Opinion: searching for the elusive targets of farnesyltransferase inhibitors. Nat Rev Cancer. 2003;3:945–951. doi: 10.1038/nrc1234. [DOI] [PubMed] [Google Scholar]
- 5.Bernhard EJ, Kao G, Cox AD, Sebti SM, Hamilton AD, Muschel RJ, McKenna WG. The farnesyltransferase inhibitor FTI-277 radiosensitizes H-ras-transformed rat embryo fibroblasts. Cancer Research. 1996;56:1727–1730. [PubMed] [Google Scholar]
- 6.Brunner TB, Cengel KA, Hahn SM, Wu J, Fraker DL, McKenna WG, Bernhard EJ. Pancreatic cancer cell radiation survival and prenyltransferase inhibition: the role of K-Ras. Cancer Res. 2005;65:8433–8441. doi: 10.1158/0008-5472.CAN-05-0158. [DOI] [PubMed] [Google Scholar]
- 7.Cengel KA, Deutsch E, Stephens TC, Voong KR, Kao GD, Bernhard EJ. Radiosensitizing effects of the prenyltransferase inhibitor AZD3409 against RAS mutated cell lines. Cancer Biol Ther. 2006;5:1206–1210. doi: 10.4161/cbt.5.9.3172. [DOI] [PubMed] [Google Scholar]
- 8.Cengel KA, Freund GG. JAK1-dependent phosphorylation of insulin receptor substrate-1 (IRS-1) is inhibited by IRS-1 serine phosphorylation. J Biol Chem. 1999;274:27969–27974. doi: 10.1074/jbc.274.39.27969. [DOI] [PubMed] [Google Scholar]
- 9.Grana TM, Sartor CI, Cox AD. Epidermal growth factor receptor autocrine signaling in RIE-1 cells transformed by the Ras oncogene enhances radiation resistance. Cancer Res. 2003;63:7807–7814. [PubMed] [Google Scholar]
- 10.Seufferlein T, Van Lint J, Liptay S, Adler G, Schmid RM. Transforming growth factor alpha activates Ha-Ras in human pancreatic cancer cells with Ki-ras mutations. Gastroenterology. 1999;116:1441–1452. doi: 10.1016/s0016-5085(99)70509-3. [DOI] [PubMed] [Google Scholar]
- 11.Toulany M, Dittmann K, Baumann M, Rodemann HP. Radiosensitization of Ras-mutated human tumor cells in vitro by the specific EGF receptor antagonist BIBX1382BS. Radiother Oncol. 2005;74:117–129. doi: 10.1016/j.radonc.2004.11.008. [DOI] [PubMed] [Google Scholar]
- 12.Buchsbaum DJ, Bonner JA, Grizzle WE, Stackhouse MA, Carpenter M, Hicklin DJ, Bohlen P, Raisch KP. Treatment of pancreatic cancer xenografts with Erbitux (IMC-C225) anti-EGFR antibody, gemcitabine, and radiation. Int J Radiat Oncol Biol Phys. 2002;54:1180–1193. doi: 10.1016/s0360-3016(02)03788-4. [DOI] [PubMed] [Google Scholar]
- 13.Toulany M, Kasten-Pisula U, Brammer I, Wang S, Chen J, Dittmann K, Baumann M, Dikomey E, Rodemann HP. Blockage of epidermal growth factor receptor-phosphatidylinositol 3-kinase-AKT signaling increases radiosensitivity of K-RAS mutated human tumor cells in vitro by affecting DNA repair. Clin Cancer Res. 2006;12:4119–4126. doi: 10.1158/1078-0432.CCR-05-2454. [DOI] [PubMed] [Google Scholar]
- 14.Bernhard EJ. Farnesyltransferase inhibition: who are the Aktors? Cancer Biol Ther. 2004;3:1099–1101. doi: 10.4161/cbt.3.11.1301. [DOI] [PubMed] [Google Scholar]
- 15.Sklar MD. The ras oncogenes increase the intrinsic resistance of NIH 3T3 cells to ionizing radiation. Science. 1988;239:645–647. doi: 10.1126/science.3277276. [DOI] [PubMed] [Google Scholar]
- 16.Bernhard EJ, McKenna WG, Hamilton AD, Sebti SM, Qian Y, Wu JM, Muschel RJ. Inhibiting Ras prenylation increases the radiosensitivity of human tumor cell lines with activating mutations of ras oncogenes. Cancer Res. 1998;58:1754–1761. [PubMed] [Google Scholar]
- 17.Bernhard EJ, Stanbridge EJ, Gupta S, Gupta AK, Soto D, Bakanauskas VJ, Cerniglia GJ, Muschel RJ, McKenna WG. Direct evidence for the contribution of activated N-ras and K-ras oncogenes to increased intrinsic radiation resistance in human tumor cell lines. Cancer Res. 2000;60:6597–6600. [PubMed] [Google Scholar]
- 18.Grana TM, Rusyn EV, Zhou H, Sartor CI, Cox AD. Ras mediates radioresistance through both phosphatidylinositol 3-kinase-dependent and Raf-dependent but mitogen-activated protein kinase/extracellular signal-regulated kinase kinase-independent signaling pathways. Cancer Res. 2002;62:4142–4150. [PubMed] [Google Scholar]
- 19.Kim IA, Bae SS, Fernandes A, Wu J, Muschel RJ, McKenna WG, Birnbaum MJ, Bernhard EJ. Selective inhibition of Ras, phosphoinositide 3 kinase, and Akt isoforms increases the radiosensitivity of human carcinoma cell lines. Cancer Res. 2005;65:7902–7910. doi: 10.1158/0008-5472.CAN-05-0513. [DOI] [PubMed] [Google Scholar]
- 20.Caron RW, Yacoub A, Zhu X, Mitchell C, Han SI, Sasazuki T, Shirasawa S, Hagan MP, Grant S, Dent P. H-RAS V12-induced radio-resistance in HCT116 colon carcinoma cells is heregulin dependent. Mol Cancer Ther. 2005;4:243–255. [PubMed] [Google Scholar]
- 21.Iannitti D, Dipetrillo T, Akerman P, Barnett JM, Maia-Acuna C, Cruff D, Miner T, Martel D, Cioffi W, Remis M, et al. Erlotinib and chemoradiation followed by maintenance erlotinib for locally advanced pancreatic cancer: a phase I study. Am J Clin Oncol. 2005;28:570–575. doi: 10.1097/01.coc.0000184682.51193.00. [DOI] [PubMed] [Google Scholar]
- 22.Moore M, Goldstein D, Hamm J, Figer A, Hecht J, Gallinger S, Au H, Ding K, Christy-Bittel J, Parulekar W. Erlotinib plus gemcitabine compared to gemcitabine alone in patients with advanced pancreatic cancer. A phase III trial of the National Cancer Institute of Canada Clinical Trials Group [NCIC-CTG] Proc ASCO. 2005;23:1. [Google Scholar]
- 23.Yokoi K, Kim SJ, Thaker P, Yazici S, Nam DH, He J, Sasaki T, Chiao PJ, Sclabas GM, Abbruzzese JL, et al. Induction of apoptosis in tumor-associated endothelial cells and therapy of orthotopic human pancreatic carcinoma in nude mice. Neoplasia. 2005;7:696–704. doi: 10.1593/neo.05193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Buck E, Eyzaguirre A, Haley JD, Gibson NW, Cagnoni P, Iwata KK. Inactivation of Akt by the epidermal growth factor receptor inhibitor erlotinib is mediated by HER-3 in pancreatic and colorectal tumor cell lines and contributes to erlotinib sensitivity. Mol Cancer Ther. 2006;5:2051–2059. doi: 10.1158/1535-7163.MCT-06-0007. [DOI] [PubMed] [Google Scholar]
- 25.Gupta AK, Cerniglia GJ, Mick R, McKenna WG, Muschel RJ. HIV protease inhibitors block Akt signaling and radiosensitize tumor cells both in vitro and in vivo. Cancer Res. 2005;65:8256–8265. doi: 10.1158/0008-5472.CAN-05-1220. [DOI] [PubMed] [Google Scholar]
- 26.Caron RW, Yacoub A, Li M, Zhu X, Mitchell C, Hong Y, Hawkins W, Sasazuki T, Shirasawa S, Kozikowski AP, et al. Activated forms of H-RAS and K-RAS differentially regulate membrane association of PI3K, PDK-1, and AKT and the effect of therapeutic kinase inhibitors on cell survival. Mol Cancer Ther. 2005;4:257–270. [PubMed] [Google Scholar]
- 27.Choi JA, Park MT, Kang CM, Um HD, Bae S, Lee KH, Kim TH, Kim JH, Cho CK, Lee YS, et al. Opposite effects of Ha-Ras and Ki-Ras on radiation-induced apoptosis via differential activation of PI3K/Akt and Rac/p38 mitogen-activated protein kinase signaling pathways. Oncogene. 2004;23:9–20. doi: 10.1038/sj.onc.1206982. [DOI] [PubMed] [Google Scholar]
- 28.Prior IA, Muncke C, Parton RG, Hancock JF. Direct visualization of Ras proteins in spatially distinct cell surface microdomains. J Cell Biol. 2003;160:165–170. doi: 10.1083/jcb.200209091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Niv H, Gutman O, Kloog Y, Henis YI. Activated K-Ras and H-Ras display different interactions with saturable nonraft sites at the surface of live cells. J Cell Biol. 2002;157:865–872. doi: 10.1083/jcb.200202009. [DOI] [PMC free article] [PubMed] [Google Scholar]